Abstract
We aimed to clarify the relation between allergic rhinitis and the serum levels of 25-hydroxivitamin D in the adult population. The study group consisted of 86 patients with allergic rhinitis who were diagnosed with the help of history of allergy, positive signs for allergy, blood samples, and positive skin prick tests; while the control group included 43 age- and sex-matched healthy volunteers with negative skin prick tests. The demographic data, medical history, findings in the physical examinations, serum levels of total immunoglobulin E (IgE) and 25-hydroxyvitamin D, and skin prick test results of the groups were noted. A total of 129 patients fulfilling the necessary criteria were enrolled. The median serum 25-hydroxyvitamin D levels in the study group were significantly lower compared to the control group (P = .014). In the study group, median serum vitamin D levels were significantly higher in men, compared to women (P = .03). There was a significant negative correlation between IgE and vitamin D levels in the allergic rhinitis group (P = .028, r = −0.246). This study showed that patients with allergic rhinitis might be more vulnerable to have lower serum levels of vitamin D. Thus, vitamin D supplementation as an adjunctive therapy may be considered in those patients.
Introduction
Allergic rhinitis (AR) is a common allergic disorder with type 1 allergic reaction of the nasal mucosa distinguished by paroxysmal sneezing, itching, serous rhinorrhea, and nasal blockage. However, ocular symptoms, postnasal drip, itching of the palate, and chronic cough can associate the nasal symptoms. The incidence rate has been increasing worldwide. 1,2 According to Allergic rhinitis and its impact on asthma (ARIA) guidelines, the prevalence of self-reported AR is approximately 1% to greater than 40% in adult population. 1 The prevalence of confirmed AR in European adults was reported as between 17% and 28.5%. 1,3 Although the morbidity and mortality are relatively low, AR has remarkable burden and costs.
In patients with AR, the antigens inhaled through the nasal mucosa are binded to immunoglobulin E (IgE) antibodies on the mast cells, located in the entire nasal mucosa. This antigen–antibody complex provokes the synthesis of various chemical mediators, especially histamine and peptide leukotrienes from the mast cells. These chemicals stimulate the nerve endings and blood vessels, leading to sneezing, rhinorrhea, and nasal mucosal congestion. Nearly 6 to 10 hours after the antigen exposure, various cytokines, chemokines, and activated eosinophils are synthesized, which are responsible for the late-phase reactions. 2
In addition to its role in skeletal development, vitamin D has various important roles in nonskeletal tissues. It has complex various immunoregulatory effects on both innate and adaptive immunities. 4,5 Vitamin D, via its nuclear receptors (vitamin D receptor) found in various immunologically active cells, such as macrophages, T cells, B cells, and dendritic cells, function both as an anti-inflammatory and an immunoprotective agent. 4,5,6
Since the past 50 years, many studies have been conducted to investigate the relation between allergy and the serum levels of vitamin D; however, the results are inconsistent and most of them were performed on the pediatric population. We aimed to clarify the relation between AR and the serum levels of 25-hydroxivitamin D in the adult population.
Materials and Methods
The research was conducted at Başkent University Medical Faculty Otorhinolaryngology Department between September 2018 and February 2019. Following the sample size calculations for both groups, a total of 129 patients fulfilling the necessary criteria were enrolled. The study group consisted of 86 consecutive patients with AR, while the control group included 43 age- and sex-matched healthy volunteers, who had no history of allergy and had negative skin prick tests. Exclusion criteria were as follows: Pediatric patients Patients with comorbid systemic diseases Gestation Being diagnosed with musculoskeletal diseases such as osteoporosis Presence of acute upper respiratory tract infections Patients using drugs, such as phenobarbital, phenytoin, and cholestyramine Patients diagnosed with non-AR Patients using hormone replacement or oral contraceptive therapy Smoking Patients already using medical therapies for AR and/or vitamin D deficiency Patients who have skin diseases or any condition that may have the possibility to interfere the skin prick test.
All the patients involved in the study were informed and asked to sign the clinical research informed consent form. This study was approved by Başkent University institutional review board and ethics committee (project no.: KA19/98) and supported by Başkent University Research Fund.
The demographic data, medical history, findings in the physical examinations, serum levels of total IgE and 25-hydroxyvitamin D, and skin prick test results of the groups were noted. The diagnosis of AR was made clinically according to ARIA guidelines. 1
The skin prick test was performed using standardized allergen extracts Dermatophagoides pteronyssinus, Dermatophagoides farinea, grass-mix pollens, grain-mix pollens, weed-mix pollens, Blatella germanica (cockroaches), cat dander, dog dander, latex, Plantain plantago, Parietaria officinalis, Cladosporium, Alternaria alternata, Artemisia vulgaris, Penicillium mix, Olea europaea, tomato, banana, and Betula betulaceae (Allergopharma D-21462 Reinbek, Germany), with the technique standardized by the European Academy of Allergy and Clinical Immunology. 7 Histamine and physiologic saline were used for positive and negative controls, respectively. The skin reactions on both forearms were recorded after 15 minutes and the wheal diameters were measured. Results 3 mm greater than the negative control were marked positive. Patients with positive skin test to at least one of the allergens were considered atopic.
Even though 1,25-dihydroxyvitamin D is an active form, serum 25-hydroxyvitamin D is the most abundant (100-fold higher serum levels than the active form) and stable form of vitamin D in human systemic circulation. Serum 25-hydroxyvitamin D is considered to reflect directly the serum vitamin D levels and preferred test to diagnose vitamin D insufficiency. Serum 25-hydroxyvitamin D concentrations were determined by a chemiluminescent microparticle immunoassay with Alinity analyzer (Abbott Diagnostics, Chicago, Illinois). The assay is standardized against National Institute of Standards and Technology Standard Reference Material 2972 (NIST SRM 2972). Levels <20 ng/mL, between 20 and 30 ng/mL, and ≥30 ng/mL were defined as group 1 (deficient), group 2 (insufficient), and group 3 (normal), respectively. 6,8,9
SPSS version 25.0 (IBM Corporation, Armonk, New York) program was used to analyze the variables. The Shapiro-Wilk test was used to determine the normal distribution of the data. The Mann-Whitney U test was used along with Monte Carlo results for comparing the 2 independent groups to each other according to the quantitative data. Kruskal-Wallis H test and the Whitney U test were used to determine the correlations of the variables and Spearman ρ test was used to examine the correlations of the variables. In the comparison of categorical variables, Pearson χ2 and Fisher exact tests were analyzed using exact results, while Fisher-Freeman-Holton test was tested with Monte Carlo Simulation technique. Quantitative variables were expressed as median (minimum/maximum) and categorical variables as n (%). The variables were examined at 95% confidence level and P value less than .05 was considered significant.
Results
The age, sex distribution, the number of patients in each subgroup, median serum vitamin D, and total IgE levels in both groups were presented in Table 1. The median serum 25-hydroxyvitamin D levels in the study group were significantly lower in the study group compared to the controls (P = .014; Table 1).
Demographic Data, Vitamin D, and Total IgE Levels in Both Groups.
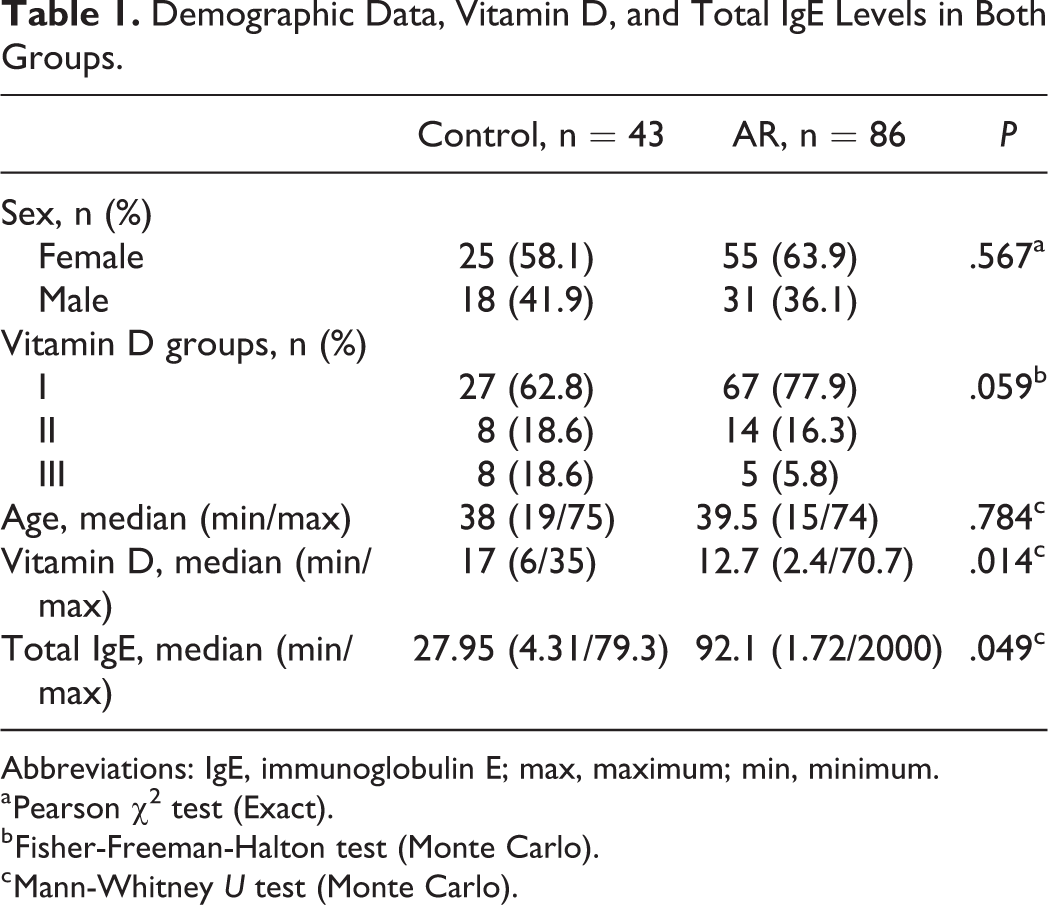
Abbreviations: IgE, immunoglobulin E; max, maximum; min, minimum.
a Pearson χ2 test (Exact).
b Fisher-Freeman-Halton test (Monte Carlo).
c Mann-Whitney U test (Monte Carlo).
In the AR group, there was no statistical difference of sex among subgroups, but in group 1 and group 2, there was statistically significant difference of age (P = .028). Total IgE levels were significantly different in group 1 and group 3 in AR group (P = .003; Table 2). In the control group, there was no difference for the parameters mentioned above (P > .05).
Age and Serum Total IgE Levels Reached Statistical Significance.
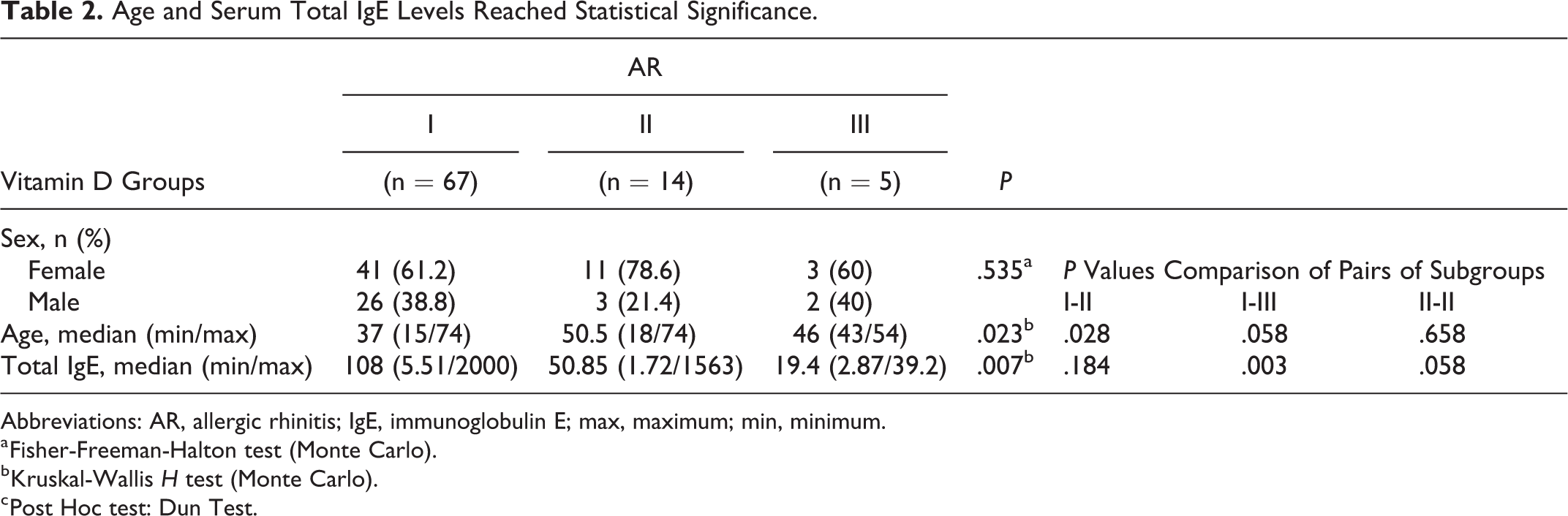
Abbreviations: AR, allergic rhinitis; IgE, immunoglobulin E; max, maximum; min, minimum.
a Fisher-Freeman-Halton test (Monte Carlo).
b Kruskal-Wallis H test (Monte Carlo).
c Post Hoc test: Dun Test.
When all the 3 subgroups were compared for age, sex, and vitamin D levels, there was no statistically significant difference among the study and control groups (P > .005; Tables 3 -5).
Comparison of Vitamin D Subgroup I Among the Study and Control Groups for Sex, Age, and Serum Vitamin D Levels.
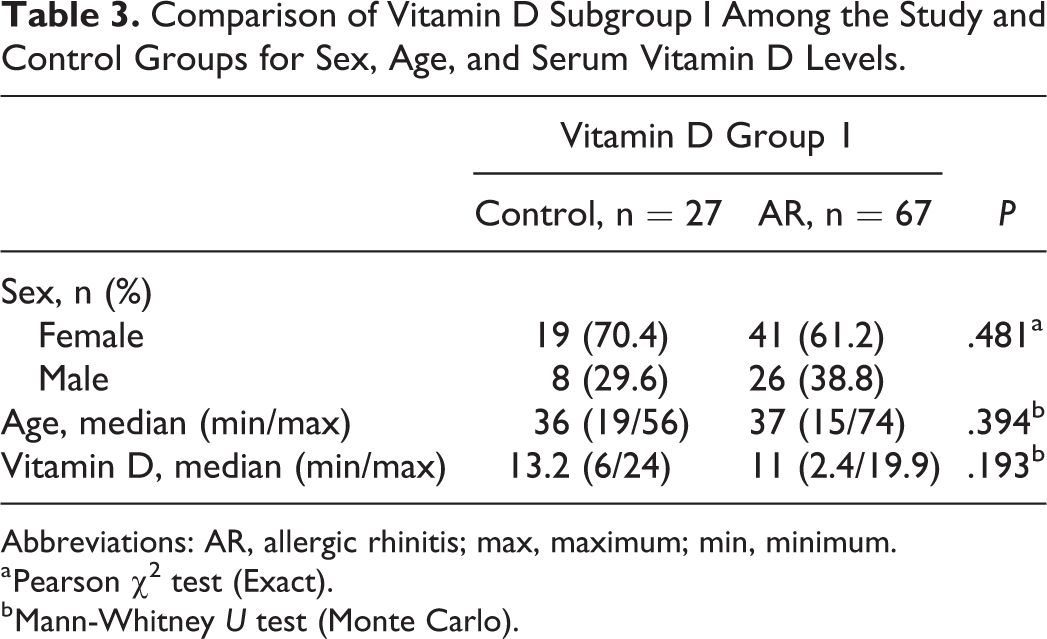
Abbreviations: AR, allergic rhinitis; max, maximum; min, minimum.
a Pearson χ2 test (Exact).
b Mann-Whitney U test (Monte Carlo).
Comparison of Vitamin D Subgroup 2 Among the Study and Control Groups for Sex, Age, and Serum Vitamin D Levels.
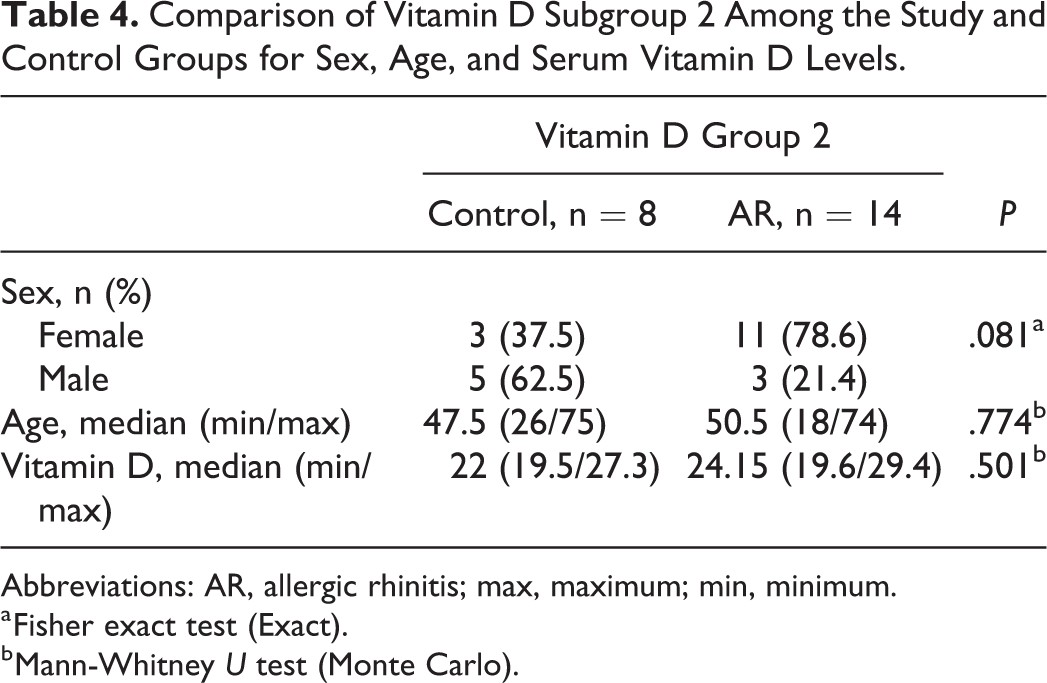
Abbreviations: AR, allergic rhinitis; max, maximum; min, minimum.
a Fisher exact test (Exact).
b Mann-Whitney U test (Monte Carlo).
Comparison of Vitamin D Subgroup 3 Among the Study and Control Groups for Sex, Age, and Serum Vitamin D Levels.
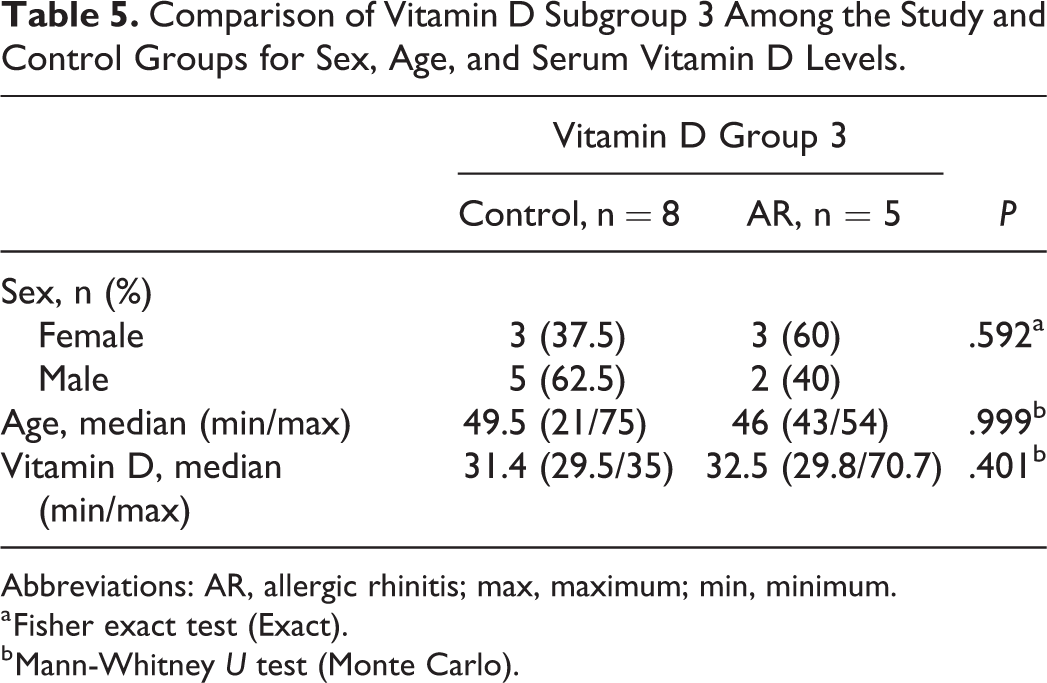
Abbreviations: AR, allergic rhinitis; max, maximum; min, minimum.
a Fisher exact test (Exact).
b Mann-Whitney U test (Monte Carlo).
In the control group, serum vitamin D levels showed no statistically significant difference among females and males (P > .05; Table 6). In the study group, median serum vitamin D levels were significantly higher in men, compared to women (P = .03; Table 7).
Comparison of Vitamin D levels Among Females and Males in the Control Group.

Abbreviations: max, maximum; min, minimum.
a Fisher-Freeman-Halton test (Monte Carlo).
b Mann-Whitney U test (Monte Carlo).
Comparison of Vitamin D Levels Among Females and Males in the Study Group.
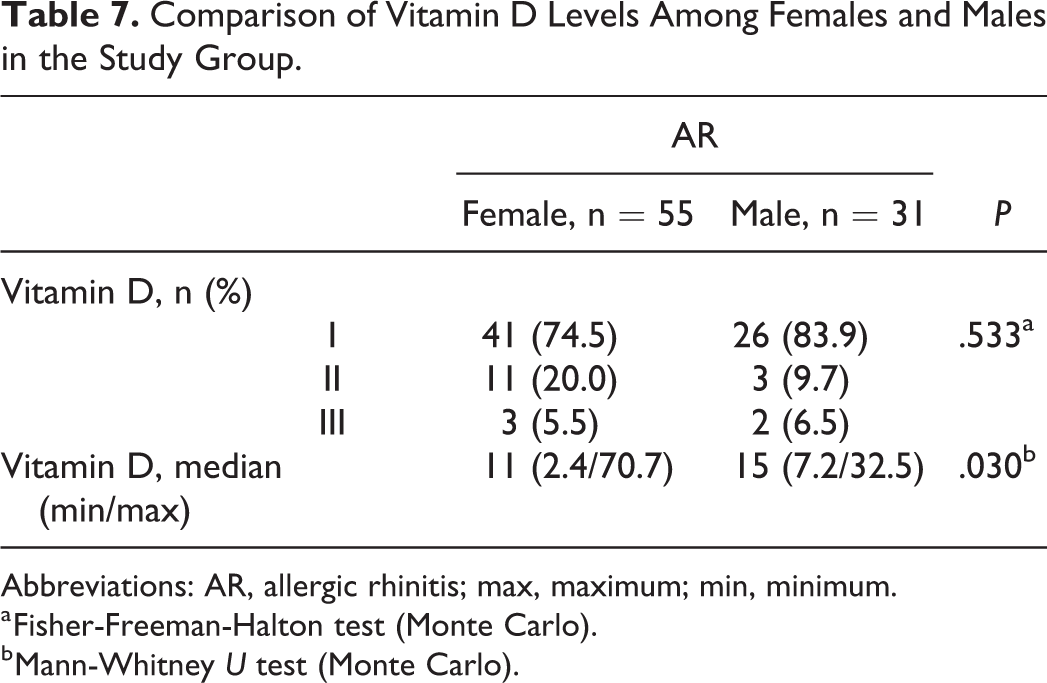
Abbreviations: AR, allergic rhinitis; max, maximum; min, minimum.
a Fisher-Freeman-Halton test (Monte Carlo).
b Mann-Whitney U test (Monte Carlo).
There was a significant negative correlation between IgE and vitamin D levels in the AR group (P = .028, r = −0.246).
Discussion
The majority of the daily vitamin D requirement is by exposure to sunlight. However, intensity of the sunlight, amount of skin exposed, duration of exposure, the zenith angle of the sun, thickness of the skin, and the skin color differ the amount of vitamin D generation among individuals. 10
Apart from its skeletal effects, vitamin D has various nonskeletal benefits, including improved immunity, decreased diabetes mellitus, prevention of cancer, decreased all-cause mortality, decreased pulmonary morbidities, and decreased cardiovascular diseases. 10
Considering the importance of vitamin D in regulating the immune system, the relation between AR and the levels of vitamin D has attracted interest in recent years.
It has been known for nearly 30 years that vitamin D has immunoregulatory effects on T cells, B cells, dendritic cells, monocytes, and macrophages. 6 1,25-Dihydroxyvitamin D inhibits the proliferation of T cells, induces a shift from Th1 to Th2, and provokes the synthesis of T regulatory cells that are important immune reactions in the course of AR. 11 -13 Additionally, vitamin D activates apoptosis of activated B cells and inhibits plasma cell differentiation and immunoglobulin secretion, including IgE. 14,15
Vitamin D increases the antimicrobial activity in monocytes; however in later stages, it provokes them to secrete anti-inflammatory agents and decreases macrophage inflammation and T-cell proliferation. 16,17 Additionally, it induces up and down regulations of various specific cytokines. 11
Various studies suggested that higher levels of vitamin D are associated with reduced risk of asthma attacks. 18,19,20 However, the studies searching the relationship between AR and vitamin D are contradictory. A survey study from Germany 21 reported that patients with high serum levels of vitamin D are at high risk for AR. The authors defined AR by the question “Did a doctor ever tell you had hay fever?.” Mai et al 22 conducted the HUNT survey and found opposite results. Aryan et al 23 found a negative correlation between AR and levels of vitamin D in pediatric population. Keleş et al 5 reported similar results in their study, in which they searched serum 1,25-dihydroxyvitamin D levels in adults. Lee et al 4 found that serum levels of vitamin D were lower in AR compared to vasomotor rhinitis and control groups in Korean children. They also noted an inverse correlation between IgE levels and vitamin D. We also detected a negative correlation between serum IgE levels and vitamin D.
Wu et al 24 found lower serum levels of vitamin D in persistent AR when compared to the controls; however, their results did not reach statistical significance. Bunjavanich et al 25 reported that each 100 IU/d of vitamin D intake during the first and second trimesters of gestation was decreasing the incidence of AR at school age. Dogru and Suleyman 26 compared serum 25-hydroxyvitamin D levels in children with AR and non-ARs and found that the mean serum levels of vitamin D in patients with AR and non-AR were lower than the controls. Jung et al 27 investigated the role of vitamin D in Korean adults and found a significant relation between vitamin D and AR. The determination of AR was achieved by having an answer “yes” to a question “Are you diagnosed AR by a doctor?”, history of rhinitis symptoms, and patients’ rhinological findings.
Another study found a significant correlation between AR in males and levels of vitamin D. 23 On contrary, in a study by Lee et al, 4 no gender differences of vitamin D status among groups were observed. In our study, the serum levels of vitamin D are significantly lower in AR group. Males had significantly higher serum vitamin D levels compared to females in our study group. Women may spend more time indoors compared to men due to sociocultural, socioeconomic causes, and child care. Therefore, the amount of sun exposure and its duration may be shortened, causing lower serum vitamin D levels.
In order to maintain a normal immunological activity, the required minimum level of 25-hydroxyvitamin was determined as 30 ng/mL. 10 For respiratory system infections and asthma attacks, levels at least between 30 and 40 ng/mL are recommended. We grouped the individuals as group 1, group 2, and group 3 according to the serum levels of vitamin D as aforementioned. We considered the levels ≥30 ng/mL as normal, which is consistent with the literature. 8,10,28
We observed significantly lower levels of 25-hydroxyvitamin D in AR group when compared to the healthy volunteers. To our knowledge, this current study has the widest patient series among the studies searching the relation between 25-hydroxyvitamin D levels and AR, which was confirmed by history of allergy, endoscopic examination, skin prick, and allergy-specific laboratory tests.
However, how vitamin D affects the development and/or exacerbations of AR is still unknown. The effects of vitamin D supplementation therapies and the optimal serum levels of vitamin D to suppress AR are still unclarified. Further prospective studies including larger patient series are necessary.
We performed our study between September and February, a time period in which exposure to sunlight is relatively low. Vitamin D levels alter in different seasons. Therefore, further studies investigating the correlation between AR and vitamin D levels in different seasons are required.
As Kim et al 29 suggested, lower vitamin D levels may co-occur with AR, instead of causing it. People with AR may refrain outdoor activities to avoid exacerbations of AR. They also suggested that use of oral steroids for atopic dermatitis and asthma may cause lower levels of vitamin D. It seems that the relationship between AR and vitamin D status is versatile and complex.
Conclusion
As the prevalence of AR is increasing, an increasing amount of studies are being conducted in order to highlight the immunological pathogenesis of the disease. Vitamin D, which is an immunomodulating agent, seems to have a role in the pathogenesis of AR. This study suggested that serum levels of 25-hydroxyvitamin D may have an impact on AR. However, there is much to clarify with further studies, regarding this topic.
Footnotes
Authors’ Note
Informed consent was obtained from all individuals included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
