Abstract
Promoting the permeability of deep, low-permeability coal seams through biological means is currently a research hotspot for enhancing the efficiency of coalbed methane extraction. In this study, we selected anthracite coal from Sihe Mine for microbial anaerobic degradation culture experiments. The effects of adding functional microorganisms on the microstructure of anthracite coal were analyzed by high-throughput sequencing of samples before and after the cultivation and microcharacterization experiments of coal samples. The results showed that the maximum methane migration during the period of biodegradation reached 0.640 mL/g coal, and the cumulative migration of the whole cycle was as high as 1.318 mL/g coal. 16S rRNA high-throughput sequencing results indicated that the bacterial community structure had undergone significant succession after the biodegradation experiments, and that the Firmicutes represented by Bacillus(82.41% of the total) occupied the dominant niche. Metabolic pathway analysis based on Kyoto Encyclopedia of Genes and Genomes (KEGG) database showed that the degradation of aromatic compounds by microorganisms appeared to be significantly enhanced by the addition of nitrogen sources. Also, the relative abundance of a number of key metabolic enzyme genes capable of catalyzing the oxygen-containing functional groups into the structure of the coal molecule and the de-cyclization reaction were increased. Fourier Transform Infrared Spectrometer (FTIR) experiments revealed that biodegradation stimulated by nitrogen source reduced the aromaticity of coal by 59.62% and enhanced the hydroxyl functional group content by 1.822 times. Mercury pressure and low-temperature nitrogen adsorption experiments showed that the micropore pore volume of the treated coal decreased by 34.09%, and the macropore pore volume accounted for an increased 168.28%, with an average pore size increment of 60.72 nm. Therefore, the nitrogen source can stimulate Firmicutes on the degradation of polycyclic aromatic hydrocarbon and increase the content of oxygen-containing functional groups, which might promote the development of pores in coal and make the difficult-to-desorption methane migrates rapidly. This study investigated the effect of nitrogen source on the degradation of coal by Firmicutes. This will help improve the efficiency of gas extraction and ensure the safety of coal mine production.
Keywords
Introduction
The gradual depletion of shallow coal resources and the ongoing shift to deeper mining operations have intensified gas-related disasters in coal production (Niu et al., 2022; Zhou et al., 2016). This issue is particularly pronounced in Chinese coal seams, which are characterized by inadequate pore and fissure development and low permeability (Wang and Du, 2020; Wang et al., 2020), These geological conditions complicate gas extraction in mines, presenting significant challenges to the safety of coal production (Lu et al., 2021). Currently, physical, chemical and biological permeability enhancement technologies are mainly applied or experimented in mines to increase the permeability of low permeability coal seams with high gas content and to enhance the level of methane transport (Duan et al., 2020; Li et al., 2022; Su et al., 2022). Physical permeability enhancement technology primarily employs methods such as water jetting, drilling, and mining protective layer technology to make the coal seam decompression rupture coal. This process facilitates the development of pore and fissure structure, enhancing the transport ability of the coal. Consequently, the permeability of the coal seam is increased, which significantly improves the efficiency of gas extraction. These techniques have found widespread application in coal mine sites (Liu et al., 2020; Wang et al., 2013; Zhang et al., 2020). For chemical penetration enhancement, one part of the study is to remove the mineral components from the coal by soaking the coal in a strong acid solution, which induces a change in the pore and fissure structure of the coal (Li et al., 2018; Yi et al., 2021); the other part is to use organic solvents, which dissolve the small molecules of soluble substances in the organic structure of coal (Zheng et al., 2023).
Compared with traditional penetration enhancement techniques, biological methods space advantages of being green, safe, economic and sustainable. Therefore, the use of microorganisms to improve the gas extraction and to develop clean energy has far-reaching and significant theoretical and practical significance in today's world, which emphasises low-carbon and safety (Ritter et al., 2015). Scott (1999) first suggested that permeability of coal bodies could be enhanced by microbial degradation. Since then, scholars around the world have carried out laboratory simulations and field experiments to verify the effect and mechanism of microbial anaerobic degradation on coal permeability enhancement. Currently, studies have been conducted to describe the effect of microbial metabolism on the microstructure of coal, some of the studies have proved that the micropore structure of coal will develop into larger pores and improve the pore fracture structure after microbial degradation. Bao et al. (2023) found that biodegradation can achieve effects such as pore and fracture expansion, which are favorable for methane seepage and transport. Pandey and Harpalani (2018) concluded that coal biotransformation resulted in a swelling of the coal matrix, decreasing fractal dimension and contributing to the formation of new pore cleavages.
In addition, some scholars have confirmed through microbiology, metabolomics and spectroscopy that the molecular structure and crystal structure of coal will evolve after the action of key products of biometabolism, and its methanophilicity will be reduced, which will help to improve the permeability of coal. Guo et al. (2014) found that after the degradation of coal by biometabolism, the oxygen-containing functional groups of coal increased, and the aromatic rings were partially opened and hydroxyl groups introduced at the breaks through the FTIR and XRD tests. Furthermore, the disorder of coal macromolecules increased, and the degree of crystallization decreased. Haider (2017) found that aromatic compounds in coal could be degraded after biodegradation experiments using lignite. In addition, the organic components produced during the degradation process can be applied in biomethane synthesis. Xia et al. (2023) concluded that microorganisms break down the covalent bonds and functional groups of the macromolecules in coal by secreting extracellular enzymes in the degradation metabolism, which ultimately increase the gas content of coal seams, change the coal's pore structure, and decrease the fractal dimensions of the coal surface to make the coal surface smooth. Currently, the traditional single permeability enhancement technology, such as drilling and hydraulic fracturing, exhibit poor practical application effect effects. In some cases, these methods may lead to areas of ineffective enhancement or localized gas accumulation, resulting in issues such as gas jetting from the hole. There is an urgent need to explore a new mode of efficient and safe coal seam penetration enhancement. In addition, most research on biodegradation of coal focuses on using microorganisms to enhance coalbed methane production. In contrast, studies aimed at controlling gas hazards via microbial methods are relatively scarce. Therefore, to addresses a critical issue in mine safety: gas hazards, it is necessary to further study whether Firmicutes can enhance the permeability of anthracite under the stimulation of nitrogen source when methane inhibitors inhibit the methane metabolism function of methanogens in coal.
In this paper, we selected the poorly developed pore space and low permeability anthracite coal from Sihe Mine to add highly efficient degrading bacterial agents for anaerobic degradation indoor cultivation experiments, and monitored the dynamic migration amount of residual methane in the coal in the process of the experiments. We analyzed the microbial community structure succession law and metabolic function variability by 16 s rRNA high-throughput sequencing; jointly characterized the developmental characteristics of the microporous pore structure of the coal samples after the experiments based on pressurized mercury and low-temperature nitrogen adsorption experiments; and investigated the structural evolution characteristics of the coal microfunctional groups by using FTIR experiments. This paper explores the microstructure evolution of coal by Firmicutes under the stimulation of nitrogen source, reveals the mechanism of microorganisms in the anaerobic degradation of coal, and provides data for increasing the methane storage and transportation characteristics of high-gas and low-permeability coal seams.
Materials and methods
Coal sample collection and coal quality parameters
Fresh coal samples were collected from Shanxi Sihe Mine No.3 Coal Seam. The freshly collected coal samples were immediately placed in sterile ziplock bags and transported to the laboratory at low temperature wrapped in ice. An appropriate amount of coal samples were taken for ultimate analysis, proximate analysis and the maximum vitrinite reflectivity (R0,max) test respectively.
The results are shown in the following Table 1. After surface excision of the samples, the samples were crushed, milled, and screened, and the 60–80 mesh size of the coal powder was collected and preserved at low temperatures for the subsequent experiments.
Ultimate/proximate/R0,max analysis of raw coal samples.

M, moisture; A, ash yield; V, volatile matter; FC, fixed-carbon; ad, air-dry basis; N, nitrogen; C, carbon; H, hydrogen; S, sulfur.
Experimental design and testing
The functional microorganisms added in the experiment were obtained by pumping and filtering the highly efficient degradation functional bacterial solvents and using sterile water to mix and then repeat the operation for 3 times. (Microbial composition in bacterial solvents by high-throughput sequencing: Bacillus accounted for 56.80%, Clostridium accounted for 23.28%, Paraclostridium accounted for 19.92%, and the bacterial concentration in the agent was 108∼109 cfu/mL).
The specific ratios of the basic salt medium used in the experiment were as follows: NH4Cl 0.81 g/L, MgCl2 · 6H2O 0.17 g/L, CaCl2 · 2H2O 0. 1 g/L ,FeCl2·4H2O 0.07 g/L,Na2HPO4·12H2O 1.5 g/L,KH2PO4 1.5 g/L,sterile water.
The control group (CK) was added 50 g of coal sample and 100 mL of sterile water. Treat-1 (T-1) takes 50 g of coal sample as the reaction substrate and places it in a 500 mL culture bottle. Functional microorganisms (120 mL of bacteriophage filtration) are added, along with 100 mL of base salt culture medium, 0.5 g of cysteine hydrochloride, and 2 ml of 10 mmol sodium 2-bromoethylsulfonate solution (BES). BES is used to inhibit the methane production potential of microorganisms and ensure that the headspace methane is produced from residual methane in coal (Webster et al., 2016). Treat-2 (T-2) was identical to T-1 except for the addition of tryptone 1 g. All three sets of experiments were sealed with nitrile plugs and three parallel experiments were performed.
The initial anaerobic environment of the culture bottles was replaced with high-purity nitrogen for three times using a vacuum filtration device and a sterile syringe to replace the headspace air. The culture bottles, centrifuge tubes and culture medium used in the experiment were autoclaved, and the experimental process was carried out on the aseptic operating table. The experiments were carried out at a constant temperature of 37 °C for 30 d. To obtain pure coal samples, the residual coal was separated from the experimental samples by suction filtration with sterile water for several times, and then the coal samples were dried in an electric blast drying oven at 37 °C until constant weight and then sealed and stored (Figure 1).

Experimental design of microbial anaerobic degradation of anthracite coal.
During the incubation experiments, the methane concentration percentage in the headspace gas inside the incubation bottles during the biodegradation process was detected using a capillary analytical gas chromatograph model GC-8900 from Shandong Lunan Xinke. The temperature of the injection chamber of the gas chromatograph was 105 °C, the temperature of the column chamber was 102 °C, the temperature of the thermal conductivity cell was 116 °C, and the bridge current was set at 150 mA. High purity nitrogen was used to replace the headspace gas in the culture flask when methane migration stabilized, that is one methane migration cycle.
High-throughput sequencing of 16S rRNA gene amplicons and gene function analysis experiments
MB AquaScreen Fast Extract was used to extract total genomic DNA from CK, T-1 and T-2 on days 0 and 30, which were then stored on dry ice and sent to UW Genetics Ltd in China, where the microbial communities in the culture system were analyzed by high-throughput sequencing of the 16S rRNA gene amplicons on the HiSeq platform.515F/907R (515F : 5′-GTG CCA GCM GCC GCG G-3′; 907R: 5′-CCG TCA ATT CMT T TR AGT TT-3′) primer pairs were used to amplify the bacterial 16S rRNA gene. Clustering was performed using UPARSE at 97% similarity, representative sequences of OTUs were obtained and chimeras were identified and deleted using UCHIME (v4.2.40), and classified and annotated according to SILVA reference data (v128). Sequences classified as “mitochondrial” or “unassigned” were removed.
Based on the Kyoto Encyclopedia of Genes and Genomes (KEGG database), we screened metabolic pathways related to the degradation of methane and molecular structures in anthracite and analyzed the differences in key gene functions.
Experiments on the determination of coal pore structure parameters
Low-temperature liquid nitrogen adsorption experiments were carried out using a high-performance specific surface area and microporous analyzer, BSD-PM, manufactured by Best Instrument Technology (Beijing) Co. About 1.5 g of dried coal samples were taken and placed in a sample tube and degassed under vacuum at 105°C for 6 hours.
The high performance fully automated mercury palyzer AutoPore IV 9500 produced by Shenzhen Huapu General Technology Co. The pressure range was from 1 to 33,000 psia, the accuracy of mercury feed was 0.1 μL, and the equilibrium time of each pressure collection point was 10 seconds. The pressure range was from 1 to 33,000 psia, and the accuracy of mercury feed was 0.1 μL.
The coal sample used in the experiment came from Shanxi Sihe Mine coal, which belongs to anthracite, so the analysis was performed using the method of pore size structure division of high coal rank coal by Qin et al. (1995), that is micropore (<15 nm), transitional pore (15∼50 nm), mesopore (50–400 nm), and macropore (>400 nm).
The results of low-temperature liquid nitrogen adsorption experiments were used for the analysis of micropore and transitional pores for raw coal, T-1 and T-2 coal samples, and the results of mercury compression experiments were used for the analysis of mesopores and macropores. When dealing with the results of low-temperature liquid nitrogen adsorption experiments, the NLDFT model, which is more accurate for analyzing micropores, was chosen for the pore size distribution at <15 nm, and the classical BJH desorption method was chosen for the 15–50 nm segment (Landers et al., 2013; Monson, 2012). In addition, due to the existence of voids between the coal samples in the mercury pressure experiment, the pressure point of 0.4 Mpa was selected as the initial mercury absorption reference point in accordance with previous studies (Suuberg et al., 1995).
Experiments on the determination of coal molecular structure parameters
FTIR experiments on residual coal from raw coal, T-1 and T-2 coal samples were performed using a Bruker INVENIO infrared spectrometer, Germany. The dried coal samples were mixed with KBr powder in the ratio of 1:100, evenly ground and put into a tablet press to make pressed tablets, which were placed in the sample bin of the infrared spectrometer for testing. The infrared spectrograms of the three sets of samples were split-peak fitted using PeakFit v4.12. In order to further quantitatively characterize the law of evolution of the internal micro-molecular structure of coal, it was calculated from the split-peak fitting data.
Results and discussion
Microbial community structure succession pattern during microbial degradation process
The changes in the methane concentration in the headspace gas inside the culture flasks during the 30-day experiments of CK, T-1, and T-2 are shown in Figure 2(a). The dynamic trend of methane concentration in CK, T-1 and T-2 was basically similar, showing a rapid increase in methane concentration at the beginning, a leveling off of the growth rate of methane concentration in the middle, and almost no growth or even a decreasing trend at the end of the period of migration. Among them, the methane concentration in the headspace gas of CK was always less than 1% during the experiment, and there was almost no methane in the headspace gas in the fourth period. In group T-1, the methane concentration in the headspace gas was always higher than that in CK. The peak methane concentration in the headspace gas was almost the same as that in the first three periods, which was 0.122 mL/g of coal, and there was still methane migration continuously in the fourth period. Coal contains many adsorbed and free methane in different pore structures (Li et al., 2022). In the T-2 group, free methane molecules were rapidly migrated from the coal at the initial stage of each period, and the methane concentration in the headspace increased rapidly, with a maximum peak value of 0.640 mL/g coal, which was 5.24 times higher than that of T-1. In addition, the methane migration in the T-2 period showed a stepwise downward trend in sequence, with a decrease of about 50% of that of the previous period. In the last period, the methane migration was significantly reduced, and its methane migration was reduced to 0.132 mL/g, and the growth rate of methane concentration tends to be gentle at 28 d.
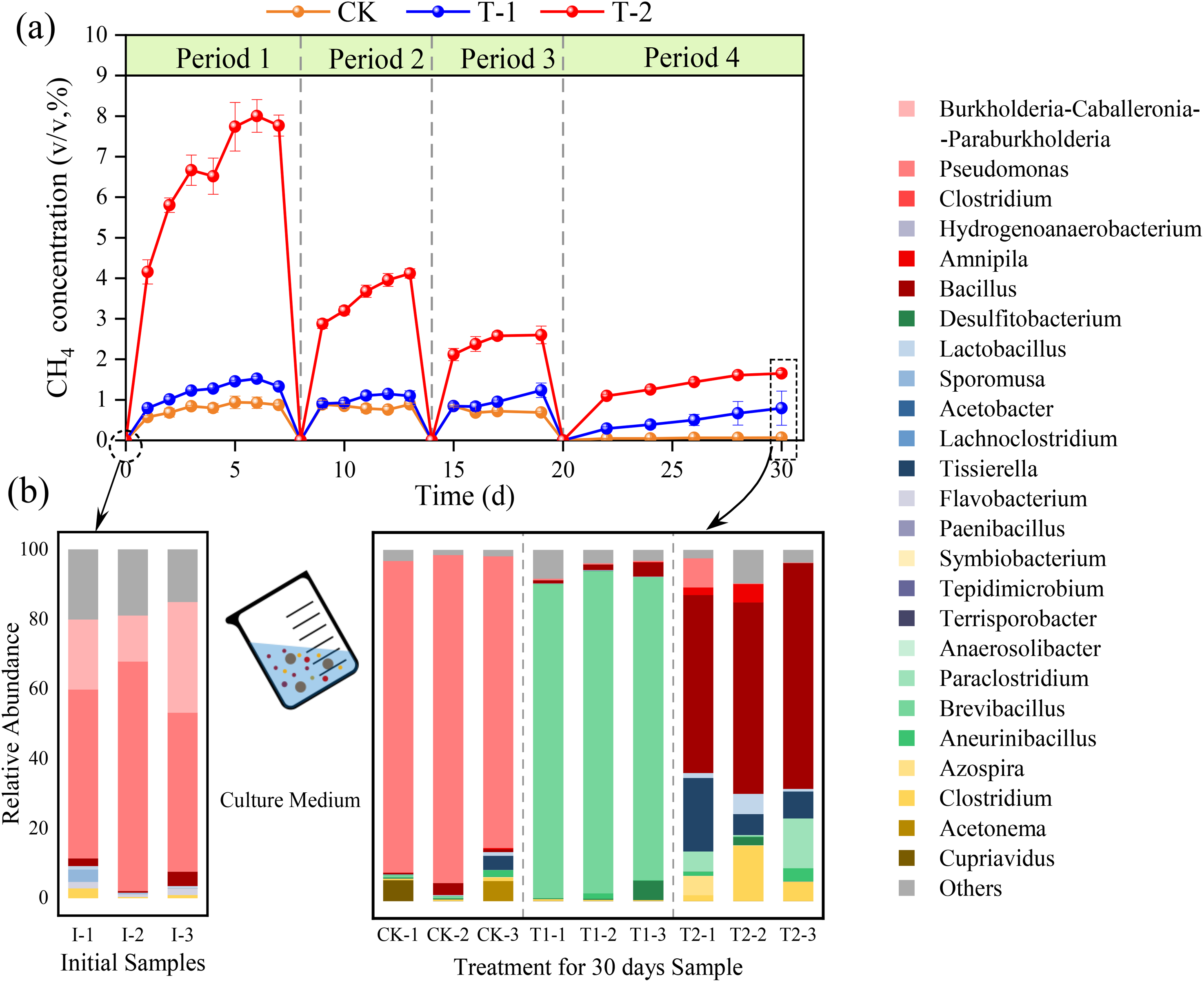
(a) Relative abundance of headspace microbial methane migration in the three experimental groups. (b) Relative abundance of microbial community composition in the initial samples on d 0 of the experiment versus the three experimental groups on d 30 of the experiment.
The dynamic trend of methane concentration in T-1 indicated that the microorganisms were able to enhance the further migration of residual methane from the coal to a certain extent under the environment of the inorganic salt medium. Still, their metabolic reactions were less active, and the effect of enhanced methane migration was weaker due to the scarcity of metabolic substrates at the initial stage. With the increase of time, the residual methane was gradually migrated from the coal, and the amount of methane migration was almost the same in different periods, which indicated that the advantageous flora to enhance the migration of residual methane evolved in the culture environment during the experimental process. The metabolism substrate was increased to improve the microbial degradation activity. The dynamic trend of methane concentration in T-2 indicated that after adding tryptone to the culture environment, it provided abundant nitrogen and amino acids for the metabolic activity of microorganisms, and its metabolic activity was obviously enhanced, and the effect of methane migration was significant. However, after a large amount of free methane is gradually migrated in larger pores, it is necessary to migration free and adsorbed methane which is difficult to desorption from small and medium-sized pores, so it is difficult to maintain the initial methane migration rate (Zhao et al., 2019).
The relative abundance of microbial community composition in the three culture environments is shown in Figure 2(b). Initial samples were taken at day 0 of the experiment, when the microbial community consisted mainly of the native microorganisms of coal, with Pseudomonas (53.17 ± 10.94%) the dominant genus of in situ microorganisms, followed by Burkholderia-Caballeronia-Paraburkholderia (21.63 ± 9.37%) and Bacillus (2.24 ± 1.81%) were more predominant. The microbial community composition within the culture system changed on the 30th day of the experiment. The original dominant genus Pseudomonas in CK in the aqueous environment was absolutely dominant with a relative abundance of 88.49 ± 5.18%. While Brevibacillus had the highest percentage of microbial community composition at the genus level in T-1, with a relative abundance of 89.26 ± 2.27%, Bacillus (56.53 ± 7.05%) was the dominant genus. T-2 had the most abundant microbial community composition due to the presence of more reaction substrates. Bacillus (56.53 ± 7.05%) had the largest percentage of relative abundance and Tissierella (11.53 ± 8.20%), Clostridium (7.59 ± 7.27%) and Paraclostridium (6.77 ± 6.97%) were the dominant genera.
The microbial community in the CK incubation environment did not change significantly due to the presence of water only, and Pseudomonas always dominated. Pseudomonas is thought to have a good solubilizing effect on low rank coal, and has the property of decomposing proteins, glucose, and acid production (Kang et al., 2021). In T-1 the microbial community changed and Brevibacillus became the dominant genus. Rasool Kamli et al. (2022) concluded that Brevibacillus has a wide range of enzymatic activities and can degrade a wide range of compounds. They are resistant to heavy metals and are effective strains for coal degradation. Thus, the increased proportion of Brevibacillus in the microbial community allowed the coal to be degraded to some extent, slightly facilitating the migration of residual methane within the coal. When a nitrogen source is added to the culture environment, the microbial community undergoes a succession in which the dominant genera Bacillus, Tissierella, Clostridium and Paraclostridium belong to the Firmicutes. Several articles have shown that these four genera are capable of degrading complex biomasses and compounds in anaerobic environments, providing metabolic substrates for other microorganisms through the decomposition of sugars or peptones to produce a mixture of organic acids and alcohols, as well as reducing the nucleation structure of the coal and enhancing the permeability and porosity of the coal (Du et al., 2020; Guo et al., 2019; Ikkert et al., 2013). Among them, Bacillus, as the most dominant genus, has been indicated to be involved in the degradation of organic matter in a variety of environments, e.g., phosphate degradation in contaminated soils, and increasing the degradation efficiency of baijiu lees by increasing cellulase activity (Meng et al., 2019; Yang et al., 2021). In addition, Bacillus exhibits active carbohydrate and amino acid metabolism, and is characterized not only by strong secretion of enzymes that stimulate the metabolic activity of other microorganisms, but also by the production of bacteriocins that inhibit pathogenic bacteria. The results of microbial community succession in T-2 showed four genera belonging to the Firmicutes, whose mutual stimulatory effects during metabolism enhanced the anaerobic degradation of the coal by microorganisms, which led to a significant increase in the migration of residual methane from coal.
Differential analysis of microbial degradation metabolic functions
Figure 3(a) demonstrates the differential analysis of major metabolic functions by KEGG level 3 for the three groups of experiments, and the effect of microbial degradation of coal may be related to functional genes for degradation of aromatic compounds, etc (Li et al., 2023). The screening of relevant metabolic pathways revealed that the metabolic activities of microorganisms enhanced the degradation of aromatic compounds and fatty acids in coal except nitrotoluene, and this metabolic degradation was more significant under the stimulation of nitrogen sources, especially in the degradation functions of xylene, styrene, and polycyclic aromatic hydrocarbons (PAHs). In addition, the methane metabolism function of the experimental group with the addition of BES showed a decreasing trend, indicating that BES inhibited the methane metabolism function of the bacteria, and the methane migrated in the experiments was derived from the coal residue, and no additional biomethane was synthesized.
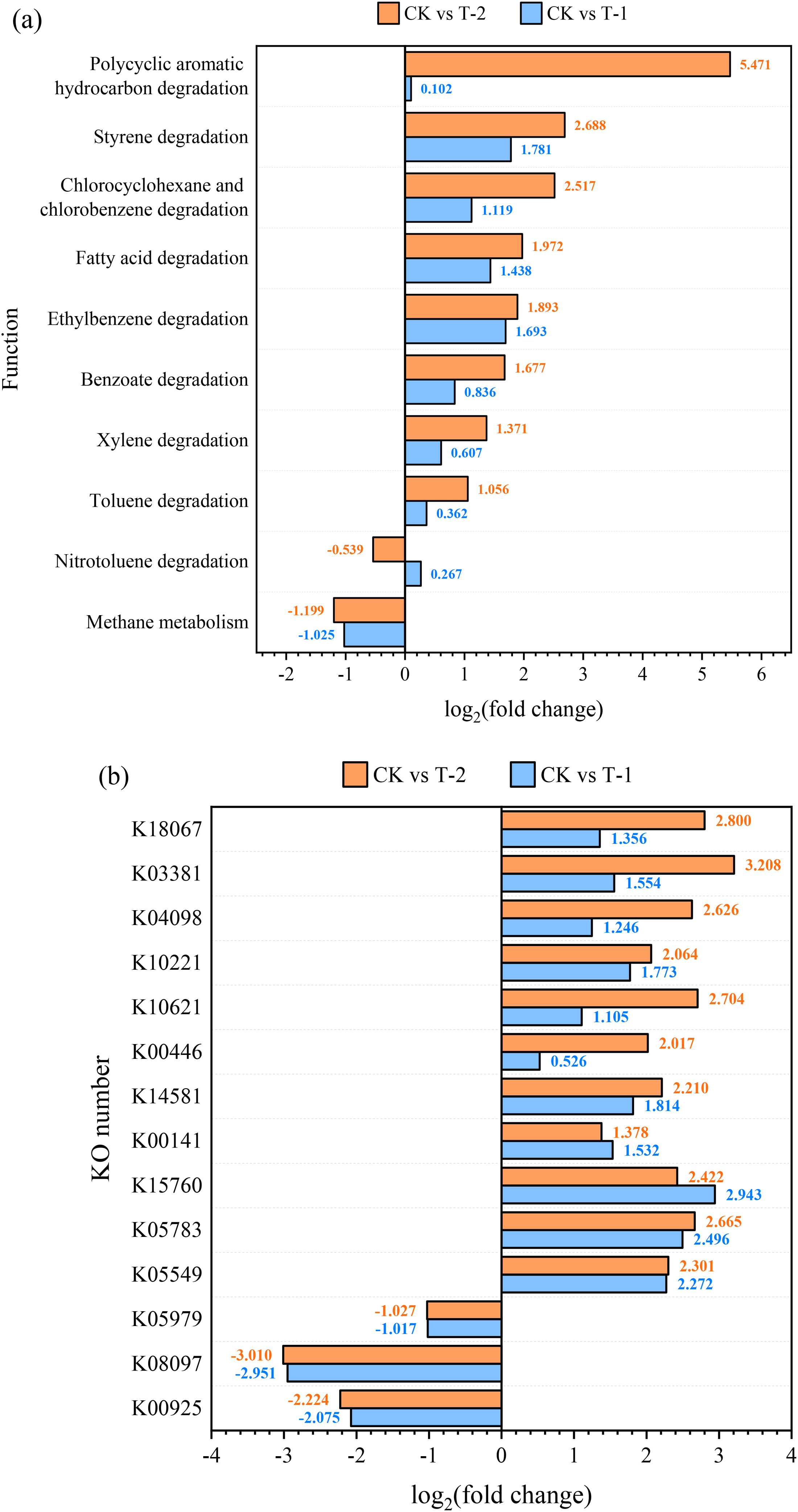
(a) Differential analysis of major metabolic functions in the three experimental groups. (b) Differential enzyme genes related to aromatic compound degradation and methane metabolism.
To further investigate the critical degradation metabolites during microbial metabolism, the differences in enzyme genes related to aromatic compound degradation and methane metabolism were analyzed in each experimental group by KEGG database as shown in Figure 3(b). In the methane metabolic pathway, acetate and coenzyme M are crucial intermediates for the synthesis of biomethane (Ferry, 1997; Wongnate et al., 2016). Acetate kinase (EC:2.7.2.1, K00925) catalyzes the reversible transfer of phosphate groups from acetylphosphate to ADP, produces acetate and ATP, and plays a central role in methane metabolism (Chittori et al., 2011). The hyperthermophilic euryarchaeon Methanococcus jannaschi is able to utilize coenzyme M as a terminal methyl carrier in methanogenesis, where phosphosulfolactate synthase (EC: 4.4.1.19, K08097), 2- phosphosulfolactate phosphatase (EC: 3.1.3.71, K05979) can catalyze coenzyme M biosynthesis sequentially (Graham et al., 2001; Graham et al., 2002). The changes in their relative abundance are consistent with Qiu et al. (2023) who showed that the addition of BES resulted in a decrease in acetate kinase activity and coenzyme M (CoM) content in the methane metabolic pathway. For degradation metabolic pathways of aromatic compounds, toluene monooxygenase system protein A (EC: 1.14.13.236, K15760) can introduce hydroxyl groups on the benzene ring in multiple degradation pathways of toluene (Takami et al., 2000); benzoate/toluate 1,2-dioxygenase subunit alpha (EC: 1.14.12.10, K05549) with dihydroxycyclohexadiene carboxylate dehydrogenase (EC: 1.3.1.25. K05783) promote the introduction of hydroxyl groups in the degradation metabolism of benzoate and xylene (Veith et al., 2004; Wrighton et al., 2008); While 2,3-dihydroxy-p-cumate/2,3-dihydroxybenzoate 3,4-dioxygenase (EC: 1.13.11.14, K10621), catechol 1,2-dioxygenase (EC: 1.13.11.1, K03381) and catechol 2,3-dioxygenase (EC: 1.13.11.2, K00446) were able to catalyze different ring-opening reactions upon the introduction of oxygen-containing functional groups, such as hydroxyl groups, respectively, and similar degradation of chlorobenzenes and styrenes was achieved by K03381 and K00446 (Benjamin et al., 1991; Marín et al., 2012; Paes et al., 2015); benzaldehyde dehydrogenase (EC: 1.2.1.28, K00141) catalyzes the synthesis of methyl benzoate from xylene and benzoates from toluene (Feng et al., 2007); naphthalene 1,2-dioxygenase ferredoxin reductase component (EC: 1.18.1.7, K14581) enhances the introduction of hydroxyl groups to PAHs and ethylbenzene (van Beilen and Funhoff, 2007). 2-pyrone-4,6-dicarboxylate lactonase (EC: 3.1.1.57, K10221) and hydroxyquinol 1,2-dioxygenase (EC: 1.13.11.37, K04098), on the other hand, can catalyze aromatic compounds in the metabolic pathway of benzoate degradation cyclization of aromatic compounds in the metabolic pathway of benzoate degradation (Kutsuna et al., 2019; Zhang et al., 2015). The increase in relative abundance of K05979, K05549, K05783, K15760, and K00141 over CK suggests that microbial metabolic functions stimulate the introduction of hydroxyl groups for a variety of aromatic compounds in coal, which enhances the hydrophilicity of coal. The increase in the relative abundance of K00446, K10621, K10221, K04098 and K03381 in T-1 and T-2 indicated that the metabolism of these genes catalyzed the de-cyclization of the coal macromolecule structure after the introduction of benzene rings into the aromatic compounds, which led to the evolution of the coal macromolecules into chains, which was conducive to the microbial pore-expansion and infiltration of the coal. In addition, the abundance of K05979, K05549, K05783, K15760, and K00141 within T-1 and T-2 were similar, while there was a significant difference between K00446, K10621, K10221, K04098, and K03381, confirming that additional nitrogen sources may not have a significant effect on promoting the introduction of oxygen-containing functional groups, such as hydroxyl, into the benzene ring. But the Firmicutes in the stimulated by nitrogen source was able to significantly catalyze the ring-opening reaction of part of the aromatic ring and enhanced biologically driven methane migration capabilities.
Coal pore structure development law under microbial action
To study the developmental characteristics of the full pore size pore structure of coal under microbial degradation, low-temperature liquid nitrogen adsorption test and mercury compression test were carried out on the treated coal samples of groups raw coal, T-1 and T-2, respectively. The pore size distributions of the three groups of coal samples are shown in Figure 4. In the range of 0–15 nm, the microporous pore size distribution of raw coal is more extensive, in which a more uniform regional distribution is presented near 1–2 nm and 7–15 nm, and bimodal pore distribution is presented out of 2.3–4 nm. The pores of the raw coal are most concentrated at 2.5 nm and 3.1 nm in particular. In the microporous range of T-1 coal samples, the distribution of pores in the range of 1–1.5 nm and 7.5 nm-15 nm is relatively average, while in the range of 1.5–7.5 nm is the distribution of many large-scale pore volume peaks, especially in the distribution of pore sizes at about 1.5 nm and 4.7 nm is the largest value. The pore distribution of the T-2 coal samples was similar to that of the residual coals of the T-1 group, with apprant peaks at 2.8 nm and 8.5 nm, where some micropores were concentrated. In the range of 15–50 nm, there was no significant difference between the coal samples before and after microbial action. In the range of 50–400 nm, the mesopore pores of coal showed a two-stage development trend. Firstly, in the 50–100 nm region, the pore size distribution pattern of the three groups of coal samples is similar, and all of them have a large number of pores distributed near 55–70 nm, in which the pore size distribution values of raw coal, T-1 and T-2 decrease in this place in turn. Secondly, in the 100–400 nm region, the raw coal pore distribution shows a decreasing trend, and from 180 nm onwards the mesopore pore distribution is tiny. In contrast the T-1 and T-2 pore size distribution patterns are similar and more evenly distributed. In the macroporous region larger than 400 nm, the pores of raw coal samples showed a three-stage distribution, mainly in the range around 550 nm, 840–1350 nm, and 2120–3122 nm. The pore size distribution of the T-1 coal sample shows an increasing and then decreasing trend, and the pores are more concentrated at the two ends of the macroporous region. The pore size distribution curve of T-2 coal sample is higher than that of the other two groups, especially after 1000 nm, the trend is significant, and the large pores of the coal sample appear to be greatly developed.
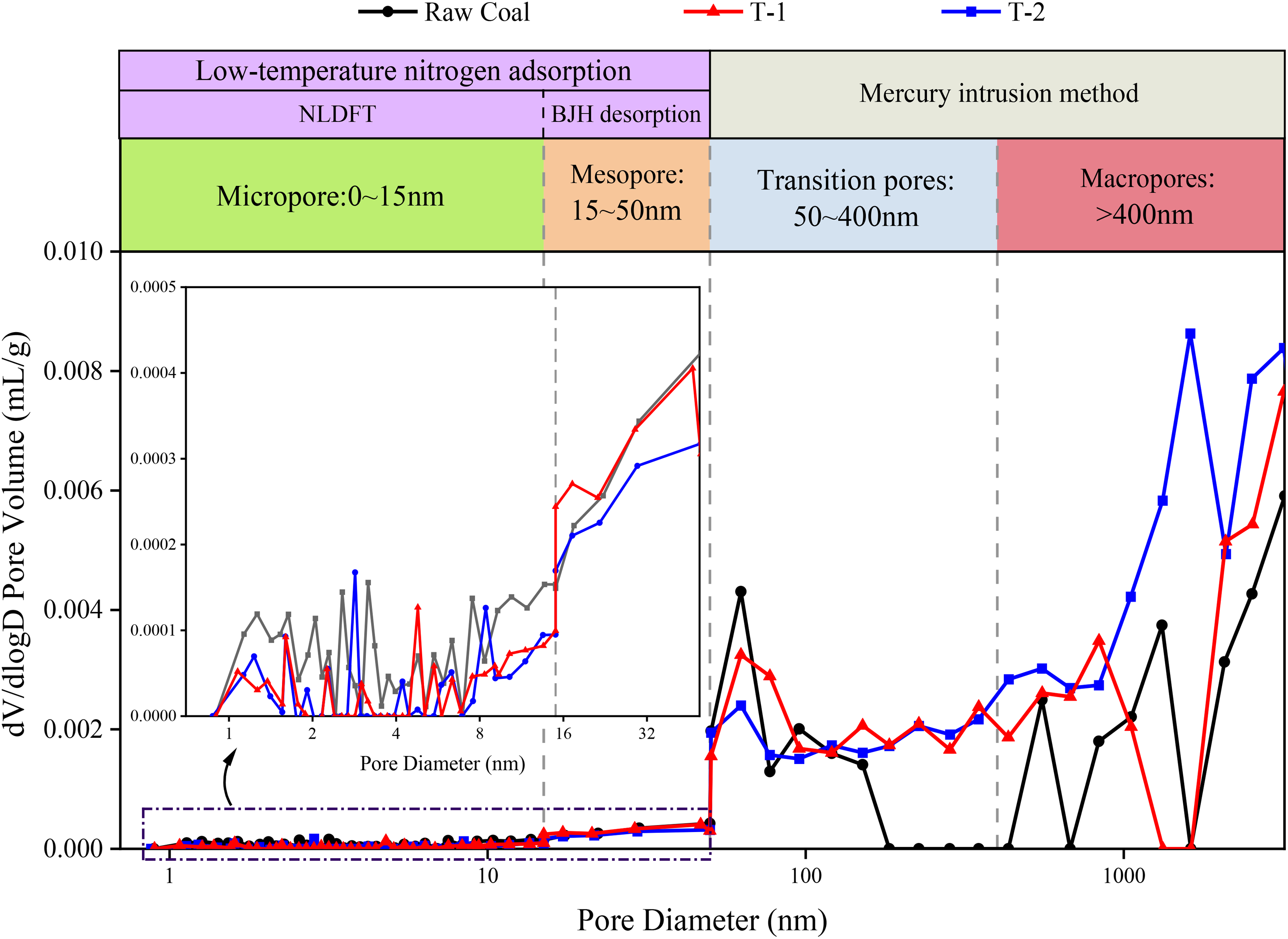
Multi-scale pore size distribution of raw coal, T-1 and T-2 coal samples.
Through the analysis of Tables 2 and 3, the developmental and evolutionary characteristics of the pore structure of anthracite under the degradation of functional microorganisms can be further explored. After coal degradation by microorganisms, the total pore volume of coal samples in both T-1 and T-2 groups increased compared to the total pore volume of raw coal, while the total specific surface area decreased and the average pore size increased significantly. Among them, the total pore volume of coal samples in T-1 and T-2 groups increased by 22.70% and 58.11%, respectively, compared with that of raw coal; the total specific surface area decreased by 44.11% and 49.61%, respectively, and the average pore diameters were 2.19 and 3.14 times higher than those of raw coal, respectively. In addition, the percentage of microporous pore volume and the percentage of specific surface area of T-1 coal sample decreased by 9.03% and 22.82%, respectively, compared with that of raw coal, while that of T-2 coal sample decreased by 9.33% and 22.58%, respectively. Since micropores are the main contributor to the specific surface area of coal and have less influence on the pore volume value of coal (Shan et al., 2015), microbial degradation has led to the development of a large number of microporous structures in coal. The proportion of micropores in the total pore structure of coal has decreased dramatically. There was no significant difference in the transitional pore of the three groups of experimental coal samples. T-1 and T-2 showed different developmental trends compared with the raw coal samples. The percentage of mesopores in the T-1 samples was increased by 5.27%. The percentage of mesopores in the T-2 samples was decreased by 15.28%. The percentage of mesopores in the T-2 samples is increased by 5.27% and decreased by 15.28%. For the macropore, the pore volume of T-1 and T-2 coal samples is 1.39 and 2.68 times of that of raw coal, and the percentage of pore volume increases by 5.08% and 27.31%, respectively.
Pore volume parameters of three groups of coal samples.

The pore specific surface area and average pore size parameters of three groups of coal samples.
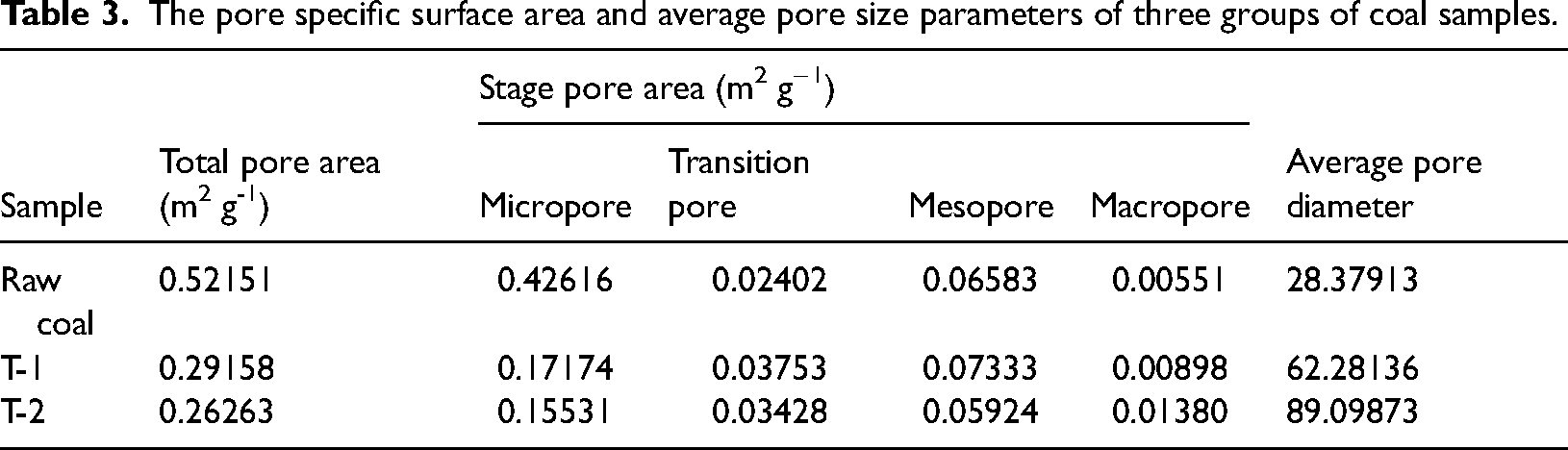
Through the joint characterization of the pore structure of the three groups of experimental coal samples, it was confirmed that microbial degradation and metabolism could promote the pore structure of anthracite coal to show a more obvious evolution pattern, which was more significant after the addition of nitrogen source. Among them, the microbial degradation of anthracite coal in the inorganic salt medium environment after its original micro-small pores to mesopore, macropore evolution and development, the average pore size increased, reducing the adsorption space of methane in coal, showing the effect of pore expansion and pore enlargement. This is consistent with the results of Guo's study (Guo et al., 2014). And although the pore structure of micropore and transitional pore had no obvious additional effect after the additional addition of nitrogen source, and the effect of mesopore development was weaker, the macropore presented a particularly significant growth. The proportion of the coal's whole pore diameter was greatly increased. This suggests that the nitrogen source further stimulated the anaerobic degradation of coal by Firmicutes and changed the evolution law of the pore structure of anthracite coal during microbial degradation. Compared with the developmental trend of micropore favoring mesopore in T-1 coal samples, T-2 micropore showed a large-scale transformation phenomenon to macropore structure, which led to a further decrease in the total specific surface area of the coal, and the effect of expanding and increasing pores was more significant. In addition, tiny pore structure is the main contributor to the adsorption of methane molecules by coal, which can provide more adsorption sites for methane, so the development of tiny pore structure will inevitably reduce the adsorption capacity of coal for methane and improve its transport characteristics (Hu et al., 2020; Li et al., 2015, 2020), which is important for the enhancement of the level of biologically driven gas migration. The coal macropore structure is regarded as the “valve that restricts the flow,” and the methane flow rate in the macropore controls the migration rate of the residual methane in the coal (Hu et al., 2023). The Firmicutes stimulated by the nitrogen source significantly increases the effective gas diffusion channels of the coal, and promoted the rapid migration of adsorbed and free methane.
Characterization of changes in the molecular structure of coal under microbial action
To study the evolution law of functional groups in low-permeability coals under microbial action, FTIR tests were carried out on three groups of experimental residual coals, respectively. According to a previous study (Ibarra et al., 1996), the infrared spectral map of coal is mainly divided into four parts, in which the range of 900–700 corresponds to aromatic hydrocarbons, the range of 1800–1000 corresponds to oxygen-containing functional groups, the range of 3000–2700 corresponds to aliphatic hydrocarbons as well as the range of 3600–3000 corresponds to hydroxyl groups as shown in Figure 5(a). To visually compare the differences in functional group content under microbial action, quantitative calculations were carried out by peak fitting and the fitted peaks of each group were R2 > 0.999, and the results obtained after peak fitting are shown in Figure 5(b).
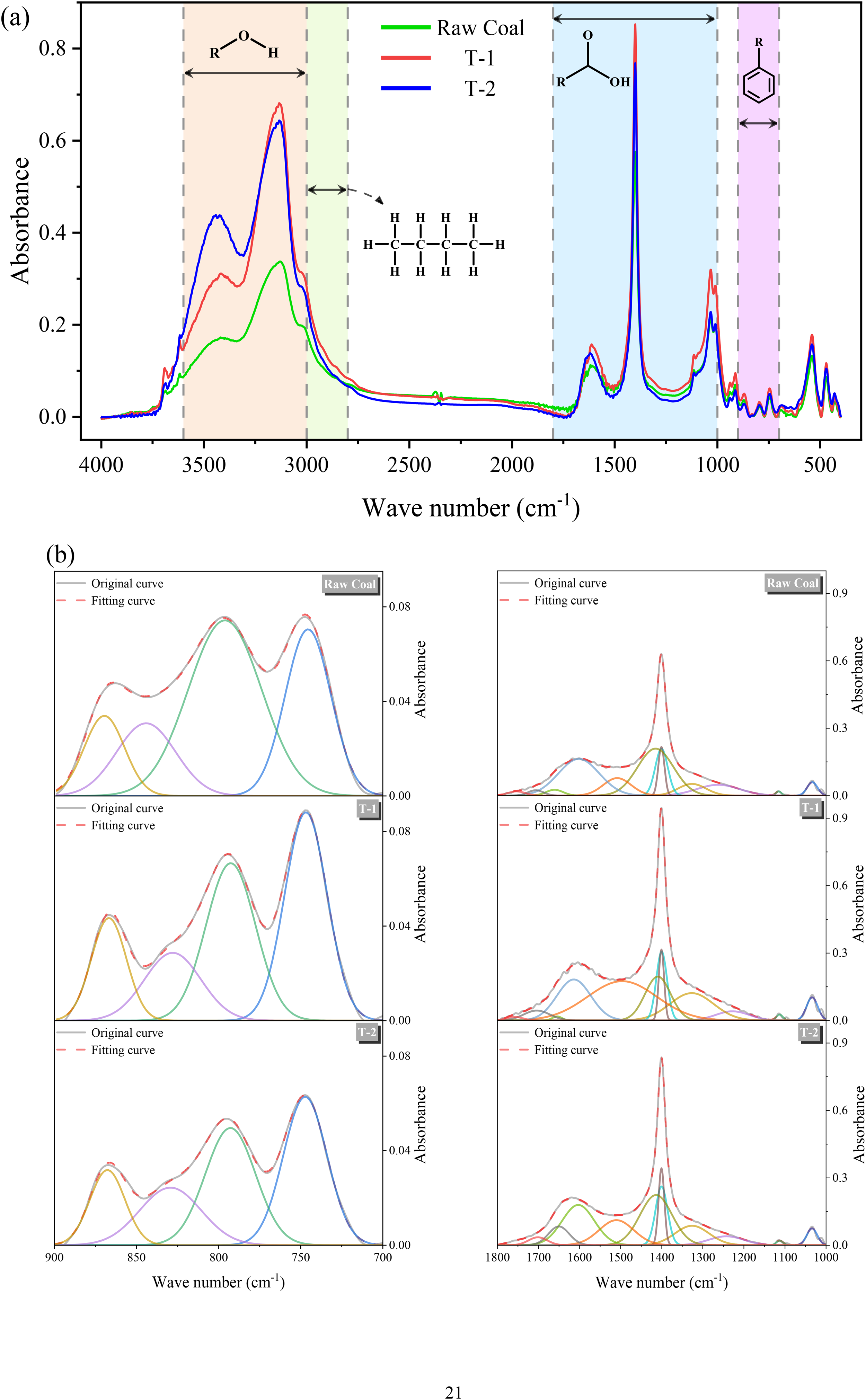
(a) Comparison of infrared spectra of raw coal, T-1 and T-2 coal samples. (b) Fitting plots of the main bands in each group of coal samples.
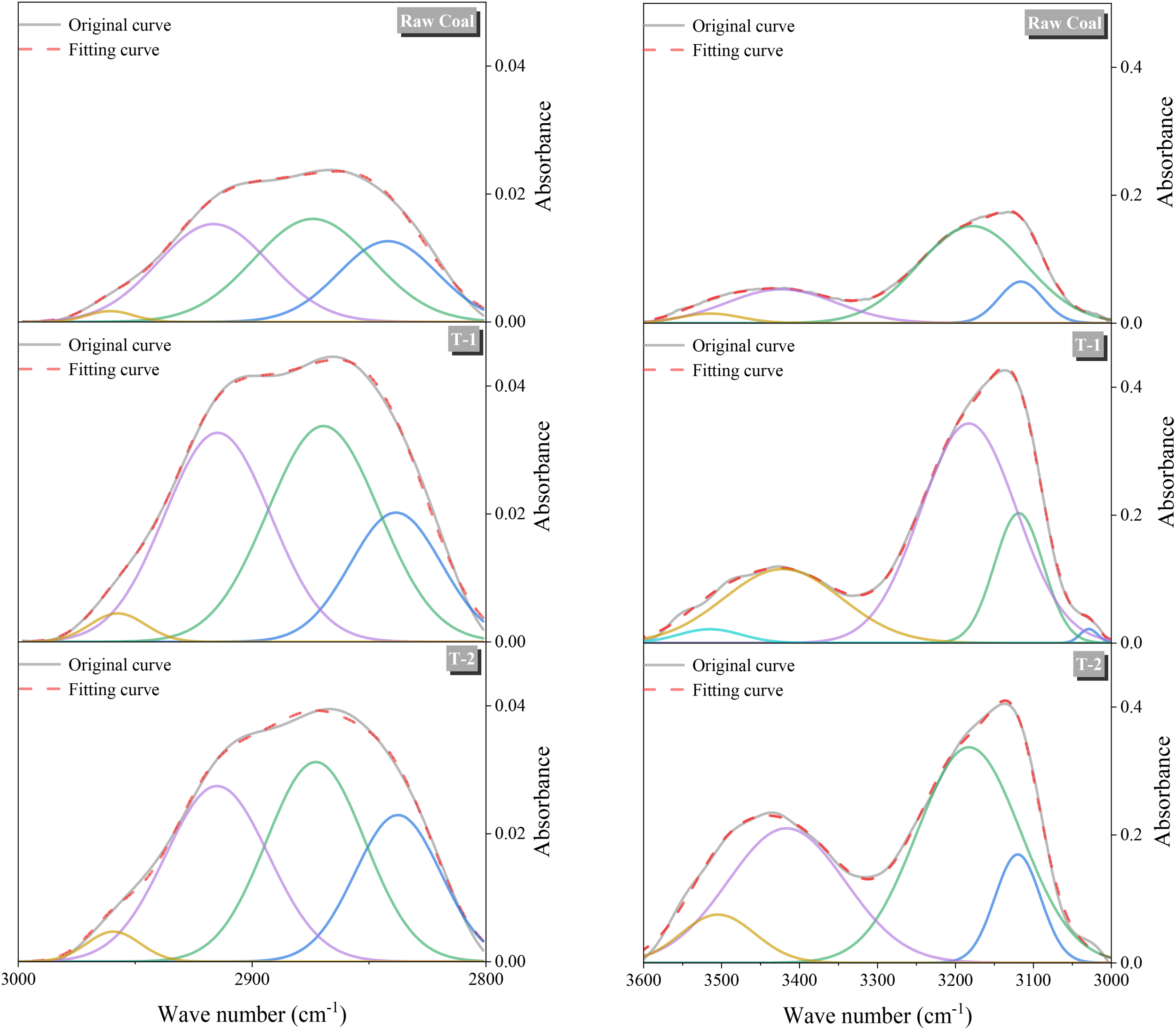
In the 3600–3000 cm−1 band, there are mainly three kinds of characteristic peaks corresponding to hydroxyl functional groups: hydroxyl-π, self-conjugated hydroxyl, and hydroxyl-N. The relative content of hydroxyl in T-2 coal samples is about 117.231, which is 2.822 times of the hydroxyl content in raw coal. The content of hydroxyl in T-1 coal samples is about 2.251 times of that in raw coal. Four groups of characteristic peaks corresponding to aliphatic hydrocarbon functional groups mainly existed in the range of 3000–2700 cm−1: symmetric stretching vibrations of aliphatic methyl and methylene groups and asymmetric stretching vibrations. According to the results of peak fitting, the content of aliphatic hydrocarbon functional groups on the surface of the coal samples showed an increasing trend of different degrees after microbial action. Compared with raw coal, the aliphatic hydrocarbon content of T-1 and T-2 was increased by 88.46% and 64.01%, respectively. In the 1800–1000 cm−1 band, the contents of oxygen-containing functional groups of T-1 and T-2 coal samples were higher than those of raw coal, which were 1.778 and 1.511 times higher than those of raw coal, respectively. And at 1600 cm−1 corresponding to aromatic hydrocarbons, the content of raw coal coal samples was the highest, and the aromatic hydrocarbons content of T-1 and T-2 decreased sequentially. In addition, carboxyl group is one of the key functional groups to enhance the hydrophilicity of coal and reduce the adsorption capacity of methane (Han et al., 2016). In the region representing the carboxyl functional group around 1700cm−1, although the carboxyl vibration of anthracite is generally weak, both T-1 and T-2 showed different degrees of growth after microbial metabolism. Within the 700–900 cm−1 region, four groups of characteristic peaks mainly existed in each group of coal samples: benzene ring five-substituted, benzene ring four-substituted, benzene ring three-substituted, and benzene ring two-substituted. Among them, the aromatic hydrocarbon content of raw coal was the highest at 9.053, and T-1 and T-2 decreased by 14.01% and 33.78%, respectively, compared with that of raw coal, which was also consistent with the changes occurred at the position of 1600 cm−1, and in addition, the percentage of benzene ring disubstitution in the coal samples of the treatment group showed an increasing trend compared with that of the original coal group (Li et al., 2023).
To further quantitatively characterize the evolutionary law of the internal microscopic molecular structure of coal, it was calculated by fitting the data with split peaks (Li et al., 2019). The calculation results are shown in Table 4.
FTIR quantitative parameterization.
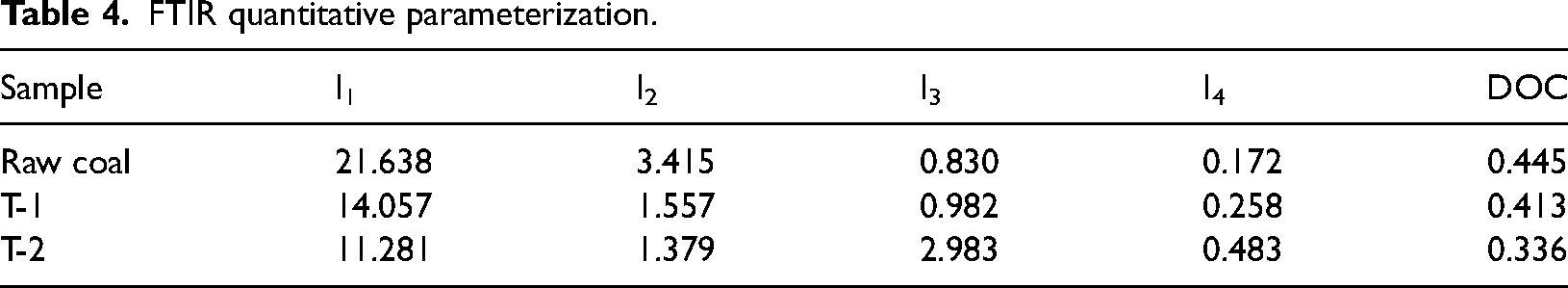
Among them, I1 represents the average chain length and degree of branching of aliphatic branched chains of the coal body, which decreased by 35.04% and 47.86% in T-1 and T-2 coal samples, respectively, compared with raw coal. The aromaticity of the coal body can be characterized by the ratio of the number of aromatic hydrocarbons to the number of aliphatic hydrocarbons, I2, which decreased by 54.41% and 59.62% in the T-1 and T-2 coal samples, respectively, compared with raw coal. I3 is the ratio of the number of hydroxyl and ether groups reflecting the interconversion of oxygen-containing functional groups within the coal, and the I3 of T-1 coal was 1.183 times that of raw coal, and the conversion of oxygen-containing functional groups prompted by T-2 due to the addition of a nitrogen source to make the microbial metabolism activity more intense was particularly significant, and its I3 was 3.594 times that of raw coal. Carbonyl is the main form of reactive oxygen present in the coal body (Xin et al., 2014), I4 is the ratio of carbonyl to aromatic hydrocarbons represents the degree of reactivity of elemental oxygen in the coal body, T-1 and T-2 coal samples I4 increased by 50% and 180.814%, respectively. The aromatic ring condensation DOC within the coals was characterized by the ratio of the out-of-plane deformation vibration of –CH in various substituted aromatic hydrocarbons to the stretching vibration of C=C in aromatic hydrocarbons, and raw coal had the highest aryl ring condensation, with a decrease in the DOC of T-1 and T-2 coals by 7.19% and 24.49%, respectively.
The results of peak fitting and quantitative calculations showed that a large number of ether groups were converted to hydroxyl groups during the microbial metabolism, which introduced various oxygen-containing functional groups, such as hydroxyl groups, into the coal macromolecular structure and thus enhanced the content of oxygens in the coal molecular structure. Because the hydroxyl group can form hydrogen bonds with hydrogen atoms and then form a water film on the coal pore surface, it can strengthen the hydrophilicity of the coal pore structure and weaken the adsorption capacity of methane (Zhou et al., 2018). The aromatic hydrocarbon functional group is an important component that constitutes the main part of coal (Xia and Zhang, 2017). In the process of microbial degradation of coal, the structure of the highly substituted benzene ring is changed to the low substituted form. Furthermore, the condensation of aromatic structures in coal decreases. The molecular structure of coal becomes more porous. Stable benzene ring structures gradually convert into loose chain-like formations, and the aromaticity of coal diminishes. At the same time, the continuous breaking of the coal's fatty chains leads to a decrease in chain length and an increase in branchedness, which can provide more metabolic sites for microorganisms (Guo et al., 2014). In addition, the above phenomenon of coal molecular structure evolution was more significant after the addition of nitrogen source, and the transformation effect within the coal oxygen-containing functional groups was significant, especially the transformation efficiency to the key oxygen-containing functional groups, such as hydroxyl group, could be increased by several times. This suggests that the nitrogen source stimulated the metabolic activity of Firmicutes to be more intense, releasing more key metabolites acting on the coal molecular structure, thus enabling the microorganisms to degrade coal more efficiently and promoting the evolution of coal molecular structure.
Conclusion
This study investigates the impact of microbial degradation metabolic functions on the microstructure of anthracite when functional microorganisms are introduced into the cultivation system under nitrogen source stimulation. The results showed that Firmicutes, mainly Bacillus, played a major role in the degradation process under the synergistic effect of coal as a biodegradable carbon source, and the additional nitrogen source. In this process, on the one hand, by shedding the alkyl side chain of coal molecular structure and introducing oxygen-containing functional groups such as hydroxyl group, the adsorption capacity of coal to methane is reduced. In this process, on the one hand, the microorganisms shed the alkyl side chain of the coal molecular structure and introduced oxygen-containing functional groups such as hydroxyl group, resulting in the reduction of the adsorption capacity of coal for methane. On the other hand, the metabolism of Firmicutes improves the degradation function of ring skeleton in coal, and the diffusion channel of methane in coal is increased; thus strengthened microbial-driven methane migration capacity. During anaerobic culture, Firmicutes, represented by Bacillus, dominated and tended to be stable in the culture group with the best methane migration. The relative abundance of genes for a variety of key degradation enzymes produced by their metabolism increased. They were able to promote the catalytic introduction of oxygen-containing functional groups, such as hydroxyls, on the structure of the benzene ring and ring-opening reaction of the coal macromolecule compounds; Secondly, the microbial metabolism reduced the aromaticity of coal, shortened the long chain of fat and increased the degree of branching. Additionally, this process elevates the content of oxygen-containing functional groups such as hydroxyl groups, thereby enhanced the hydrophilicity of coal, reduced the adsorption capacity of methane, and loosened the skeleton of coal macromolecules. As a result, the degradation by microorganisms led to the development of the coal's micropore structure towards mesopore and macropore. This change led to the improvement of the effective diffusion channels and methane transportation state within the coal, and ultimately led to the migration of methane in large quantities.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key R&D Program of China, Independent Research fund of Joint National-Local Engineering Research Centre for Safe and Precise Coal Mining (Anhui University of Science and Technology), Graduate Innovation Fund of Anhui University of Science and Technology, National Natural Science Foundation of China, The Key Research and Development Projects in Anhui Province (grant numbers 2023YFC3009002, EC2023006, 2022CX2020, 52274171, 2022l07020020).
