Abstract
The present research was carried out with the objective to establish the clinical effect and safety of betahistine (48 mg daily), for the management of peripheral vestibular vertigo, in patients treated by primary care physicians in Colombia. An observational prospective cohort study was conducted including patients older than 15 years with clinical diagnosis of peripheral vestibular vertigo who were candidates to be treated with betahistine (48 mg daily). A sample size of 150 individuals was calculated, and weekly follow-ups were planned for 12 weeks. Rotatory movement sensation, loss of balance, and global improvement scale from 0 to 100 points were evaluated. Complete improvement was defined when the patient reached a level of 100 points. We calculated average weekly improvement, cumulative incidence of complete improvement, incidence rate of complete improvement, and the probability of complete improvement as a function of time. After the first week, the average improvement was 56.6 points (95% confidence interval [CI]: 50.4-62.7). At the end of week 12, it was 89.3 points (95% CI: 86.5-92.2). Sixty-one percent of the patients had achieved complete improvement at the end of the second week. After the sixth week, the percentage of cumulative improvement was 72%, and after 12 weeks of follow-up, the cumulative incidence of complete improvement was 73% (95% CI: 65%-80%). Based on the follow-up times, a complete improvement incidence rate of 16 cases per 100 people/week was calculated (95% CI: 13-19). We concluded that Betahistine (48 mg daily) has a positive effect, controlling the symptoms associated with benign paroxysmal vertigo, with an adequate safety profile.
Introduction
The international classification of vestibular disorders defined vertigo as an illusion of movement, generally rotating, either of oneself or of the environment, associated with feelings of instability, 1 which can be classified as central or peripheral according to the location of the possible lesion in the vestibular system. 2 Regarding the frequency of this condition in medical practice, some authors have estimated that it causes about 11 consultations per 1000 people per year and that the main associated diagnoses, according to the findings described in a study conducted with the participation of 16 general practitioners in Ireland, are benign positional paroxysmal vertigo (42.2%), acute vestibular neuritis (40.8%), and Ménière’s disease (10%) whose sum corresponds to 93% of the associated diagnoses with this disorder. 3
Although most of the pathologies responsible for vestibular vertigo are of a benign nature and have a good prognosis, each patient requires a process of periodic evaluation and follow-up, and simultaneously, it is recommended to provide the patient with a treatment that helps control the associated symptomatology. 4 According to the results of the study conducted in 13 countries by Agus et al, 5 including data from 4294 patients, betahistine was the most prescribed medication for the control of symptoms in these patients with a percentage close to 87%, and according to the results of a systematic review published by the Cochrane Collaboration in 2016, the evidence suggests that in patients with vertigo, betahistine can reduce symptoms with a low percentage of adverse events. 4
The previous conclusion is based on a synthesis of clinical trials, and although these have been considered the “gold standard” to establish the effectiveness of a treatment, nowadays limitations of external validity linked to the high selection of studied populations are recognized and, therefore, may be insufficient to guide clinical practice. 6 It is for this reason that real-world data have become important arguments for health decision-making, since they can be used to understand the effectiveness of treatment and to provide an idea of the patterns of care, the safety of medicines in the long term, the use of health resources, and the epidemiology of diseases. 7,8
Based on the described problem, where on the one hand is vertigo as a frequent condition in primary care, which affects the quality of life of those who suffer from it 9 and on the other hand, the need to have results obtained from the real world, this investigation was developed with the objective to establish the clinical effect and the safety of betahistine for the management of vertigo in the context of an observational study, developed in real-life conditions, in patients treated by a group of general primary care physicians in Colombia.
Patients and Methods
Design
An observational prospective cohort study was carried out within the framework of the activities of a primary care program for the surveillance of adverse events and clinical outcomes in frequent pathologies in primary care.
Population and Sample
Between July and December 2017, in 12 cities of Colombia, within the usual clinical practice of a group of 21 general primary care physicians, through sequential sampling, resulting in the inclusion of patients older than 18 years, with clinical diagnosis of vertigo peripheral vestibular, who were candidates to be treated—according to medical criteria—with betahistine 48 mg/d. Patients were informed of the objectives of the investigation, and their informed consent to make use of the clinical follow-up data was requested. It was defined by protocol that the data of the patients who did not accept to participate would be excluded; however, all the patients granted their consent and participated voluntarily.
For the sample size calculation, the formula proposed by Lemeshow et al was used to estimate a proportion, including the desired precision. 10 Based on the results of the study carried out by Parfenov et al, 11 we assume a 90% expected clinical effect for betahistine, with a level of accuracy of 5% and a type I error of 5%, plus a 10% adjustment for potential losses, we obtained a final sample size of 150 patients.
Enrollment and Follow-Up Procedure
In real-life conditions, each doctor provided care to their patient, made the corresponding diagnosis, and assigned the treatment according to their clinical criteria. When patients met the criteria to be part of the registry, they were asked for their informed consent to be able to document and analyze the data on the clinical evolution of their condition. In each case, the professional defined the behavior according to his criteria and allowed the research group to know the baseline and follow-up data. The patients were followed weekly for a period of 12 weeks.
Variables of Interest
The sociodemographic variables of age and sex were recorded, and as outcomes of interest, the sensation of rotatory movement and loss of balance, recorded in a dichotomous scale (presence or absence), were evaluated during the initial visit and in the follow-up of control. Additionally, the level of improvement obtained was evaluated, measured by the attending physician at each visit, on a scale of 0 to 100. This variable was reclassified in a dichotomous scale (complete clinical improvement: yes or no), considering a positive result when the value reported by the doctor was 100 points. The time to event of complete improvement was documented, calculating the difference in weeks between the date of diagnosis and the date of complete improvement (100 points of improvement) or the last recorded follow-up. Finally, serious adverse events and adverse events that would have forced the suspension of treatment were evaluated.
Statistical Analysis
A general description of the study variables was made, using measures of frequency, as well as central tendency and dispersion statistics according to the scale of measurement. A baseline evaluation of the dependent variables was carried out, and it was compared to the results obtained during the clinical follow-up. The average improvement obtained from 0 to 100 was calculated in each visit, and the data are described in a comparative chart, with their respective 95% confidence interval (CI). The percentages of positive findings were compared at the initial visit and at the final follow-up, by means of a proportions difference test. The cumulative incidence of response to treatment was calculated, using the quotient between the number of patients who responded to the therapy (100 improvement points), and the total number of patients who completed the follow-up. For this estimator, the respective 95% CI was calculated. Given that the time to event was documented for each case from the onset of symptoms to the improvement or completion of follow-up, the incidence of improvement was calculated with its respective 95% CI, and Kaplan Meier’s function was also estimated, with the purpose of estimating the probability of complete improvement as a function of time of a case of peripheral vestibular vertigo treated with betahistine 48 mg/d. Serious adverse events were described through absolute and relative frequencies. For hypothesis testing, an α value of .05 was defined, considering that a P value below this cutoff point would be statistically significant.
Ethical Aspects
This project complied with all the national and international research ethics regulations and was approved by an independent ethics committee. The patients were informed of the research objectives, and their informed consent was requested to make use of the clinical follow-up data for research purposes. By protocol, the data of the patients who did not accept to participate would be excluded, notwithstanding the above, all the patients granted their consent and participated voluntarily.
Results
After giving their informed consent, 170 patients entered the cohort. Of the 170, a total of 150 patients went to the programmed follow-up appointments and completed the treatment prescribed by the treating doctor for an adherence percentage of 88%. This group included a total of 110 (73%) women and 40 (27%) men, with ages ranging from 20 to 94 years, with a median age of 61 years and 50% between 43 and 75 years old. The time during which patients were under treatment with betahistine 48 mg/d ranged between 4 and 12 weeks, with a median of 6 weeks. The description and analysis of clinical outcomes are presented on the denominator of 150 patients who followed the recommendations of their treating physician and completed the 12-week clinical follow-up.
Description of Baseline Findings and Follow-Up
Upon admission to the cohort, 100% of the patients reported episodes of rotating movement sensation of the body or environment and 66% reported a sensation of loss of balance or instability (99/150). At the end of the follow-up, absolute control of the sensation of rotary movement was achieved in 110 cases, corresponding to 73% (P value: .000), and only 12 patients, corresponding to 8%, continued with sensation of instability or loss of balance (P value: .000).
After the first week all the patients reported some level of improvement, which on a scale of 0 to 100 ranged from 5 to 100 points, with an average of 56.6 points (95% CI: 50.4-62.7). At the end of week 12 follow-up, the level of improvement ranged between 60 and 100 points, with an average of 89.3 points (95% CI: 86.5-92.2). The detail of the weekly behavior is presented in Figure 1.
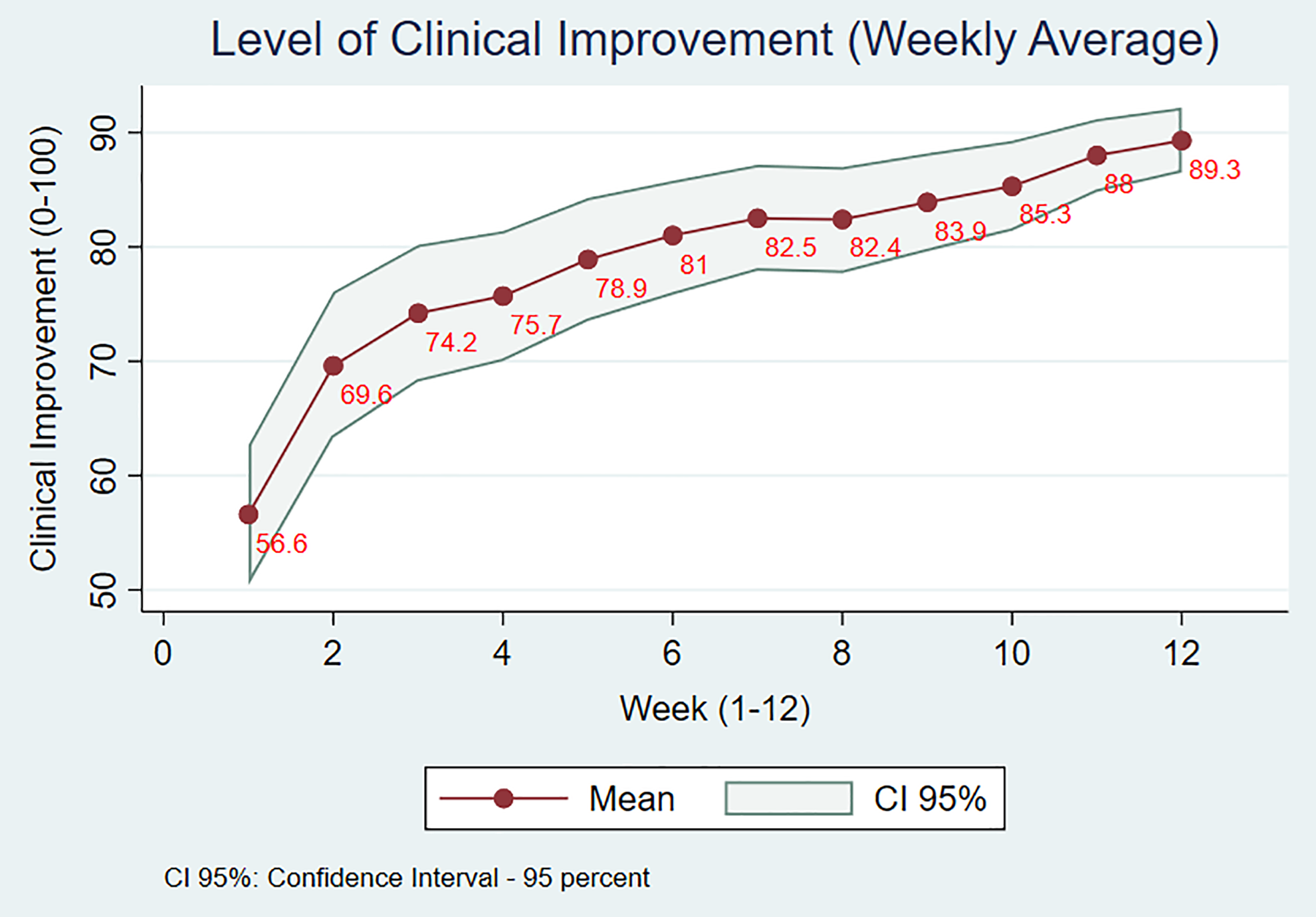
Behavior of clinical improvement level related to the follow-up time (scale: 0 to 100; 100 means total resolution of symptoms).
The outcome of complete clinical improvement was documented between the first and the ninth week of follow-up. By the end of the second week, 61% of the sample had already reached this outcome. At the end of week 6 of follow-up, the percentage of cumulative improvement reached 72%, and after 12 weeks of follow-up, the outcome of complete clinical improvement was documented in 110 patients, for a cumulative incidence of 73% (95% CI: 65%-80%). In Figure 2, the probability function of complete improvement as a function of time is illustrated with the Kaplan-Meier method. Based on the documented follow-up to the study patients, 698 weeks of follow-up were documented for an incidence rate of 16 cases per 100 people/week (95% CI: 13-19 per 100 people per week).
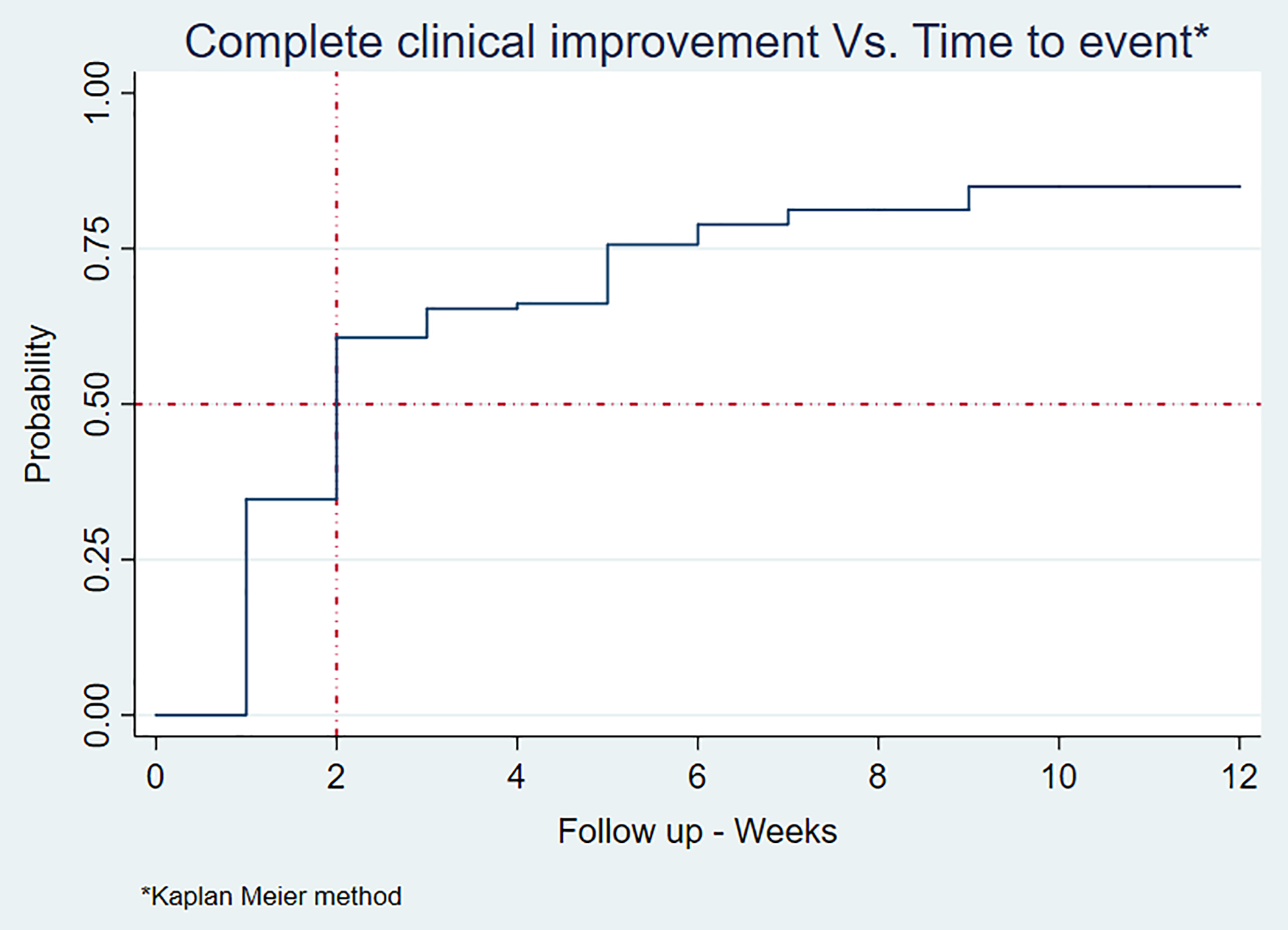
The graph shows the probability of complete improvement related to the follow-up time.
Serious Adverse Events
During the follow-up of the cohort, no serious adverse events were recorded and in no case the suspension of treatment on the occasion of an adverse event was documented.
Discussion
In the literature, different studies have been published regarding the evaluation of the clinical effectiveness of betahistine for the control of symptoms associated with peripheral vestibular vertigo. 12 –14 Additionally a Cochrane systematic review published in 2016 concluded that betahistine can reduce the symptoms associated with vertigo. 4 However, the evidence described comes from controlled clinical trials, considered of low quality, with small sample sizes and with highly selected populations. For this reason, the results of this study are important given that they provide evidence of usual clinical practice, able to complement, confirm, or contrast the previously described findings, but based on a sample of patients taken from the routine consultation of a group of doctors of primary care, which probably resembles the population that general practitioners receive in daily practice, and which does not necessarily correspond to the selection of candidates to participate in a controlled clinical trial. 8
In the present study, it was documented that betahistine 48 mg/d completely controlled the clinical picture associated with peripheral vestibular vertigo in 73% of treated patients at a follow-up of 12 weeks. It is worth noting that although not all patients achieved complete improvement, the average improvement at the end of the follow-up was 89 points on a scale of 0 to 100. These results in addition to supplementing the clinical effectiveness data published in some previously cited studies 12 –14 are consistent with what was reported in a multicenter observational study published in 2017, conducted in Russia and Ukraine by Parfenov et al, 11 in which 305 patients were included of which 74% achieved a clinical response considered good, very good, or excellent. The findings documented in previous research, as well as those described by us in the present work, can be explained by the mechanism of action of betahistine, which includes among others a histaminergic vasodilator effect of the cerebral and inner ear microcirculation as well as by its action at the level of the central histaminergic system, which improves the process of vestibular compensation and reduces the spontaneous activity of peripheral vestibular receptors. 15
Some authors have suggested that the maximum effect of betahistine is achieved with prolonged treatment periods of 3 to 8 weeks 16 and that its effect can be increased gradually during the first 2 months. 11 However, our results, despite going in the same direction, are complementary, thanks to the fact that the weekly measurement, which included a global evaluation by the attending physician, allowed us to document an incremental and sustained effect, up to week 12. From the clinical perspective, these findings are relevant, since in the previous studies identified, the probability or prognosis of improvement in a patient with peripheral vestibular vertigo as a function of time was not presented. In this sense, based on our findings, a doctor can predict that, by the second week of treatment with betahistine, at least 61% of their patients will have improved and that by the end of the ninth week, a percentage close to 73% may have achieved this outcome. Regarding the safety profile of betahistine, our findings are consistent with those published by different authors 11,17 –19 who agree that it is a safe molecule with a low percentage of serious adverse events or dropouts during therapy.
Finally, it is important to recognize that despite the virtues of observational studies based on real-life data, there are some limitations that must be taken into account given that the main advantage of clinical trials is given by their potential to control confusion factors and limit the biases derived from the measurement; however, the consistency of the findings with both observational and experimental studies gives us an indication of the validity of the described findings. It is important to collate evidence from controlled clinical trials, with evidence from real-life studies, and in that sense they should be understood as complementary findings based on the advantages of each design.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a research grant from Abbot-Lafrancol Colombia.
