Abstract
Objective
This study analyzed the possible effects of intratympanic steroid (ITS) therapy in the symptomatic treatment of vertigo attacks in patients with Ménière’s disease.
Methods
Thirty-five patients treated with ITS (dexamethasone) plus betahistine (Group A) and 35 patients treated with betahistine alone (Group B) were enrolled in this investigation. Complaints were analyzed using medical records and vertigo diaries. Statistical analysis was conducted using IBM SPSS V24 software.
Results
Based on the analysis, there were no significant differences in baseline features between the two groups. When the occurrence of vertigo attacks was compared using the Kaplan–Meier method, no significant difference was detected between Groups A and B (odds ratio [OR] = 1.051, 95% confidence interval [CI] = 0.965–1.067; p = 0.972). In addition, no difference in the incidence of vertigo attacks was noted in group A between the periods of treatment with betahistine alone and betahistine plus ITS when the groups were analyzed via logistic regression (OR = 1.07, 95% CI = 0.065–1.467; p = 0.614).
Conclusion
It can be concluded that the addition of ITS therapy to betahistine did not improve outcomes in patients with Ménière’s disease. Further prospective studies should be conducted to analyze the results in a more detailed manner.
Keywords
Introduction
Ménière’s disease (MD) is an inner ear disorder characterized by episodes of vertigo, sensorineural hearing loss, tinnitus, and vegetative symptoms. 1 The treatment of MD usually involves lifestyle changes and oral pharmacologic therapies, such as betahistine dihydrochloride, a histamine-like drug (H1 agonist and H3 receptor antagonist). 2 Using conservative treatment options, remission can be achieved in 80% of patients with MD. If these treatments fail to control symptoms, intratympanic therapies are used.3,4 Intratympanic steroid (ITS) therapy is a more symptomatic treatment option for vertigo attacks 5 and a first-line treatment for sensorineural hearing loss in patients with MD. 6 The possible mechanism of action of ITS therapies is based on autoimmune etiology and anti-inflammatory immunosuppressive effects or the regulation of Na+ absorption and osmotic regulation in the inner ear.7,8 Using ITS therapies, a higher concentration of corticosteroids can be achieved in the perilymph while avoiding systemic adverse effects. 9 Some previously published studies presented encouraging results for ITS therapies for vertigo attacks; 10 however, other studies indicated lower effectiveness of these treatments in the management of vertigo.11,12 Therefore, the present study investigated the effects of ITS therapy in the treatment of vertigo attacks in patients with MD.
Materials and methods
Patients
The study was a retrospective clinical research project with long-term patient follow-up. In this investigation, patients treated with an ITS plus an individualized daily dose of betahistine (IDBH, Group A) and those treated with IDBH alone (Group B, control) were selected. All patients in Group A who received the ITS had previously failed to respond to betahistine monotherapy and experienced severe vertigo attacks. Each patient completed at least one course of ITS therapy consisting of five injections, but some patients completed a second round of ITS treatment. No additional treatments were provided to either group. The patients were diagnosed with unilateral definite MD based on the criteria of the Bárány Society 13 as follows: 1) two or more spontaneous episodes of vertigo each lasting 20 minutes to 12 hours; 2) audiometrically documented low-to-moderate sensorineural hearing loss in one ear on at least one occasion before, during or after an episode of vertigo; 3) fluctuating aural symptoms (hearing, tinnitus, or fullness) in the affected ear; and 4) the absence of another explanatory vestibular diagnosis. Patients with incomplete medical history data, those with bilateral MD, those who received intratympanic gentamicin, and those who underwent surgical treatment were excluded. MRI was performed to exclude vestibular schwannoma. Information about symptoms was taken from medical records and vertigo diaries, especially those related to the attacks, and contrasted with the characteristics of the medications used since the previous check-up. The control of vertigo attacks was calculated using the American Academy of Otolaryngology–Head and Neck Surgery (AAO–HNS) index. 14 In this index, class A denotes complete control of the symptoms, whereas class B implies limited control. The research project was approved by Semmelweis University Regional and Institutional Committee of Science and Research Ethics (approval number: 47/2018), all patients provided verbal informed consent.
Treatment protocol
A dose of 4 mg/mL dexamethasone phosphate was injected into the middle ear through the anteroinferior part of the tympanic membrane under a microscope. Each patient held the syringe in his or her hand for approximately 5 minutes to avoid a temperature gradient in the inner ear. In addition, 10% lidocaine pump spray was used as local anesthesia. After ITS administration, the patient was instructed to not swallow or move for 30 minutes to permit the passage of dexamethasone from the middle ear into the inner ear. The treatment protocol consisted of daily injections for 5 days.
Patients in the control group received conservative management with IDBH. IDBH indicates that the daily dose of betahistine was individualized for each patient with necessary changes during the follow-up period based on the characteristics of the symptoms.
Statistical analysis
Statistical analysis was performed using IBM SPSS V24 software (IBM, Armonk, NY, USA). Because the Shapiro–Wilk test revealed that the studied parameters did not have a normal distribution, the Mann–Whitney U test was used. Categorical analysis was conducted using the chi-squared test. Kaplan–Meier curves were created, and logistic regression was performed. Statistical significance was specified by P < 0.05.
Results
The baseline characteristics of the parameters of the two groups, each of which included 35 patients with MD, presented in Table 1. No statistically significant differences in patient demographics, including the numbers of men and women and age, were detected between the two groups (p = 0.32). The AAO–HNS index did not differ between the groups at baseline. The mean daily dose of betahistine was also not statistically significantly different between the groups (p = 0.12). The similar mean follow-up times of the groups suggested that long-term follow-up was achieved.
Baseline features of the two groups.

Group A, intratympanic steroid plus individualized daily dose of betahistine; Group B, individualized daily dose of betahistine (control group)
*Chi–squared test, **Mann–Whitney U test
IQR, interquartile range; Q1, first quartile; Q3, third quartile; AAO–HNS, American Academy of Otolaryngology–Head and Neck Surgery
To analyze the long-term outcomes of treatment, Kaplan–Meier curves were drawn. As illustrated in Figure 1, there was no obvious difference in the curves for Groups A and B. Logistic regression analysis also identified no differences between the groups (odds ratio [OR] = 1.051, 95% confidence interval [CI] = 0.965–1.067; p = 0.972).

Occurrence of vertigo attacks in Group A (ITS + betahistine) and Group B (betahistine only). The number of events (i.e., vertigo attack) that occurred over time was determined as follows: 0, ≤3 attacks/month; 1, >3 attacks/month. ITS, intratympanic steroid.
Kaplan–Meier analysis was also performed to assess whether the addition of an ITS to betahistine reduced the frequency of vertigo attacks in patients with MD in Group A (Figure 2). No obvious difference in outcomes was noted after the addition of the ITS (OR = 1.07, 95% CI = 0.065–1.467; p = 0.614), implying that ITS therapy does not improve the control of vertigo.
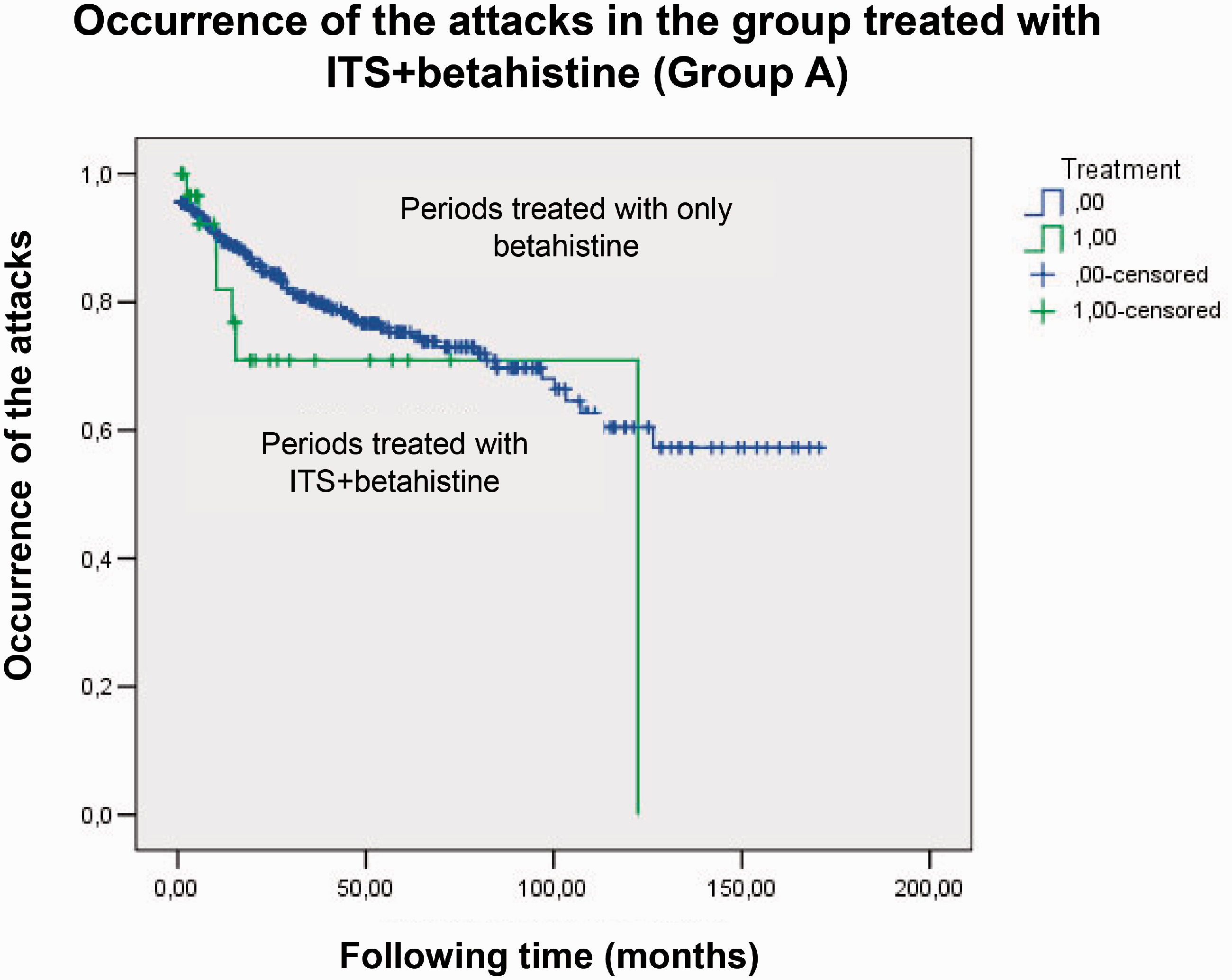
Occurrence of vertigo attacks in Group A. Kaplan–Meier analysis was performed to assess the efficacy of the addition of an ITS to betahistine in the same group. ITS, intratympanic steroid.
Discussion
ITS therapy has displayed good efficacy in the treatment of hearing loss in patients with MD15,16 and sudden sensorineural hearing loss. 17 Studies have been undertaken to clarify the effectiveness of ITS therapy in the management of vertigo attacks. Leng et al. 18 concluded that at 2 years, 73.9% of patients achieved complete (class A) vertigo control. McRakan et al. 5 also stated that 81.1% of their patients experienced complete control of vertigo (Class A or B). Meanwhile, a prospective study indicated that ITS therapy provided complete vertigo control in only 15.1% of patients and substantial control in 32.1% of patients. 11 Based on a systematic review, it was concluded that the efficacy of ITS therapy is questionable in patients with MD, and further research is needed. 19 Meanwhile, another review stated that only one of six studies described potential benefits of ITS therapy in the treatment of vertigo attacks. 20
Previous studies also investigated the effects of ITS and betahistine therapy in patients with MD. Albu et al. 21 compared the effectiveness of ITS therapy and daily high-dose betahistine (144 mg/day), finding that class A vertigo control was achieved in 44% of patients in the ITS group, versus 73.3% in the combination group, supporting the questionable effectiveness of ITS therapy for vertigo attacks. In another study, the long-term outcome of ITS therapy was investigated. It was concluded that in contrast to the preoperative values (when betahistine and diuretics were used), the number of patients without vertigo decreased over time. 22 In another investigation, patients were assigned to treatment with ITS therapy and oral placebo or betahistine and an intratympanic saline injection. The researchers concluded that high-dose betahistine (144 mg/day) provided similar vertigo control as ITS therapy. 23 In a study by Paragache et al., vertigo control was contrasted between patients treated with ITS therapy and betahistine. The vertigo control rates were nearly equivalent between the two groups. However, it was concluded that ITS therapy is more effective in patients with severe vertigo or a shorter duration of MD. 24 Our study also failed to find a benefit of the addition of ITS therapy to IDBH over long-term follow-up. The use of IDBH was based on previously published studies, which indicated that based on the metabolism of betahistine, metabolic features should be considered using long-term follow-up and necessary changes in the dosage. 25
The side effects of betahistine were mild, and symptoms such as nausea, gastrointestinal distension, and headache occurred. As previously reported, 26 ITS therapy also carries certain complications, but these effects were negligible in the current study. In some cases, short-lived vertigo occurred, but other complications, such perforation of the tympanic membrane, were not typical.
Finally, it can be concluded that effects of the combination regimen were not significantly better than those of conservative therapy. Further prospective studies are needed to analyze the results in a more detailed manner. However, based on the long-term follow-up in this study, ITS therapy does not appear effective in the management of vertigo attacks. Hence, if conservative therapy fails to control the symptoms, other therapies should be considered.
Footnotes
Acknowledgements
This study was supported by the EFOP-3.6.3-VEKOP-16-2017-00009 Project and the ÚNKP-20-4-I New National Excellence Program of the Ministry for Innovation and Technology from the National Research, Development and Innovation Fund.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
