Abstract
To deepen the understanding of chlorite and laumontite mineral characteristics within the Lower Wuerhe Formation in the Madong area of the Junggar Basin, a detailed investigation was carried out using thin sections, electron probe microanalysis (EPMA), and X-ray diffraction (XRD). Through this multi-method approach, their chemical compositions, occurrences within thin sections, and the impacts on the physical properties of the reservoir were illustrated. The results indicate that the chlorite minerals exhibit a trioctahedral crystal structure and are found in four distinct forms: as pore linings, grain coatings, pore fillings and chlorite pseudomorph. They belong to the iron-magnesium transitional type with a magnesium-rich composition. The octahedral sites primarily feature Fe substituting for Mg, with an Al/(Al + Mg + Fe) ratio ranging from 0.25 to 0.37. Chlorite formation is attributed to the alteration of argillaceous rocks and mafic rock transformations, with significant material input from the hydrolytic dissolution of tuffaceous volcanic materials and interconversions among clay minerals. Chlorite has been shown to significantly enhance reservoir physical properties, contributing to the development of high-quality reservoirs. Laumontite occurs in three states: crystalline aggregates, filling materials, and through replacement processes. Laumontite crystalline aggregates are often surrounded by debris, which promotes their formation. In its filling state, laumontite coexists with chlorite, calcite, and other minerals, competing for material sources, which can partially inhibit its formation. Replacement-type laumontite forms mainly by replacing feldspar and rock fragments, leading to a high Si/Al ratio and acid resistance, making it less susceptible to dissolution. However, the effect of laumontite on reservoir properties is relatively limited compared to that of chlorite. The diagenetic environment of the Lower Wuerhe Formation varies with increasing burial depth, transitioning from alkaline to acidic and then back to alkaline conditions.
Keywords
Introduction
In recent years, with the continuous development of oil and gas exploration and development technology, compact sandy conglomerate oil and gas reservoirs have received more and more attention(Tan et al., 2010; Xiao et al., 2021; Qin et al., 2023). Breakthroughs in the Tarim, Junggar, Bohai Bay, and other basins show broad exploration prospects in deep oil and gas exploration (Feng et al., 2014; Hua et al., 2016; Lihong et al., 2024). After extensive research, it has been established that minerals such as chlorite and laumontite are significantly abundant in alkaline deep formations within key geological basins(Yi et al., 2024). The formation mechanisms and occurrence of these minerals have a profound impact on the physical properties of the reservoir, influencing factors such as porosity, permeability, and overall reservoir quality(Wuren et al., 2010; Jiayang et al., 2020; Chaowei et al., 2023). Therefore, clarifying the genesis mechanisms and distribution characteristics of chlorite and laumontite in the study area is of significant importance for oil and gas exploration in this region.
In the 1960s, Heald identified the development of authigenic chlorite in clastic reservoirs and proposed that the formation of authigenic chlorite contributes to the preservation of primary pores(T. 1950). Following this, extensive research has been conducted on the microscopic characteristics, genesis mechanisms, formation periods, and protective mechanisms of authigenic chlorite regarding primary pores, leading to the following achievements: (1) Based on their occurrence characteristics, authigenic chlorite can be classified into four types of microscopic occurrences: grain-coating chlorite, pore-lining chlorite, pore-filling chlorite, and pseudomorphic chlorite(Tian Jianfeng et al., 2008; Xiaofeng et al., 2017). (2) The development of authigenic chlorite can inhibit the secondary enlargement of quartz, enhance the reservoir's resistance to compaction, and play a constructive role in both the preservation of primary pores and the development of secondary pores within the reservoir(V et al., 2003; Sijing et al., 2008). Studies on laumontite have shown the following progress: (1) The formation of laumontite is attributed to mineral transformation and the alteration of volcanic materials (L 1996; Hay R L and A 2001; Haiyan et al., 2020). (2) Laumontite forms in an alkaline diagenetic environment rich in calcium ions. It tends to block pore throats, leading to a significant loss of primary intergranular pores and degrading reservoir properties(B et al., 1989; Chipera S J et al., 2008). However, it also enhances the reservoir's compressive strength to some extent, thereby protecting the original pore structure(A. 2001; Chaowei et al., 2023).
For this study, samples from Lower Wuerhe Formation in Mahu depression were selected. A detailed analysis about the types, occurrence and its impact for the physical properties of the sandy conglomerate reservoir is carried out. Which may shed light for the further exploration in Junggar Basin.
Regional geological settings
The Mahu Depression is located in the central depression of the Junggar Basin. It borders the Wuxia Fault Zone to the north, the Zhongguai and Dabasong Uplifts to the south, the Kebai Fault Zone to the west, and the Luliang Uplift to the east (Figure 1) (Hai-bo et al., 2004; Dengfa et al., 2018). During Late Carboniferous to Early Permian, the subduction and eventual collision of the Kazakh plate beneath the Junggar Basin resulted in structural uplift along the northwestern margin of the basin and the formation of a large adjacent intra-continental sag(Tang et al., 2021a, 2021b). These tectonic movements laid the foundation for the shape of the Mahu Sag. Concurrently, the region experienced frequent volcanic activity, characterized by extensive volcanic eruptions and magmatic intrusions, as well as multi-phase hydrothermal activities(Tang et al., 2021b). Our study area is located in the east of the Mahu Sag, and on the west side of the Yingxi Sag, the Sanquan Uplift and the Xiayan Uplift. The structural shape is basically a southwest-dipping gentle monoclinic structure, with an average stratigraphic dip angle of 2°∼4°. Affected by the continuous compressive tectonic stress of the Mahu Depression and Xiayan Uplift from the Permian to the Triassic, a series of nose-shaped structures, steep slopes, low-amplitude anticlines, and wide and gentle platform structures developed(Haitao et al., 2020).
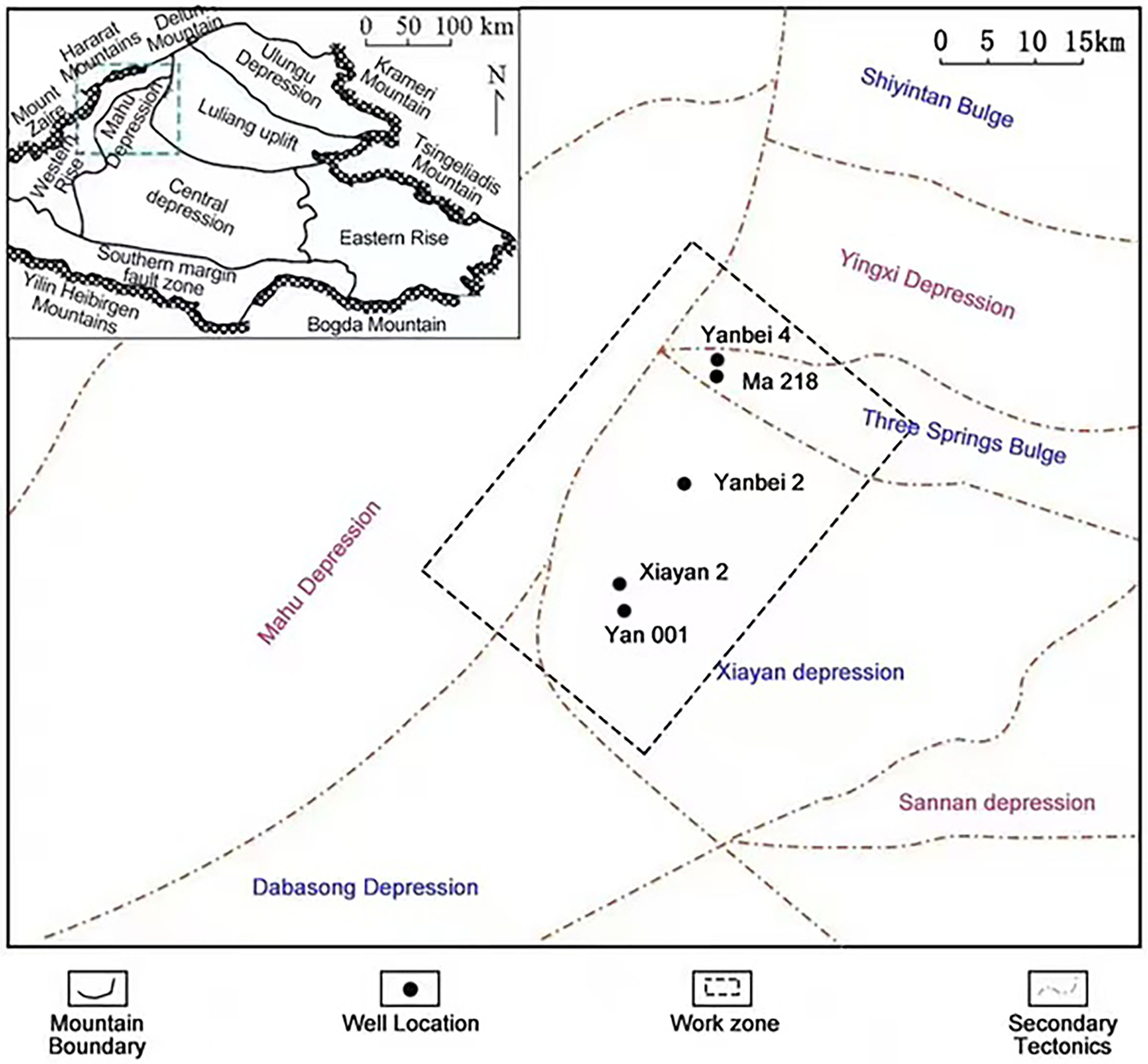
Structural location map of Madong area in the Junggar Basin (revised according to Qian Haitao et al., 2020).
Methods
In total 77 samples from four key wells: Yanbei-4, Xiayan-2, Ma-218, and Yan-1in the Lower Wuerhe Formation were collected. Among which, 77 thin sections were made from each sample for the analysis of petrological characteristics by a polarizing microscope and scanning electron microscopy (SEM). Meanwhile, the porosity and permeability of all the 77 sample were analyzed by Smart-Por and Smart-Perm system, respectively, based on the standard of SY/T 6385–2016 Testing methods for rock porosity and permeability under confining pressure.
Both the clay mineral and chlorite, laumontite composition were measured in Test Center of the Beijing Research Institute of Uranium Geology. 43 samples were selected and analyzed by X-Ray diffraction (XRD) for the composition of clay minerals. The samples were measured by a PANalytical X’Pert diffractometer equipped with a Bragg–Brentano goniometer (copper beam), each sample was scanned at 40 kV and 30 mA for 2 h. The start angle was 2.5 degree, the end angle was 70.0 degree and a step size of 0.008 degree per second was applied.
To measure the composition of chlorite and laumontite in the study area, 21 samples were selected and analyzed by a JXA-8100 electron probe (EPMA) under 20 kV and 1 × 10-8 A, with a take-off angle of 40°. The correction method is ZAF (Z: Atomic Number Effect, A: Absorption Effect, F: Fluorescence Effect). Chlorite compositions were calculated based on O14, acknowledging the complexity of its mixed-layer structure and potential for beam-induced damage. As a result, minor measurement errors are possible, but the selected oxide content consistently exceeds 80%. In the deep burial environment where Fe3+ levels are minimal, total iron measurements serve as a reliable proxy.
With these analysis methods, a workflow for this study is as follows (Figure 2):
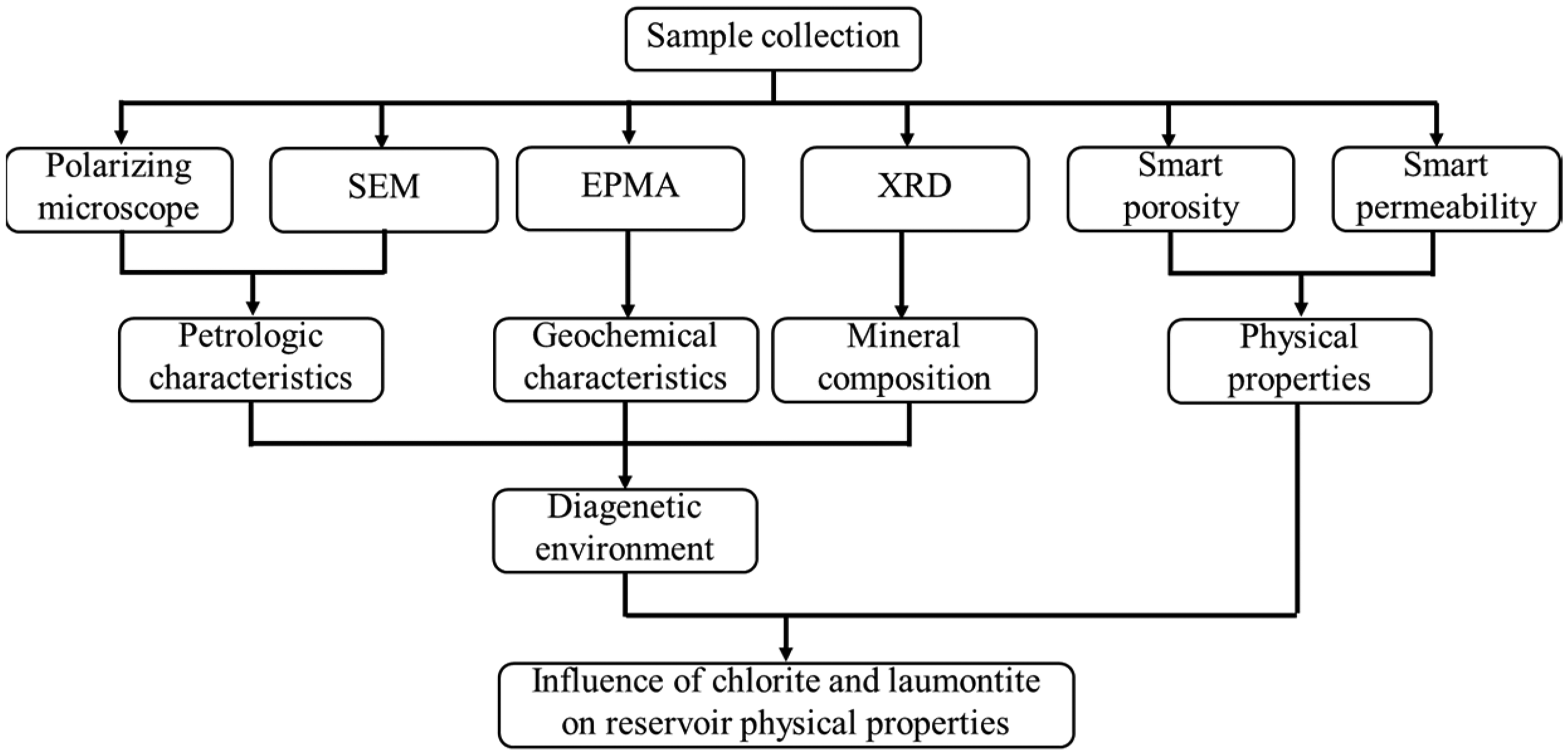
Workflow for this study.
The petrologic characteristics were analyzed by polarizing microscope and SEM, geochemical characteristics and mineral composition were measured by EPMA and XRD, respectively, with the petrologic, geochemical and minerals characteristics, the diagenetic evolution along with depth is demonstrated. Together with physical properties were measured by Smart porosity and permeability, the effects of laumontite and chlorite on reservoir are clarified.
Results
Petrological and mineralogical characteristics
Based on the polarizing microscope analysis with these 77 thin sections, it was found that clastic components of the Lower Wuerhe Formation in Madong area are diverse and mainly develop rock fragments; among them, tuff clasts are most widely distributed and abundant (Figure 3A), followed by felsic extrusive rock clasts and medium-basic extrusive rock fragments, content of quartz and feldspar are relatively less. There are more than 10 types of interstitial materials (Figure 3B), among which chlorite and laumontite are the most developed, they both take up more than 20%, followed by tuffaceous and calcite (>10%), and the content of iron and ilmenite is low. The particle size of rocks in the study area varies a lot with poor sorting. The particles are mainly in line contact with pore cementation, and the composition maturity and structural maturity are generally low.
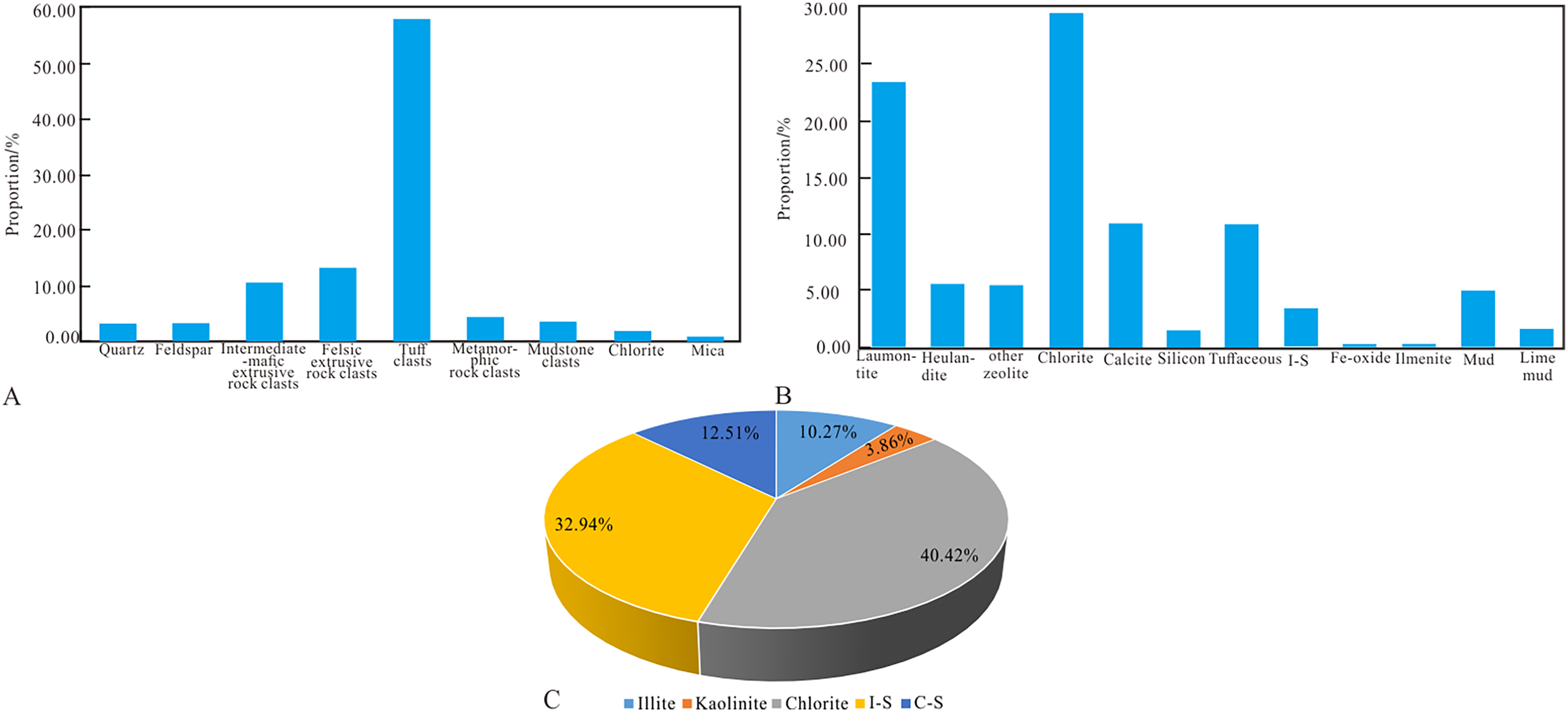
Petrological characteristics of Madong area in Junggar Basin; A. Proportion of clastic component; B. Proportion of interstitial material; C. Clay mineral content pie chart.
According to XRD results, the clay minerals in the target layer in the study area mainly include chlorite, illite, mixed layer of illite and smectite (I-S), mixed layer of chlorite and smectite (C-S) and kaolinite, among which chlorite accounts for 40.42% of the clay minerals, with the highest content, illite accounts for 10.27%, I-S accounts for 32.94%, C-M accounts for 12.51%, and kaolinite accounts for 3.86%, with the lowest content (Fig. 3C).
Chemical composition and occurrence of chlorite
Occurrence of the chlorite
Through thin-section identification, it was found that a large number of thin ilmenite films (Figure 4A, C, E) and tuffaceous were developed around the chlorite in the study area. The thin ilmenite films not only promote the formation of chlorite, but also compete with the chlorite in terms of material sources, they influence each other. The chlorite mainly has four types of occurrence mode (Figure 4): pore-lining type (Chl-I), particle-coated type (Chl-II), pore-filling type (Chl-III) and chlorite pseudomorph type (Chl-IV).
Pore-lined type (Chl-I) (Figure 4A, B, F) is the most common form of chlorite in the study area. It is generally 5μm∼20μm thick and usually found at locations of large pores and the edge of pores outside the particle contact points. The former presents a shape of bamboo leaves or continuous sheets; while the shape of the latter looks like a comb shell, and the closer it is to the larger pores, the better it is the degree of euhedral. The particle-coated type (Chl-II) (Figure 4C) develops in a form of thin film around the entire particle. The thickness is generally less than 5μm, and the crystals are arranged in a disorderly manner and have a poor degree of euhedral. The pore filling type (Chl-III) (Figure 4C, D) is filled with single particles or particle aggregates in the pores, often in the shape of rosettes or dispersed flakes, with a high degree of euhedral. The chlorite pseudomorph type (Chl-IV) (Figure 4E) is transformed from mafic mineral grains, the mafic mineral grains were altered and transformed into chlorite leave only the shape of the mafic mineral grain.

Characteristics of the occurrence of chlorite. A: Xia Yan 2 well, 4620.1 m, plane-polarized light, pore lined chlorite (continuous), thin layer of titanium iron; B: Yan Bei 4 well, 3913.67 m, backscatter image, pore lined chlorite; C: Xia Yan 2 well, 4854.67 m, plane-polarized light light, particle coating chlorite and pore filled chlorite, thin layer of titanium iron; D: Xia Yan 2 well, 3868.5 m, backscatter image, pore filled chlorite; E: Xia Yan Well 2, 4718.56 m, plane-polarized light, chlorite pseudomorph, pore filled chlorite and potassium feldspar; F: Yan Bei 4 well, 3869.37 m, plane-polarized light, pore lined chlorite (comb shell like), zeolite and potassium feldspar; Chl-I- Pore lined chlorite; Chl-II- Particle coated chlorite; Chl-III- Pore filled chlorite; Chl-IV- chlorite pseudomorph; Ilm- Thin layer of titanium iron; Anh- anhydrite; Qtz-quartz; Fsp- feldspar; Kfs- k-feldspar; Hul- Heulandit; Lmt- laumontite.
Chemical composition
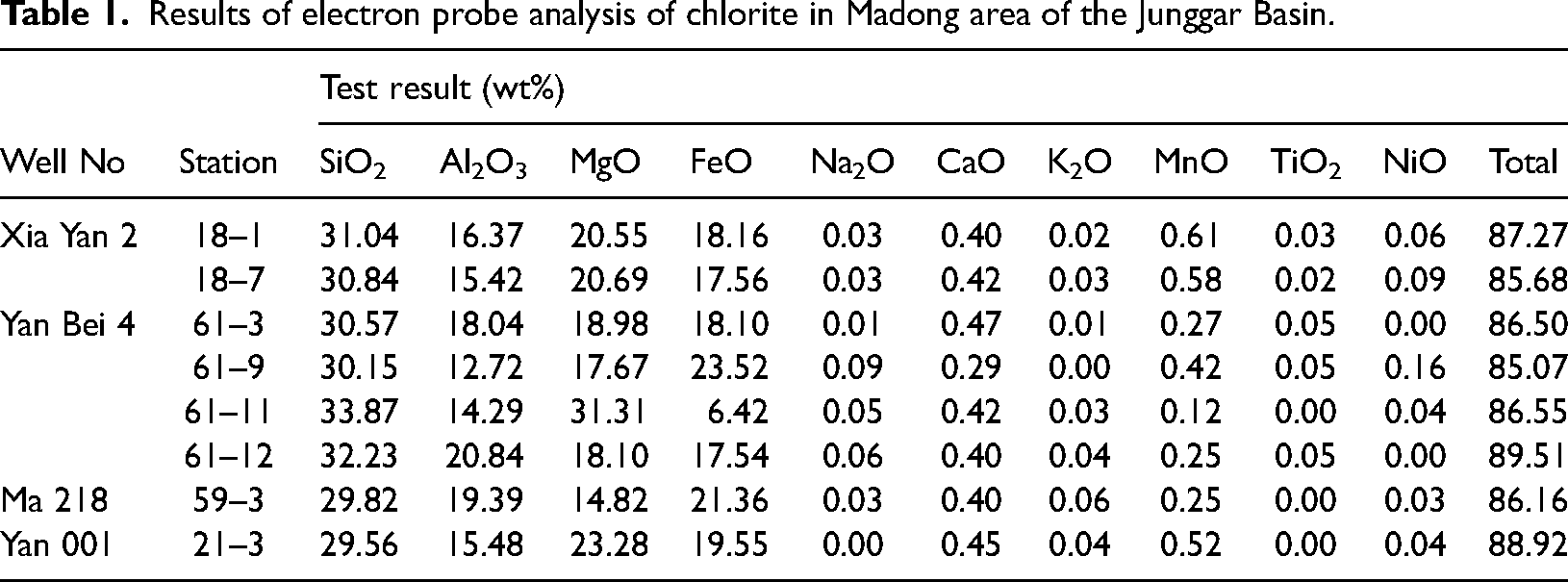
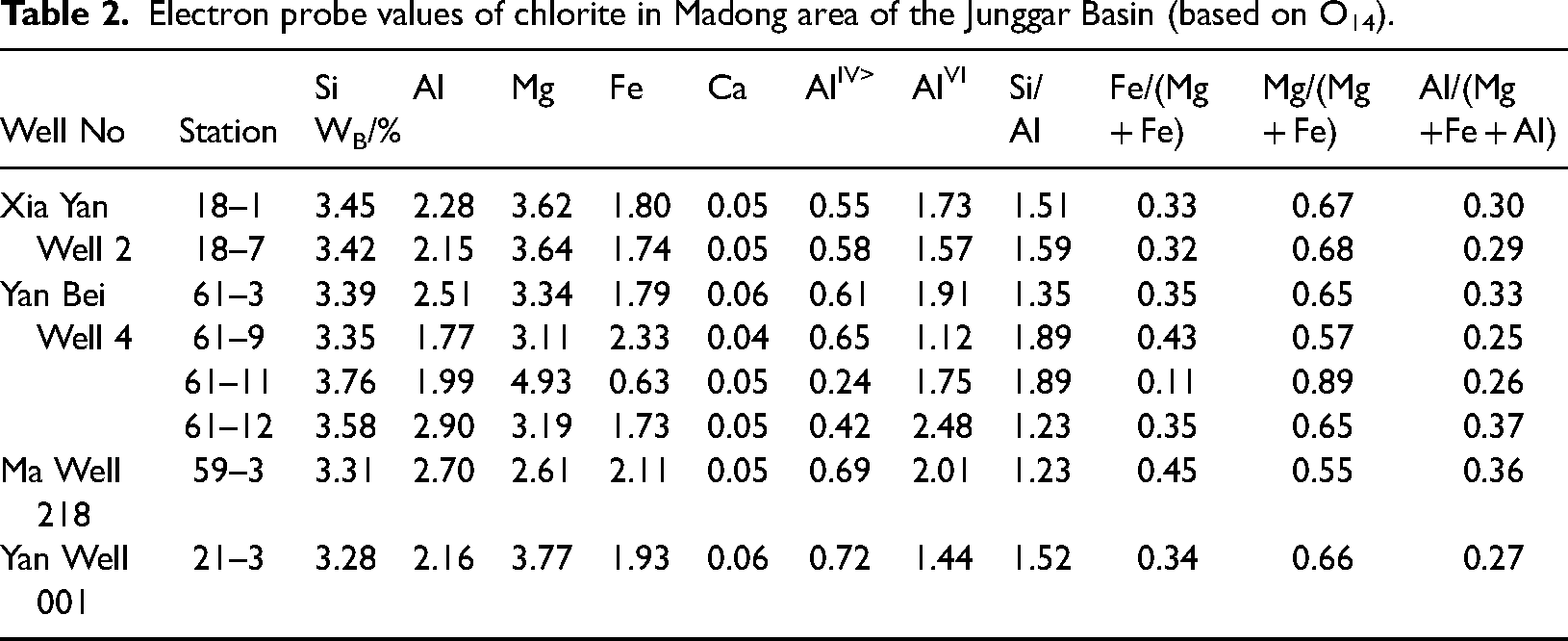
To ensure the purity of chlorite samples in EPMA, a criterion of (Na2O + CaO + K2O) < 0.5% is typically applied to exclude contamination by other minerals(D, 1962; W and WS, 1995). After filtering out any anomalous data, the detailed chemical composition of chlorite from the Madong area in the Junggar Basin is presented in Tables 1 and 2. The content of SiO2 ranges from 29.56% to 33.87%, with an average of 31.01%, which is relatively high. The Al2O3 content spans from 12.72% to 20.84%, averaging 16.57%. The MgO content ranges from 14.82% to 31.31%, with an average of 20.68%, indicating a notably high content. The FeO content varies from 6.42% to 23.52%, with an average of 17.78%.
Results of electron probe analysis of chlorite in Madong area of the Junggar Basin.
Electron probe values of chlorite in Madong area of the Junggar Basin (based on O14).
Chemical composition and occurrence of laumontite
Occurrence of laumontite
The laumontite is an aluminosilicate mineral. Based on the thin section identification, it was found that there are three occurrence states for the laumontite in this area: continuous crystals (Lmt-a), filled form (Lmt-b) and metasomatic form (Lmt-c), which are discussed as follows:
Crystal aggregates laumontite (Lmt-a) (Figure 5A, B, C): often develops in large continuous crystals, and the clastic particles are floating or in point contact[28] . This state is found in samples with high laumontite content. Filling-type laumontite (Lmt-b) (Figure 5C, D, F): fills the intergranular pores in forms of crystal clots or plate-like crystals. Metasomatic type laumontite (Lmt-c): often has irregular metasomatism of feldspar or rock fragments, with strong local metasomatism.

Characteristics of zeolite occurrence. A: Yan Bei 4 well, 3913.67 m, plane-polarized light, Crystal aggregates laumontite and metasomatic laumontite (metasomatic rock fragments); B: Yan Bei 4 well, 3915 m, plane-polarized light, Crystal aggregates laumontite and metasomatic laumontite (metasomatic rock fragments); C: Yan Bei 4 well, 3915 m, backscattered image, Crystal aggregates laumontite and filling-type laumontite; D: Xia Yan Well 2, 4717.76 m, plane-polarized light, filling-type laumontite and metasomatic type laumontite; E: Yan Bei 4 well, 3868.5 m, backscattered image, metasomatic type laumontite; F: Xia Yan Well 2, 4850.75 m, crossed polars, filling-type laumontite; Lmt-a- Crystal aggregates laumontite; Lmt-b- filling-type laumontite; Lmt-c metasomatic type laumontite; Chl-chlorite; Fsp-feldspar; Ilm-titanium iron thin film; Cal-calcite.
Chemical composition
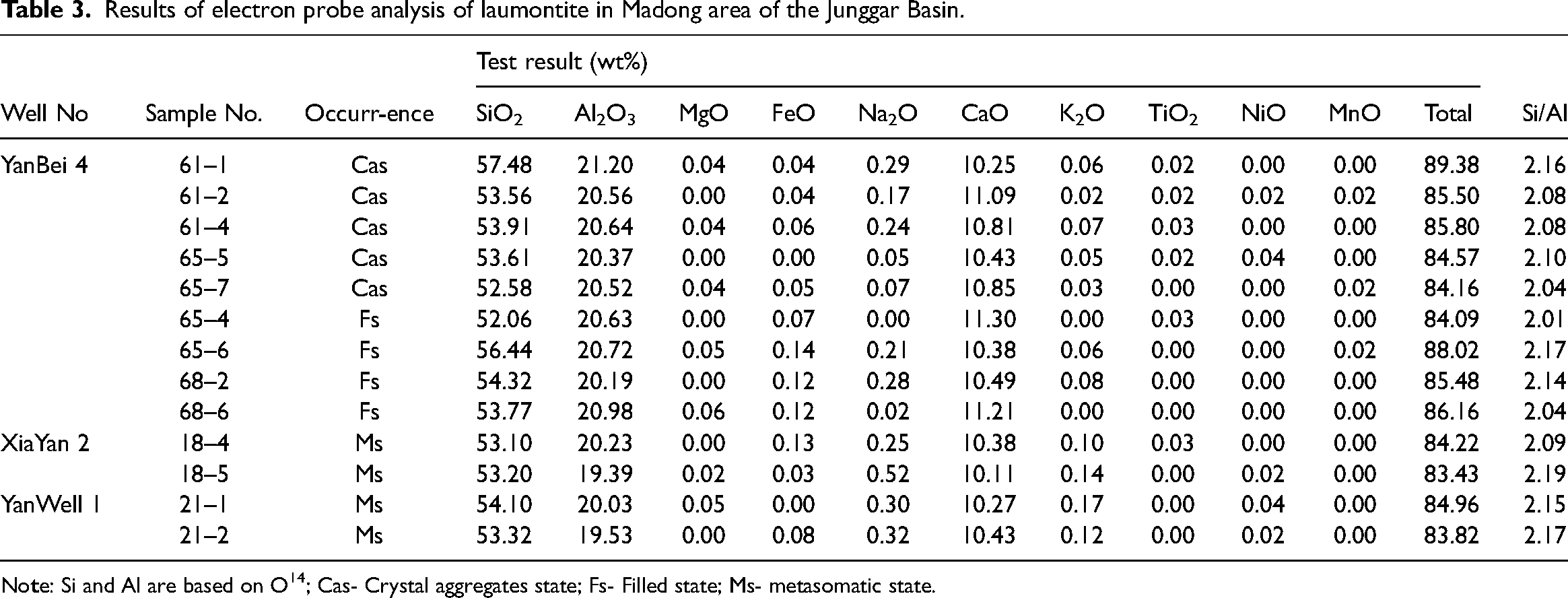
Based on the results from EPMA (Table 3), with difference occurrence, the laumontite has different chemical composition, for crystal aggregates laumontite (Lmt-a), the SiO2 is 52.58%∼57.48%, with an average of 54.23%, Al2O3 is 20.37%∼21.20%, with an average of 20.66%, CaO is 10.25%∼11.09%, with an average of 10.69%, the Si/Al value is 2.04∼2.16, with an average of 2.09, and the silicon and aluminum content are relatively high. Calcium content is relatively low. The filling-type laumontite (Lmt-b) shows slightly different characteristics, the SiO2 is 52.06%∼56.44%, with an average of 54.15%, and Al2O3is 20.19%∼20.98%, with an average of 20.63%. CaOis 10.38%∼11.30%, with an average of 10.85%. The Si/Al value is 2.01∼2.17, with an average of 2.09. The silicon and aluminum content are relatively low, and the calcium content is high. While for the Metasomatic type laumontite (Lmt-c), the SiO2 is 53.10%∼54.10%, with an average of 53.43%, and Al2O3 is 19.39%∼20.23%, with an average of 19.80%, CaO is 10.11%∼10.43%, with an average of 10.30%, the Si/Al value is 2.09∼2.19, with an average of 2.15, and the silicon, aluminum, and calcium contents are low.
Results of electron probe analysis of laumontite in Madong area of the Junggar Basin.
No
Discussion
Identification of chlorite types
Chlorite is an aluminum silicate mineral with a crystallochemical formula of ((Fe2+, Mg2+, Mn2+)x(Fe3+, Al3+, Cr3+)y□z)VI(Si4−uAlu)IVO10 + w (OH)8−w, in which x + y +z = 6; □ in the formula signifies an octahedral vacancy within the layer structure, indicating positions where cations are absent(F et al., 2013). The most widely used classification methods for chlorite are Si-Fe diagram which performs the classification based on the composition, and Fe-Mg-Al+□ triangle diagram which classifies the chlorite according to its structure and uses O14 as the criterion(W.A et al., 1962; A and Z, 1998; Deer et al., 2013). From the Si-Fe diagram (Figure 6A) it is obvious that the chlorite in the target layer of the study area is mainly ferroan clinochlore. According to the Fe-Mg-Al+□ diagram (Figure 6B), all chlorite samples exhibit a trioctahedral structure, indicating that they belong to the iron-magnesium transitional type with a partial magnesia-rich composition. This classification suggests that these chlorites have a significant magnesium content relative to aluminum and iron, but also contain substantial amounts of iron, reflecting their transitional nature between iron-rich and magnesium-rich varieties.
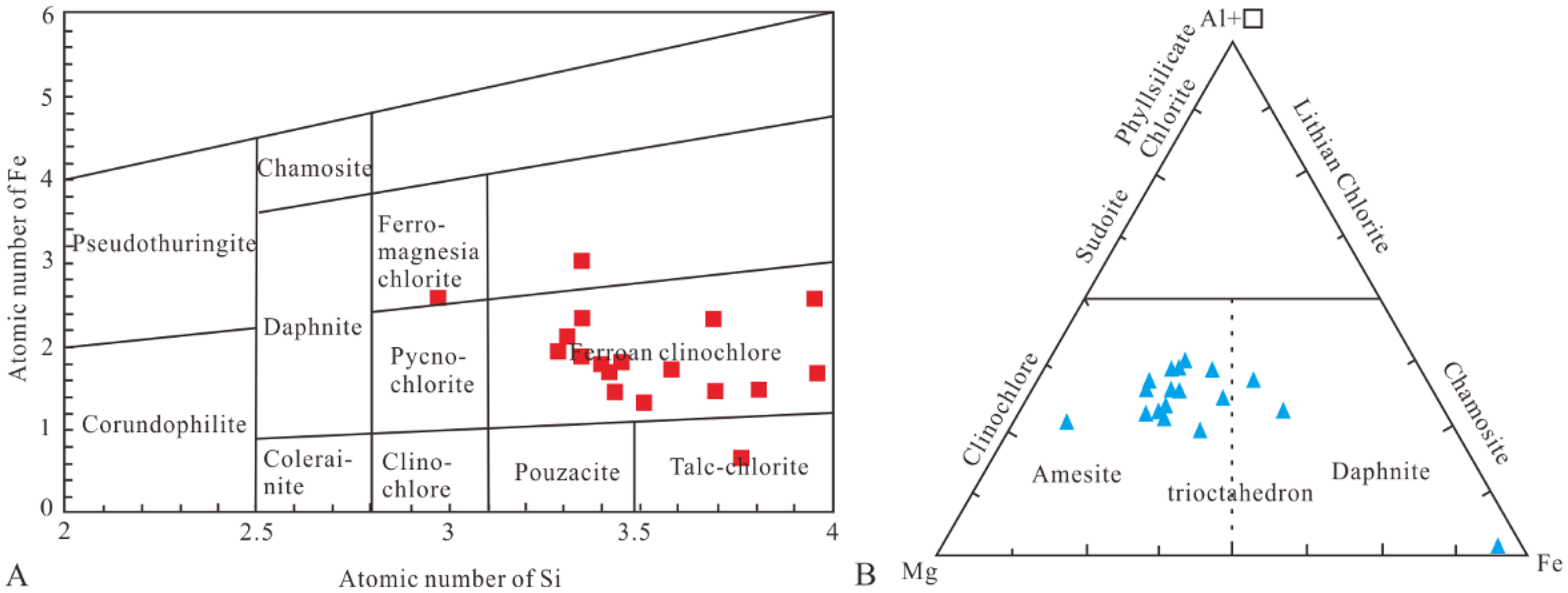
Classification diagram of chlorite in madong area of the junggar basin.
The chlorite samples exhibit significant variability in their iron and magnesium content, highlighting the prevalence of iron-magnesium substitution within the chlorite lattice. This substitution phenomenon is consistent with the observed trioctahedral structure and supports the classification of these chlorites as iron-magnesium transitional types, partially magnesia-rich.
Correlation of major cations in chlorite
To express the substitution relationships of chlorite in tetrahedral and octahedral positions, mutual relationship diagrams of AlIV, AlVI, Mg, Fe, Si, Fe/(Mg + Fe), and Mg/(Mg + Fe) are commonly utilized (W and WS, 1995; Chunrong et al., 2020). In the Madong area of the Junggar Basin, the value ranges for AlIV and AlVI are 0.24 to 0.72 and 1.12 to 2.48, respectively (Table 2). The AlIV-AlVI relationship diagram (Figure 7A) shows a weak negative correlation (R2 = 0.14). The observed relationship suggests that the tetrahedral position substitution in chlorite is not exclusively of the calcium-magnesium-amphibole type but involves more complex mechanisms. Specifically, it indicates that AlIV replaces Si at the tetrahedral position, accompanied by AlVI replacing Fe or Mg in the octahedral position to balance the charge(Songyan et al., 2024). The generally higher AlVI values compared to AlIV values reflect that substitution of Fe or Mg by AlVI in the octahedron is more common than the substitution of Si by AlIV in the tetrahedron. This pattern implies that chlorite formation in this area may be influenced by multi-stages diagenetic activities, leading to heterogeneous substitution behaviors.
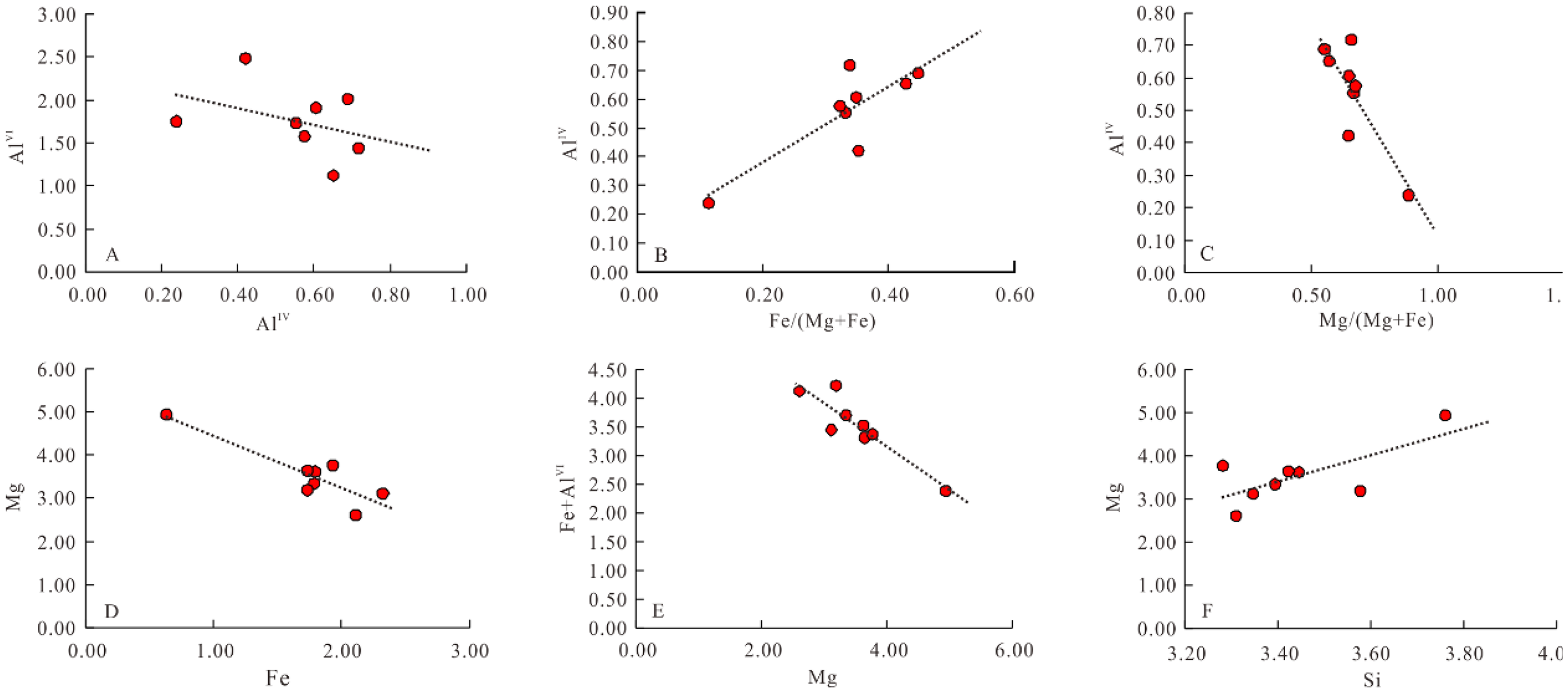
Correlation diagram of main cations of chlorite in madong area of the junggar basin.
The relationship diagrams (Figure 7B, C), show that there is a positive correlation (R2 = 0.7) between Fe/(Mg + Fe) and AlIV, and a negative correlation (R2 = 0.7) between Mg/(Mg + Fe) and AlIV. These correlations indicate that as AlIV substitutes for Si, there is concurrent substitution between Fe and Mg in the octahedral positions. Specifically, an increase in AlIV corresponds with an increase in Fe relative Meanwhile, a strong negative correlation (R2 = 0.8) between Fe and Mg (Figure 7D) confirming that Fe substitution for Mg predominates in the octahedral positions of chlorite in this area.
The diagram of the relationship between Mg and Fe + AlVI (Figure 7E) also shows a negative correlation. Combined with Figure 4(d), this reinforces the observation that the primary substitution in the octahedral positions is Fe substituting Mg, followed by Mg being replaced by AlVI.
The diagram of relationship between Si and Mg (Figure 7F) shows a relatively weak positive correlation (R2 = 0.5). This suggests that the chlorite in this area was formed through multiple stages of diagenetic activity which corresponds well with the relationship between AlIV and AlVI(Wei et al., 2014).
The indicative significance of chlorite for diagenetic environments
The ratio of the mass fractions of Al, Mg, and Fe in chlorite can be used to indicate the formation environment of chlorite. When the mass fraction ratio of Al to Al + Mg + Fe is greater than 0.35, it indicates that the chlorite was formed from the alteration of clay minerals. Conversely, when this ratio is less than 0.35, it suggests that the chlorite was derived from the transformation of mafic rocks (J, 1988). The Al/(Al + Mg + Fe) value (Table 2) of the chlorite in the target layer in the study area is 0.25∼0.37, which also shows that the chlorite in the area has different origins. The occurrences of the chlorite under the polarizing microscope include both the chlorite filled in the void and chlorite pseudomorph, this implies that, in the study area, there are at least two pathways for the formation of the chlorite: transformed from mafic minerals and transformed from other clay minerals such as kaolinite. This explanation corresponds well with the geological background that was affected by the Yanshan Movement, it is volcanically active in the basin, and the mafic volcanic rock fragments were transported to the study area through rivers or in the form of volcanic ash.
After sedimentation, these volcanic materials have a long time of hydrolysis and dissolution, providing a large amount of ions, such as Mg2+, Fe2+, Ca2+ and K + (Liqi et al. 2023). This leads to the mutual transformation of clay minerals: in a K-rich environment, kaolinite (Kao.) transforms into illite (I) (Equation (1)), and in a mafic-rich environment, kaolinite transforms into chlorite (Chl.) (Equation (2)) (Tian Jianfeng et al., 2008; Jianfeng et al., 2022).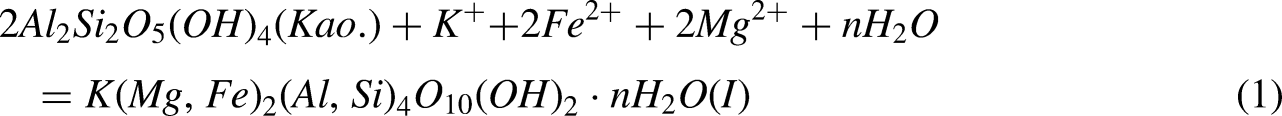
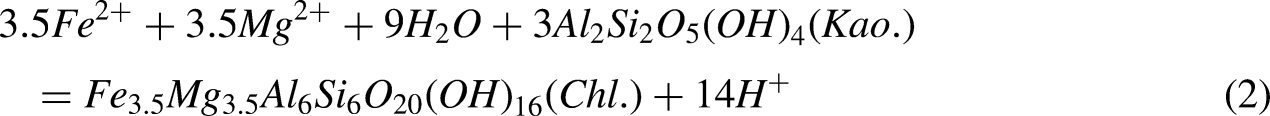
Transformation diagram of clay mineral components.
Factors influencing the formation of laumontite
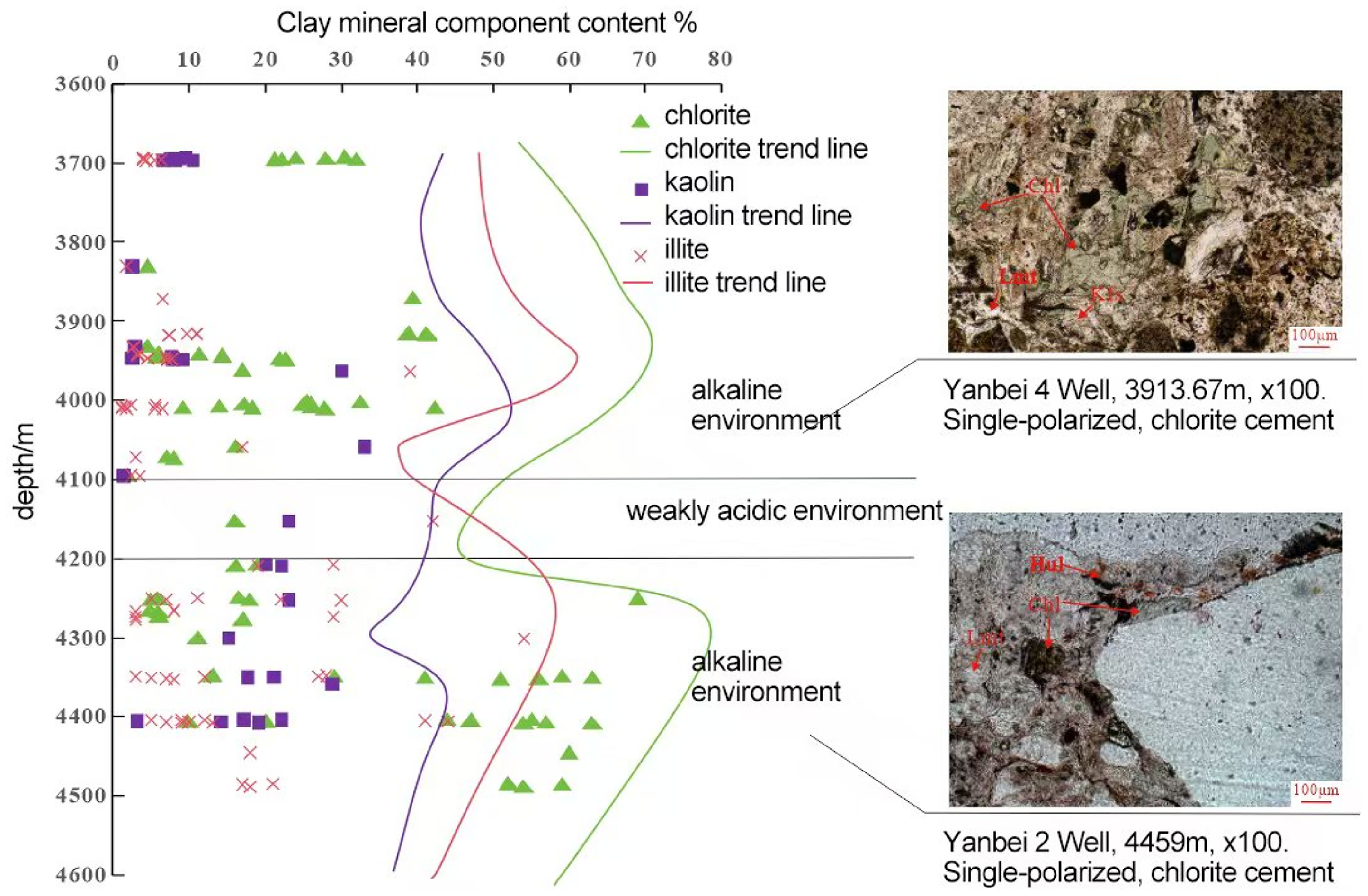
According to the rock thin section identification (Figure 5) and the chemical characteristics of major elements (Figure 9), it can be seen that the laumontite in the study area mainly develops in the form of crystal aggregates and metasomatic forms. With the accumulation of the overlying sediments, the diagenetic system will gradually turn from opening to closing (Yang et al., 2020). Under the closed diagenetic system, the occurrence of laumontite is closely related to the surrounding minerals (Chen et al., 2024; Zhang et al., 2024). In this study, the types and contents of the symbiotic minerals around the laumontite in different occurrences also vary, which indicates that its formation is closely related to symbiotic minerals. The continuous crystalline laumontite is relatively rich in aluminum and silicon elements and is surrounded by a large number of rock fragments. The dissolution of the rock fragments provides it with sufficient ions such as Si4+, Al3+, Ca2+, which promotes the formation of laumontite; the filled laumontite is surrounded by chlorite, calcite and other minerals, and these minerals compete with the laumontite in terms of material sources (Figure 9A), resulting in relatively low silicon and aluminum content in the laumontite. Since a large amount of H+ is released during the formation of chlorite, a large number of dissolution pores appear in the laumontite (Mozhen et al., 2016); Metasomatic laumontite mainly metasomatizes feldspar and rock fragments. Taking albite as an example (Figure 9B), the precipitation of laumontite occurs almost simultaneously with the albitization of plagioclase (Long et al., 2022). There is an obvious trade-off between the main elements in metasomatic laumontite and albite, resulting in a higher Si/Al value in the metasomatic state, which means strong acid resistance, and not easily corroded (Yuanhua et al., 2020).
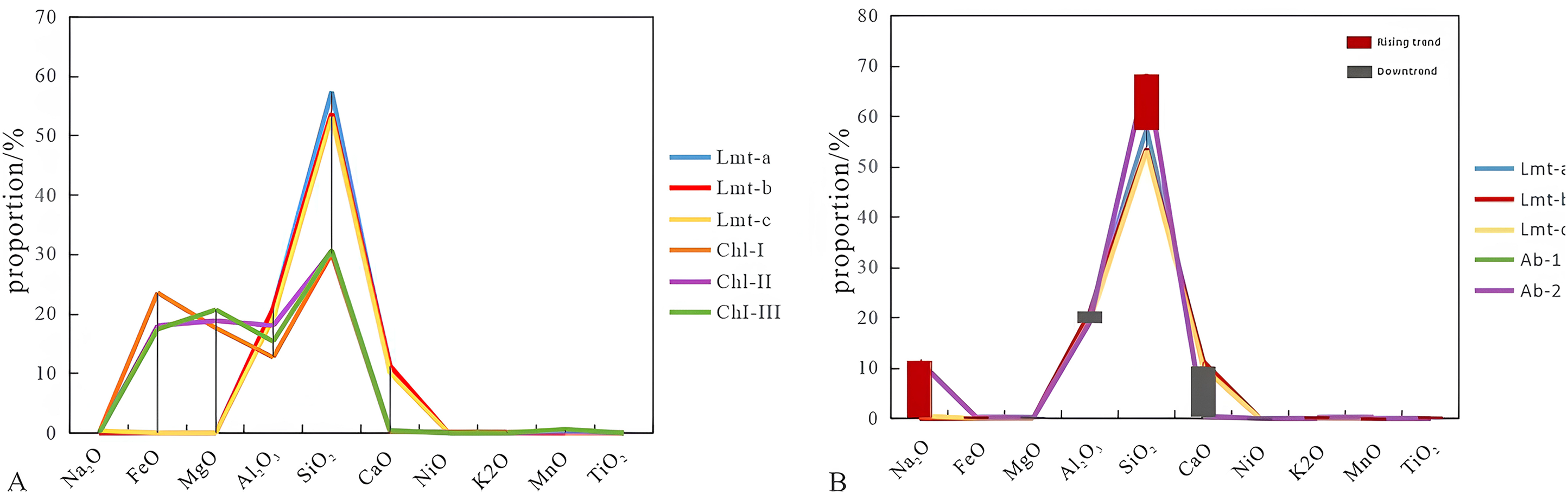
Chemical characteristics of laumontite with chlorite and albite.
The indicative significance of laumontite for diagenetic environments
The formation of laumontite requires not only an alkaline diagenetic environment but also takes into account the competitive equilibrium among various components (He et al., 2023; Chen et al., 2024). Conditions favorable for laumontite precipitation include lower CO32− concentration, pore waters rich in Na+, and high pH values. When the diagenetic fluid becomes acidic, laumontite tends to transform into illite. Furthermore, if the fluid has a high CO32− content during this acidic phase, laumontite will instead convert to kaolinite (Xuehua et al., 2011; Zhang et al., 2024). Between depths of 3800 m to 4120 m, laumontite is relatively enriched (Figure 10), indicating an alkaline diagenetic environment, during which feldspar content is relatively abundant. From approximately 4120 m to 4220 m, laumontite is less developed, while kaolinite (Figure 7) and illite are abundantly present, with a reduction in feldspar content. This indicates the transformation of laumontite to illite along with the dissolution of feldspar, suggesting that this depth segment has an acidic diagenetic environment. Between 4220 m and 4600 m, laumontite reappears, indicating a shift back from an acidic to an alkaline diagenetic environment. As pH values and formation temperatures increase, plagioclase undergoes sodic alteration. This corresponds well with the formation of chlorite and further confirms that within the Lower Wuerhe Formation of the study area, as depth increases, there is a change in the diagenetic environment from alkaline to acidic and back to alkaline.
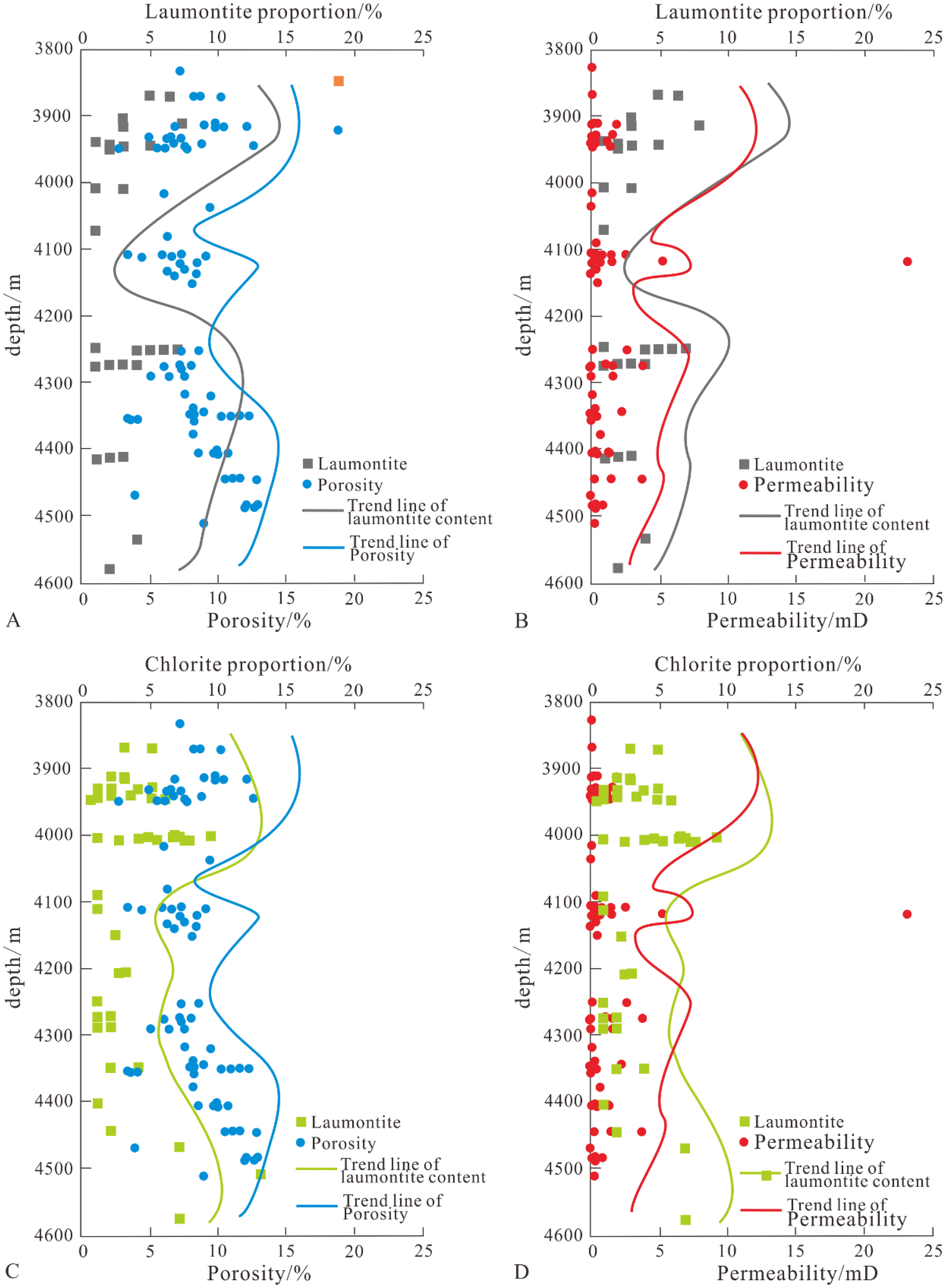
Analysis of the influence of chlorite and zeolite on reservoir properties.
The influence of chlorite and laumontite on reservoir physical properties
Based on the characteristics of thin sections (Figures 6 and 8), it can be seen that chlorite and laumontite in different occurrence states have dual effects on the physical properties of the reservoir: constructive and destructive.
Constructive effects
Chlorite and laumontite enhance the reservoir pressure resistance to a certain extent. Pore-lined chlorite (Figure 6A, E, F) and particle-coated chlorite (Figure 6C) develop around the particles in large quantities, which suppresses secondary quartz[32] and protects pores and throats. Many secondary pores are formed due to the acidic dissolution of the crystal aggregates laumontite, which improves the physical properties of the reservoir.
In sandy conglomerates that develop pore-lined chlorite, particle-coated chlorite and laumontite, the particles show point-line contact and line contact; in those that do not develop these, the particles show line contact, concave-convex contact, and a small amount of suture line contact. The secondary pores in the target layer in the study area are mainly produced by the dissolution of acid-soluble substances such as feldspar, rock fragments and silicate cement by acidic fluids, while the development of pore-lined chlorite and particle-coated chlorite provides channels for the flow of acidic fluids and promotes dissolution.
Destructive effects
Both chlorite and laumontite further densifying the reservoir. The pore-filling chlorite is in the form of crystal grains and completely fills the pores, destroying the pore structure of the reservoir, shrinking the throat, and dividing the pores into many tiny pores (Xiao-feng et al., 2017; Tao et al., 2022), seriously affecting the permeability of the reservoir; under the alternating action of multiple weakly acidic to alkaline diagenetic environments, some of the dissolved pores of the crystalline laumontite will be secondary cemented by the zeolite again, destroying the reservoir structure; metasomatic laumontite is rich in silicon and poor in aluminum, which restrains the dissolution of minerals such as feldspar and rock fragments; filled laumontite destroys a large number of pores and throats, further reducing the permeability of the reservoir.
Influence on reservoir physical properties
Both laumontite and chlorite are developed between 3900 and 3950 m. Within this segment of the reservoir, kaolinite transforms into chlorite, releasing a certain amount of H+, which causes feldspar and other materials to generate secondary pores, thereby increasing porosity and permeability (Figure 10). As chlorite again accumulates in large quantities at a depth of 4000 meters, it blocks pores and fills pore throats, leading to a decrease in both porosity and permeability.
Between depths of 4120 to 4220 meters, chlorite is scarce, and laumontite is almost non-existent, indicating an acidic diagenetic environment. During this period, acidic fluids cause feldspar and other acid-soluble materials to produce a significant number of secondary pores, thus increasing porosity and permeability.
From 4220 to 4600 meters, porosity and permeability initially decrease and then increase, with overall trends varying according to the content of chlorite. This suggests that within this segment of the reservoir, some chlorite has played a protective role for pore throats, while laumontite has had limited improvement effects on the reservoir properties.
Conclusion
Based on analyses using polarizing microscopy, SEM, XRD, EPMA, and physical property analysis, this study investigates the characteristics of chlorite and laumontite in the Lower Wuerhe Formation of the Madong area and their impact on the properties of conglomeratic sandstone reservoirs. The findings and conclusions are as follows:
In the study area, chlorite within the Lower Wuerhe Formation is primarily ferrichlorite, a type that belongs to the Fe-Mg transitional category but is magnesium-rich. Chlorite occurs mainly in three forms: pore-lining, grain-coating, and pore-filling. Laumontite exhibits three types of occurrences: crystal aggregates, fillings, and replacements. The dissolution of feldspar and lithic fragments promotes the formation of laumontite. As depth increases, the diagenetic environment changes from alkaline to weakly acidic and then back to alkaline. Pore-lining and grain-coating chlorite protect pores and throats, whereas pore-filling chlorite completely fills the pore spaces, damaging the reservoir's pore structure. Crystal aggregate laumontite forms numerous secondary pores through acid dissolution in weakly acidic conditions. Replacement laumontite inhibits the dissolution of feldspar and lithic fragments, while filling laumontite destroys a significant number of pores and throats.
Footnotes
Acknowledgements
The authors would like to thank the editor and the two professional reviewer to help improve this paper.
Author contributions
LF and NJ were involved in conceptualization, methodology and investigation, HJ and WC collected and analyzed the samples; writing – original draft preparation by LF and NJ; writing – revised by LF
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Xinjiang Uygur Autonomous Region Natural Science Foundation – Prefecture-level City Science Fund Project, Karamay City – Innovation Talent Program, Research Foundation of China University of Petroleum-Beijing at Karamay, Basic Research Fund of the Autonomous Region for the Karamay Campus, (grant number 2022D01F59, 2024hjcxrc0046, XQZX20240017, XJEDU2024P105).
