Abstract
Phase change materials (PCM) possess the issues of leakage, low thermal conductivity, lack of rigidity and poor photo thermal conversion ability. To overcome these challenges, we have successfully designed and prepared flexible PW/PU/CuNWs/MXene (PPCuM) composite phase change materials with high thermal conductivity by solution blending and vacuum impregnation by using polyurethane (PU), one-dimensional copper nanowires (CuNWs) as high thermal conductivity, and two-dimensional transition metal carbide/nitride (MXene) as fillers. Furthermore, PU based PCM is developed by adding MXene at different weight fractions and the influence of MXene content on PPCuM properties was studied. As expected, benefiting from the high aspect ratio and strong capillary force of the MXene as well as the interfacial interaction between the PCM molecule and MXene, the resulting PPCuM exhibit a large thermal conductivity (0.705 W·m−1·K−1), which is 332.5% higher than that of pure PW, high latent heat (155.42 J·g−1), mechanical strength reaches 81.83 KPa, and are form-stable upon heating up to 75°C. The solar simulator and thermal conductivity demonstrated a superior photo-thermal conversion capability and thermal networks for the developed PU based composite sample at 83.3 wt% of MXene. At the same time, MXene effectively improved the photo-thermal conversion capacity (91.35%) and heat transfer rate (2.91 J·s−1) of PPCuM, which was further confirmed by the surface temperature distribution results. In addition, PPCuM has excellent photo-thermal conversion efficiency, good chemical compatibility, crystallization properties, and acceptable thermal stability.
Keywords
Introduction
The shortage of fossil energy sources and the emission of greenhouse gases is increasingly becoming a global issue, which can be resolved through the development of renewable energy sources and energy conservation systems.1,2 Thermal energy and solar energy are used as alternatives to fossil fuel energy owing to their advantages of being renewable and inexhaustible sources. In recent years, the use of PCM to absorb and release heat has attracted much attention owing to their large latent heat and fixed phase change temperature, which result in enhance energy utilization and efficiency.3,4 Up to now, it has been shown that solid-liquid organic PCM possess a constant phase transformation temperature range, large energy storage density, low corrosion, and chemical stability. 5 It has been widely used in the fields of solar energy utilization, 6 built environment, 7 wearable electronic devices 8 and battery thermal management. 9 Nevertheless, most organic solid-liquid PCM have problems such as easy leakage of phase change, poor light adsorption capacity and strong rigidity, which make it difficult to achieve good contact in limited thermal energy management devices. 10
To overcome these challenges, extensive researches have been focused on impregnating the PCM into porous materials.11,12 In this regard, many researchers have studied the shape-stable PCM with energy storage ability and photo-thermal conversion by using graphene aerogel, 13 graphite foam, 14 carbon nanotube sponge, 15 and porous carbon 16 as photo-thermal conversion materials and supporting scaffolds. Compared with the above carbon materials, MXene is a two-dimensional transition metal carbon/nitride with superior thermal conductivity and energy conversion capabilities on the surface. Furthermore, MXene has a high specific surface area and numerous oxygen-containing groups, which facilitate its adsorption of PCM. Recently, PCM composites modified by MXene have also been developed, and good encapsulation as well as solar-to-thermal energy storage properties were achieved.17,18 For instance, Gong et al 19 Successfully prepared polyurethane/MXene-based SSPCM with enhanced thermal stability and photo-thermal conversion capability. It was inferred that MXene at a weight fraction of 2% delivered a melting enthalpy of 128 J·g−1 and the photo-thermal conversion efficiency of 2.0-MPH is as high as 90.45%. Furthermore, the performances and structures of 2.0-MPH have basically unchanged after 200 times thermal cycling and 50 times light cycling, the excellent comprehensive mechanical properties also broaden the scope of application. Wang et al 20 reported that MXene has an excellent light-to-heat conversation ability (∼100%) because of its superior light absorption capacity. Jin et al 21 fabricated a poly (vinyl alcohol) (PVA)/MXene film via multilayered casting, in which the thermal conductivity of the PVA/MXene filris almost 23 times than that of PVA due to the presence of the continuous MXene layer. Idumah 22 prepared MXene (M-X) polymeric nanocomposites (M-X@PNC) and performed in depth morphological characterization of M-X@PNC and evaluated their overall effectiveness at improving the nanocomposites properties, vis-a-viz mechanical, heat dissipation as well as heat deflection properties along with potential applications. Kalidasan et al 23 developed a low-temperature MXene@SSD/SPDD composite phase change material with phase transition temperature of 27.6°C and melting enthalpy of 216 J·g−1. The synthesized MXene nanocomposites showed better chemical stability, high absorbance (1.1) and reliable thermal conductivity (0.621 W·m−1·K−1). Nevertheless, because of lower mechanical properties, these PCM composites exhibited strong rigidity and easy frangibility. This would result in difficult installation of PCM composites due to the insufficient surface contact with devices, which greatly restricts their practical applications. 18 At present, according to the concept of integration of structure and function, the selection of flexible PU skeleton is of great significance for the preparation of PCM composite materials with shape memory properties. The modified of polymers with MXene is an effective method to obtain composite materials with high porosity and excellent elasticity.24,25 Therefore, the design and development of flexible PU composite PCM with excellent comprehensive properties such as solar energy storage and shape stability has become an urgent problem to be solved.
In this study, inspired by the structural features and comprehensive properties of PU/MXene composite materials, MXene dispersions were prepared by in situ hydrofluoric acid etching method, and then treated with MXene by solvent exchange method 26 to obtain CuNWs/MXene concentrated solutions dispersed in isopropanol, and paraffin wax (PW) was encapsulated into PU/CuNWs/MXene (PCuM) three-dimensional flexible composite matrix materials prepared by solution blending, so as to prepare PCM composites with flexibility and photothermal conversion capabilities. Flexible composite PCM with different amounts of MXene were prepared by controlling the amount of MXene added, and the changes in mechanical properties and thermal properties of the composites were studied. Therefore, the obtained PPCuM has high porosity, large storage capacity and good resilience. The nanosheets of MXene loaded on the PPCuM enable the PCM composites with desirable solar-thermal conversion capability as well as encapsulation property. More importantly, the PPCuM exhibit satisfactory shape memory functions and self-healing properties by integrating the phase change of PW and the elasticity of PCuM. These composites have a broad application prospect in solar-to-thermal energy utilization.
Experimental methods
Materials
PW (pathological grade) with a melting point of 55°C, NH4Cl (PT grade >99.7%), CuCl (AR grade 97%), and oleylamine (90%) were provided by China Aladdin Reagent (Shanghai) Co., Ltd. PU with a void density of 50PPI (98% porosity) from Yuanshengxing Electronic Materials Factory, Suzhou, China. Tri (hydroxymethyl) aminomethane (biotech grade 99.9%), dopamine hydrochloride (98%), LiF (AR grade 99%), Ti3AlC2 (98%, 200 mesh), and glycerol (AR grade 99%) were purchased from McLean Company (Shanghai, China). Acetone and anhydrous ethanol (AR grade) were purchased from China Tianjin Yongsheng Fine Chemical Co., Ltd. Ethylene glycol (AR grade), HCl (AR grade 37%), H2SO4 (∼98%), NaNO3 (AR grade), KMnO4, and H2O2 (AR grade 30%) were provided by Sinopharm Chemical Reagent Co., Ltd. N-hexane (>95%) and isopropyl alcohol (>99.7%) were purchased from Tianjin Guangfu Technology Co., Ltd.
Pretreatment of PU
Commercial PU was repeatedly ultrasonic washed with acetone, anhydrous ethanol, and deionized water three times to remove the oil on the surface. Then, it was immersed in a mixed solution of tri (hydroxymethyl) aminomethane (0.446 g), dopamine hydrochloride (0.258 g), and deionized water (100 mL) for 24 h.
Synthesis of CuNWs
CuNWs were synthesized using the salt-assisted polyol method. Measure 100 mL ethylene glycol solvent, add 0.054 g of NH4Cl and 0.6 g of CuCl in turn, stirred for 20 min to form a light green solution; slowly add 8.4 mL of oleylamine to the solution, stir vigorously with a magnetic agitator until the solution turns dark blue, and pour it into a 200 mL reaction kettle and heated it at 180°C for 10 h. After heating, the solution in the reactor was poured into an Isopropanol (IPA) mixture (n-hexane: isopropanol = 3:1) in the centrifugal pipe for washing and centrifuging. The solution was discharged, and only CuNWs in the centrifugal pipe were retained. Repeated the wash with the IPA mixture three times, and the CuNWs were dispersed into isopropyl alcohol.
Synthesis of MXene
The Ti3C2TX was obtained by etching the Al layer in Ti3AlC2 (MAX phase) with LiF/HCl mixed solution. 27 Firstly, 6.00 g LiF was dissolved in a preconfigured 120 mL HCl solution, then 6.00 g Ti3AlC2 powder was slowly added to the above mixed solution and stirred in a water bath at 35°C for 36 h. Next, the suspension was repeatedly washed with deionized water by centrifugation at a speed of 10000 r·min−1 for 10 min until the pH was about 7. Then, the pellet was collected in the centrifuge tube, deionized water was added, the exfoliation of the multilayer Ti3C2TX nanosheets were facilitated with the assistance of sonication. Finally, the above mixture was centrifuged at 3500 r·min−1 for 10 min to obtain Ti3C2TX colloid. The colloidal concentration of Ti3C2TX could be calculated by the mass of Ti3C2TX solid obtained by freeze-drying at −45°C for 48 h.
Preparation of PPCuM
PPCuM was prepared by a simple physical blending method, as shown in Figure 1 First, 100 mg of CuNWs and MXene with different mass fractions (x = 50 wt%, 66.6 wt%, 75 wt%, 80 wt% and 83.3 wt%) were added to 20 mL of isopropanol and stirred for 10 min at room temperature. Then, the above mixture is sonicated for 60 min. The pretreated PU was immersed in the mixed solution for 30 min by a simple cyclic impregnation method and repeatedly extruded at the same time to ensure that the PU was in full contact with the mixed solution, and then vacuum dried at 85°C for 2 h, and repeated impregnation and drying for 3 times to ensure that the composite solution was evenly introduced into the PU frame, and the PU frame composite material (PCuM) wrapped by MXene/CuNWs was obtained, named PCuMX. Finally, the PW was placed on the PCuM composite material and placed in an oven at −0.1 MPa at 75°C for 1 h, and the liquid PW would continue to penetrate the pore structure of the PU under negative pressure conditions. Finally, the sample was polished with sandpaper to remove excess PW on the surface of the composite, and the PPCuM composite phase change material was obtained. PPCuM preparation flow chart.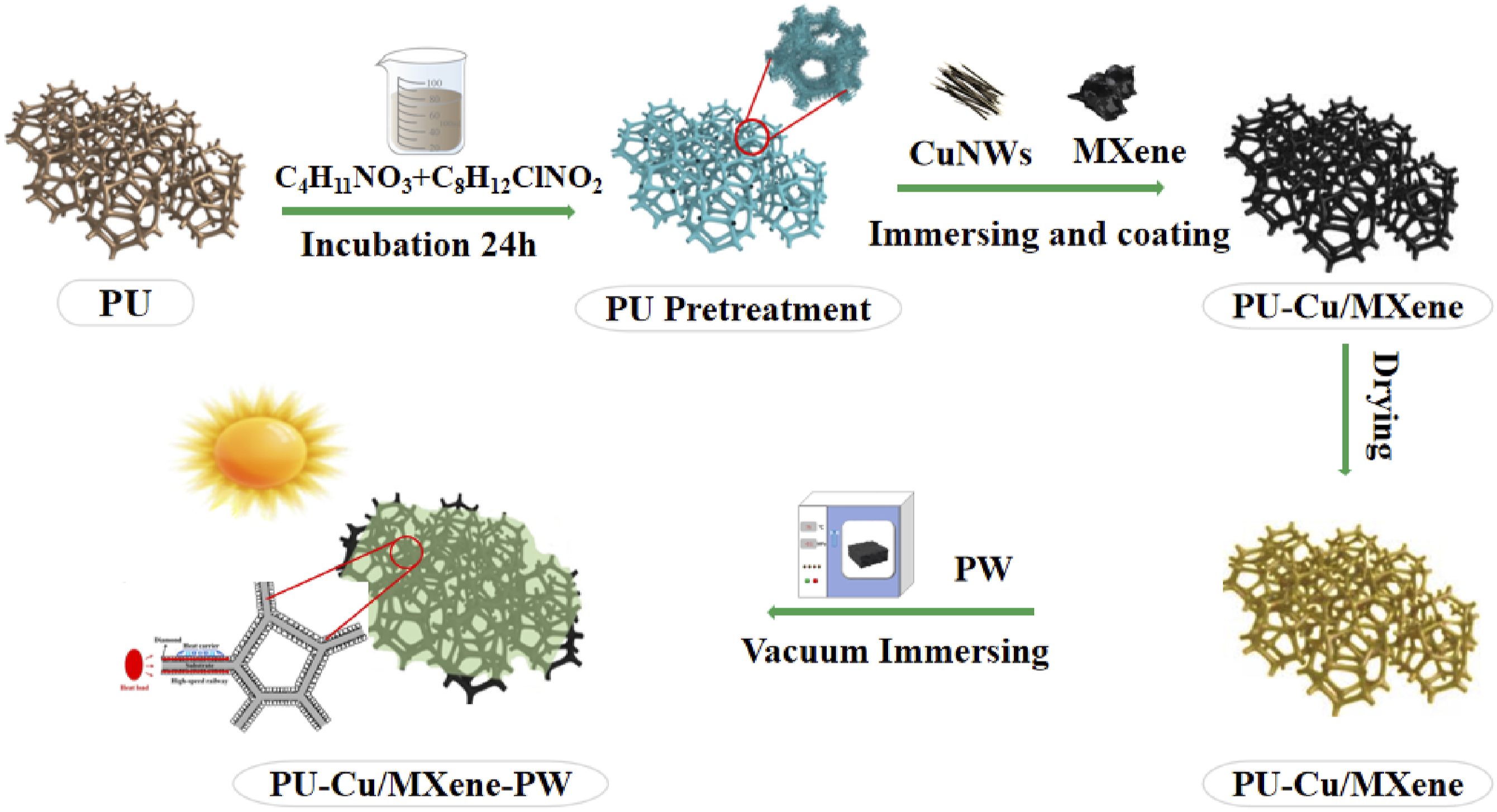
Characterization
The morphology and microstructures of the PCM were characterized by scanning electron microscopy (SEM,thermo scientific Apreo 2S,FEI Corporation, USA). The Fourier studied the chemical structures of the samples transform infrared spectra (FT-IR) (Thermo Fisher Scientific, Nicolet IS50) and X-ray diffractometer (XRD, Bruker, D8A A25X), and the composition and crystal form of the resulting products were analyzed. The thermal conductivity difference between PW and PCM was compared with a laser thermal conductivity meter (C-THERM/TCI type). In the 20–800°C temperature range, The Gravity Analysis of the sample was carried out with a TG instrument (STA2500, NCI Scientific Instrument Trading Co., LTD.) at a heating rate of 10°C·min−1 under N2 atmosphere. The infrared thermal imaging spectrometer recorded the prepared sample’s surface temperature changes, heat storage, and heat release (EVERTE, EN60825-1). A general testing machine (CMT2502, China MTS System Co., LTD.) was used for mechanical deformation testing at a 0.5 mm·min−1 speed. The phase transition characteristics (phase transition temperature and enthalpy values) were characterized by differential scanning calorimetry (DSC, pyris1DSC, PerkinElmer) at heating and cooling rates of 5°C·min−1 in an N2 atmosphere. The temperature-time curves under the simulated solar light illumination were recorded by a solar-thermal test system (solar simulator and data collector).
Results and discussion
Morphology and structure of PPCuM
The microstructure of PU and PPCuM was observed by SEM. As shown in Figure 2(a), a hard and smooth CuNWs surface was prepared. Figure 2(b) shows the MXene monolayer structure obtained after etching, and Figure 2(c) and (d) shows that PU has a three-dimensional continuous network structure. With the increase of magnification, the skeleton surface presents a smooth structure. After surface pretreatment, wrinkling and rough texture appeared on the skeleton surface from smooth, but hardly affected the porous PU structure (Figure 2(e) and (f)). It can be seen from Figure 2(g) that the microstructure of PU did not change after the addition of CuNWs/MXene, and CuNWs were coated by MXene and wound on the micropore surface of PU skeleton gap. No significant agglomeration was found with the increase of MXene filling which attribute to many hydroxyl groups on MXene can form hydrogen bonds with PU matrix,28,29 thus improving the compatibility of MXene and PU. A continuous 3D network is generated between CuNWs/MXene and PU, resulting in a high aspect ratio. Figure 2(h)–(l) shows the morphology and structure of CuNWs/MXene composite phase change materials with different mass ratios under higher magnification by SEM. PU porous structure can be seen in the entire CuNWs/MXene filler. CuNWs was gradually coated with MXene and wrapped around the skeleton of PU. It can be observed that the area covered by MXene expanded, the pore structure decreased, and MXene layer began to appear on the pore surface. This is because the high concentration of MXene gathered into more two-dimensional layered structures, thus intensifying the densification between PU pore structures in the limited space. Therefore, a relatively uniform distribution of CuNWs/MXene in the PU skeleton can still be achieved through a simple physical mixing method. Microstructure of PPCuM; (a) CuNWs; (b) MXene; (c and d) smooth surface of PU; (e and f) pretreated PU; (g) PCuM; (h) PCuM1; (i) PCuM2; (j) PCuM3; (k) PCuM4; (l) PCuM5.
The interaction between PW and PPCuM was confirmed through the FT-IR spectra, which are presented in Figure 3(a). In the spectrum of PW, the absorption peaks at 2914 cm−1, 2845 cm−1, 1459 cm−1 and 720 cm−1 are C-H asymmetric stretching vibration, C-H symmetrical stretching vibration, C-H bending vibration and -CH2 swaying vibration, respectively.30,31 PU has a wide absorption band at 3450 cm−1, which is due to the stretching vibration of the O-H group on the polymer.
32
In the spectrum of MXene, the stretching vibrations of the C-O and Ti-O fragments are centered at 1102 cm−1 and 548 cm−1, respectively. And there is an obvious absorption peak at 3430 cm−1, which is mainly attributed to the stretching vibration of O-H groups.
33
Furthermore, no clear new signals can be detected in the FT-IR spectrum of the PPCuM compared with the spectra of PW and MXene. This indicates the physical interaction between PW and MXene. Particularly, a slight shift of the hydroxyl peak can be observed in the PU-based PCM, implying that an intermolecular hydrogen-bonding interaction can take place between PW and MXene.
34
Accordingly, these results indicate that the form-stable characteristics of the PPCuM can be enhanced during the phase transition process.
35
(a) FT-IR spectra of PW and PPCuM; (b) XRD patterns of PU, PW, MXene, and CuNWs; (c) XRD patterns of PPCuM.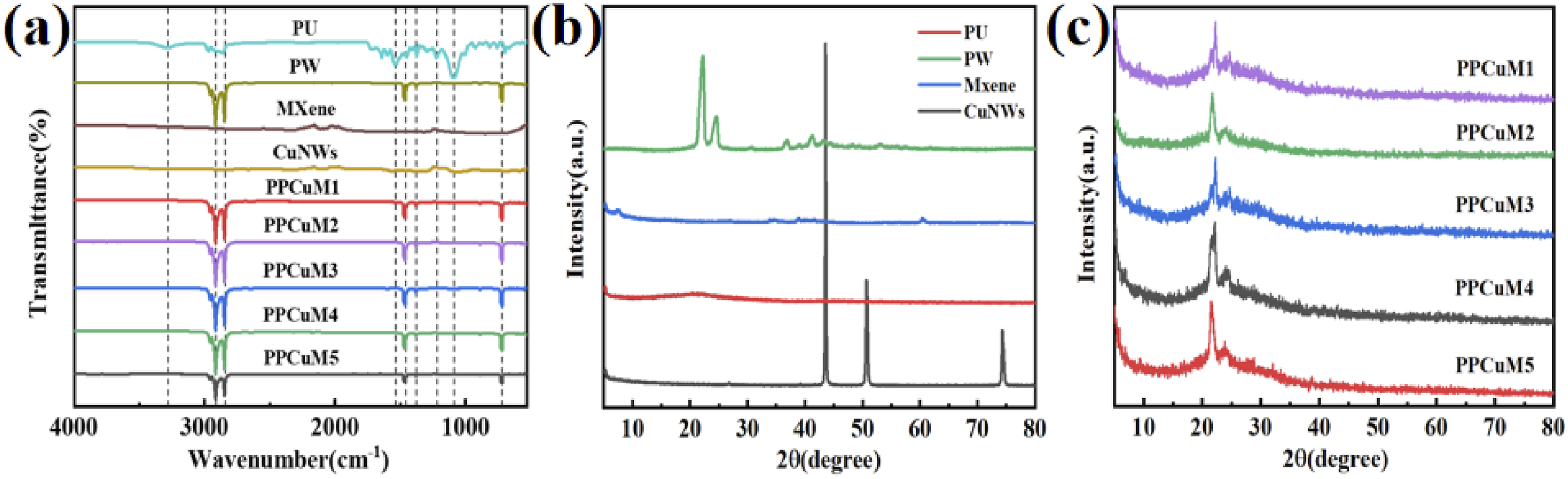
To study the phase and crystal structure of PPCuM, the crystallization behavior of PW and PPCuM was analyzed by XRD, and the results were shown in Figure 3(b). The XRD pattern of PW showed two intense and sharp diffraction peaks at 21.4° and 23.8° indicating the good crystallinity of PW. 27 In the XRD pattern of CuNWs, diffraction peaks appeared at 43.4°, 51.5° and 74.5°, indicating that the prepared CuNWs was relatively pure and unoxidized. 36 The characteristic peak representing MXene appears at 6.2°, indicating that the etching of Al in the raw material is basically complete. 37 As shown in Figure 3(c). The diffraction peak intensity of CuNWs/MXene film coatings on PU surface decreased. Due to the strong interaction between the large specific surface areas of the MXene film and the pores in the PU, it helps the PW to be closely absorbed by the pores of the PU. Two diffraction peaks of PW were observed in all samples of PPCuM, and the positions of diffraction peaks did not shift significantly, indicating that the PW was still in a crystalline state in the PPCuM, which was a guarantee for the release of latent heat. Furtherly, since the diffraction peak intensity of PPCuM depended on the content of MXene, the diffraction peak intensity increased with the increasing of MXene content, and the diffraction peak intensity of PPCuM5 was the strongest. In the composite PCM, the diffraction peak of PW was strong, and no obvious characteristic peak of carbon material is found. At the same time, the skeleton of PU has no chemical effect on the crystal structure of PW and has good chemical compatibility.
Mechanical properties of PCuM
Good mechanical properties were the prerequisite for the practical application of PPCuM. Usually, the simultaneous enhancement of mechanical strength and heat storage property of PPCuM was contradictory due to the solid-phase rigidity of PW. The Figure 4(a) shows the stress-strain curves of PCuM with different MXene loadings, and the corresponding stress-strain parameters are summarized in Table 1, respectively. As shown in Figure 4(a) and Table 1, the mechanical strength of PCuM regularly increased with the increasing MXene loadings, which was related to the content of MXene. When the compressive strain fraction reaches 90%, PCuM containing 50 wt% Mxene can withstand higher compressive strength (56.47 KPa) than PU without Mxene (48.36 KPa). When the load of MXene increases to 83.3 wt%, the compressive stress of PCuM increases to 81.83 KPa. Revealing that the existence of MXene could substantially enhance the mechanical strength of the PPCuM and effectively improve their flexibility. At the same time, all the samples can rebound quickly and have no plastic deformation when the compressive strain fraction reaches 90%. Consequently, it was possible to construct the PPCuM with simultaneously enhanced mechanical strength, light-to-heat conversion and heat transfer while maintaining available latent heat, The reason why the mechanical strength of PPCuM was enhanced by MXene was as follows: the improved flexibility and stretchability of PPCuM originated from the abundant surface functional groups (-OH, -F, -O, etc.) on MXene nanosheets, which provided additional hydrogen bond binding sites, thereby promoting the cross-linking of PPCuM. Thereby improving the overall mechanical strength of the PPCuM.38–40 Based on the above discussion, although the distribution status of MXene was irregular, the cross-linking structures with hydrogen bonds could still be formed so that the mechanical strength of PPCuM was effectively improved. (a) Compressive stress-strain curves of PU and PCuM at 90% strain; (b) digital photographs of PCuM deformation tests; (c–f) Photographs of PPCuM with different shapes. Stress-strain parameters of PCuM under different MXene loads.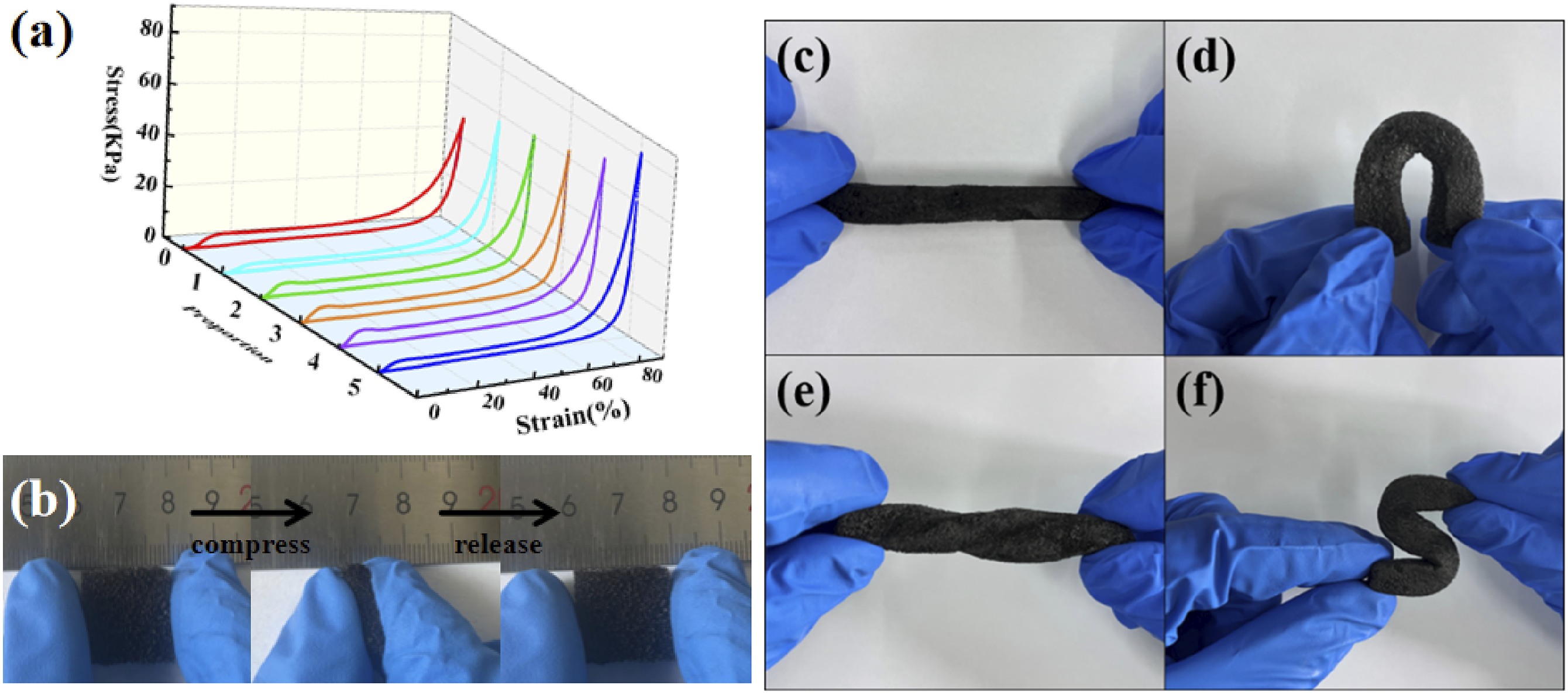
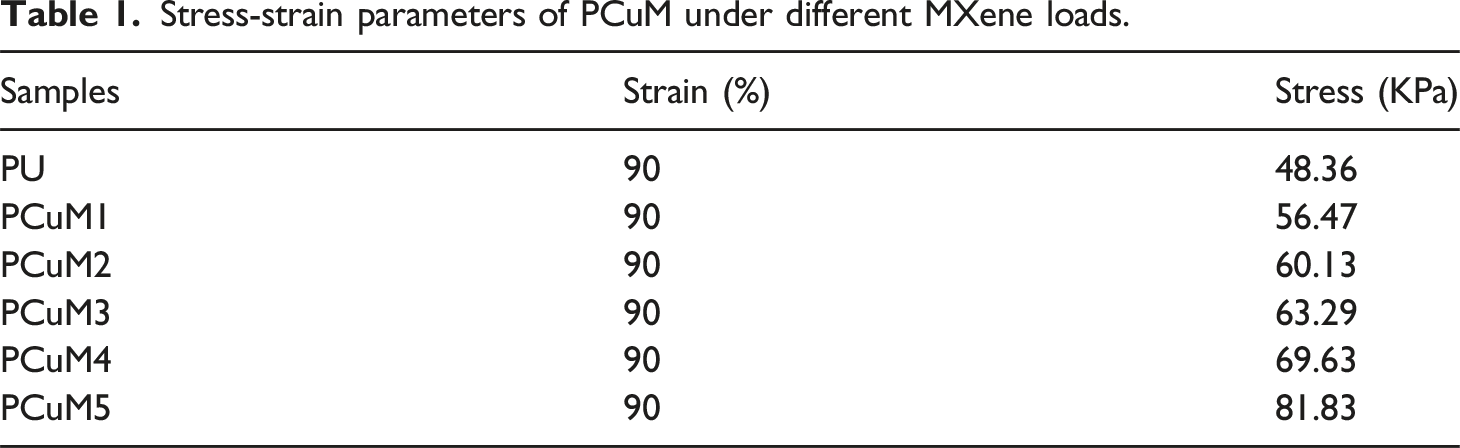
Figure 4(b) shows the digital photographs of the deformation test. When the PCuM carrier is compressed and released with fingers, its porous material is quickly recovered without shedding any impurities, and the nanomaterials are tightly attached to the surface of PU. In addition, the good mechanical properties of the PU skeleton can maintain the stability of PCuM, such as preventing the volume contraction of PW during the melting/solidification process and damaging the structure of the entire composite material. The exceptional plasticity of PU can realize the synchronous contraction of the nanomaterial CuNWs/MXene, effectively avoiding the structural collapse of PCuM. The results show that the PCuM carrier can maintain the thermal conductivity pathway of PCM stably under deformation conditions.
Flexibility is a critical characteristic of PPCM in thermal energy storage and management applications, as it denotes the material’s ability to deform without breaking under external forces. In addition, PW exhibits a large volume expansion during melting/solidification, and the use of a flexible polymer can prevent PW from leaking in the composite. As shown in Figure 4(c)–(f). The superior flexibility of the as-fabricated PPCuM is demonstrated by their bending process It was found that the PPCuM could be easily folded into complex shapes at 60°C without destroying its structure. Furtherly, the PPCuM could withstand the external forces of folding, bending and twisting without breaking, indicating good flexibility and mechanical strength, which was closely related to its structure. Furthermore, all the PPCuM with different MXene loadings showed superior foldability and flexibility, which was beneficial to prevent the volume expansion of PW in solar thermal systems.
Shape stabilization properties
Shape stabilization properties represent an important criterion to determine the service life of PCM. For this reason, measurements to quantify shape stabilization properties were performed. As depicted in Figure 5, none of the samples exhibited any leakage at 50°C. However, PPCuM1 and PPCuM2 samples showed minor leakage when the temperature increased to 60°C, Moreover, as the temperature was further increased to 75°C, the PPCuM1, PPCuM2 and PPCuM3 samples exhibited a leakage phenomenon. Notably, no leakage was found during the entire heating process for the PPCuM5 sample. The reasons for such phenomenon have been related to the flexibility of 3D supporting skeleton and strong capillary force of PU as well as to the intermolecular hydrogen-bonding interactions between PW and MXene.35,36,38–41 The 2D MXene nanosheets act as a protective shell around the PCM to impede the leakage from the confined cages. Leaked digital photos of PPCuM under different MXene loads.
Transient temperature response
To further confirm the effect of MXene on the light-to-heat conversion performance, the surface temperature distribution taken by the infrared thermal camera of the PPCuM was analyzed (Figure 6). As the irradiation time increased, the surface temperature of the PPCuM increased continuously. Furtherly, the higher MXene content, the higher surface temperature of the PPCuM at the same irradiation time. For example, after irradiation for 60 s, the average surface temperature of PPCuM with 50 wt%, 66.6 wt%, 75 wt%, 80 wt% and 83.3 wt% MXene was respectively 34.23°C, 39.76°C, 42.83°C, 45.36°C and 48.25°C, and the corresponding maximum surface temperature was 77.25°C, 79.42°C, 83.61°C, 84.38°C, 86.53°C and >88.50°C, respectively. The above results further verified that the light-to-heat conversion performance of PPCuM was effectively improved by MXene. During the cooling process, the surface temperature of the PPCuM decreased with the increasing MXene content at the same cooling time. The above results demonstrated that the MXene not only increased the light-to-heat conversion ability of the PPCuM, but also improved their heat transfer rate. Transient temperature response of PPCuM under infrared thermal imager.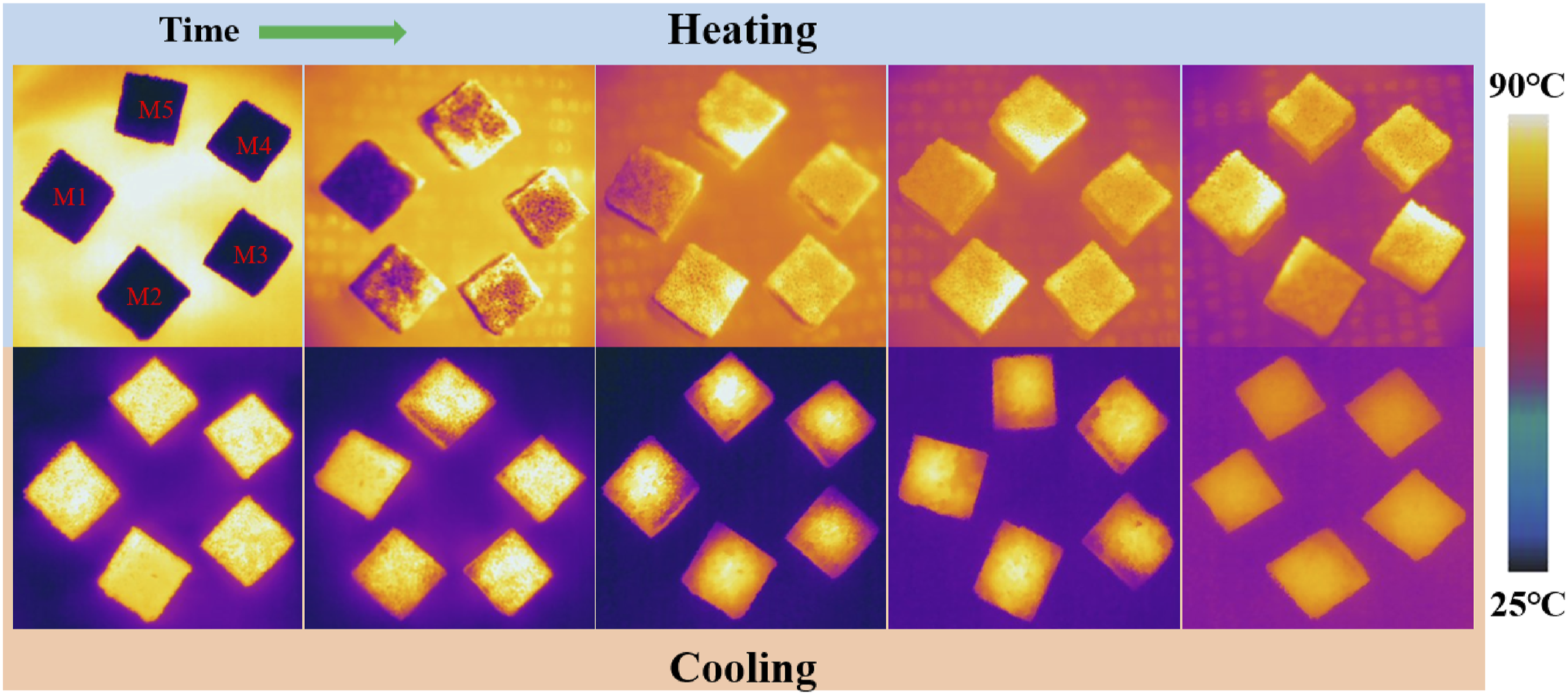
Phase change properties
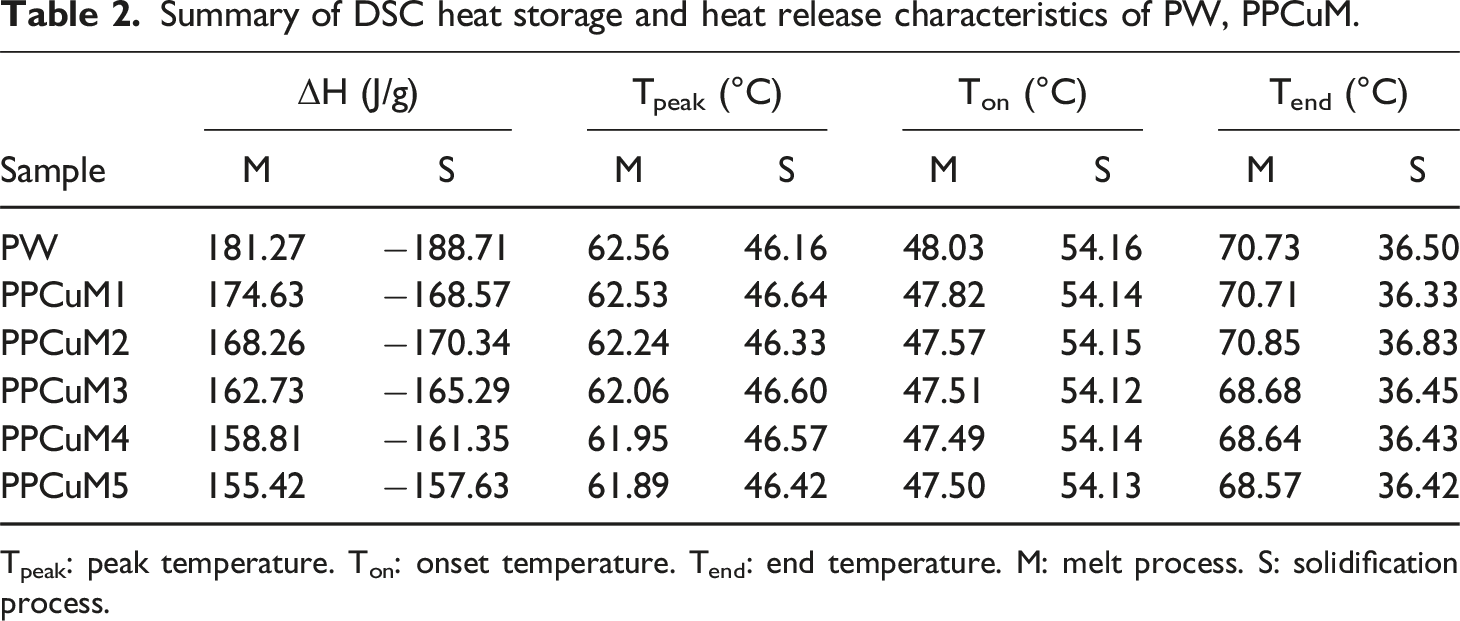
Latent heat is a parameter that plays a crucial role in the thermal energy storage capacity of PPCuM. Figure 7(a) illustrates the differential scanning calorimeter (DSC) melting and cooling curves of PW and PU-based PPCuM. The corresponding thermal parameters are illustrated in Table 2. As shown in Figure 7(a), the DSC curve of PW displayed two obvious phase transition peaks during the melting and solidification processes, indicating that the PW exhibited good heat storage and release characteristics. The first phase transition peak around 30°C was related to the solid-solid phase transition, while the second one around 50°C was attributed to the solid-liquid phase transition.
42
The DSC curves of PPCuM with different MXene content loadings were highly overlapping, showing similar phase transition behavior, and the endo-thermic and exothermic peaks could be clearly observed, demonstrating that the latent heat was stored and released, and the PW filled in the cross-linked network did not undergo chemical reaction and could normally undergo a phase transition process, which was consistent with the conclusion that PW showed good chemical compatibility confirmed by FT-IR result. The difference between melting and solidification temperature of PPCuM was ascribed to the super cooling behavior of PW. However, the melting temperature of PPCuM was slightly lower than that of PW (Table 2), which was attributed to the limited volume expansion of PW particles during the melting process caused by the cross-linking effect and dense structure of PPCuM.
43
The phase transition temperature of PPCuM slightly changed with the increasing MXene loadings, it was proved that the ΔH value of the PPCuM reduces to a certain extent because of increases with the proportion of MXene, which was beneficial to simultaneously enhance the light-to-heat conversion and thermal conductivity of PPCuM while maintaining available latent heat. (a) DSC curves of PW and PPCuM; (b) DSC curves of PPCuM3 measured after a different number of thermal cycles. Summary of DSC heat storage and heat release characteristics of PW, PPCuM. Tpeak: peak temperature. Ton: onset temperature. Tend: end temperature. M: melt process. S: solidification process.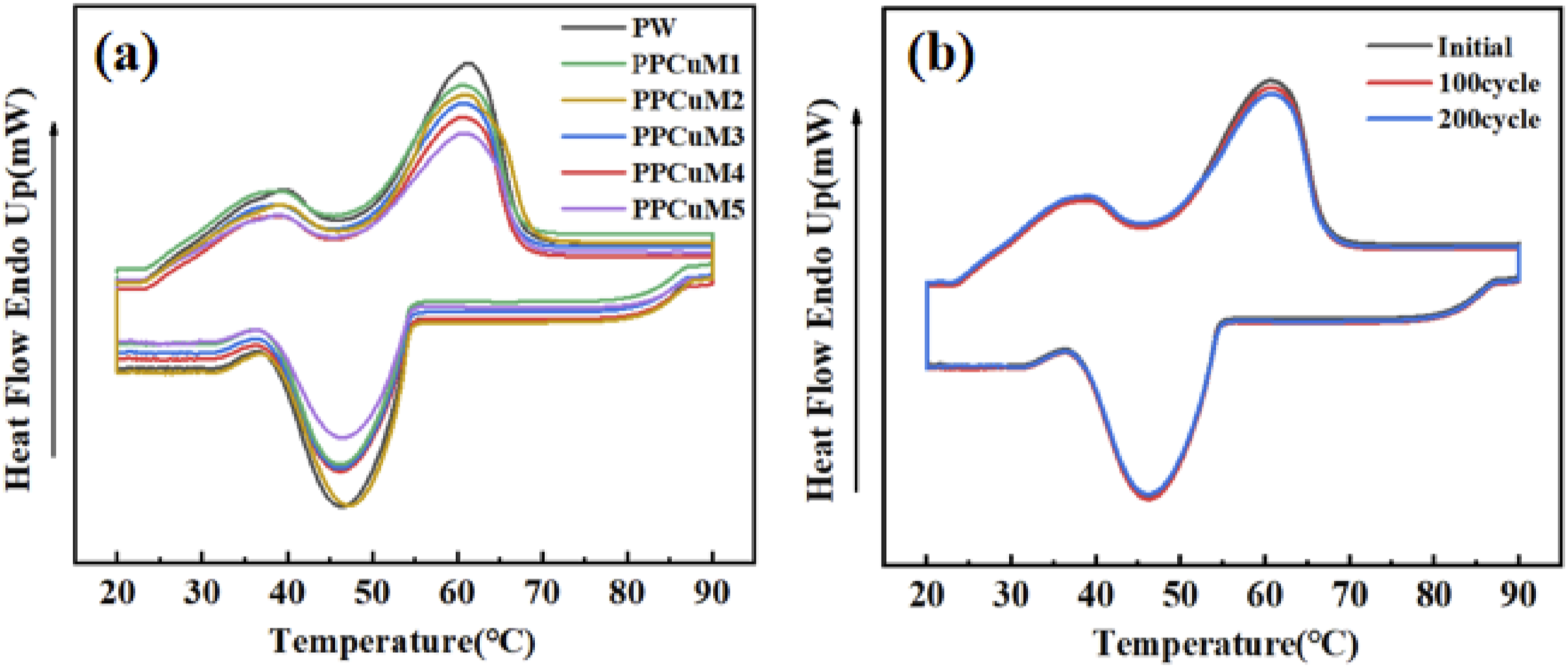
Furthermore, the thermal reliability and reusability of the PU-based PPCuM are critical for practical applications. The DSC curves of PPCuM3 obtained after going through 1, 100, and 200 thermal cycles are presented in Figure 7(b). As it can be clearly seen, the phase change properties of PPCuM3 exhibit negligible change after 200 thermal cycles, indicating a superior thermal reliability and reusability of the PU-based PCM.
Entropy generation is the energy dissipation of a system in an irreversible process. For composite PCM, entropy generation is due to the multi-scale irreversible process such as phase change hysteresis, interfacial thermal resistance, cyclic damage and environmental interaction. Entropy generation is directly related to its energy storage efficiency and thermodynamic performance. The introduction of additives may significantly change the entropy generation path through interface effects, dispersion state and recovery complexity while improving functionality. Entropy generation is expressed as follows: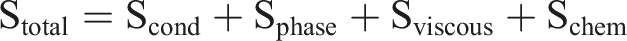
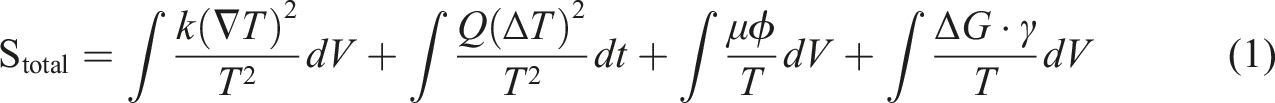
Scond is the entropy generation of heat conduction, Sphase is the kinetic entropy generation of phase transition, Sviscous is the Viscous dissipation entropy generation, Schem is the entropy generation of chemical reactions, k is the thermal conductivity, ∇T is the temperature gradient, T is the local temperature, ΔT is the degree of supercooling, μ is the dynamic viscosity, Φ is the viscous dissipation function, ΔG is the gibbs free energy change, r is the reaction rate.
In the PPCuM, the phase change is dominated by kinetic entropy generation, ignoring viscosity and chemical terms. Additives are usually added to enhance thermal conductivity, prevent leakage, improve stability, etc. By adding high thermal conductivity inert nanomaterials to homogenize the temperature field, increase the thermal conductivity (k), reduce the temperature gradient (∇T), reduce Scond, Schem, and use ultrasonic dispersion and surface modification technology to increase the additive dispersion to reduce the degree of supercooling and interface thermal resistance, and reduce Scond. The PU porous structure skeleton is selected to optimize the porosity and pore size distribution, balance the thermal conductivity increase and phase transition inhibition, and reduce Scond and Sphase. Therefore, the total entropy generation is reduced.
Photo-thermal energy conversion and storage
Owing to their excellent properties, including the excellent encapsulation effect, thermal conductivity, and large phase change enthalpy, the obtained PU-based PCM can be expected to have good application prospects in sunlight energy management, Hence, it is necessary to evaluate the light-to-thermal energy conversion and storage through a simulated solar-thermal conversion test. The results of this test are shown in Figure 8(a). The PPCuM samples were exposed to a light irradiation of 1.5 KW·m−2. Figure 8(a) shows the temperature-time curve during the test. The temperature of the PPCuM first increases sharply and then slowly until it reaches a plateau, which indicates the melting of the samples, the temperature then increases rapidly again under light irradiation. As the light source is removed, the temperature of the PPCuM samples declines quickly, and a phase transition platform emerges between 40°C and 50°C, indicating the release of the stored latent heat due to the liquid-to-solid phase transition of the PW. It can be found that with the increase of MXene load, the heating rate and platform temperature increase with the increase of MXene load. For example, the time for PPCuM containing 50 wt%, 66.6 wt%, 75 wt%, 80 wt% and 83.3 wt% MXene to reach the predetermined temperature 50°C is 270 s, 220 s, 190 s, 170 s and 150 s, respectively, indicating that the heating rate increases. The corresponding maximum temperatures reached after 500 s of illumination were 63.18°C, 68.83°C, 73.52°C, 78.32°C and 83.41°C, respectively. The above results show that a lower loading of MXene will absorb much less solar energy. MXene can effectively improve the photo-thermal conversion performance of PPCuM. At same time, MXene acts as an effective photon capture and molecular heater under solar irradiation, forcing the lattice of PPCuM to vibrate, thus completing the conversion of solar thermal energy.44,45 The above results demonstrate that the solar-thermal conversion efficiency is related to the load capacity of MXene. The photo-thermal energy conversion and storage are feasible through an efficient thermal transmission from the continuous conductive CuNWs/MXene network structure to the adjacent PW via the PCM, and vast amounts of stored thermal energy can be released. (a) Temperature-time curves of PPCuM under simulated solar illumination; (b) heat storage and change of heat storage rate of PPCuM; (c) solar-thermal conversion efficiency of PPCuM and peak temperature; (d) thermal conductivity of PPCuM.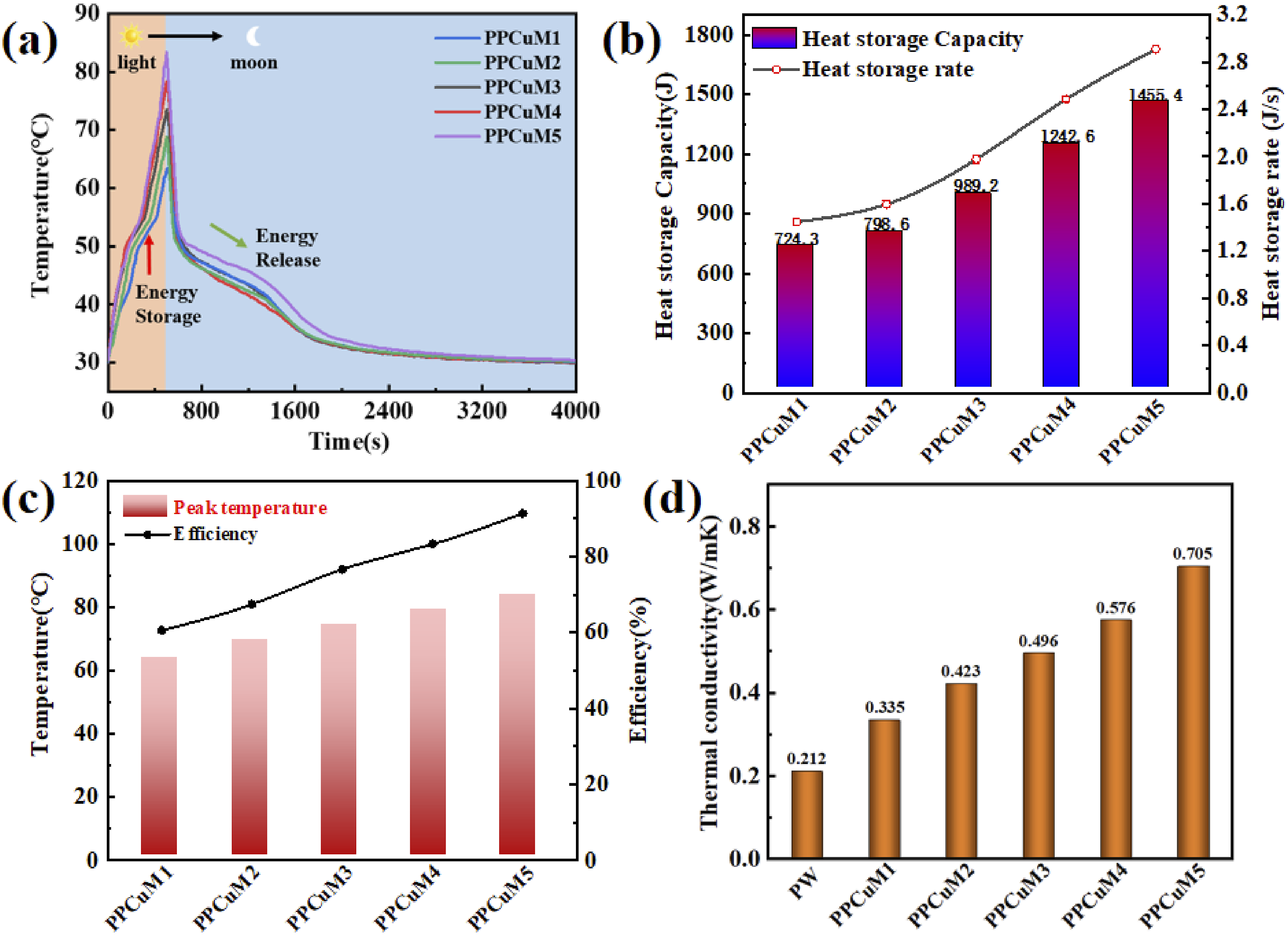
Considering that the PPCuM is affected by the error of simulated sunlight irradiation, this paper adopts the average algorithm of each temperature point to analyze and calculate its storage efficiency and other fundamental evaluation indexes. The heat storage capacity of PPCuM can be expressed as follows
46
: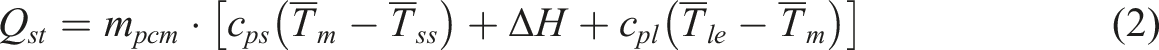



The average heat storage rate of the equipment q
st
is given by
47
:
Solar-thermal conversion efficiency (η) is another crucial evaluation index of solar heat utilization in the process of heat storage, and it was calculated as follows
48
:
G is the solar irradiation intensity, S sc is the sample’s top surface area, and t e is the duration of the phase transition.
As shown in Figure 8(b), when the light intensity of the solar simulator is 1.5 KW·m−2, the photo-thermal energy storage of PPCuM reaches 724.4 J, 798.6 J, 989.3 J, 1242.6 J and 1455.4 J, respectively. The heat storage rates were 1.45 J·s−1, 1.59 J·s−1, 1.97 J·s−1, 2.48 J·s−1 and 2.91 J·s−1, respectively. After 500 s of irradiation, the highest temperature of PPCuM1 was 63.18°C, while the temperatures of PPCuM2, PPCuM3, PPCuM4 and PPCuM5 were 68.83°C, 73.52°C, 78.32°C and 83.41°C, respectively Figure 8(c). Compared to PPCuM1, the thermal response time required at 50°C based on PPCuM5 is greatly reduced. In addition, PPCuM also conducted a study on the storage efficiency of solar thermal energy, and found the following: The η of PPCuM1, PPCuM2, PPCuM3, PPCuM4 and PPCuM5 reached 60.56%, 67.42%, 76.61%, 83.27% and 91.35%, respectively, when the simulated light intensity was 1.5 KW·m−2. This is due to the ultra-high photo-thermal conversion capability of the MXene. 49 The results show that the content of MXene in the composite has a significant effect on the photo-thermal absorption and conversion under simulated lighting conditions. Specifically, the high thermal conductivity of both MXene and CuNWs has a significant effect on enhancing the solar energy absorption of PPCuM, that is, it has better photo-thermal conversion performance under the same light, and the above research shows that the composite PPCuM can quickly convert solar energy into heat energy, promoting the storage and release of heat energy.
The thermal conductivity of all samples is shown in Figure 8(d). It can be seen that PW shows a low thermal conductivity of 0.212 W·m−1·K−1, while PPCuM1 shows a thermal conductivity of 0.335 W·m−1·K−1. Interestingly, the thermal conductivities of the PPCuM1 are enormously enhanced. In particular, the PPCuM5 sample shows a significantly higher thermal conductivity value (0.705 W·m−1 K−1), which is 332.5% higher than that of PW. The observed trend indicates that CuNWs and MXene can facilitate the formation of heat channels via its continuous network structure, which is beneficial to heat conduction. In addition, the strong interface interaction between MXene and PW causes a reduction of the interfacial thermal resistance, owing to the large aspect ratio of MXene and hydrogen bonding between MXene and PW. 50 Consequently, MXene acts as a supporting material with superior heat conduction, visibly boosting the thermal conductivity of PCM. This enhanced thermal conductivity could be beneficial for solar-thermal conversion and storage.
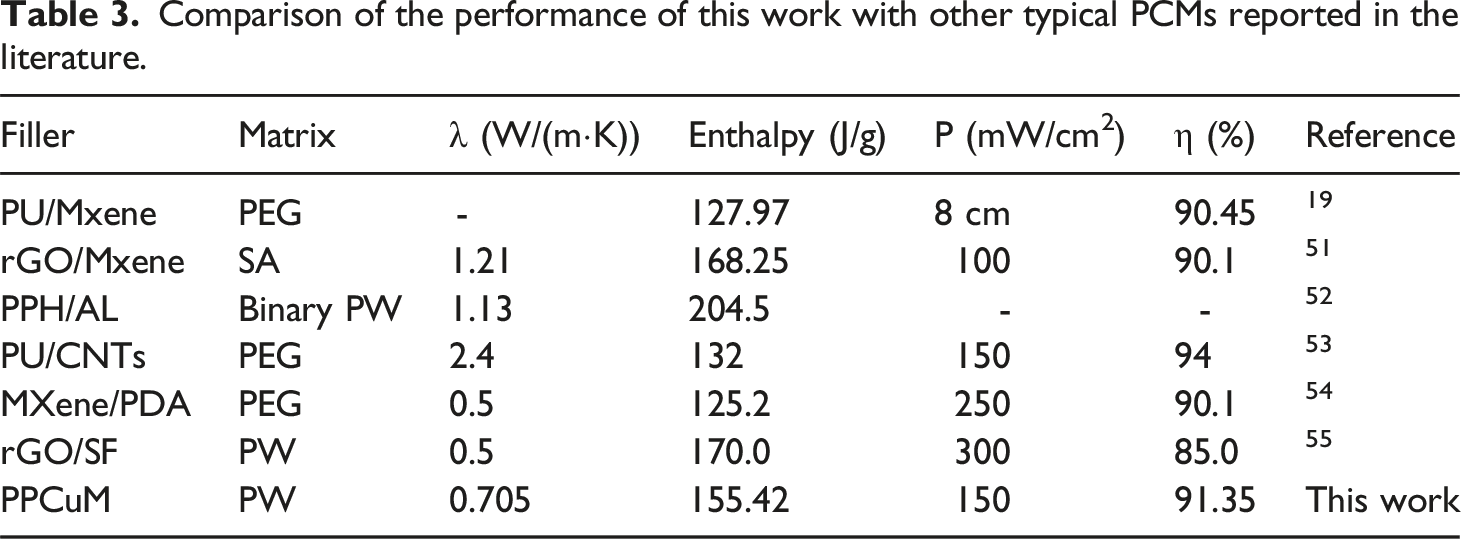
Comparison of the performance of this work with other typical PCMs reported in the literature.
Thermogravimetry analyses
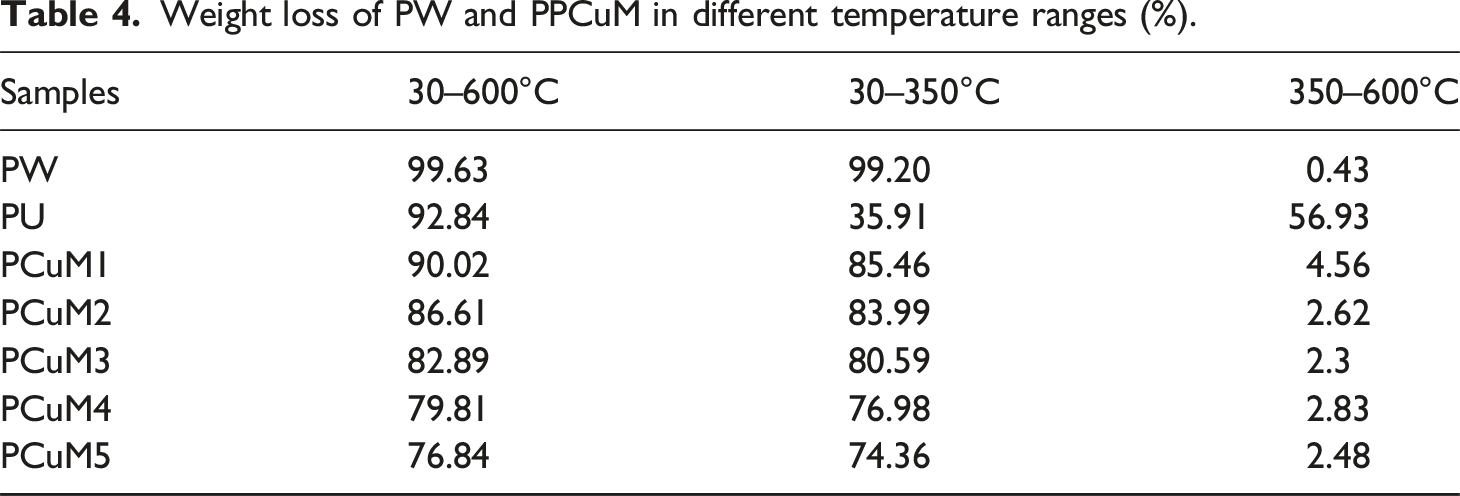
The thermal stability of PCM is also an important parameter for the evaluation of the thermal energy storage capability. The thermal properties of pure PW, PU and PPCuM under nitrogen were investigated using thermogravimetry analysis (TG), and the corresponding results are illustrated in Figure 9 and Table 4. Referring to Figure 9, pure PW shows a one-stage thermal degradation corresponding to the pyrolysis of PW chains. Due to the almost complete thermal decomposition, the weight loss of PW within 30–600°C reached 99.63%. The PPCuM exhibited two-step weight loss within 30–600°C, indicating similar thermal stability characteristic which was mainly ascribed to the combined thermal decomposition of PW, removal of adsorbed water within 30–350°C (first step), and oxidative degradation of PW above 350°C (second step). Importantly, the onset temperatures with substantial weight loss of PPCuM were much higher than their phase change temperature range (∼60°C), which may be due to the limitation of the evaporation and pyrolysis of the PW segments because of the layered MXene nanosheets. The char residues at 600°C for the PU-based PCM are improved as the loading of MXene increases, which is attributed to the ultrahigh thermal stability and catalytic charring effect of the MXene nanosheets.
56
Furthermore, the high char residues can be conducive to the prevention of heat and mass transfer between the gaseous and condensed phases. In addition, the maximum weight loss rates (Rmax) of the PPCuM shift toward lower values with the incorporation of MXene in comparison with PW, demonstrating the reinforced thermal resistance of these PCM. The superior thermal stability of these PCM favors their practical application in thermal energy storage. Revealing that the PPCuM showed reasonable and acceptable thermal stability within the phase change operating temperature range. Thermogravimetric curves of PU, PW, MXene and PPCuM. Weight loss of PW and PPCuM in different temperature ranges (%).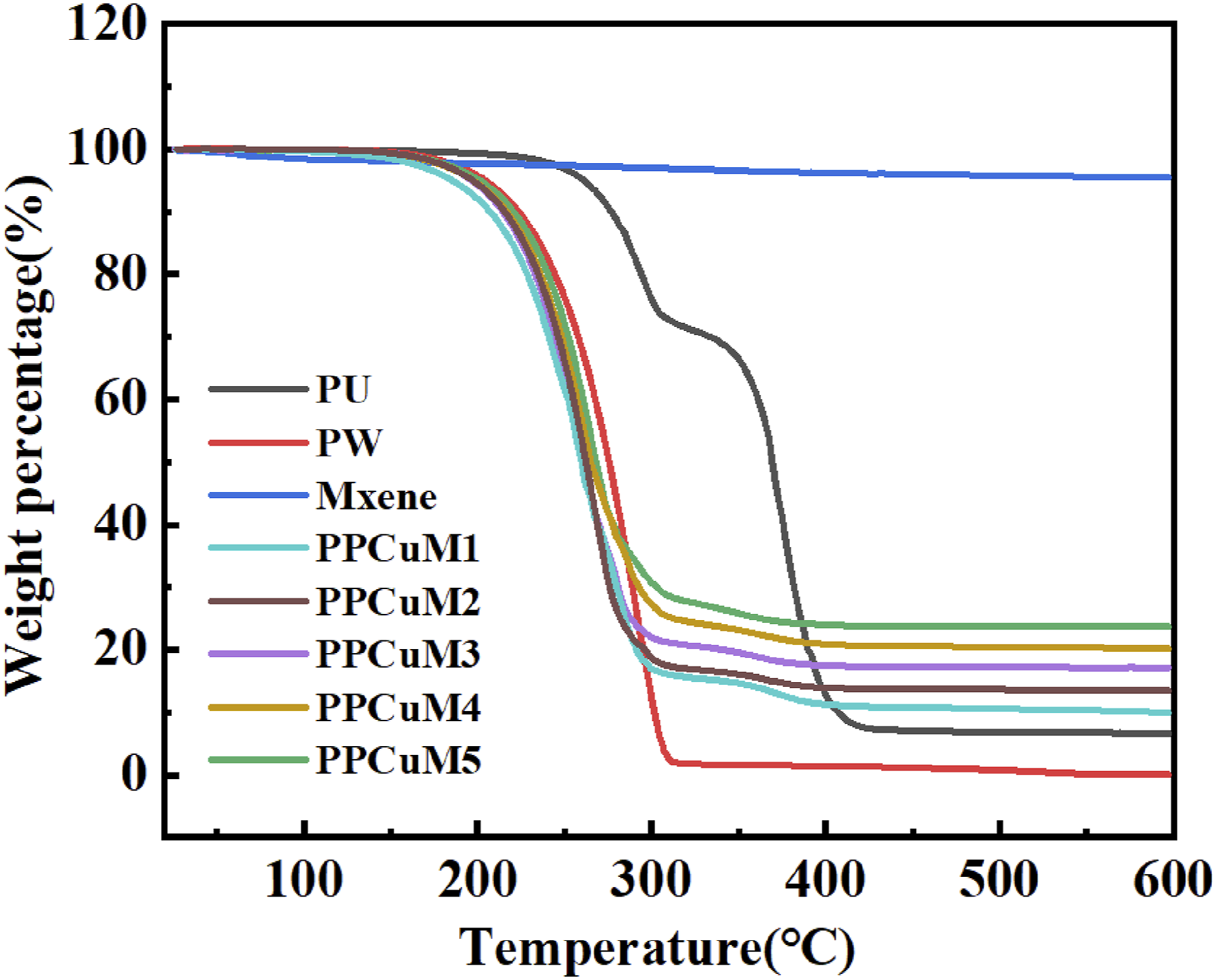
Conclusions
In summary, Flexible PCM composites with stable morphology were prepared by solution blending,in which PU was selected as the supporting skeleton for the PCM, CuNWs acts as the core bendable thermal conductivity path. MXene acts as the thermal conductivity carrier and adsorption site, and PW is introduced into the PU skeleton through a simple vacuum impregnation method. The prepared PPCuM composite has high latent heat (155.42 J·g−1) and an excellent leakage resistance. The MXene network endows the PPCuM composites with improved thermal conductivity and superior light-to-thermal storage capacity, The TG results show a distinct enhancement of the thermal stability and charring ability for these Composite materials at high temperature. Furthermore, as the loading of MXene in the PCM increases, photo-thermal conversion efficiency (91.35%) and Thermal conductivity (0.705 W·m−1 K−1) values of the PCM increase significantly, suggesting strong thermal energy conversion and storage. The obtained PU-based PCM composites are promising candidates in the thermal energy storage field.
Footnotes
Acknowledgements
The authors would like to acknowledge the support of Basic Research Foundation for Universities directly under the Inner Mongolia Autonomous Region (2023QNJS060; 2023QNJS050).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Basic Research Foundation for Universities directly under the Inner Mongolia Autonomous Region (2023QNJS060; 2023QNJS050).
