Abstract
The objective of this study is to evaluate stapes surgery in patients with otosclerosis and “profound” hearing loss. This means they meet hearing threshold criteria for cochlear implantation (CI). We performed a retrospective study and patient questionnaire. The results from 33 patients (35 ears) were recorded (mean age: 63.6, range: 40-85). The primary outcome measure was hearing thresholds recorded before and after surgery at 0.5, 1, 2, 3, and 4 kHz. Hearing thresholds at 2 and 4 kHz were also analyzed. Glasgow Benefit Inventory (GBI) was used in 21 patients to assess life quality changes. Hearing thresholds improved in 80% of ears (mean improvement, 26.3 dB), were unchanged in 11.4%, and worsened in 8.6%. Mean GBI score was +20.7. Hearing aid use decreased in 23.8% and ceased in 28.6%. One patient subsequently underwent CI. For patients with profound otosclerosis, stapes surgery provides a quantitative improvement in hearing thresholds and improvement in quality of life, with reduced reliance on hearing aids. This avoids CI, auditory rehabilitation, and a change in quality and tonality of sound.
Introduction
There is no universal consensus regarding the optimum management of appropriately hearing aid–supported patients with profound hearing loss secondary to otosclerosis. Stapes surgery aims to alleviate a fixed footplate by performing a stapedotomy and inserting a prosthesis to bypass stapes fixation. Audiological function can then be enhanced using a hearing aid. The main alternative surgical treatment, cochlear implantation (CI), is used to bypass the middle ear entirely using digital sound detection and electrical stimulation of the cochlear nerve.
Traditionally, patients with severe hearing loss secondary to otosclerosis are offered CI in the first instance. For example, in the United Kingdom, the National Institute for Clinical Excellence (NICE) recommends CI for patients in the following circumstances: Profound hearing loss defined as hearing thresholds greater than 90 dB at 2 and 4 kHz. Adequate benefit from hearing aids, defined as Bamford-Kowal-Bench (BKB) scores greater than 50% at 70 dB, is not achieved.
1
Clinical evidence supports this recommendation, describing high success rates using CI in patients with severe hearing loss. 2 –4
However, there is also much evidence to suggest that stapes surgery can restore hearing to acceptable levels in patients with similar “profound” hearing loss, especially when combined with a well-fitted hearing aid. 5 –10 Furthermore, whereas CI can be time-consuming and physically invasive, stapes surgery is minimally invasive and cost-effective. If stapes surgery is not beneficial, CI or revision stapes surgery can be performed. No study has investigated the quantitative and qualitative effect of stapes surgery in patients with otosclerosis and profound hearing loss. This article uses a validated tool, the Glasgow Benefit Inventory (GBI), alongside audiometric data to assess the person-centered quality-of-life improvements conferred by stapes surgery on patients with profound hearing loss.
Patients and Methods
Study Design
Retrospective study with quality of life questionnaire. The project was registered with the divisional audit lead, and as a service evaluation, no external approval was necessary.
Study Location
The study was conducted at a specialist ear, nose and throat tertiary care hospital, the Royal National Throat, Nose and Ear Hospital (London, United Kingdom).
Inclusion Criteria
Patients were selected according to the following criteria: Postoperative air conduction hearing thresholds measured 6 weeks postoperatively. Surgery occurred between 2013 and 2017. Preoperative air conduction hearing thresholds >90 dB at 2 and 4 kHz.
The NICE guidelines recommend CI in patients with “profound” hearing loss and low BKB scores with a hearing aid in situ. However, it is important to note that in United Kingdom it is not routine practice to undertake speech discrimination testing (such as BKB assessment) in the workup for stapes surgery. We are aware that this is in stark contrast to the situation in many other countries. In United Kingdom, it would be the exception to find these tests used for stapes surgery, despite their integral role in preassessment for CI. This article focuses on the use of stapes surgery before CI, which is before speech recognition scores would be routinely performed, so they were not available for this cohort of patients and were not included in this study.
Exclusion Criteria
All patients were included for audiometric analysis. In GBI analysis, patients were excluded according to the following criteria: Deceased. Could not be contacted. Underwent CI following stapes surgery.
Patients who chose to have CI after stapes surgery were included for audiometric analysis, but excluded from GBI analysis, because undergoing CI between completing the questionnaire and receiving stapes surgery was judged to be a significant confounding factor.
Patient Demographics
Thirty-three (35 ears) patients were included. The mean age was 63.6 years (range, 40-85). Twenty were female and 13 were male (ratio 1:54). Two patients had bilateral surgery. Eleven patients had received prior stapes surgery on the contralateral ear.
Surgical Technique
In all cases, the same surgical technique was used, as described previously by our unit. 11 A standard permeatal stapedotomy was performed under local anesthetic with the option of sedation. The stapedius tendon and posterior crus were incised, and the stapes superstructure downfractured. A rosette stapedotomy was performed using a potassium titanyl phosphate laser. The ossicular chain was reconstructed with a SMart 360 nitinol fluoroplastic piston (Olympus, Southend-on-Sea, United Kingdom). Finally, the tympanic membrane was replaced to facilitate clinical hearing testing prior to completion of the procedure.
Audiometry
Audiometry was conducted according to the recommended procedure published by the British Society of Audiology at a minimum of 6 months prior to surgery and at 6 weeks following surgery. 12 A pure-tone average (PTA) was calculated from these data as the mean of air conduction thresholds at 0.5, 1, 2, and 3 kHz, according to the guidelines of the American Academy of Otolaryngology–Head and Neck Surgery. 13 Air conduction hearing threshold change was calculated as the preoperative PTA subtracted from the postoperative PTA obtained at routine follow-up. Air conduction thresholds at 2 and 4 kHz before and after surgery were also compared. Bone conduction thresholds were not included as the profound nature of hearing loss meant that the thresholds of many patients were greater than vibrotactile limits.
Glasgow Benefit Inventory
The GBI is a person-centered quality of life questionnaire that has been validated for assessing outcomes after a wide range of otolaryngologic procedures. Consisting of 18 questions, it measures quality of life over 3 areas of experience: social, physical, and general. Three questions related to social improvement in quality of life, 3 to physical improvement, and 12 to general improvement. Patients indicate their response for each question on a 5-point Likert scale. A response of 1 represents the most negative outcome possible, 5 represents the most positive outcome, and 3 indicates no change. A mean is calculated from the responses and plotted as a graph where 1 correlates to −100 and 5 correlates to +100. 14
Patients were followed up at least 6 weeks after the operation with a questionnaire based on the GBI. 15 An additional assessment of hearing aid use was made. Our questionnaire response rate was 77%, which is high and supports the accuracy of our data.
Results
Air Conduction Hearing Threshold Results
Air conduction hearing thresholds improved in 80% of ears (28/35) following stapes surgery. The mean air conduction hearing threshold improvement from 35 ears was 26.3 dB (range, −12.5 to 62.5 dB). Pre- and postoperative air conduction hearing thresholds are compared in Figure 1.
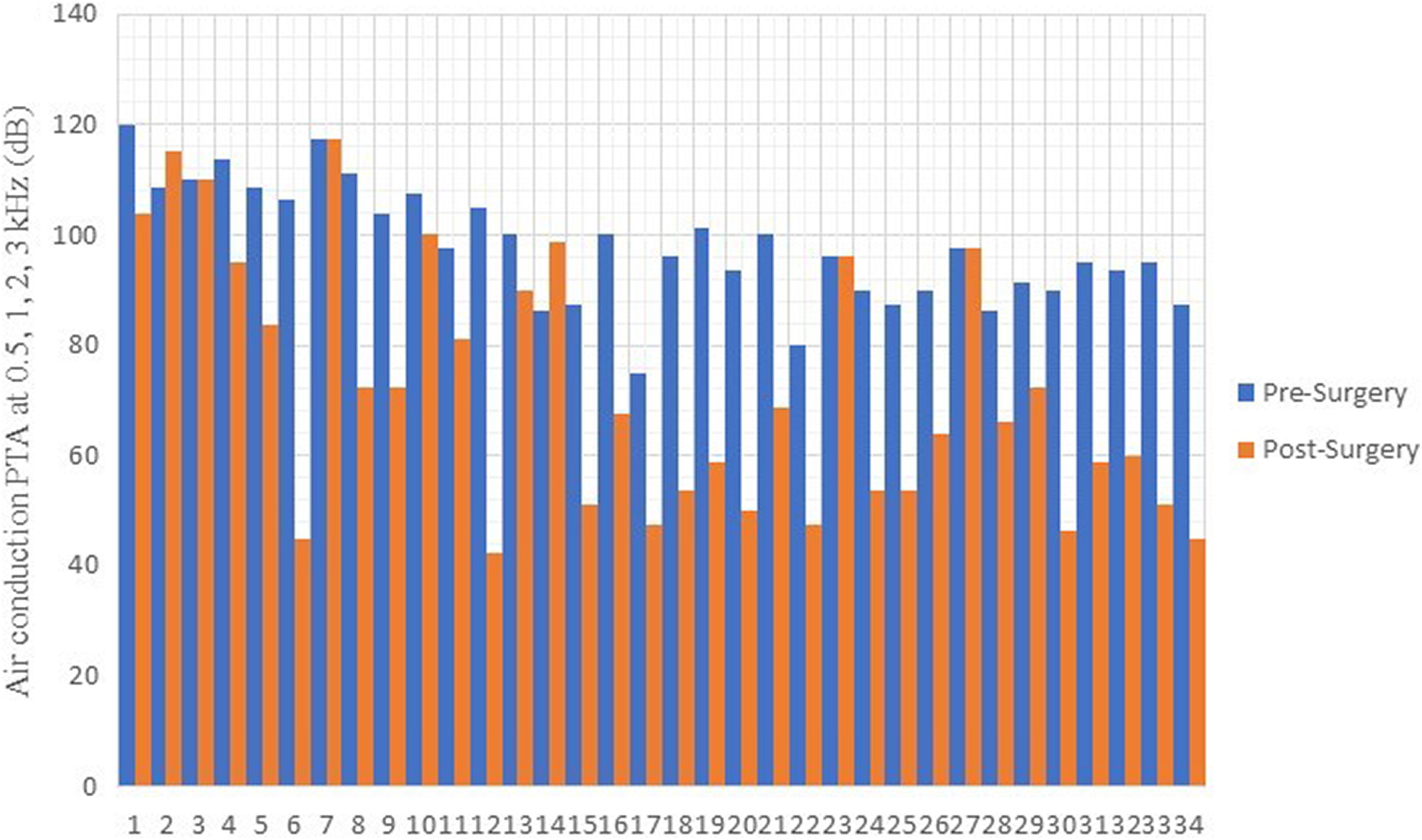
Graph comparing air conduction hearing thresholds before and after stapes surgery.
Thresholds for one ear could not be recorded due to instrumental limitation: this “dead ear” was excluded from numerical analysis. For 22.9% (8 ears), this improvement was greater than 40 dB. Of those that did not benefit, 11.4% (4 ears) experienced no change in air conduction hearing thresholds, while 8.57% (3 ears) experienced worsened thresholds. Out of these, one patient went on to have CI, one more was awaiting the operation, and one was rejected once their BKB scores were recorded and found not to meet the criteria. For the remaining four, CI was deemed inappropriate due to comorbidities or was declined by the patient.
Air Conduction Hearing Thresholds at 2 and 4 kHz
We also specifically compared air conduction hearing thresholds at 2 and 4 kHz before and after surgery to assess the influence on the hearing thresholds used by NICE in assessment for CI. Results were similar at 2 and 4 kHz to those at 0.5, 1, 2, and 3 kHz. Eighty (n = 28) percent of ears experienced air conduction hearing threshold improvements that moved them outside the hearing thresholds that NICE would recommend for CI.
Glasgow Benefit Inventory Results
Twenty-one (80.8%) of the 26 patients surveyed completed the postoperative questionnaire. This response rate is high and supports the accuracy of our data. The mean GBI score was +20.7 (range, −66.7 to +94.4). Fourteen (66.7%) patients demonstrated functional improvement (range, +8.33 to +94.4). Four (19.0%) experienced very significant improvement with scores of +75 or greater. Six (28.6%) demonstrated an overall decrease in function (range, −5.56 to −66.7). One patient experienced no overall change following the operation. In this group, the audiometric data showed 71.4% (n = 15) experienced improved air conduction hearing thresholds, 14.3% (n = 3) stayed the same, and 14.3% (n = 3) experienced worsened thresholds. These outcomes are poorer than those in the broader cohort.
The 3 domains of GBI (general, social, and physical) were analyzed separately, and we found that mean social benefit was greatest at +29.4 (range, −33.3 to +100), followed by general benefit at +21.7 (range, −91.7 to +95.8) and physical benefit at +7.94 (range, −33.3 to +100).
When average GBI score is plotted against air conduction hearing threshold improvement (Figure 2), there is a positive linear regression. The R2 value is 0.409, which suggests a moderate positive correlation.
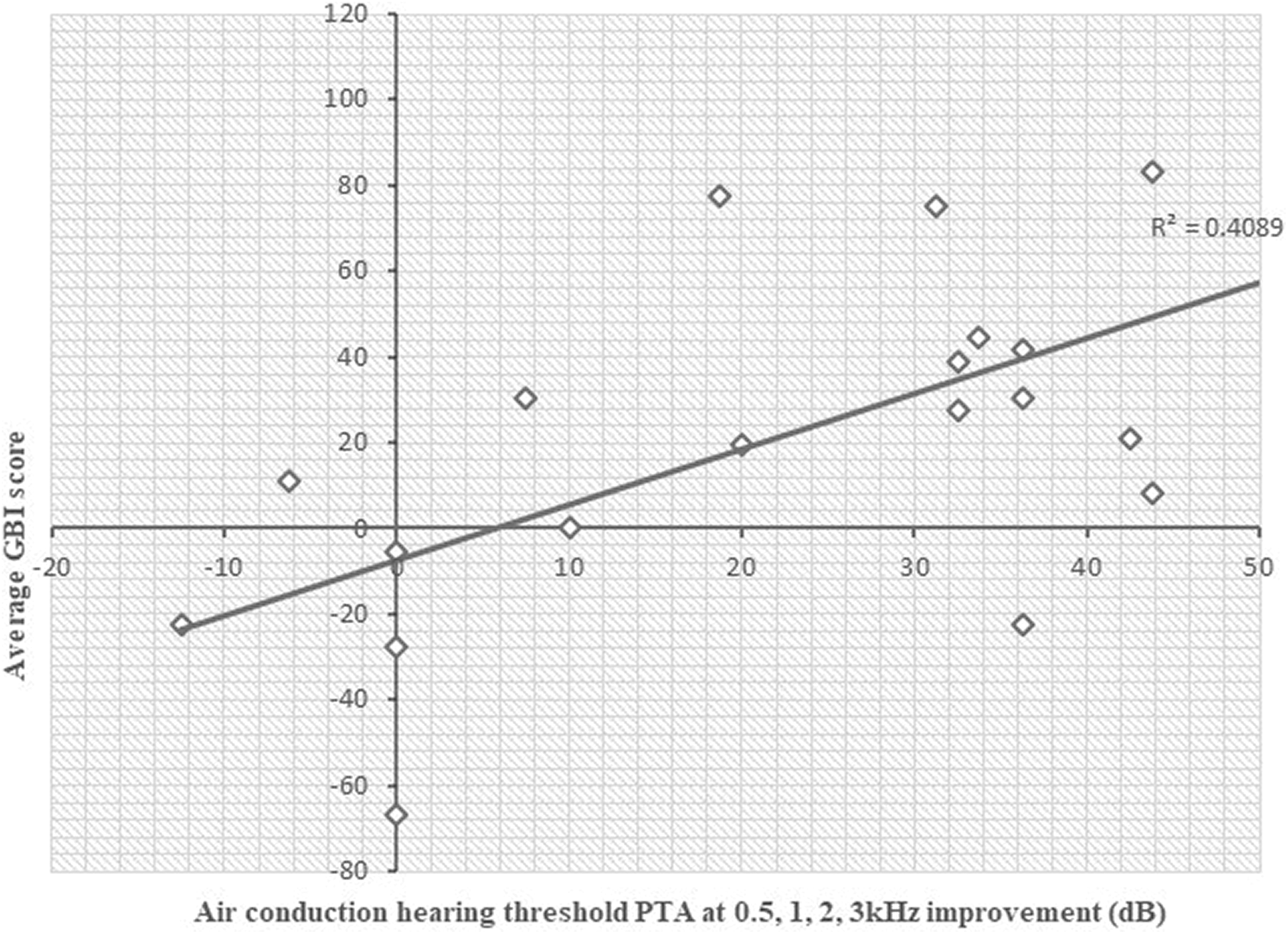
Scatter graph showing average Glasgow Benefit Inventory (GBI) against air conduction hearing threshold improvement.
Comparison Between Patients Who Had Undergone Previous Contralateral Surgery and Those Who Had Not
We analyzed air conduction hearing thresholds and GBI separately in the groups who had undergone previous contralateral stapes surgery and those who had not. These results are displayed in Table 1. Those who had undergone previous contralateral stapes surgery experienced a greater average PTA improvement than those receiving their first operation (29.8-24.6 dB). The magnitude of difference was greater between the subgroups who qualified for GBI analysis than the overall samples (37.8-22.5 dB). However, the GBI was comparable between the groups who had undergone previous surgery and those who had not (+23.3 compared with +23.0).
Comparing Air Conduction Hearing Threshold Improvement and Average Glasgow Benefit Inventory (GBI) Score in Patients Who Did and Did Not Undergo Previous Stapes Surgery.
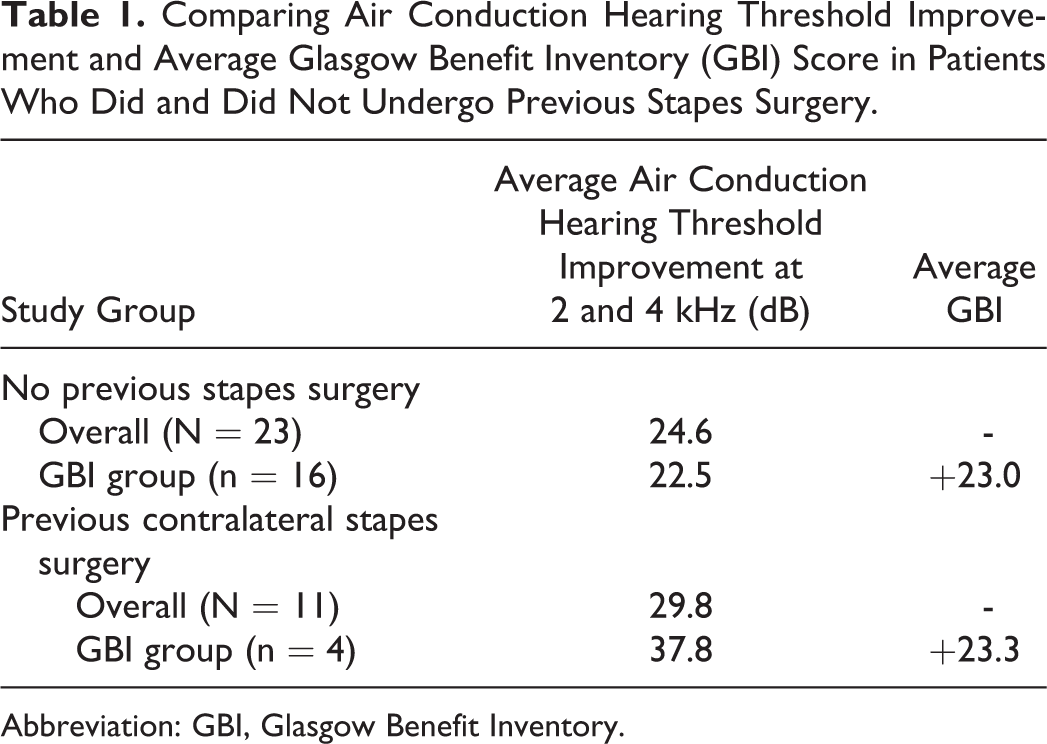
Abbreviation: GBI, Glasgow Benefit Inventory.
Hearing Aid Use
Furthermore, 28.6% (n = 6) of patients who used hearing aids before were able to stop their use completely. A further 23.8% (n = 5) had reduced the time they used their hearing aid, the amplification setting, or both. For the remaining 47.6% (n = 10), the use of their hearing aid had not changed. These data are displayed in Table 2.
Comparing Air Conduction Hearing Threshold Improvement and Average GBI With Relation to Change in Hearing Aid Use.

Abbreviations: GBI, Glasgow Benefit Inventory; NA, not applicable.
Discussion
Quality of Life Measures
Satisfaction scores are increasingly being used to evaluate otolaryngologic procedures. They are being used by clinicians and health-care administrators alike to decide which treatments to offer and fund, by isolating those interventions that have the biggest impact on quality of life. 15,16 This study suggests that for many patients with profound hearing loss, stapes surgery is an appropriate first intervention, conferring significant improvement in quality of life. 8 More extensive use of validated, multifactorial tools such as the GBI would enable more thorough and sophisticated analysis of the impact of stapes surgery on the aspects of quality of life of patients.
Glasgow Benefit Inventory
Our GBI outcomes for stapes surgery show a smaller benefit than is usually described in the literature. 17 This may be accounted for by the setting of this study, which was a tertiary referral hospital. The cohort may have included more difficult and severely affected cases than are usually seen in secondary care, as well as revision cases that have previously failed.
Interestingly, our results for the separate GBI domains differ from previous studies where general GBI > social > physical. 15 In our study, the pattern showed social > general > physical. This may be a result of the severe level of hearing loss featured in this group of patients, whereby improving hearing makes the greatest impact on their social lives, above their general life experience. These results do support the findings of Robinson et al in 1996, which suggested that the physical domain correlates most poorly with objective hearing measures. 15
Glasgow Benefit Inventory and Objective Measures of Hearing
The hearing threshold improvements following stapes surgery in this group were comparable to others in the literature. 8,18,19 The positive linear regression (R 2 = 0.4089) between average GBI score and air conduction hearing threshold improvement indicates a moderate correlation, which supports the validation of GBI with objective measures of hearing.
One patient reported a negative GBI despite experiencing improvement in air conduction hearing thresholds, and the 3 patients who experienced no change in their thresholds all reported negative GBI outcomes rather than neutral. Furthermore, although the group who had undergone previous contralateral stapes surgery experienced a greater improvement in hearing objectively, they reported comparable GBI scores to those who had not had a prior operation. This reminds us to consider the pros and cons of medical interventions when planning patient management and highlights the importance of using patient-centered outcome measures. Speech recognition tools such as the BKB may shed light on this variability in GBI score. Unfortunately, as these were not recorded for most patients in this cohort, we cannot perform any meaningful analysis of this here. More investigation into the relationship between hearing thresholds, speech recognition scores, and patient-centered outcome measures, including specific quality of life domains, is warranted and would help us better understand and select our interventions accordingly.
Stapes Surgery Compared to CI
Since the 1990s, the literature has supported the use of stapes surgery in severe hearing loss caused by otosclerosis. 20,21 Stapes surgery is safe, minimally invasive, and low cost. 7 It can be performed under local anesthetic, which is especially beneficial when considering the age and comorbidities of many patients with profound hearing loss. Unlike CI, stapes surgery does not require extrusive rehabilitation and avoids the need to change quality and tonality of sound. 10
As other studies have suggested, our findings show that some patients respond very well to stapes surgery, while for a minority, the results are unsatisfactory. In most cases, hearing thresholds are improved to a level where hearing aids are of benefit, and in 80% of patients, their thresholds improved to move them out of the category of “profound” hearing loss, so they would be no longer eligible for CI. This study represents the largest cohort of patients with profound hearing loss secondary to otosclerosis who are evaluated qualitatively and quantitatively. We also hope it will contribute to future meta-analyses, increasing the evidence available; the 2 published meta-analyses looking at this clinical question contain 4 and 9 articles, respectively. 10,22 These studies show that CI is more likely to improve hearing to a good standard overall, but that when stapes surgery is effective, it produces hearing that is nearly equivalent. 7,10,19,22 As yet, there is no complete consensus on the preoperative factors that can predict successful stapes surgery, although variables such as word recognition scores, benefit derived from hearing aid use, and extent of retrofenestral sclerosis have been suggested. 9,10,23 If the outcome from stapes surgery is unsatisfactory, CI can be performed in the same ear with no additional risk of failure or adverse outcomes (see Figure 3). 10,22
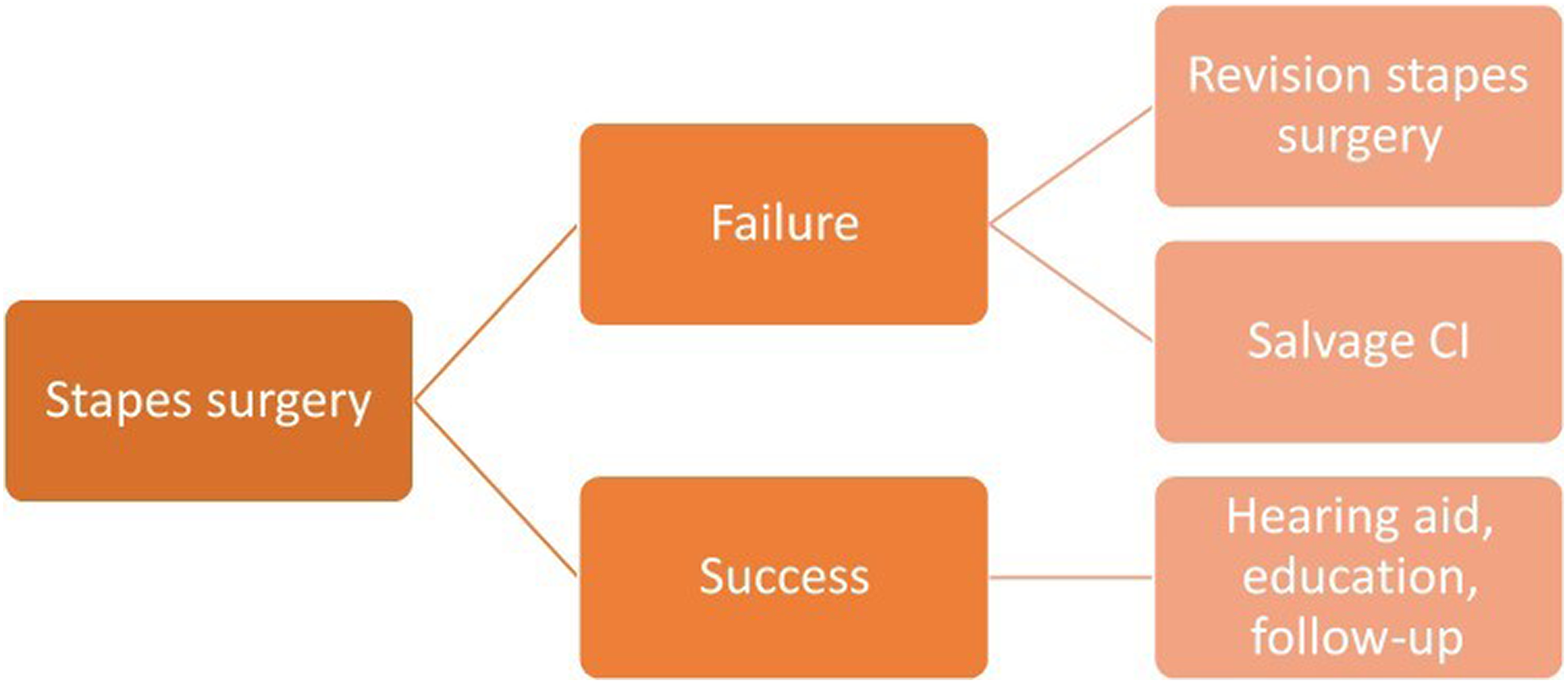
Proposed algorithm for surgery in patients with profound hearing loss secondary to otosclerosis.
Conclusion
Our study shows that stapes surgery in patients with profound hearing loss secondary to otosclerosis can produce significant improvements in air conduction hearing thresholds, decrease reliance on hearing aids, and increase quality of life as measured by GBI. This leads us to agree with the several other authors who have suggested that stapes surgery should be used in the first instance as surgical management for this patient group. 7,9,10,19,22 Due to the high chance of a positive outcome, stapes surgery should be offered first, with CI reserved for patients who show an unsatisfactory response.
Footnotes
Acknowledgments
The authors thank all the patients for their cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
