Abstract
In this work, the iron-modified Ni-Co composite catalyst was synthesized, and its catalytic effect on product distribution, behaviour and liquid characteristics was deeply studied. The result showed that the synthesized catalyst significantly improved the pyrolysis behaviour of cellulose, and the decomposition of cellulose occurred earlier. The highest yield of liquid product was 56.79wt%, which was obtained by adding the NCF-6 catalyst. The optimal ratio of catalyst in the sample was 5%, and the optimal pyrolysis temperature was 530°C. The characteristics of liquid product showed the introduction of the synthesized catalyst enhanced the decomposition of anhydrosugars and significantly promoted the generation of ketones in the liquid. The highest content of ketones in the liquid was 25.12%. Further analysis showed that the selectivity of 2(5H)-Furanone in the liquid component was strongest. These works provided an effective way to produce ketone-rich bio-oil from biomass.
Introduction
Energy is the necessary material basis for socio-economic development (Siddiqi et al., 2021, 2022). Fossil energy is still the main energy source in the world at present, but the rapid development of social economy has led to a sharp increase in the consumption of fossil energy, and the energy crisis has seriously threatened the normal production and life safety of human beings (Mishra et al., 2021; Siddiqi et al., 2020). More importantly, fossil fuels will produce SO2, NO2 and other gases during the combustion process, causing serious damage to the environment. Therefore, it is extremely urgent to find and develop a clean alternative energy source. Biomass has the characteristics of the wide source and renewable. The development and utilization of biomass can effectively alleviate the shortage of fossil energy and at the same time reduce the huge pressure on the environment caused by fossil fuels.
The conversion of biomass into bio-oil by pyrolysis technology is an important way to realize its high-value utilization. In the process of biomass pyrolysis, catalysts play an important role in improving the behaviour of biomass pyrolysis and increasing the selectivity of specific components in liquid products (Dai et al., 2020; Alayont et al., 2022). Researches have reported that metal catalysts have excellent catalytic effects (Lu et al., 2010; Park et al., 2005; Mora et al., 2007; Kuriyama et al., 2013; Chen et al., 2022). Among various metal catalysts, nickel and cobalt catalysts exhibit good catalytic performance in volatile reforming (Sun et al., 2021; Qiu et al., 2022; Huang et al., 2002; Yin et al., 2012). The catalysts for nickel-based and cobalt-based catalysts applied in biomass pyrolysis have been summarized in Table 1. For example, Tian et al. (2022) pointed out that nickel-based catalyst had good catalytic activity for hydrogenation and dehydrogenation reactions. The work of Iliopoulou et al. (2012) indicated that cobalt-based catalyst had good deoxidation ability. Moreover, Cho et al. (2017) noted that cobalt salt promoted the generation of H2 and CO during lignin pyrolysis. Based on the above studies, the synergistic effect between nickel and cobalt can produce unexpected effects on biomass pyrolysis. However, most of the current research focuses on the analysis of the catalytic effect of a single metal component, and there are few reports on the effect of the composite metal catalyst synthesized by cobalt and nickel on the pyrolysis behaviour of biomass.
Catalytic effect of nickel-based/cobalt-based catalyst on biomass pyrolysis.

In addition, some studies have shown that iron salts have excellent performance in volatile reforming reactions. For example, the work of Li et al. (2016) indicated that iron facilitated deoxygenation and hydrocarbon reactions during biomass pyrolysis. Liu et al. (2022) applied iron oxide and cerium oxide to the catalytic pyrolysis process of maize straw, and the results showed that the catalyst had high activity for converting L-glucose and acetic acid. Moreover, Yang et al. (2020) studied the fast pyrolysis of sawdust with the Fe-Ca catalyst and found that the catalyst significantly promoted the decomposition of lignin in biomass. Therefore, the introduction of iron into the synthesized Ni-Co catalyst could enhance the catalytic activity of the catalyst, but the catalytic effect of iron-modified Ni-Co composite catalysts on the distribution of biomass pyrolysis products, behaviour and liquid product characteristics is still unclear.
In this work, a series of nickel–cobalt binary catalysts and iron-modified nickel–cobalt composite catalysts were synthesized and applied to the pyrolysis process of cellulose, and the effects of pyrolysis temperature, catalyst ratio and iron loading on pyrolysis behaviour, product distribution and liquid product composition were analyzed. Furthermore, the formation pathways of ketones in bio-oils were explored in depth. These works provided meaningful ideas for improving the pyrolysis behaviour of biomass and preparing high-quality bio-oil.
Experimental
Raw materials
Microcrystalline cellulose (CE), Ferric nitrate (Fe(NO3)3·9H2O), nickel nitrate (Ni(NO3)2·6H2O) and cobalt nitrate (Co(NO3)2·6H2O) were obtained from China National Pharmaceutical Group Corporation. Before use, biomass sample was dried at 80°C for 9 h.
Preparation of iron-modified Ni-Co composite catalyst
The iron-modified Ni-Co composite catalyst was prepared by using high-temperature calcination method. The synthesis steps of the Co-Ni binary catalyst were as follows: a certain amount of metal salts were dissolved in 100 ml deionized water, and then fully stirred at 40°C for 5 h. Afterwards, the mixing solution was placed in a thermostatic oven at 90°C until the moisture was completely evaporated. The obtained solid sample was heated from room temperature to 900°C at a heating rate of 3°C/min and held at 900°C for 3 h. After cooling, the Ni-Co binary catalyst was obtained. The catalyst was mixed into the cellulose sample in mass fractions of 1%, 3%, 5%, 7% and 9%, which were marked as NC-1, NC-3, NC-5, NC-7 and NC-9, respectively. The iron-modified Ni-Co composite catalyst was prepared by the same method (Figure 1). The mass ratios of Ni(NO3)2·6H2O, Co(NO3)2·6H2O and Fe(NO3)3·9H2O were 5:5:1, 5:5:2, 5:5:3, 5:5:4 and 5:5:6, which was marked as NCF-1, NCF-2, NCF-3, NCF-4 and NCF-6, respectively. The X-ray diffraction (XRD-7000, Japan) was used to detect components of the catalyst.
The preparation of iron-modified Ni-Co composite catalyst.
Micro-scale catalytic pyrolysis experiment
To study the effect of the prepared catalyst on the pyrolysis behaviour of cellulose, the thermogravimetric analysis was performed by using a thermogravimetric analyzer (SHIMADZU, Japan). In each experiment, 15 mg of sample was evenly placed into a ceramic crucible, and then the temperature was raised from room temperature to 700°C. The heating rate of the sample was 20°C/min. Nitrogen was used as an inert atmosphere, and the flow rate was 50 ml/min. By analyzing the law of weight loss of the sample, the influence of the catalyst on the pyrolysis behaviour of cellulose was analyzed and summarized.
Lab-scale catalytic pyrolysis experiment
The catalytic pyrolysis experiment of cellulose was carried out in a lab-scaled fixed-bed reactor. About 10 g of sample was put into a push tray and then pushed to the middle of the reactor. The catalytic pyrolysis temperature was set at 430°C, 480°C, 530°C, 580°C and 630°C, respectively, and the flow rate of nitrogen was 100 ml/min. The generated liquid product was collected by using a liquid collector, and the solid residues were weighed after the experiment was completed. In this work, the mass loss of catalyst decomposition was neglected, so the mass of biochar was obtained by the following formula (1):


Liquid characteristics
The chemical compounds in the liquid product were detected by using GC-MS (Agilent, 7890, 5975N). The sample was heated from room temperature to 50°C for 3 min, then raised to 200°C for 15 min. The flow rate of helium was 1 ml/min, and the separation ratio was 1:10. The National Institute of Standards and Technology Library was used to define the component of liquid product.
Results and discussion
Catalyst characterization
Figure 2 showed the XRD patterns of the Co-Ni binary catalyst and iron-modified Ni-Co composite catalyst. The characteristic peaks of Co3O4, Fe2O3 and NiO were found by comparing with standard cards. The obtained results showed that the iron-modified Ni-Co composite catalyst had been successfully synthesized, and it mainly consisted of metal oxides.
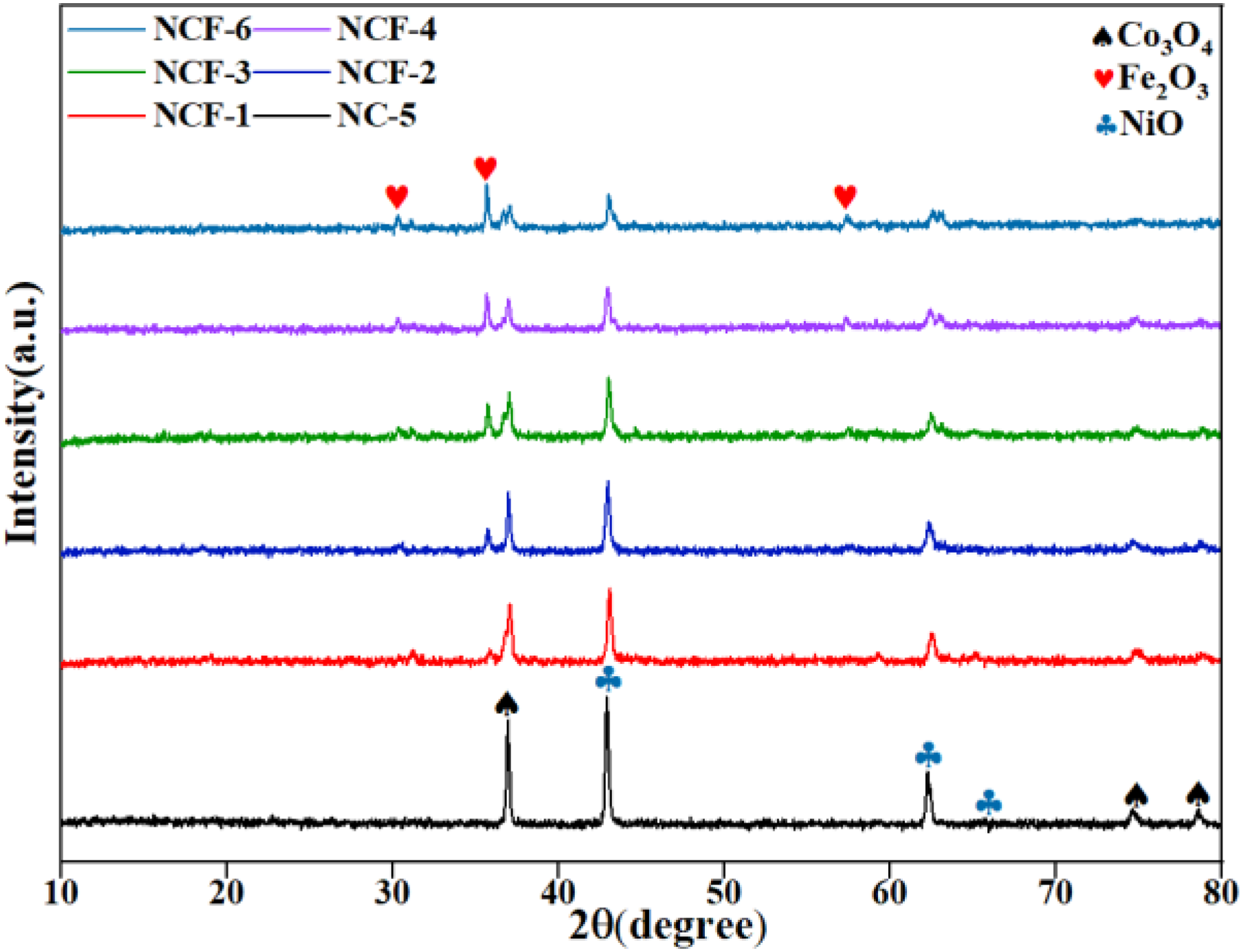
The XRD patterns of the prepared catalysts.
Effect of pyrolysis temperature on products distribution
The pyrolysis temperature not only changed the product yield but also significantly affected the catalytic effect of the catalyst. In this work, the mass fraction of nickel salt and cobalt salt in the catalyst was 5%, and the mass fraction of catalyst in the sample was also 5%. Figure 3A and B gave the yields of products obtained from catalytic pyrolysis of cellulose at different temperatures. As seen from Figure 3A, it was found that as the temperature increased, the syngas yield increased, and the solid yield decreased. The yield of syngas increased from 26.88wt% to 30.68wt%, and the yield of biochar decreased from 24.11wt% to 14.76wt%. The yield of liquid product showed an increase first and then a decrease. The maximum of liquid yield was 55.25wt%, which was obtained at 530°C. The results showed that the increase of pyrolysis temperature was beneficial to improve the pyrolysis efficiency of cellulose, and there was an optimum temperature for catalytic pyrolysis of cellulose.
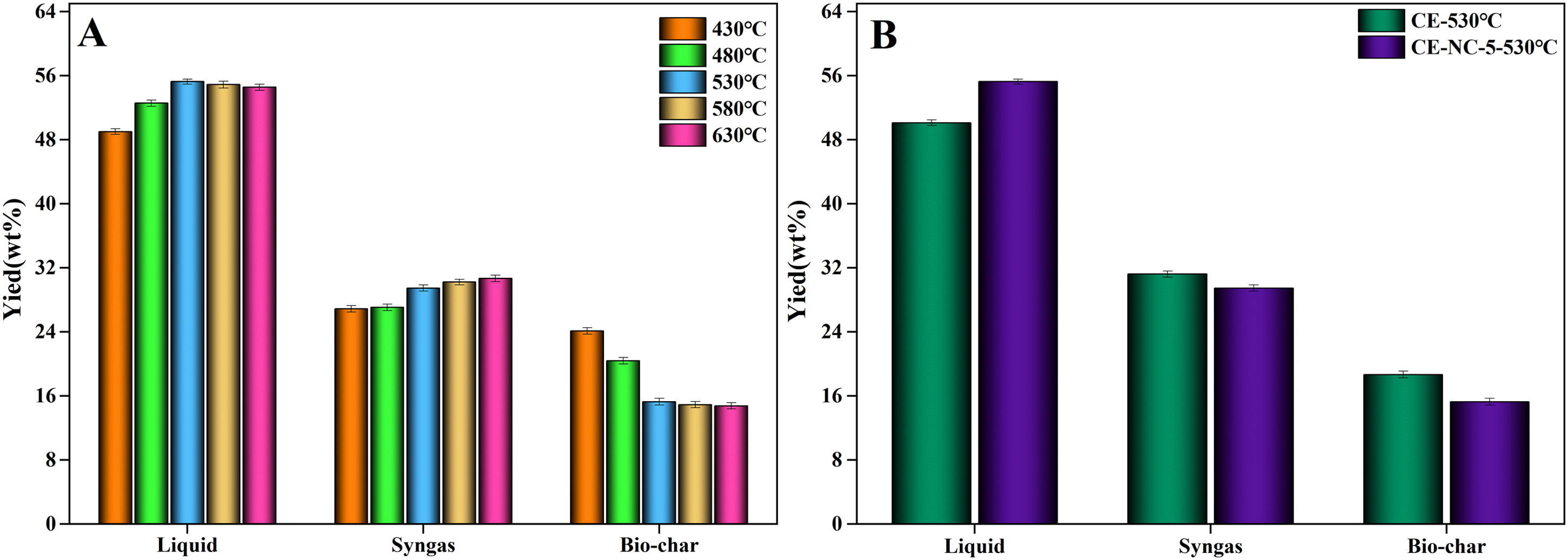
Products distribution obtained from pyrolysis of cellulose with the catalyst at different temperatures.
The product yields from cellulose pyrolysis were compared with/without the addition of catalyst to obtain the effect of the catalyst. As seen from Figure 3B, the liquid yield increased about 5.13wt%, and the biochar yield decreased about 3.38wt% after adding the catalyst. Thus, it could be concluded that the synthesized catalyst had a good catalytic cracking ability, which promoted the decomposition of cellulose to generate liquid product at a suitable temperature.
Effect of catalyst ratio on products distribution
According to the obtained results, the pyrolysis temperature of 530°C was selected to investigate the effect of catalyst ratio on product distribution, and the results were given in Figure 4. With the increase in the amount of catalyst added to the sample, the liquid production first increased and then stabilized. The maximum liquid yield was 55.25wt%, and the optimal proportion of catalyst in the sample was 5%. The work of Zheng et al. (2017) reported that the nickel-based catalyst had strong catalytic cracking activity, and Jiang et al. (2020) noted that cobalt-based catalyst was very active in the catalytic pyrolysis process of biomass and others, which was beneficial to promote the depolymerization of biomass macromolecule. Therefore, the cobalt and nickel could act as active centers to promote the cracking reactions during the cellulose pyrolysis, and the synergistic effect between cobalt and nickel promoted the generation of liquid production. In addition, the catalyst resulted in a decrease in terms of solid yield, but no increase in syngas yield. The yield of biochar decreased from 18.66wt% for the non-catalyzed pyrolysis to 15.28wt% after adding NC-5 catalyst. All in all, the optimum proportion of catalyst in the sample was 5%.
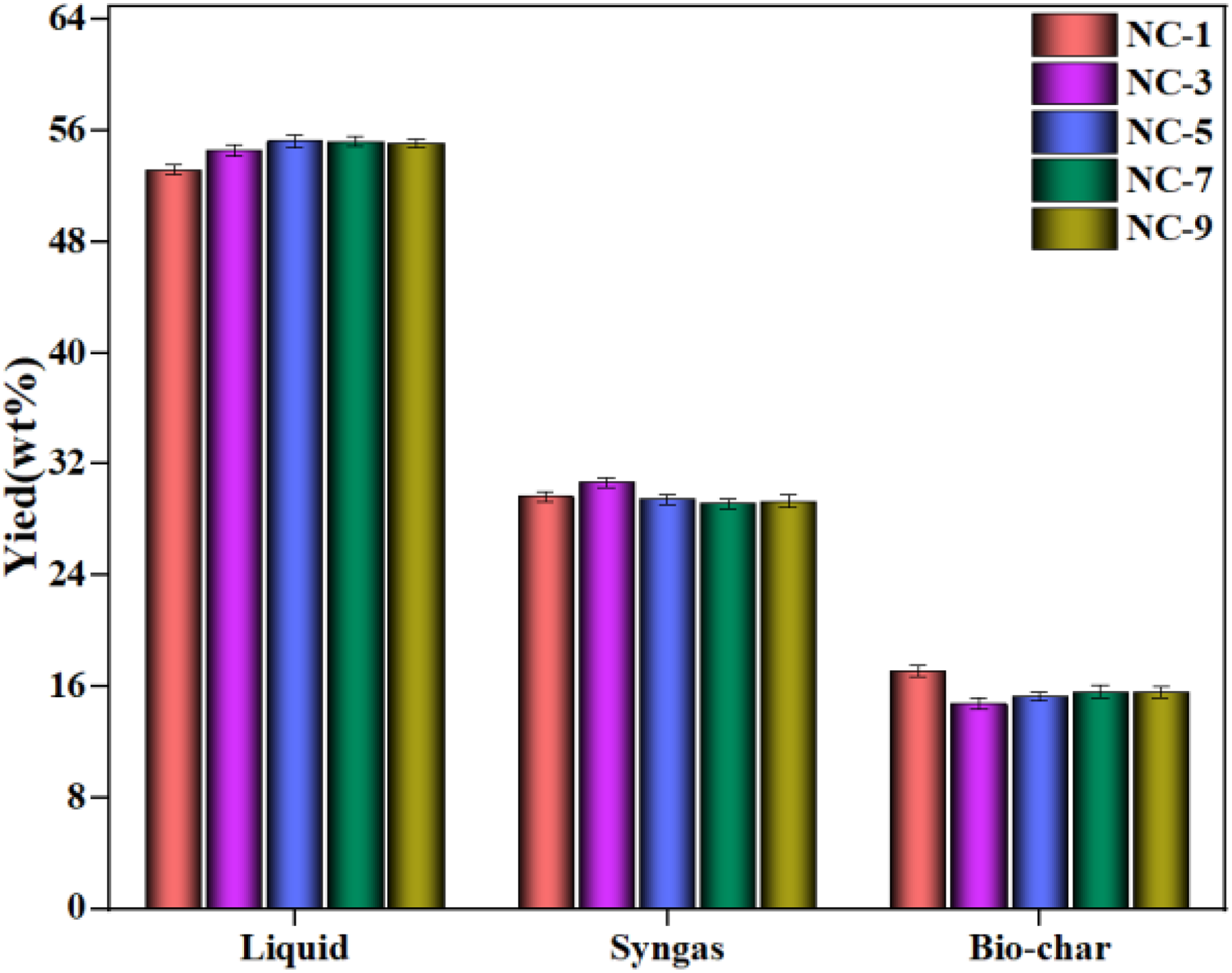
Products distribution derived from catalytic pyrolysis at different mixing ratios.
Effect of iron modification on products distribution
After determining the relevant characteristics of the NC-5 catalyst, iron was added to the catalyst to investigate the enhancement effect of iron on the catalytic performance. The amount of iron salt was 1%–6%, and the obtained product yields from the catalytic pyrolysis of CE with the NCF composite catalyst were given in Figure 5. It was observed that the introduction of iron further increased the yield of liquid product. Compared with that of the NC-5 catalyst, the liquid yield increased about 1.54wt%. As mentioned above, the role of the NC-5 composite catalyst was mainly reflected in the inhibition of the formation of solid products. Moreover, it could be found that the presence of iron mainly promoted the conversion between the generated syngas and liquid. Under the action of the iron component, the secondary reaction of syngas components produced by cellulose pyrolysis was enhanced, which in turn promoted the repolymerization of the gaseous components, leading to the increase of more liquid yield (Lu et al., 2022; Hong et al., 2022; Shao et al., 2020). The obtained results indicated that the reforming reactions of volatile matters were significantly improved by the introduction of the iron-modified NC-5 composite catalyst.
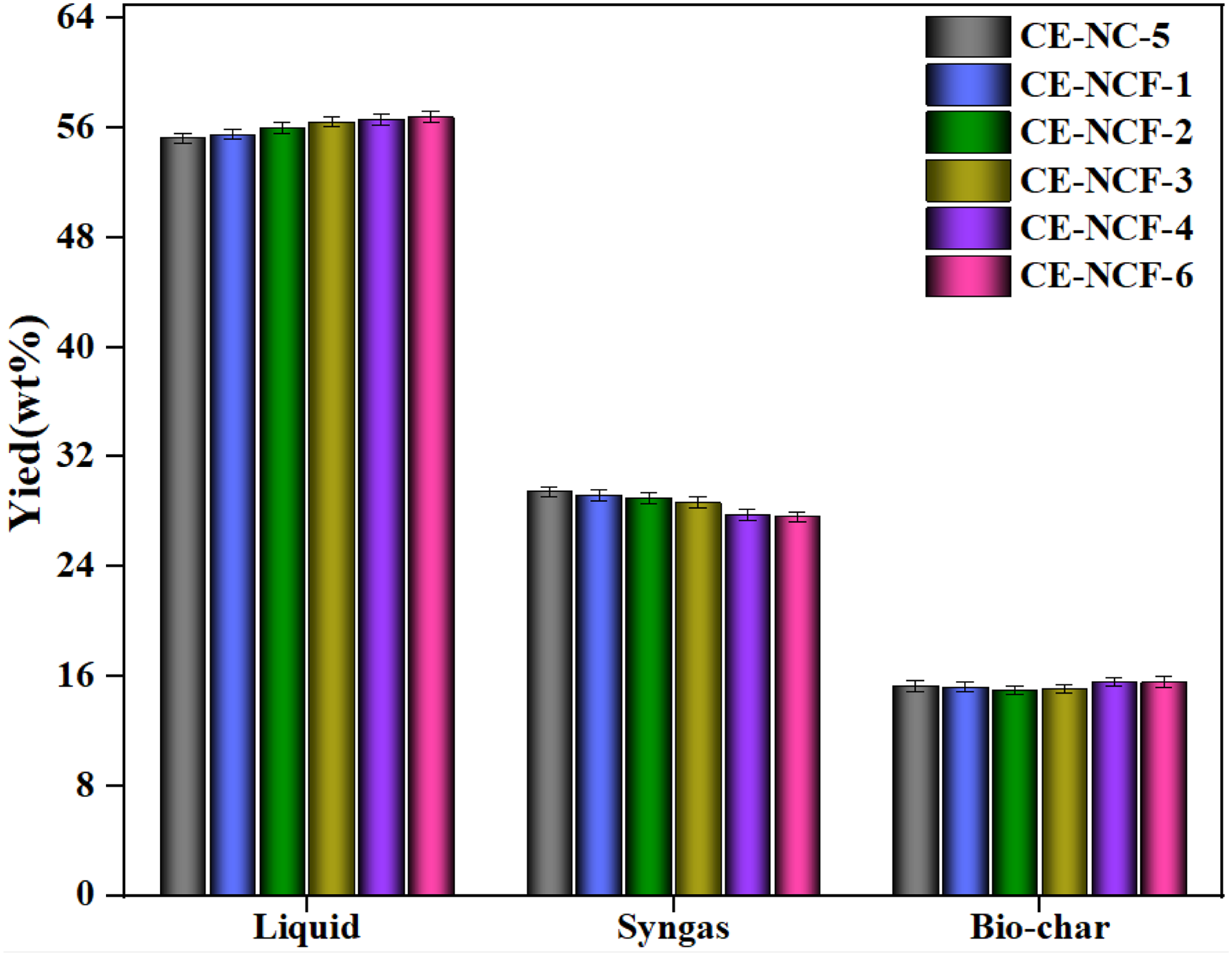
Products distribution catalyzed by the iron-modified Ni-Co composite catalyst.
Catalytic pyrolysis behaviour of cellulose
To further reveal the catalytic effect of the synthesized catalyst, the catalytic pyrolysis behaviour of cellulose was analyzed. In addition, the pyrolysis characteristics of cellulose with/without catalysts were compared, which was given in Figure 6. For the pyrolysis of cellulose, a significant mass loss occurred at a temperature range of 280–380°C, and the temperature corresponding to the maximum weight loss (Tmax) was about 355°C. After adding the NC-5 catalyst, the value of Tmax had a decrease. The obtained results indicated that the prepared catalyst had positive effects on the pyrolysis of cellulose. In addition, the iron-modified Ni-Co composite catalyst further improved the pyrolysis behaviour of cellulose compared with that catalyzed by the Ni-Co catalyst. It was found that the initial temperature was lower than that for the non-catalyzed pyrolysis of cellulose, and the Tmax value was the smallest among others.
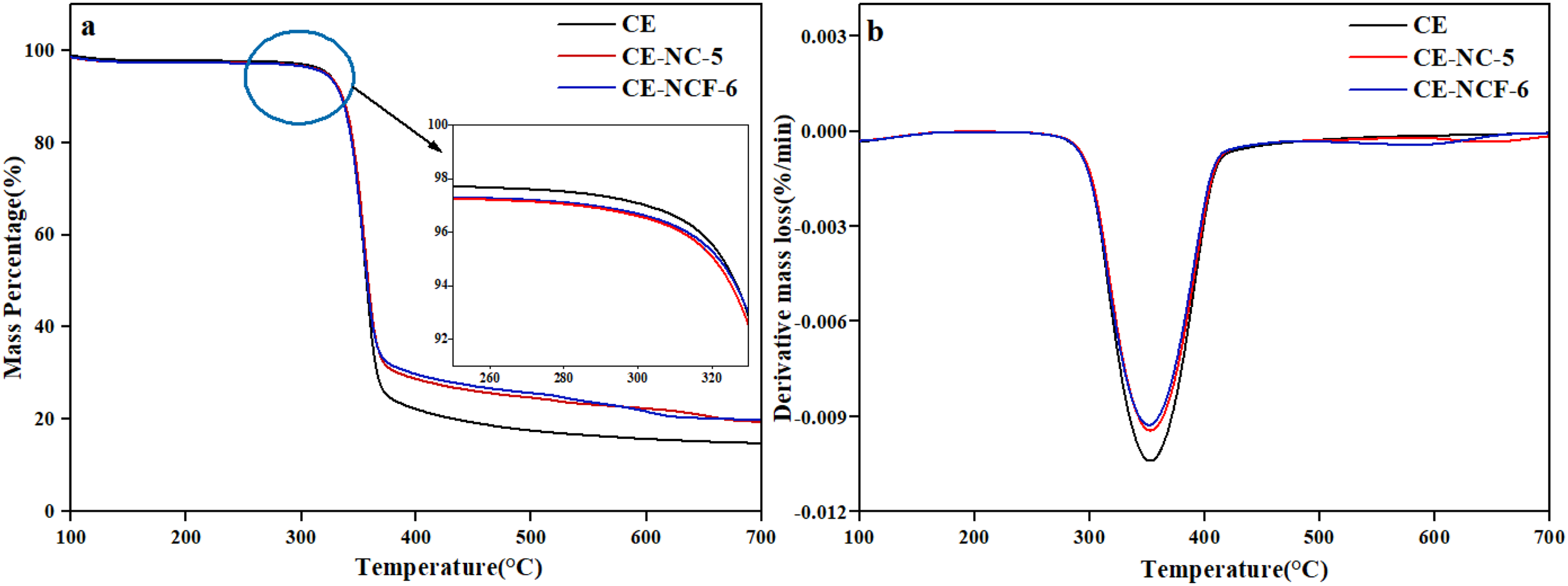
The TG and DTG curves of cellulose pyrolysis with/without catalyst.
Evolution of ketones obtained by catalytic pyrolysis
A lot of compounds were detected in the bio-oil, such as phenols, alcohols, anhydrosugars, ketones and others. This work focused on the change of ketone content in the bio-oil under different catalysts. The relative contents (peak area,%) of chemical compounds in the liquid products derived from catalytic pyrolysis of cellulose were given in Figure 7. For the pyrolysis alone of cellulose, there were a large number of oxygenated compounds in the bio-oil. It was found that the relative content of anhydrosugars was the highest in the selected compounds, and it was about 33.63%. After adding the NC-5 catalyst, the relative content of anhydrosugars was significantly reduced, which decreased about 14.27%. Some work (Banyasz et al., 2001a, 2001b) reported that anhydrosugars and their derivatives (ADs) were mainly derived from the decomposition of cellulose, and the generated ADs could be further composed to form ketones and other oxygenated compounds. It was observed that the relative content of ketones was the highest among these compounds, and the relative content of ketones was 20.74% obtained by the catalytic pyrolysis of cellulose. These results indicated that the presence of the catalyst facilitated the decomposition of anhydrosugars and their derivatives and the generation of ketones.
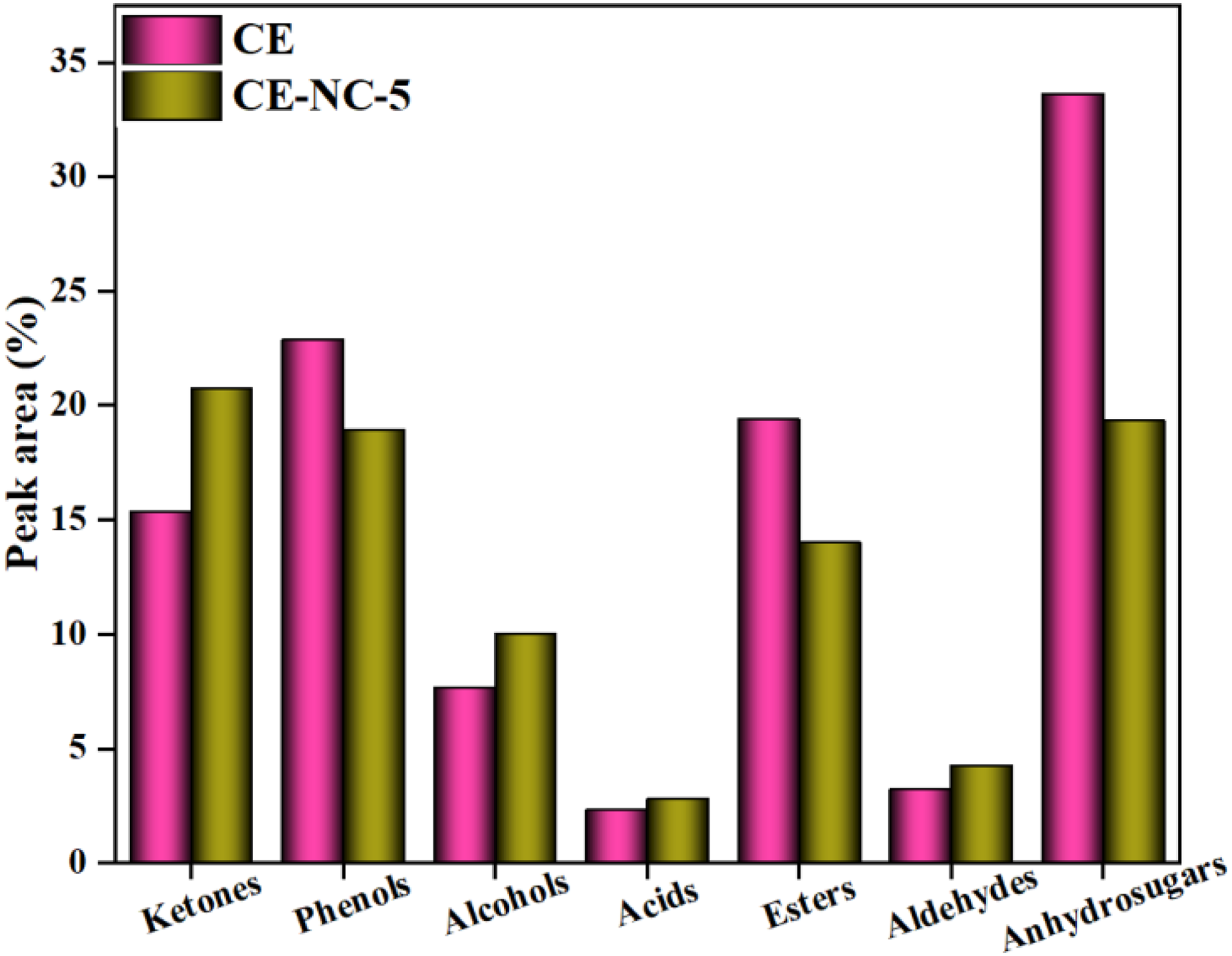
Relative contents of compounds in the bio-oils.
The effect of iron modification catalyst on the relative content of ketones in the bio-oil was investigated, and the results were given in Figure 8. It was found that the content of anhydrosugars was significantly decreased, reduced from 19.36% for the CE to 11.02% for the NCF-5 catalyst. But, the relative content of ketones increased from 20.74% to 25.12%. Compared with that of the NC-5 catalyst, the ketones in the bio-oil increased about 4.38%. These results indicated that the iron modification catalyst significantly promoted the decomposition of cellulose and further promoted the formation of ketones. As mentioned above, dehydration reactions and cracking reactions of ADs were the main reasons for generating ketones. Thus, the introduction of the iron component further enhanced the occurrence of the above reactions and then promoted the decomposition of ADs to generate ketones. In addition, the work of Jiang et al. (2022) indicated that oxygen species on the catalyst surface participated in the ketylation of compounds during biomass pyrolysis. Based on the XRD results, the prepared iron-modified Ni-Co composite catalyst was mainly composed of metal oxides. It could be conjectured that the catalyst had rich oxygen vacancy on the surface of the catalyst, and oxygen species could combine with intermediates derived from the cracking of organic polymers (Yang et al., 2023). These reactions promoted the formation of ketones in the bio-oil.
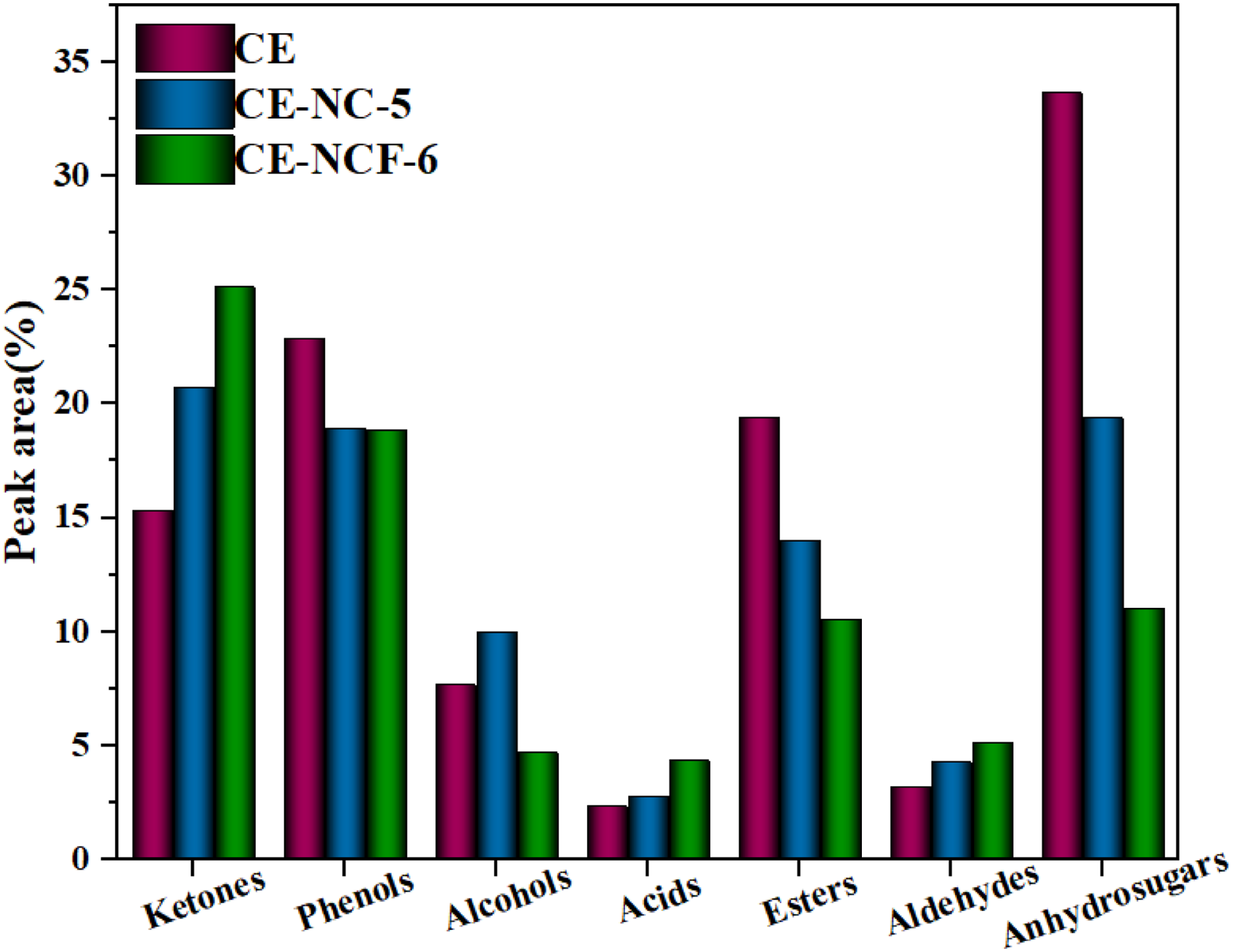
Compounds in the obtained bio-oils from pyrolysis of cellulose with catalyst.
Selectivity of ketone compounds in the bio-oil
Figure 9 gave the relative contents of ketones in bio-oils obtained from the pyrolysis cellulose with different catalysts. For pyrolysis alone of cellulose, the compound with the highest content in the bio-oil was 3-methyl-1,2-cyclopentanedione, followed by 3-ethyl-2-hydroxy-2-cyclopenten-1-one. After adding the NC-5 catalyst, 2(5H)-Furanone showed the strongest selectivity in the ketone compounds. Furthermore, the addition of the NCF-5 catalyst further promoted the generation of 2(5H)-Furanone. Compared with that obtained by the NC-5 catalyst, the relative content of 2(5H)-Furanone increased about 3.46%. The results indicated that the iron-modified Ni-Co composite catalyst was conducive to the enrichment of ketones in bio-oil production. Ketones were important chemical raw materials, and it had a wide range of applications in the fields of energy, chemical industry and biomedicine (Li et al., 2014; Yoritate et al., 2017; Zhang et al., 2017). Therefore, this work provided an efficient method for obtaining ketones from biomass.
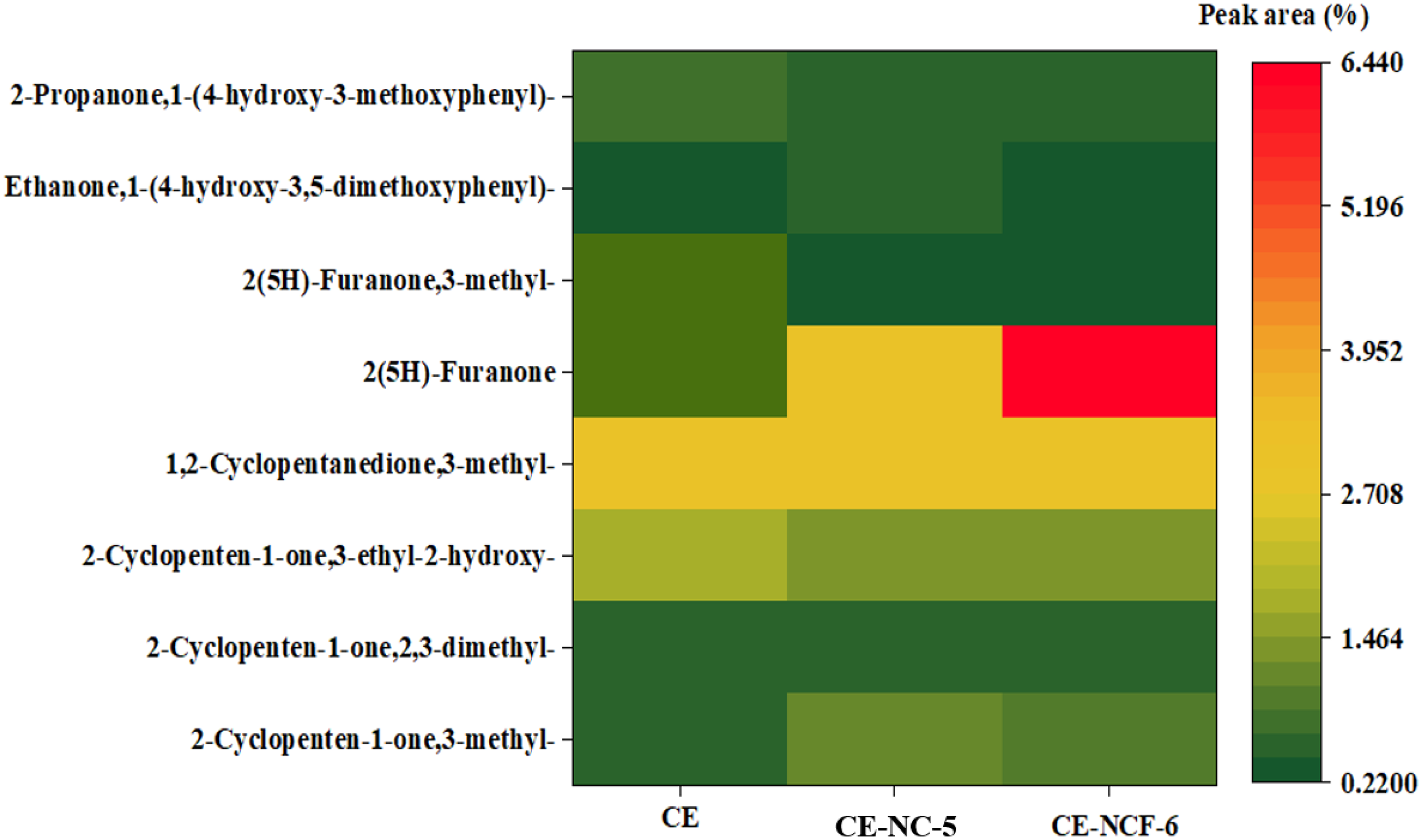
The selectivity of ketone compounds in the bio-oils.
Catalytic pyrolysis mechanism of cellulose with iron-modified Ni-Co composite catalyst
A brief reaction mechanism based on the obtained experimental results was given. During the pyrolysis alone, cellulose was decomposed into ADs and other organic macromolecular intermediates by dehydration, thermo-cracking, decarboxylation and other reactions (Leng et al., 2022), which was further decomposed to form oxygenated compounds such as phenols, alcohols, esters and ketones. In the absence of catalysts, a few of ketones were generated in the bio-oil production. After adding the Ni-Co catalyst, the ability of cellulose macromolecules to ketones was improved and the relative content of ketones in the liquid was increased. The Ni and Co had good catalytic activity, which was conducive to promote the decomposition of ADs to ketones by ring-opening reaction and enhance the conversion of other oxygenates to ketones through the synergistic effect between Ni and Co. Specifically, the synergistic effect was conducive to the formation of oxygen vacancy, which enhanced a series of reactions such as dehydration, cyclization and rearrangement reaction to form ketones (Stefanidis et al., 2016). The loading of iron further enhanced the above reactions, which led to the increase in the yield of ketones. Therefore, iron-modified Ni-Co composite catalyst had a superior catalytic performance for producing ketones.
Conclusions
In this work, the iron-modified Ni-Co composite catalyst was synthesized and applied to the cellulose pyrolysis process. The effects of pyrolysis temperature, catalyst ratio and iron ratio on product distribution, behaviour and liquid characteristic were analyzed. The selectivity of ketones in the bio-oil obtained from catalytic pyrolysis was highlighted. The highest yield of bio-oil was 56.79%. The maximum relative content of ketones was 25.12%. In addition, the iron-modified Ni-Co catalyst was conducive to the formation of 2(5H)-Furanone. All in all, synergistic effects among Ni, Co and Fe effectively improved the pyrolysis behaviour of cellulose and promoted the generation of ketone compounds, and this work provided an effective method for the enrichment of ketones in bio-oil and the high-value utilization of biomass.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
