Abstract
Introduction
Allergic rhinitis (AR) is one of the most important allergic airway diseases in children. It is an allergic disorder of the mucous membranes of the nasal passages. Allergic rhinitis is caused by an immune-mediated inflammatory reaction characterized by raised serum immunoglobulin E (IgE), mucus hypersecretion, and eosinophilic cell infiltration. 1,2 Patients usually presented with paroxysmal sneezing, watery rhinorrhea, and nasal blockage. 3 At present, AR is considered a major health problem affecting up to 40% of children worldwide, which may interfere with sleep and quality of life in AR children and impose a considerable burden on public health in economic terms. 1,3 The exact etiology of AR remains poorly understood, and it is an active area of research. 2 Vitamin D deficiency is progressively documented as a possible etiologic or disease-modifying factor in many allergic disorders, including asthma, 4 eczema, 5 and food allergy. 6 However, a few pieces of research have studied the relationship between vitamin D status and AR in children, especially in the Middle East. The epidemiologic pattern for the deficiency of vitamin D is coupled with the rising epidemic of atopic and allergic diseases in the developed countries so raises the likelihood of an association between the 2 variables. 2 This study aimed to assess the serum levels of vitamin D in a large Egyptian cohort of children with AR and to evaluate any correlation of vitamin D status with the disease severity.
Materials and Methods
Compliance With Ethical Standards
The procedures of our study were following the regulations of the relevant clinical research Ethics Committee and with those of the code of ethics of the world medical association Declaration of Helsinki). Written informed consents of parents of all children were taken after explanation of the study objectives and its benefits for their children. Faculty of Medicine, Assiut University Ethical Committee approved the study protocol.
Study Design
This was a case-controlled study undertaken in Assiut and Al-Azhar University Hospitals, Assiut city, Egypt.
Patients
This study included 120 patients aged 7 to 13 years with AR who were recruited to the study from 2 outpatient clinics in Al-Azhar and Assiut University Hospitals, Assiut, Egypt during winter 2016 (from September to December to avoid the seasonal variation in vitamin D levels.). The diagnosis of AR was performed according to Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines and supported by a skin prick test. 7 Skin prick tests were performed using the same antigens for all patients with a wide panel of most widespread aeroallergens in our locality, which included house dust, hay dust, mixed mites, mixed molds, mixed pollens, and cat epithelium. In addition to the allergens tested, there were positive and negative controls. The positive control, was a histamine solution, should become itchy within a few minutes, then red, and swollen with a wheal in the center. The negative control was a saline solution and should show no reaction. A senior otorhinolaryngologist evaluated all patients and controls before the children were recruited into the study. The severity grading and classification of all children with AR were performed based upon ARIA criteria into mild or moderate-severe rhinitis 7 and total nasal symptom score (TNSS, nasal congestion, runny nose, itchy nose, and sneezing). Total nasal symptom score was assessed in all patients depending on the severity of AR manifestations. For every symptom TNSS has the following scores: 0 = no symptom; 1 = mild, unremarkable symptoms; 2 = moderate, disturbing but tolerable; and 3 = severe, disturbing, interfere with routine activities and/ or sleep. 8 One hundred age- and sex-matched nonatopic, healthy children were recruited as normal control group. Any treatment with systemic corticosteroids and antihistamines were stopped 2 weeks before the study. The exclusion criteria were: (1) any accompanying disease that could affect vitamin D status, for example, rheumatologic diseases, chronic gastrointestinal and renal diseases, and endocrine disorders; (2) children who had received some drugs, for example, vitamin D, calcium-containing drugs, and immunotherapy; (3) patients with other types of rhinitis, for example, infectious, drug-induced, hormonal, or another non-AR.
Laboratory Investigations
Venous blood samples were collected from all patients and controls after 12-hour fasting between 8:00 and 10:00
Statistical Analysis
We analyzed our data by the Statistical Package for Social Sciences version 22 (SPSS Inc, Chicago, Illinois, USA). Data were expressed as the mean (standard deviation [SD]). The mean values in AR and healthy groups were compared by the unpaired Student t test and Mann-Whitney U test. The correlation coefficient r was generated by Spearman rank correlation. Statistical significance was defined as p < .05.
Results
Table 1 shows demographic and laboratory criteria of all study participants. Table 2 shows the classification and nasal manifestations of all patients. Our study was conducted on 220 children. There were 120 children in the AR group (mean age 6.29 [4.03] years; 54 [45%] males) and 100 children in control group (mean age: 7.08 [2.91] years; 43 [43%] males). There was no statistical difference between both groups in age, gender, socioeconomic status, and anthropometric data. No significant differences were found on comparison of birth month and season of birth between children with AR and healthy controls.
Demographic and Laboratory Characteristics of Study Patients.a
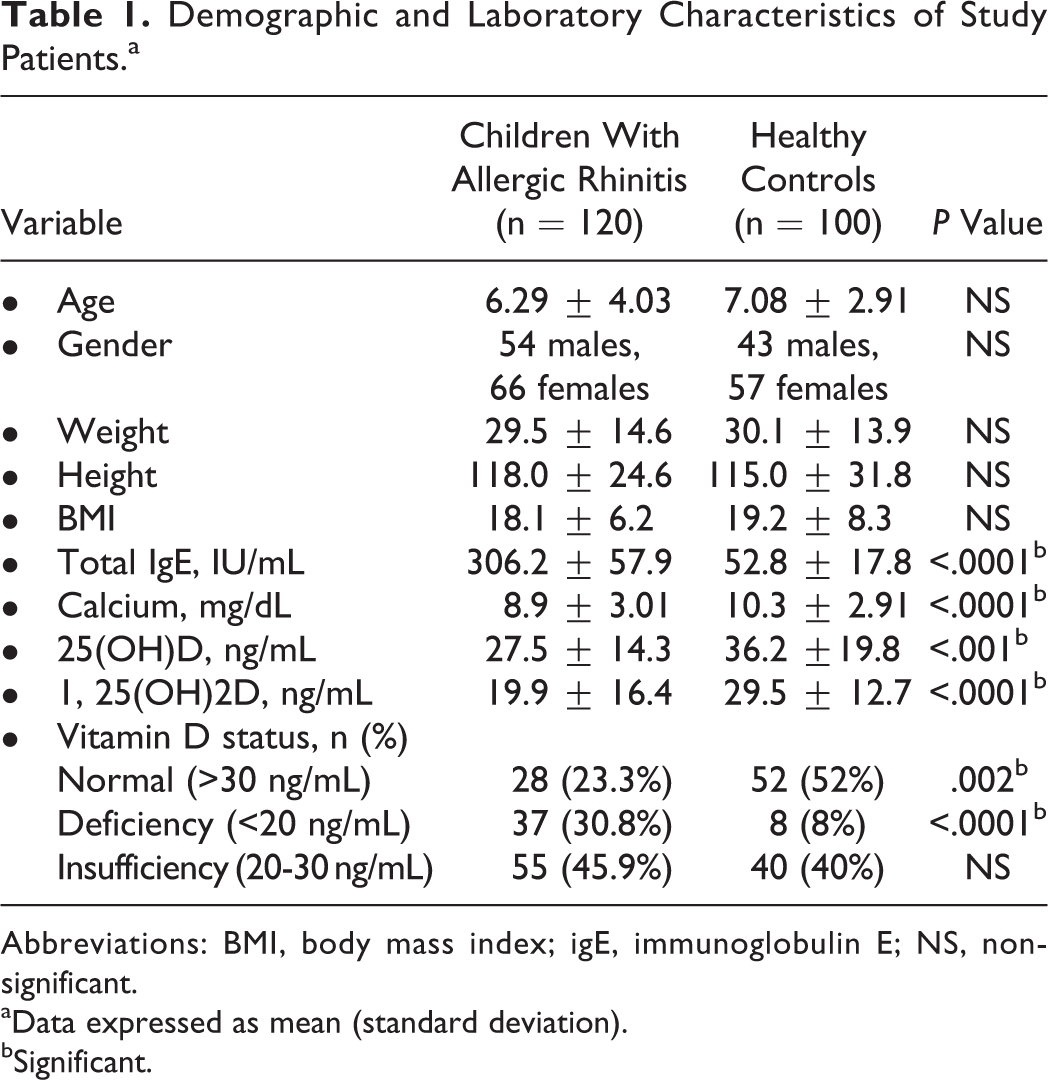
Abbreviations: BMI, body mass index; igE, immunoglobulin E; NS, non-significant.
aData expressed as mean (standard deviation).
bSignificant.
Classification, Nasal Signs, and Symptoms of Patients With Allergic Rhinitis.
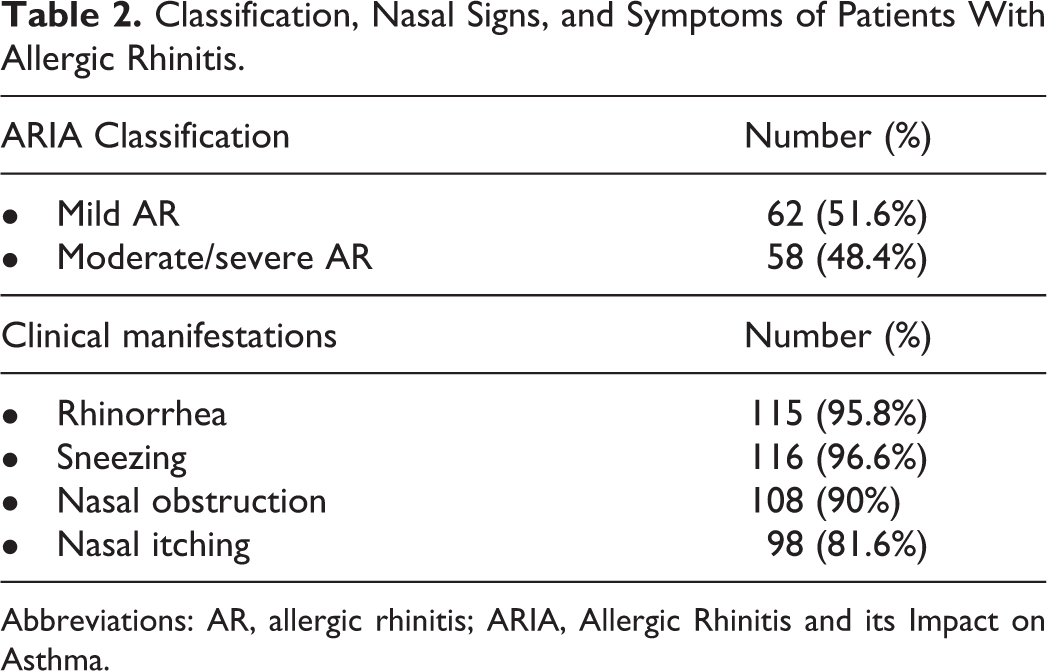
Abbreviations: AR, allergic rhinitis; ARIA, Allergic Rhinitis and its Impact on Asthma.
Children with AR had significantly higher total serum IgE and calcium levels than those of the healthy control group (p < .0001 for each). The mean serum levels 25(OH)D and 1,25(OH)2D of were significantly lower in AR group than in healthy children ( p < .001 and p < .0001 respectively, Table 1).
In the present study, only 23.3% of AR children had normal serum 25-OHD concentration (>30 ng/mL) versus 52% of controls (p = .002), 45.9% of the patient group had vitamin D insufficiency (20-30 ng/mL) versus 40% in healthy controls (p = .4). Vitamin D deficiency (<20 ng/mL) was found in 30.8% of AR group versus 8% of healthy children (p < .0001, Table 1).
According to AR severity between both groups, we found the mean 25-OHD3 levels in patients with moderate/severe AR were significantly lower than those with mild AR (p < .001). Correlation analyses between vitamin D levels and various parameters were done in our study. We found a significant positive correlation between 25(OH)D and calcium (calcium levels (r = .29, p = .023). Besides, we found significant negative correlations between mean 25(OH)D levels and TNSS (r = −0.62, p = .002, Table 3) and total IgE levels (r = −0.27, p = .013) in AR group. No significant difference was found in comparison of birth month and season of birth between children with AR and healthy controls.
Relationship Between Total Nasal Symptom Score and Vitamin D Levels.
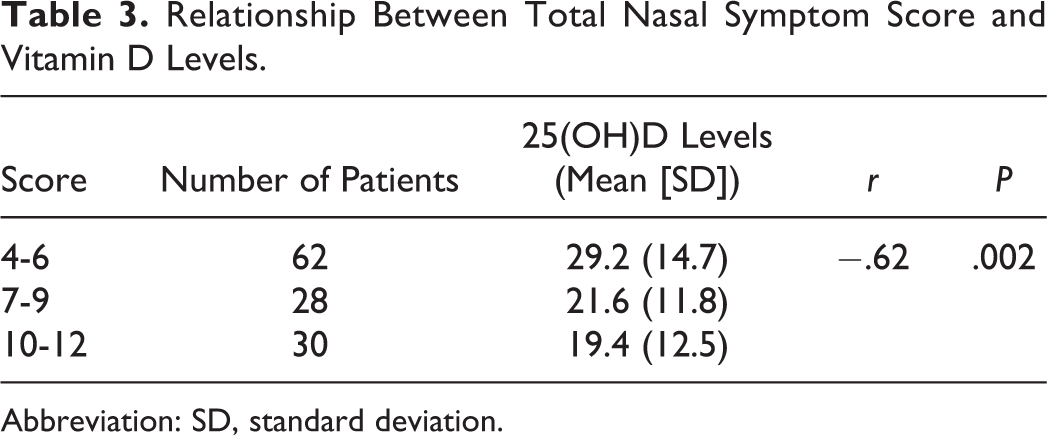
Abbreviation: SD, standard deviation.
Discussion
Our study identified essential results in the growing area of vitamin D levels and allergic children. This study investigated vitamin D status in a large cohort of children with AR. We observed that vitamin D deficiency is a frequent finding among children with AR when compared to healthy controls. In line with our report, Dogru and Suleyman 10 studied 76 children with AR and 65 healthy patients. They evaluated 25(OH)D3 levels in both groups, and the mean serum 25(OH)D3 levels of AR children were significantly lower than healthy group (18.07 ± 6.1 ng/mL vs 24.03 ± 9.43 ng/mL; p = .001). However, contrary to our finding, they failed to find any significant relationship between vitamin D deficiency and the severity of AR. Another study (11) compared the levels of 1,25(OH)2D3 in 30 children with AR having recurrent tonsillitis and 30 controls. The median levels of 1,25(OH)2D3 were significantly lower in AR group when compared to healthy children (34.65 pg/mL vs 52.86 pg/mL; p = .001). In the same report, 11 the researchers found significantly lower 1,25(OH)2D3 values (p < .05) in children with severe nasal symptoms. Yenigun et al 12 and Goksugur et al 13 investigated 2 small groups of children with allergic rhinoconjunctivitis; they found significantly lower vitamin D levels in patients with allergic rhino-conjunctivitis when compared to controls. In recent years, the importance of vitamin D in the immune system and allergic diseases has received much interest in research. There is a general agreement that the shift from a Th1 to Th2 phenotype in CD4+ T cells is the primary pathogenesis of AR, but the precise mechanism of AR is still not clear. Current researches showed that Th17 and T-regulatory cells are essential in the pathogenesis of AR. 12,14 Vitamin D inhibits the proliferation of T-lymphocytes and increases Th1 to Th2 switch by stimulation of the development of Th2 cells. In addition, it enables the induction of T-regulatory cells and suppresses the development, transcription, and bioactivity of Th17 cells. 12,14 These data indicate that there is a relationship between vitamin D and AR morbidity. 15 Deficiency of vitamin D leads to contraction of the smooth muscles and disturbance of prostaglandin regulation with impairment of Th1–Th2 balance which increases airway inflammation. 16
Some clinical and epidemiological studies have reported significant associations between vitamin D intake during pregnancy and its serum level with the incidence of AR in different ethnic groups. Two studies 17,18 reported a positive relationship between maternal vitamin D intake and AR. Erkkola et al 17 reported an inverse relationship between maternal intake of vitamin D during late pregnancy and the risk of asthma (confidence interval [CI]: 0.64-0.99) and AR (CI: 0.75-0.97) in 5-year-old children. Bunyavanich et al 18 studied a cohort of 1248 mother–child pairs, and they found that the intake of each 100 IU of vitamin D/d during the first and second trimesters was associated with 21% (odd ratio[OR]: 0.79 [95% CI: 0.67-0.92] and 20% (OR: 0.80 [95% CI: 0.68-0.93] reduced odds of ever AR at school age. Bener et al 19 reported that vitamin D deficiency was significantly associated with AR children. Another report by Jung et al 20 used the data from the annual Korean National Health and Nutrition Examination 2009 Survey. They reported lower mean vitamin D levels in patients with AR when compared to the healthy adult group, after adjusting demographic data, body mass index, sun exposure, socioeconomic status, exercise, and body fat percentage. Arshi et al 16 measured vitamin D levels in adult patients with AR and compared their data to the general population (no control group). The prevalence of severe 25(OH)D deficiency was significantly higher in patients with AR than the normal population, (30% vs 5.1% respectively; p = .03). Inconsistent with our results and the previous studies, there were few studies that reported no association between AR and vitamin D levels or positive association with the use of vitamin D. 21 -23 Hyppönen et al 21 found that regular use of vitamin D supplementation was associated with an increased risk of atopy and AR. Other studies 22,23 observed no significant association between serum vitamin D levels and AR. Our data were not consistent with the previous findings; this may be due to differences in study designs, the age of the patients selected, different methods of clinical assessment of AR, and laboratory investigations of vitamin D levels.
Recently, Aryan et al 2 performed a systematic review and meta-analysis about vitamin D status and AR. They found that the association of vitamin D status with AR was age- and sex specific. Children with serum 25(OH)D more than 30 ng/mL had lower odds for having AR. However, this was not significant (OR: 0.91, 95% CI: 0.81-1.01), while this figure was significant in adults (OR: 0.64; 95% CI: 0.51-0.81).
Conclusions and Recommendations
Vitamin D deficiency is a frequent finding among Egyptian children with AR when compared to the healthy group. A significant inverse association was observed between vitamin D levels and AR disease severity, therefore determining vitamin D levels should be considered in the assessment of children with AR. Additional studies are required to determine the effects, dose, and duration of vitamin D therapy for the management of AR. Moreover, more studies are essential to fully elucidate the actual mechanism of action of vitamin D in AR.
Footnotes
Authors’ Note
All authors have read and agreed with the content of this study and no portion of this work has been published previously or is under consideration for publication elsewhere and is an original article. Khaled Saad, Abobakr Abdelmoghny, Mohamed Diab Aboul-Khair, Yasser Farouk Abdel-Raheem, Bahaa Hawary conceptualized the study design, writing of the paper, collection of sample and interpretation of data. Asmaa M. Zahran and Mohamd A. Alblihed did all the laboratory tests for our studied children. Amira Elhoufey and Ahmed El-Sayed Hammour did the statistical analysis of data. The protocol of this study was in accordance with the regulations of the relevant clinical research Ethics Committee and with those of the code of ethics of the world medical association Declaration of Helsinki. Written informed consents of caregivers of all children were taken according to the Ethical Committee of Faculty of Medicine, Assiut University, Egypt, the date of approval January 2, 2015 (number 126-1-2015).
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
