Abstract
To study the influence of vibration on gas desorption and diffusion in particle coal, gas desorption experiments on soft coal with outburst risk under different frequency vibrations were carried out by using a self-designed gas adsorption and desorption platform under vibration conditions, and the influence of different frequency vibrations on the diffusion kinetic parameters was quantitatively analyzed by using a dynamic diffusion coefficient model. The influence mechanism of vibration on gas desorption and diffusion in coal was further analyzed from the three aspects of gas molecules, energy conversion and pore structure through theoretical analysis and mercury injection experiments. The results showed that with increasing vibration frequency, the gas desorption of the coal samples first increases and then decreases. The initial diffusion coefficient of gas in the coal samples increases linearly with increasing vibration frequency, but the attenuation coefficient of the diffusion coefficient decreases first and then increases with increasing vibration frequency. The "throwing effect" and thermal effect of vibration on the gas molecules are both conducive to the desorption of gas molecules. In addition, vibration causes changes in the pore structure in coal, increasing gas diffusion paths and reducing diffusion resistance.
Introduction
Coal and gas outbursts are phenomena where large amounts of coal and gas are ejected in a very short timeframe, which can result in casualties and economic losses (Cao et al., 2001; Lama and Bodziony, 1998; Tang et al., 2016). As a special porous medium, coal has a strong ability to store gas. Gas escape from the coal body to the outside environment is a complex diffusion-seepage-desorption process. Scholars have investigated different factors involved in the mechanism of gas adsorption and desorption, such as the degree of coal metamorphism (Laxminarayana and Crosdale, 1999; Levy et al., 1997), coal pore structure (Clarkson and Bustin, 2000; Talu, 1998), coal seam gas pressures (Zhang et al., 2004), ambient temperature (Azmi et al., 2006; Liu et al., 2013; Pan et al., 2012), moisture (Krooss et al., 2002; Pan et al., 2010; Zhao et al., 2018), microwave fields (Hu et al., 2017; Wang et al., 2020; Wen et al., 2015), and electromagnetic fields (He and Zhang, 2000). As one of the common phenomena in coal mines, vibration has attracted scholarly attention.
Using a low-frequency vibration generation system, Li et al. analyzed the fracture mechanics of coal and found that vibrations encouraged large numbers of cracks in coal to expand and become unstable, thereby inducing coal and gas outbursts. They concluded that vibrations reduced the ability of coal to adsorb gas (Li et al., 2011). Jiang et al. studied thermal and cavitation effects on mechanical (ultrasonic) vibrations and found that different frequencies and intensities increased the permeability of coal seams, decreased the internal diffusion resistance of a coal body, and enhanced the mass transfer ability in coal, which were factors beneficial to the desorption and diffusion of methane in coal (Jiang et al., 2008, 2015). Wang et al. used a low-frequency excitation system composed of low-frequency vibrations and sound waves to study the effects of changes in frequency excitation from 70-120 Hz and found that the gas desorption rate and desorption depth increased (Wang et al., 2016). Li et al. studied the characteristics of gas adsorption and desorption under low-frequency mechanical vibration conditions from 0-30 Hz and used an adsorption-diffusion model to analyze gas diffusion in coal samples. It was concluded that low-frequency mechanical vibration will hinder gas desorption and diffusion in coal samples(Li et al., 2010, 2012b). Zhang studied the desorption characteristics of gas-containing coal under vibration at frequencies of 10, 20, 50 and 100 Hz and proposed that with increasing vibration frequency, the stripping effects, tearing effects and thermal effects caused by mechanical vibration would increase, thereby promoting gas desorption (Zhang, 2016). Chen et al. also concluded that the shear force and thermal effect of a 30–100 Hz mechanical vibration on gas molecules can promote gas desorption and diffusion (Chen et al., 2020).
In the aforementioned studies on the influence of vibration on the gas adsorption and desorption characteristics of a coal body, different results have been reported due to the different types of vibrations and conditions. For mechanical vibration, the results indicated that mechanical vibration both promoted and suppressed gas desorption (Li et al., 2010, 2012b; Zhang, 2016). Obviously, these two conclusions were in opposition. Moreover, in the literature, vibration has been reported to affect pore fissures in a coal body; however, these observations have been qualitative in nature. To describe the effects of vibration on gas desorption diffusion, scholars have proposed a series of theoretical models. Relevant models using constant diffusion coefficients have been adopted for the study of gas diffusion characteristics of a coal body under vibration conditions. With the prolongation of desorption time, gas diffusion gradually develops from macropores to micropores (Li et al., 2015), and different diffusion resistances of gas in pores with different pore size ranges will cause a change in the effective diffusion coefficient during the diffusion process. Therefore, it is inaccurate to use a correlation model with a constant diffusion coefficient to analyze the diffusion law of gas in coal. After summarizing and analyzing the shortcomings of existing gas diffusion mechanism models in coal, Li et al. proposed a dynamic diffusion coefficient model based on a multistage pore system of a coal body, which improved the quantitative accuracy of models for studying the dynamic diffusion process of coal gas(Li et al., 2015, 2016b).
Based on the differences and shortcomings in the literature, this paper investigated the effects of vibration frequency on gas desorption using the dynamic diffusion coefficient model to quantitatively analyse relevant coal gas diffusion parameters under vibration conditions. The mercury intrusion porosimetry (MIP) method was used to quantitatively determine the pore characteristics of experimental coal samples. The influence of different vibration frequencies on the gas desorption and diffusion in the coal body was explored, and the effects of vibration on gas molecules and coal body pore structure were analysed to further develop existing theory and guide mitigate gas disasters.
Materials and methods
Coal samples
A coal sample was selected from the 15021 coal face of the Wangxingzhuang Coal Mine in the western Henan coal field. Fresh coal samples were collected and sent to the laboratory. The coal seam was soft and cracked, exhibiting typical characteristics for coal and gas outburst risk. The fresh coal samples were screened to 0.25-0.5mm and 1–3 mm diameters and divided into 5 groups weighing 300 g each. The coal samples were dried in a vacuum dryer at 40 °C for 12 h. Then, 200 g of each group was selected for adsorption and desorption experiments. The basic physical parameters of the coal sample are shown in Table 1.
Basic physical parameters of coal sample.

Mad denotes the yields of moisture on an air-dried basis; Ad denotes the yields of ash on a dry basis; Vdaf denotes the yields of volatile matter on a dry-ash-free basis; a denotes Langmuir volume; b denotes Langmuir pressure.
Experimental equipment
The test platform for examining gas adsorption and desorption of coal under vibration conditions comprised a self-designed constant temperature adsorption and desorption tester, a high pressure gas injection system, a vacuum degassing system, a vibration-excitation system, and a signal and data acquisition system as shown in Figure 1. The mechanical vibration generation subsystem comprised a YE1311 signal generator (USA), a YE5873A power amplifier (USA) and a JZK-20 electric vibration generator (USA). The signal generator provided 60 W to generate signals from 2 Hz to 2 kHz. The signal can be amplified to 500 W by the power amplifier. Different vibration forms and vibration frequencies were generated by connecting the vibration generator. The vibration generator had a maximum vibration amplitude of 10 mm, a maximum vibration force of 200 N, and a maximum acceleration of 25 g. The vibration generator was connected to a coal sample tank through a connecting rod to impact coal samples. The gas adsorption-desorption subsystem consisted of a gas adsorption and desorption tester (China), a gas adsorption device (China), a coal sample tank and a thermostat. The coal sample tank was placed in the thermostat. By adjusting a shut-off valve, the device provided vacuum pumping and gas adsorption functions. The gas desorption and desorption tester measured gas desorption, which was recorded on an external data acquisition system. The data acquisition system included an acceleration sensor, a gas pressure sensor, a computer, a measuring cylinder, and other related equipment. The acceleration sensor was a CA-YD-186 piezoelectric acceleration sensor (USA) with a maximum transverse sensitivity less than 5%. The accuracy of the gas pressure sensor was 0.1%. The test platform carried out the gas adsorption and desorption experiments under wide ranges of vibration conditions (different frequencies and vibration amplitude). The experiments were monitored in real-time by the data collector, and the test accuracy was high.
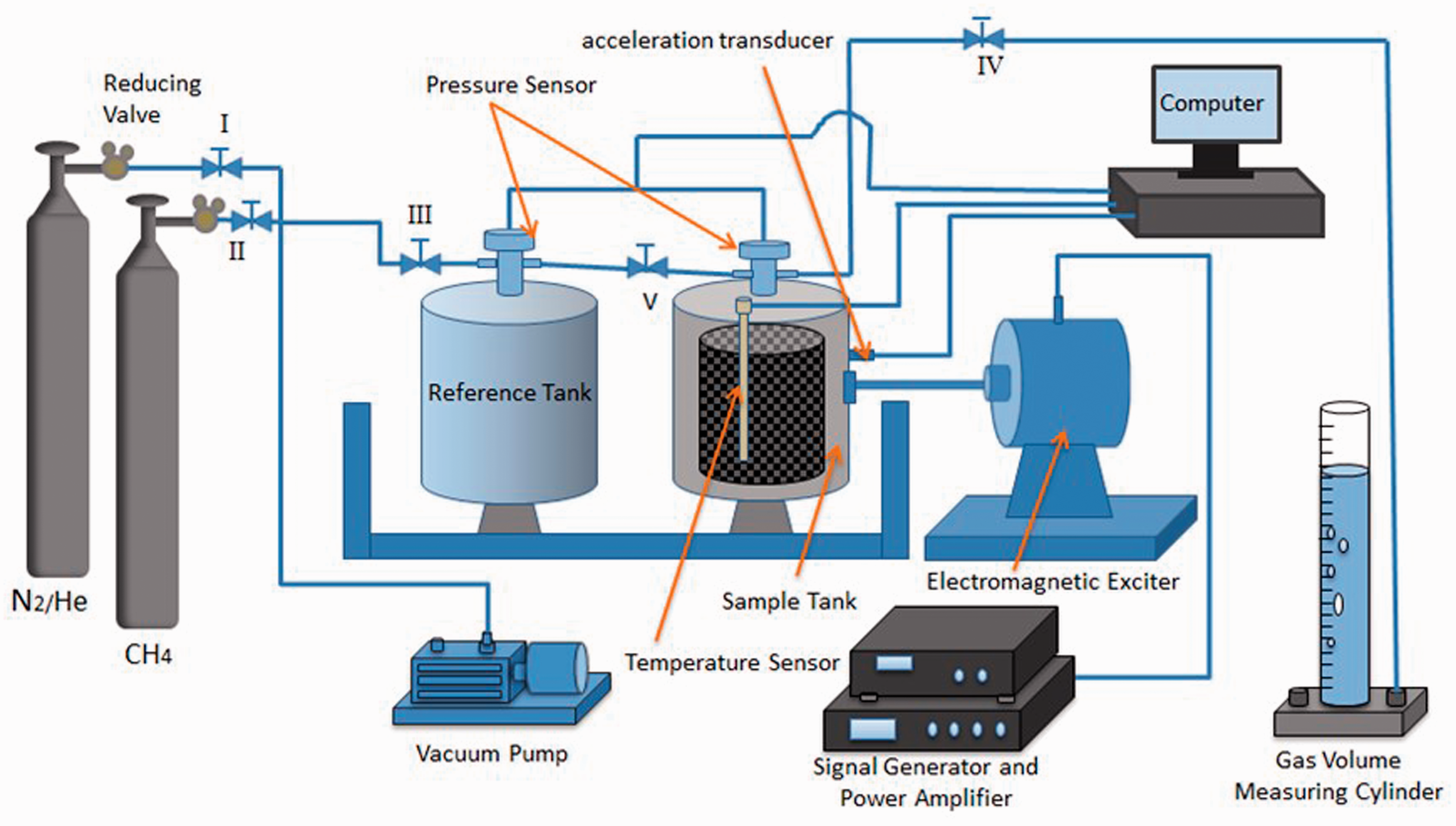
The principle diagram of the experimental apparatus.
Gas adsorption desorption experiment
At coal mines, vibrations that induce coal and gas outburst are mainly due to blasting, drilling, coal cutting by shearers, and tunnelling by road headers. The outburst strength and induction modes of coal and gas outburst accidents examined in the literature (Zhao, 2011) occurred at a vibration frequency range of 5-100 Hz. Thus, mechanical vibration frequencies of 0Hz, 25Hz, 50Hz, 75Hz, and 100 Hz were selected to simulate the effects of vibrations on the coal body to model gas desorption and diffusion in coal particles.
The experimental process was divided into 5 steps:
Temperature, pressure test system debugging. System air tightness test. Close valve IV and open valves I, III, and V after adding 2 MPa helium to the system; then, close the valves and maintain the pressure for 12 h. If the pressure remains steady, good air tightness is achieved. Determination of free volume. Free volume was measured using helium gas before the adsorption and desorption experiments. Conduct gas adsorption experiments. The temperature was maintained at 30°C, the adsorption time was 12 h, and the adsorption equilibrium pressure was 1 MPa. Conduct desorption experiments. When the adsorption equilibrium is balanced, open valve IV to drain the free gas, open the vibration system at predefined frequency and amplitude, and record data for 1 h.
The calculation of the maximum gas desorption quantity
The maximum gas desorption quantity is another important parameter in the law of gas desorption and diffusion in coal and is calculated by the following formula:
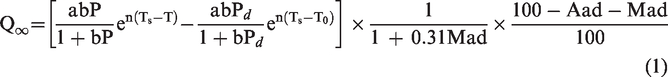
The calculation of gas diffusion parameters
The diffusion coefficient is an important parameter in characterizing the diffusivity of gas in the coal body. For studying the relationship between the diffusion coefficient and vibration frequency, it is helpful to determine the influence of vibration on gas diffusion samples. Based on a dynamic diffusion coefficient, the gas diffusion characteristics of coal samples were quantitatively analysed (Li et al., 2015, 2016b)
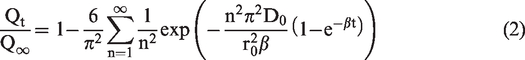
The model assumes that the coal matrix pore system is composed of heterogeneous, multi-scale, distributed pores. The diffusion coefficient is considered a quantity that varies over time:

Pore volume test by MIP
After the adsorption and desorption experiments, coal samples treated with different vibration frequencies were degassed, and some coal samples were reserved for mercury intrusion porosimetry (MIP) measurements to determine pore structure characteristics. MIP was conducted using an AutoporeIV 9500 Instrument (Micrometics, USA).
Results
The influence of mechanical vibration of different frequencies on gas desorption characteristics
The influence of mechanical vibration on gas desorption quantity
At an adsorption equilibrium pressure of 1 MPa, the desorption volume and the gas desorption percentage of coal samples at different vibration frequencies are shown in Figure 2. In the early stages of desorption, the volume of desorption greatly increased and gradually weakened over time. The desorption quantity of vibrating coal samples was larger than that of nonvibration coal samples over the same period, and the desorption amount was largest at 75 Hz. For the 0.25–0.5 mm coal samples, the cumulative gas desorption volume of the vibrating coal samples at vibration frequencies of 25, 50, 75 and 100 Hz increased 2.09%, 9.17%, 16.17% and 10.22%, respectively, compared with the nonvibration coal samples. For the 1–3 mm coal samples, the cumulative gas desorption volume of the vibrating coal samples at vibration frequencies of 25, 50, 75 and 100 Hz increased 9.31%, 13.38%, 26.98% and 16.83%, respectively, compared with the nonvibration coal samples. The desorption percentage is an important parameter reflecting the degree of gas desorption in coal samples. Changes in the desorption percentage of coal samples with different vibration frequencies are shown in Figure 2. As the vibration frequency increased, the gas desorption percentage initially increased and subsequently decreased. For coal samples with particle sizes of 0.25–0.5 mm and 1–3 mm, the desorption percentage is maximized at 75 Hz, and the desorption percentage is minimized at 0 Hz. The gas desorption percentage of the vibration coal sample was larger than those without vibration, which indicated that vibration conditions promoted the gas desorption percentages. However, at the same vibration frequency, the desorption volume and desorption percentage of the 0.25–0.5 mm coal samples were greater than those of the 1–3 mm coal samples.
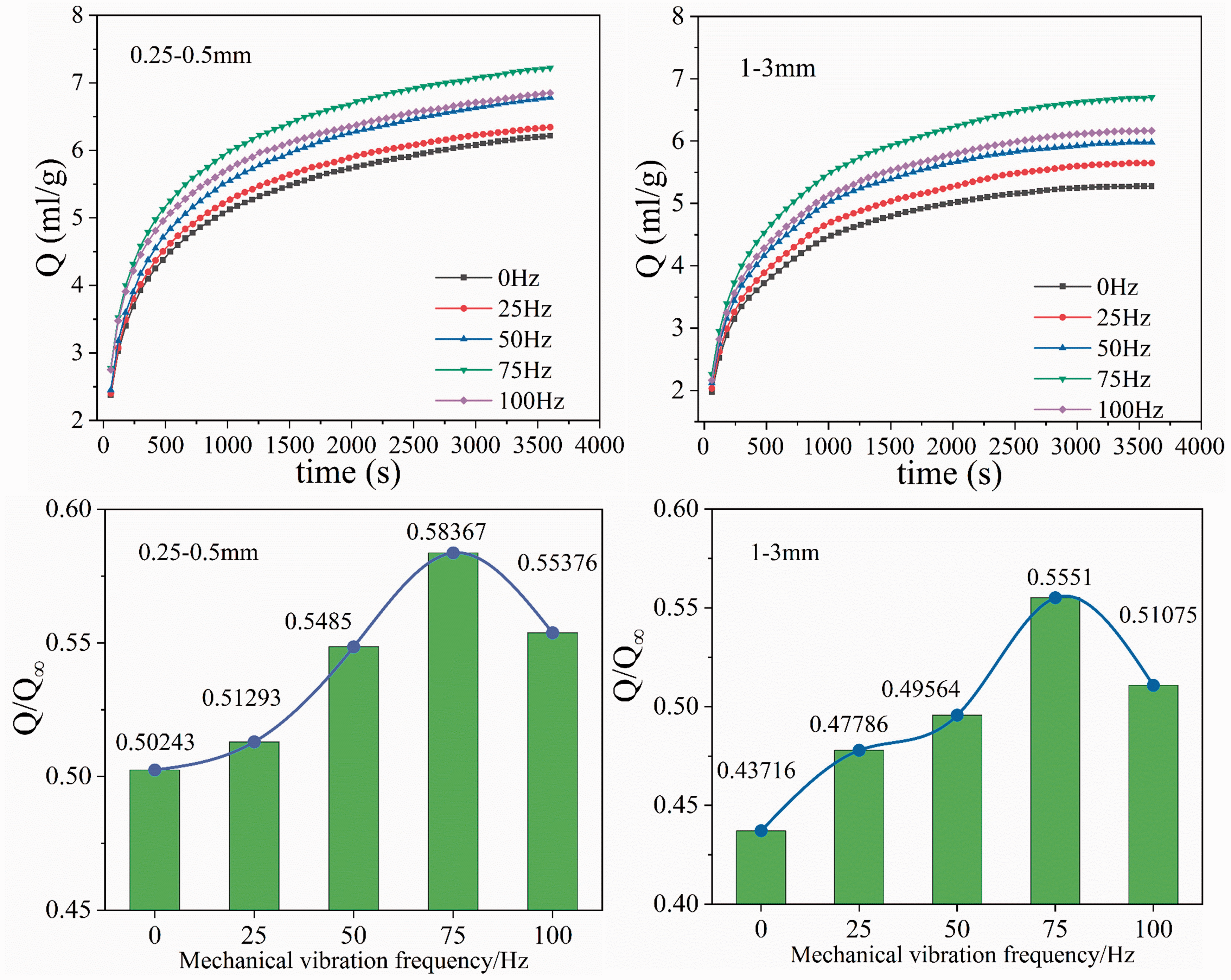
The curves of gas desorption volume and the gas desorption percentage with different vibration frequencies.
The influence of mechanical vibration on gas desorption rate
As shown in the variation curve of the cumulative gas desorption volume in Figure 2, the desorption volume increases rapidly during the early stages of desorption, which indicates that the desorption intensity is large. The variations in coal desorption rates under different vibration frequencies were compared and plotted in Figure 3.
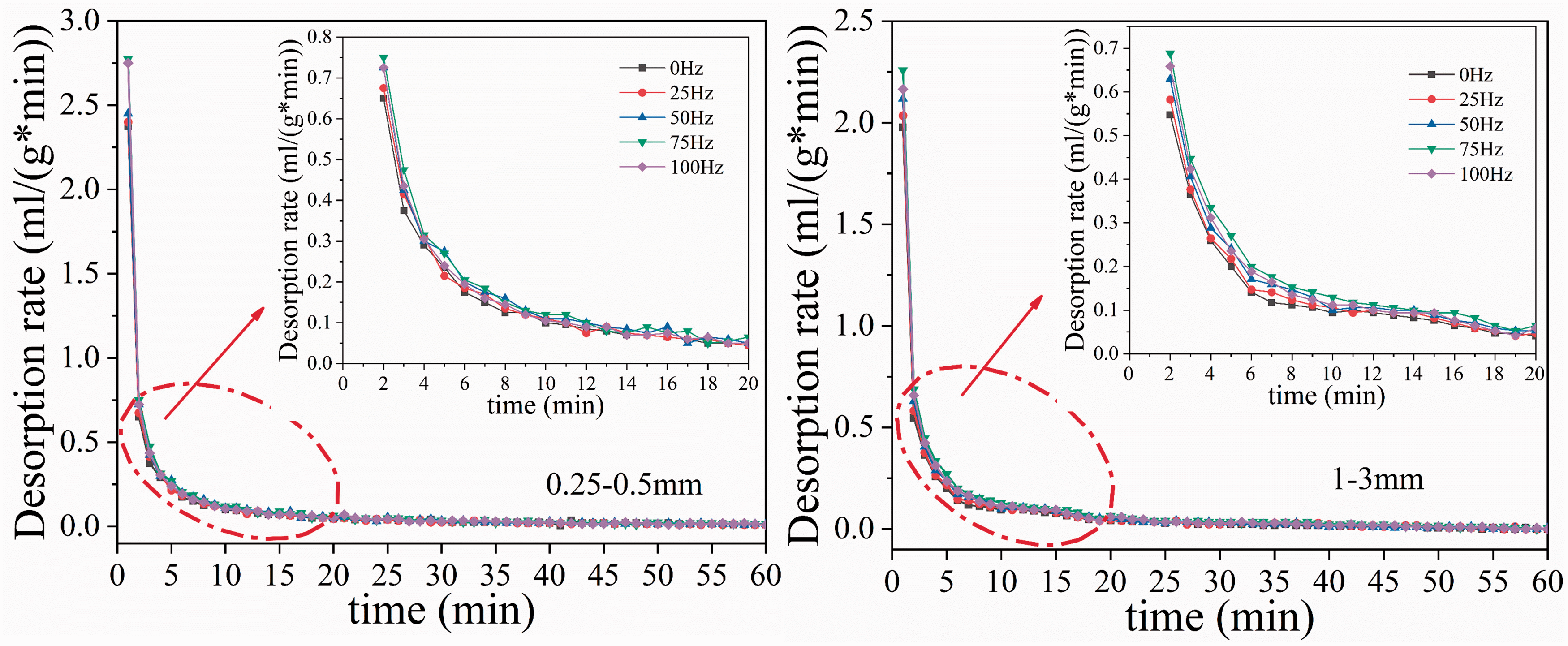
Changes in the gas desorption rate of coal samples with time under different vibration frequencies.
Figure 3 shows that the desorption rate of all coal samples decayed rapidly during the early stages and gradually stabilized with time. The coal samples of 0.25–0.5 mm and 1–3 mm have similar variation characteristics. The desorption rate of all coal samples can be divided into three stages with the extension of desorption time. In the first stage (0–2 min), the desorption rate sharply dropped; this is identified as a rapid decay period. Then, in the second stage (2–10 min), the desorption rate continued to decline, but the degree of decay weakened; this was identified as a transition decay period. Finally, in the third stage (after 10 min), the desorption rate stabilized; this was identified as a stable period. During rapid decay, both the 0.25–0.5 mm and 1–3 mm coal samples had a maximized desorption initial rate at 75 Hz. The initial desorption rate increased with increasing vibration frequency at 0, 25, 50, and 100 Hz for both particle sizes. During the transition decay period, the gas desorption rates of each vibrating coal sample and nonvibrating coal sample were similar and continued to decay. This indicated that the surface diffusion process gradually changed to pore diffusion inside the coal matrix with the extension of desorption time. Vibration constantly affects the pore structure within the coal particles, which in turn affects the gas diffusion process in coal particles and leads to fluctuations in desorption rates. During the stable period, the desorption rates of all coal samples decayed slowly, and the numerical values of the desorption rates of the five frequency coal samples with the same particle size were close to each other. However, the gas desorption rate of vibrating coal samples was larger than that of 0 Hz coal samples overall. It can be seen that vibration has a significant impact on the gas desorption rate, especially the effect on the initial desorption rates, which is more significant.
Effects of mechanical vibration of different frequencies on gas diffusion in coal particles
To study the influence of vibration conditions of different vibration frequencies on the diffusion parameters of the gas diffusion process in coal, the data of this experiment are fitted with a dynamic diffusion coefficient model, and the relevant diffusion parameters are obtained for comparative analysis. The coal sample sizes used in the experiment were 0.25–0.5 mm and 1–3 mm. We used the sieve method to calculate the average particle sizes of 0.25–0.5 mm and 1–3 mm coal samples, which were 0.3764 mm and 1.8968 mm, respectively, by taking the mass ratio of coal particles with different particle size ranges as the weight. The fitting results are shown in Figure 4, and the corresponding parameters in the model are shown in Table 2.
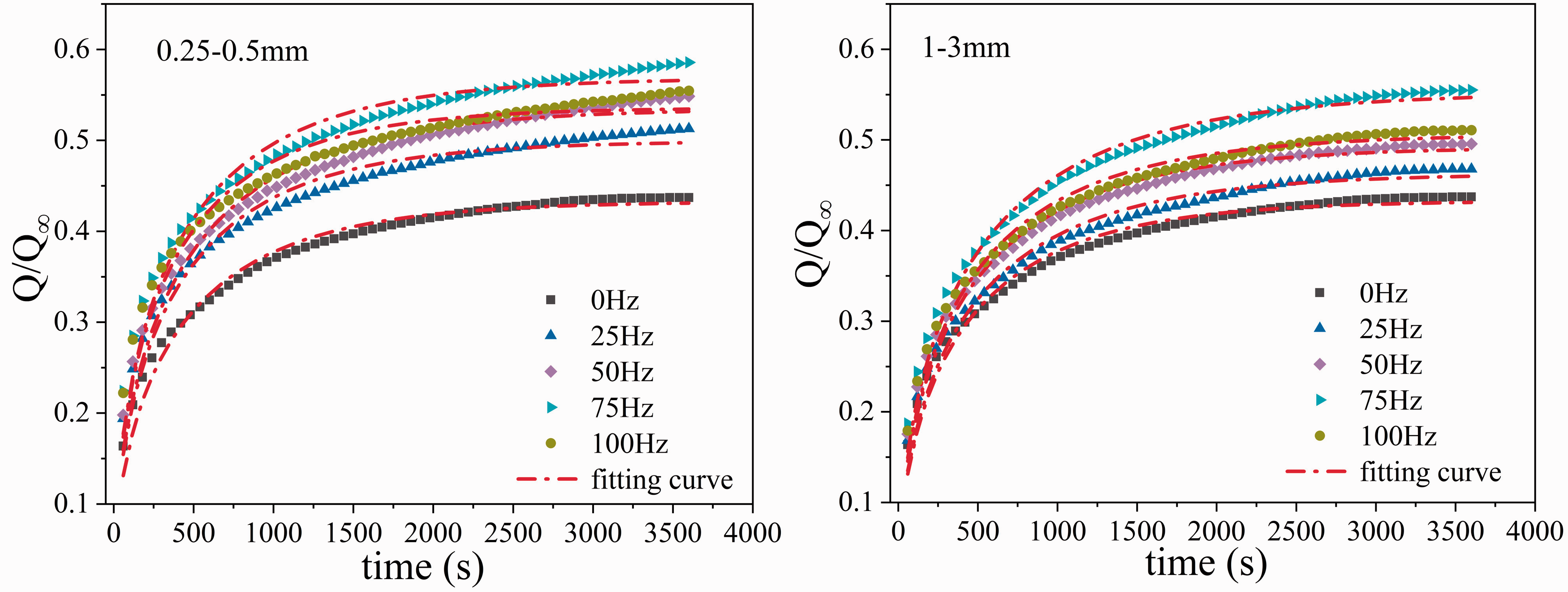
Fitting results of gas desorption data at different vibration frequencies based on dynamic diffusion coefficient model.
Gas diffusion parameters of coal samples under different vibration frequencies based on dynamic diffusion coefficient model.
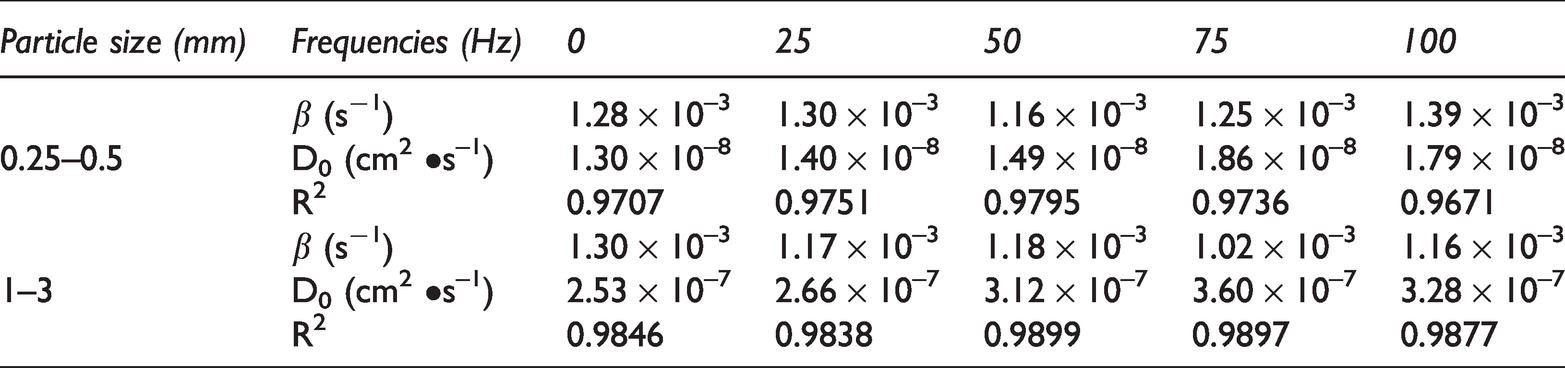
Figure 4 shows that the desorption data of the 0.25–0.5 mm and 1–3 mm coal samples at different vibration frequencies have a good fit with the dynamic diffusion coefficient model, as well as their correlation coefficients in the ranges of 0.9671–0.9795 and 0.9838–0.9899, respectively. This result indicated that the dynamic diffusion coefficient model can well describe the characteristics of gas desorption and diffusion in vibrating coal samples at different frequencies. As shown in Table 2, under experimental conditions, vibration affected the initial gas diffusion coefficient D0 and the attenuation coefficient of the diffusion coefficient β. As the vibration frequency increased, D0 gradually increased. For the 0.25–0.5 mm coal samples vibrated at 25Hz, 50Hz, 75Hz, and 100 Hz, the initial gas diffusion coefficients were 1.08, 1.15, 1.43, and 1.38 times that of the 0 Hz coal sample, respectively. For the 1–3 mm coal samples vibrated at 25Hz, 50Hz, 75Hz, and 100 Hz, the initial gas diffusion coefficients were 1.05, 1.23, 1.42, and 1.30 times that of the 0 Hz coal sample, respectively. The results showed that vibration enhanced the diffusion of gas on the outer surfaces of coal particles. For the 0.25–0.5 mm coal samples, β initially decreased and then increased as the vibration frequency increased, having the lowest values at 50 Hz and the highest values at 100 Hz. For the 1–3 mm coal samples, β initially decreased and then increased as the vibration frequency increased, having the lowest values at 75 Hz and the highest values at 0 Hz.
To study the relationship between D0, β, and vibration frequency, D0 and β were fitted and analyzed with vibration frequency. The fitting results are shown in Figure 5. D0 showed a linear relationship with the vibration frequency, and the correlation coefficients of the 0.25–0.5 mm and 1–3 mm coal samples reached 0.8599 and 0.8233, respectively. Although the D0 values of both particle size coal samples at different frequencies reached a maximum value at 75 Hz, the overall D0 increased with increasing frequency. The reason why both reached a maximum at 75 Hz may be because the vibration at 75 Hz is close to the natural frequency of the coal sample and resonance occurs. A quadratic polynomial relationship between β and vibration frequency was observed with a correlation of 0.6986 for 0.25–0.5 mm coal samples. For the 1–3 mm coal samples, the correlation coefficient of the fit between b and vibration frequency was 0.7251. Overall, β initially decreased and then increased with vibration frequency.
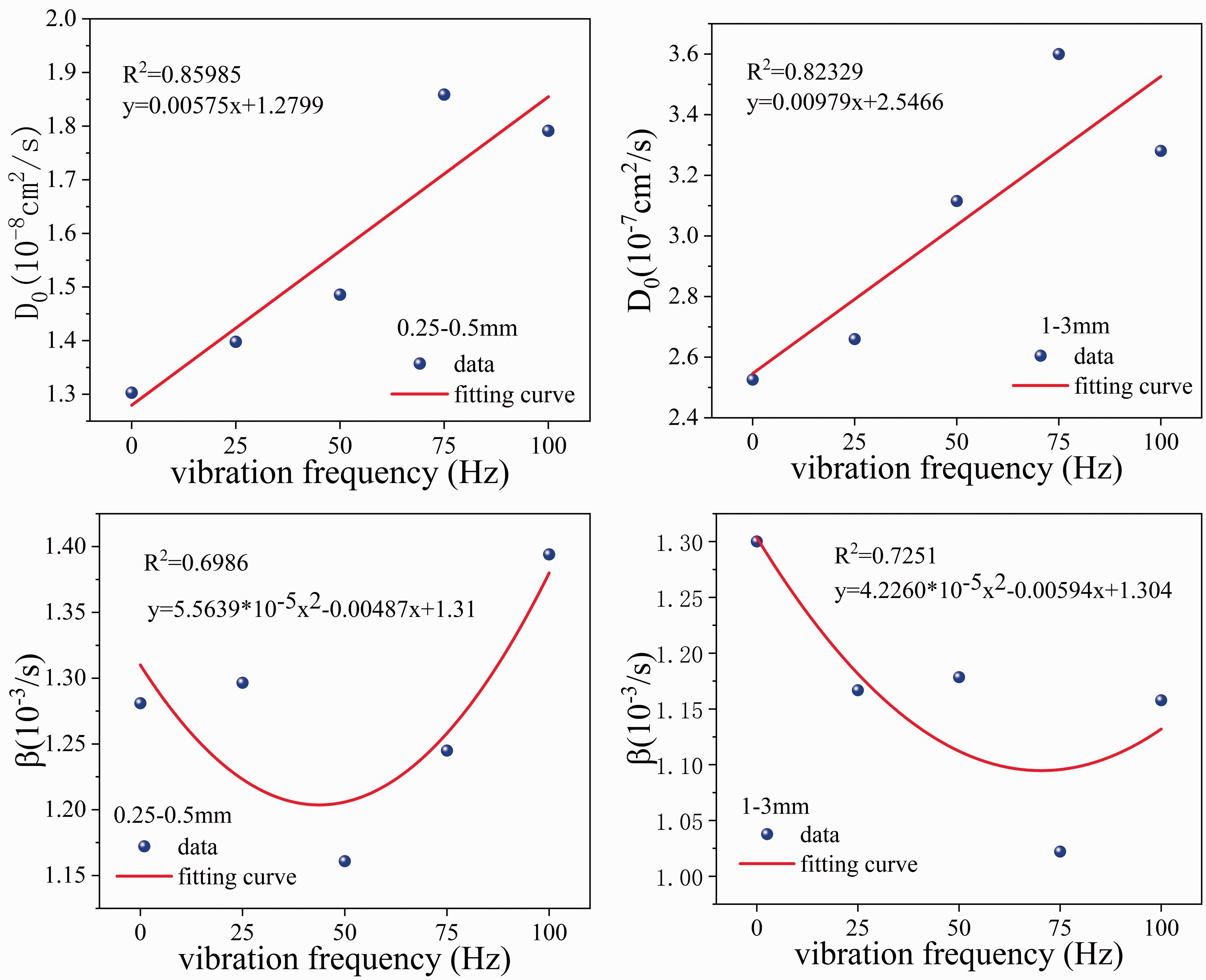
D0, β and vibration frequency fitting diagram.
As shown in Table 2, the gas diffusion coefficient function D(t) of 0.25–0.5 mm and 1–3 mm coal samples at different vibration frequencies can be obtained. Figure 6 shows the relationship between dynamic diffusion coefficient and time of two particle size coal samples at different vibration frequencies. It can be seen that the diffusion coefficients of both the vibrating coal sample and the non-vibrating coal sample decrease with time, but the diffusion coefficients of the vibrating coal sample at each moment are greater than that of the non-vibrating coal sample. This shows that in the desorption process under experimental conditions, the diffusion capacity of gas in the vibrating coal sample is stronger than that of the non-vibrating coal sample. For 0.25–0.5 mm coal samples, after desorption for a period of time, the diffusion coefficients of vibration coal samples of 50 Hz and 100 Hz appear to cross, which is because the values of β are the minimum and the maximum value respectively. The diffusion capacity declines the slowest at 50 Hz and the fastest at 100 Hz. For 1-3mm coal samples, The 75 Hz vibrating coal sample is always larger than other vibrating frequency coal samples. Moreover, the diffusion coefficients of coal samples of other frequencies at the same moment increase with increasing vibration frequency. For the 0.25–0.5 mm and 1–3 mm coal particle samples, the gas diffusion coefficients of the coal samples with different frequencies gradually move close to each other with the extension of desorption time, indicating that the influence of vibration on gas diffusion in coal is weakened as the desorption time increases.
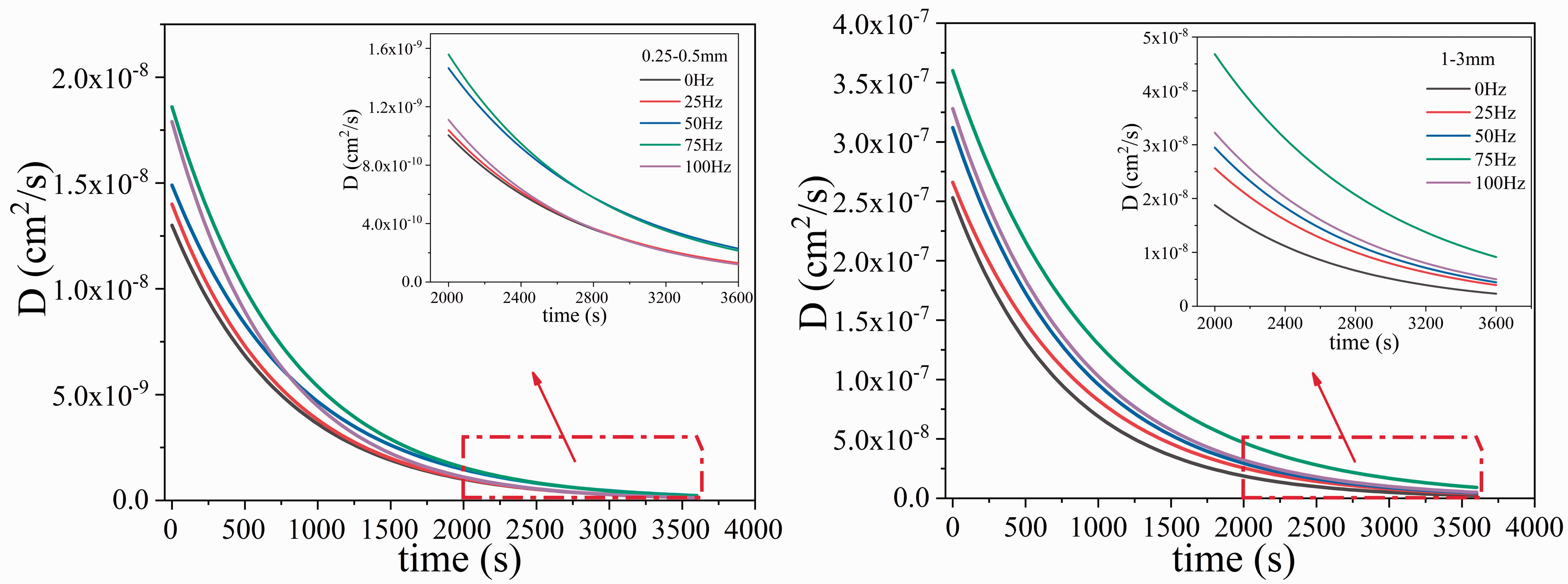
Dynamic diffusivity function D(t) of gas diffusion in coal samples with different vibration frequencies.
Discussion
Effect of mechanical vibration on gas molecules adsorbed in coal and its influence on desorption and diffusion
The adsorption of gas molecules by coal is a physical process, whereas the force causing this physical adsorption is the van der Waals force (Cai et al., 2013; Peng et al., 2018; Wang et al., 2019). On the one hand, at the beginning of the desorption process, due to exposure to vibration conditions, the coal matrix and the gas molecules adsorbed correspondingly on the matrix surface are subjected to periodic movement. In each movement cycle, when the coal sample tank and the coal sample that have adsorbed gas reach the maximum amplitude, the coal sample tank is immediately affected by the reverse acceleration (Zhao, 2011). However, the coal sample will maintain the original motion state due to inertia, resulting in relative acceleration between the coal sample and the coal sample tank, and the relative displacement of the coal sample to the coal sample tank. Different coal particles have different masses, and the inertia between coal particles is different, resulting in relative sliding and friction occurring between coal particles. The gas molecular layer adsorbed on the outer surface of coal particles will also be subjected to extrusion, collision, and friction, resulting in the fracture of the gas molecular layer. Under constant amplitude conditions, the faster the vibration frequency is, the shorter the period is, the greater the relative acceleration between coal particles and the coal sample tank is, and the relative acceleration between coal particles is also increased. Therefore, the faster the vibration frequency is, the more intense the relative sliding between coal particles is, the stronger the friction and collision on the outer surface of coal particles are, and the greater the possibility of gas molecular layer fracture is. On the other hand, when relative sliding occurs between the coal particles, the gas molecules adsorbed on the inner and outer surfaces of the coal particles will also generate relative acceleration with the coal particles due to inertia. At this time, gas molecules are subjected to inertial force relative to coal particles. The van der Waals force that causes physical adsorption is a weak interaction force (Wang, 2016; Wang et al., 2019). When the resultant force of inertial force, friction force, mechanical shear force and other external forces on gas molecules in the direction of van der Waals force is greater than that of van der Waals force, some gas molecules quickly “throw out” from the surface of coal particles and become free state. Under a constant amplitude, the higher the vibration frequency is, the greater the relative acceleration between the gas molecule and the coal particle, the greater the inertial force generated by the gas molecule relative to the coal particle, and the more likely the gas molecule is to be thrown away.
These two mechanisms make it easier for the adsorbed gas molecules to spread into the free space of the coal sample tank at the start of the desorption process. The gas desorption rate, desorption quantity and diffusion coefficient of the vibrated coal samples were greater than those of the nonvibrated coal samples during this initial period. This mechanism is consistent with the experimental results shown in Figure 3, wherein the vibration coal sample desorption speed within the first minute was greater than that of the nonvibrated coal sample. Similarly, the initial effective diffusion coefficient of the vibration coal samples was greater than that of the nonvibrated coal sample (the difference was more pronounced for higher vibration frequencies). These results agreed with those shown in Figure 5, and the initial effective diffusion coefficient of vibrating coal samples was larger than that of nonvibrated coal samples.
As the desorption process continues, gas desorption gradually shifts from the outer surface of coal particles to the inner pore surface. In the noninertial system with coal particles as a reference, the gas molecules adsorbed on the outer surface of coal particles are not only subjected to inertial force but also subjected to friction and extrusion caused by the relative sliding of adjacent coal particles, which is beneficial to the desorption of gas molecules from the bondage of van der Waals forces and forms a strong throwing effect. However, the gas molecules adsorbed on the inner surface of coal particles cannot receive external friction and extrusion, and the “throwing effect” is weak. Therefore, we believe that the “throwing effect” inside coal particles is weaker than that of outside coal particles. Therefore, the “throwing effect” mainly affects the desorption process of gas adsorbed on the outer surface of coal particles in the early stage of desorption.
Change in the pore volume of coal particles caused by mechanical vibration and its influence on gas desorption and diffusion
According to the B.B. Hotdot pore diameter classification system, coal matrix pores can be divided into four categories: micropores (pore diameter d < 10 nm), transition pores (10 nm < d < 100 nm), mesopores (100 nm < d < 1000 nm) and macropores (d > 1000 nm) (Fu et al., 2017). Generally, micropores and transition pores provide storage space for gas, whereas mesopores and macropores provide channels for gas diffusion migration (Li et al., 2012a; Yao et al., 2009).
Mechanical vibration has a mechanical effect on the coal body, inducing changes in the fracture pores. Past studies have indicated that vibration had a “softening effect” and a “damage effect” on coal (Ren et al., 1997, 1998), stress waves produced by mechanical vibration accelerated the development of cracks (Song et al., 2018), and the porosity of coal increased with increasing vibration frequency (Li et al., 2016a). These studies indicated that mechanical vibration is beneficial to the development of diffusion, seepage, and desorption paths of gas in coal.
Vibration can produce periodic extrusion stress on the coal matrix (Li et al., 2016a; Pan et al., 1999; Ren et al., 1998), which will cause the parts of the coal matrix skeleton with weak anti-extrusion ability to collapse or fracture. The pore volume decreases due to the collapse of the coal matrix skeleton blocking some of the pores. At the same time, the pore volume increases due to the expansion and development of pore fractures caused by the fracture of the coal matrix skeleton. The matrix pores are also squeezed due to the deformation of the part of the coal matrix skeleton caused by periodic extrusion stress, resulting in the bulge or depression of pore surfaces, which then leads to a decrease in pore diameter.
Figure 7 shows the pore volume of macropores, mesopores, transition pores, and micropores in the 0.25–0.5 mm and 1–3 mm coal samples at different vibration frequencies as measured by MIP. Overall, the pore volume and volume proportion of macropores and mesopores of the coal samples after vibration were larger than those of the nonvibrated coal samples, both for the particle size of 0.25–0.5 mm and for 1–3 mm. However, the pore volume and volume proportion of transition pores decrease after vibration. The reason may be that the deformation and damage of the coal matrix skeleton caused by vibration lead to an increase in the pore size of the original transition pore and develop into mesopores and macropores. At the same time, the deformation and failure of the coal matrix skeleton also lead to the expansion and development of mesopores and macropores, thus increasing the volume of both. For the 0.25–0.5 mm coal samples, the micropore volume and proportion of other coal samples are larger than those of nonvibration coal samples except for the 100 Hz coal sample. For the 1–3 mm coal samples, the micropore volumes of the other coal samples are larger than those of the nonvibration coal samples except for the 100 Hz coal sample. However, the volume proportion of micropores in the 50 Hz and 100 Hz coal samples decreases. The possible reason for the increase in micropore volume is that the deformation of the coal matrix skeleton caused by vibration will squeeze part of the pores, which leads to a decrease in pore diameter and an increase in micropore volume.
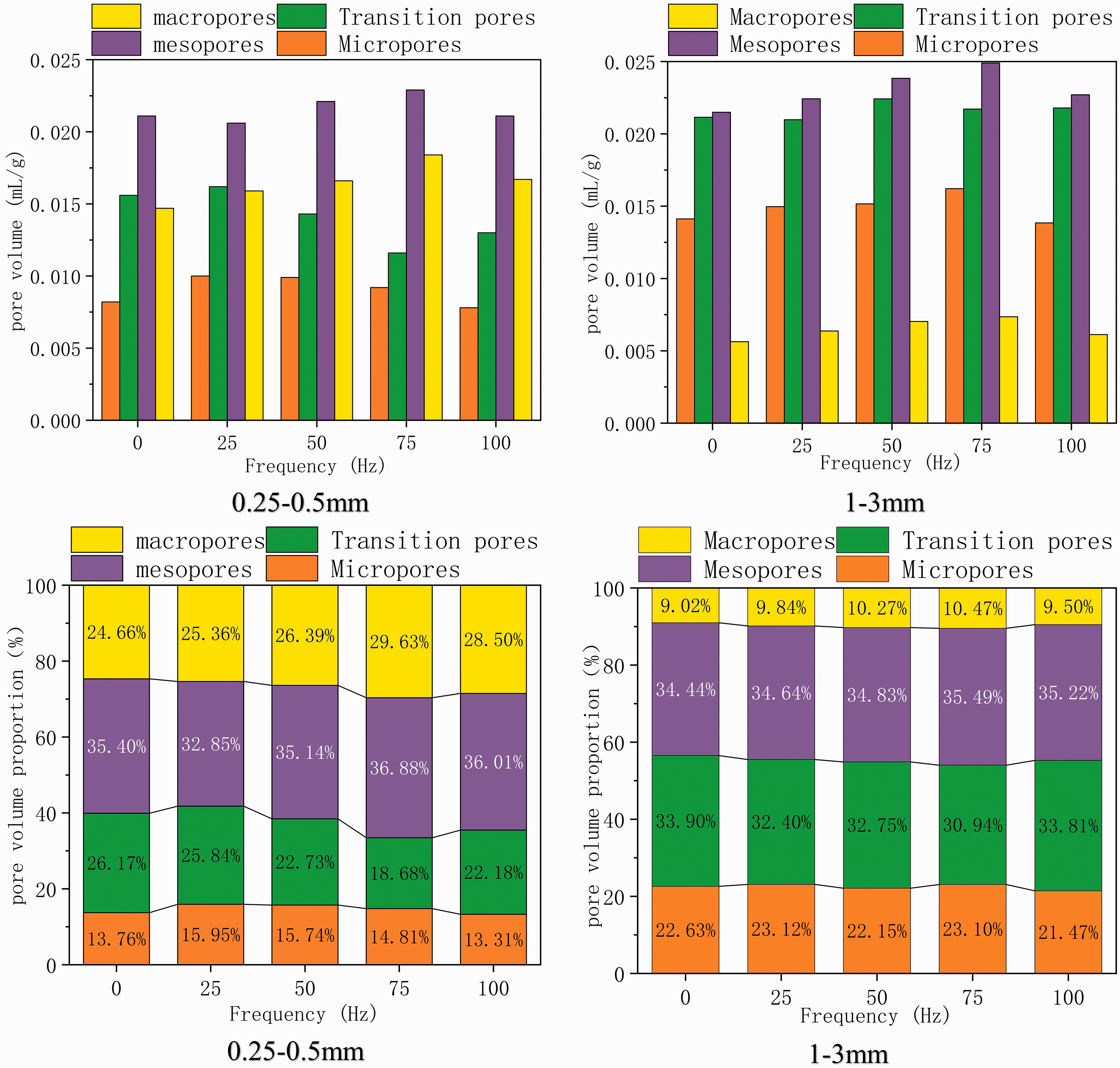
Comparison of pore volume and proportion of pore volume in coal samples with different vibration frequencies.
Figure 8 shows the relationship between the total pore volume of pores with diameters less than 10,000 nm and the vibration frequency. For the 0.25–0.5 mm coal samples, the total pore volume (3 nm<d < 10000 nm) of the vibrating coal samples was larger than that of the nonvibrated coal samples except at 100 Hz; the maximum value was observed at 50 Hz. For the 1–3 mm coal samples, the total volume of all vibrated coal samples was larger than that of the nonvibrated coal samples. The total pore volume initially increased and then decreased with increasing vibration frequency, which roughly fit a quadratic polynomial relationship with correlation coefficients of 0.9902 and 0.8973, respectively. The total pore volume first increased and then decreased with increasing vibration frequency for particle sizes of 0.25–0.5 mm and 1–3 mm. The total pore volumes of the 0.25–0.5 mm and 1–3 mm coal samples after vibration at different frequencies were fitted with the vibration frequencies. The total pore volume and vibration frequencies approximately conformed to a quadratic polynomial relationship, and their correlation coefficients reached 0.9902 and 0.8973, respectively.
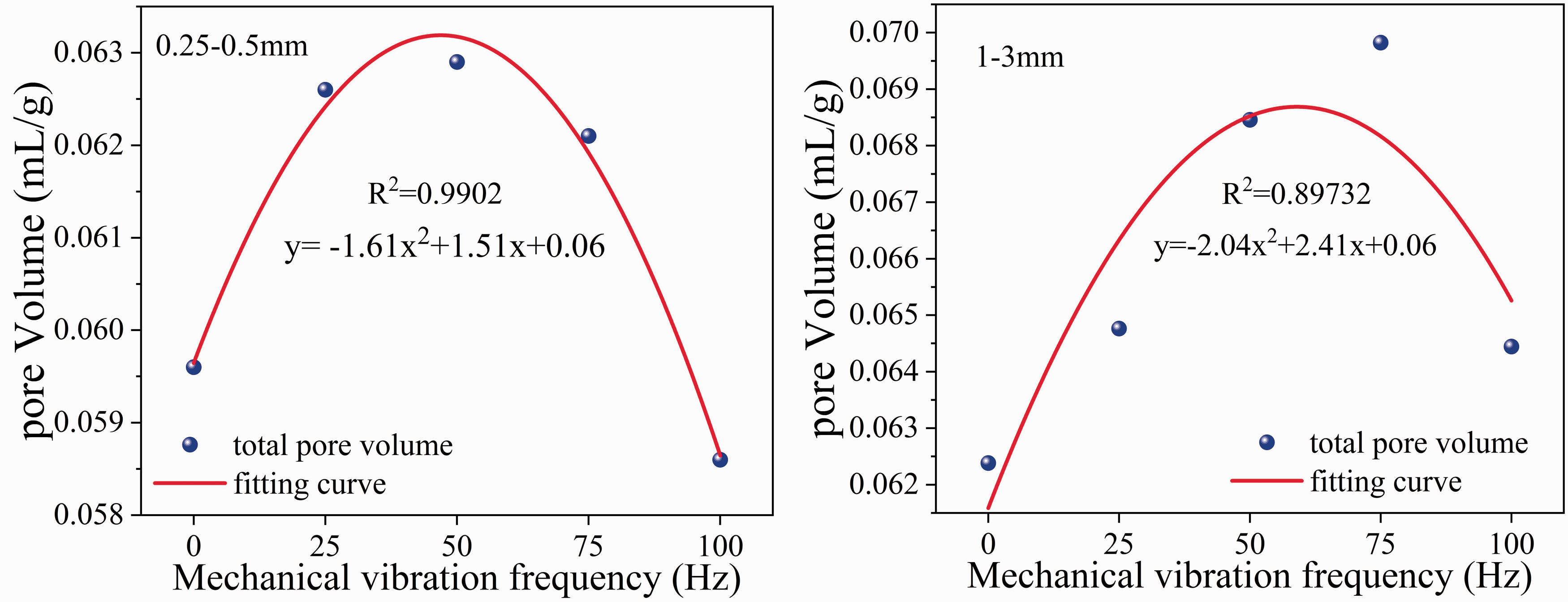
Fitting curve between total pore volume and vibration frequency.
In this study, regardless of whether the particle size was 0.25–0.5 mm or 1–3 mm, the volume of macropores and mesopores of all vibrated coal samples increased, which increased the number of diffusion paths. Thus, the diffusion resistance decreased, which led to an increase in the diffusion coefficient and the gas diffusion rate. Whether the particle size is 0.25–0.5 mm coal sample or 1–3 mm coal sample, coal pore fissures rapidly developed when the vibration frequency was 75 Hz, because of resonance so that the gas diffusion resistance decreased more rapidly than those of coal samples vibrated at other frequencies. This indicated that there was a maximum diffusion rate at 75 Hz, and the D value was larger than those of the other coal samples. For the 0.25–0.5 mm coal samples, the diffusion coefficient curves at 50 Hz and 100 Hz crossed with the extension of time. The reasons for this are that the mesopore and macropore volume proportion of the 100 Hz vibrated coal samples was more than 50 Hz at the initial stage of desorption, so the gas diffusion resistance of the 100 Hz coal sample was weak. As desorption continued, the gas desorption process gradually shifted from gas diffusion in the macropores and mesopores to gas diffusion in the micropores and transition pores. However, the micropore and transition pore volume proportion of the 100 Hz vibrating coal samples was less than 50 Hz, and the gas diffusion resistance was strong. Thus, the diffusion coefficient of the 100 Hz coal sample decays faster than that of the 50 Hz coal sample. Consequently, for all coal samples, the diffusion resistance increased, decreasing the gas desorption rate and diffusion coefficient to values similar to those in the later stages of desorption.
Energy exchange during mechanical vibration
In the process of vibration, the coal sample is in a steady-state forced vibration state (Li et al., 2010, 2016a). The coal sample tank and coal sample are regarded as a vibration system with mass m. During the vibration process, the vibration system works by overcoming the viscous resistance and increases the internal energy of the system at the same time. The periodic driving force and the viscous resistance are as follows (Bai, 1984)



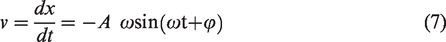
The frequency and amplitude of steady-state forced vibration have the following relationship (Zhao, 2011)
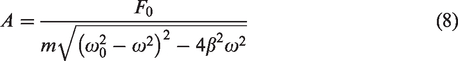
The amplitude in the vibration system decreases with increasing frequency when the power is constant. Therefore, the experimental conditions of a constant amplitude and different frequencies are produced by adjusting the power amplifier to increase the driving force. However, in the process of steady-state forced vibration, the general mechanical energy is not conserved except resonance because part of the energy is converted into the internal energy of the system during the vibration process (Bai, 1984; Zhang, 2016). The following is a specific analysis of the functional relationship.
The work done by the driving forces in a period during steady-state forced vibration is (Li et al., 2010)

The average kinetic energy of the system can be expressed as follows:
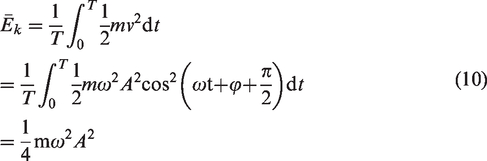
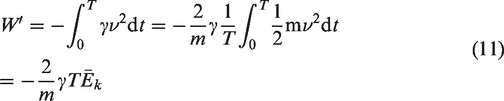
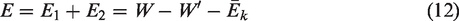
Gas desorption is a complex endothermic process (Feng et al., 2017; Yang et al., 2018), and mechanical thermal effects can be generated during the propagation of mechanical waves generated by vibrations (Jiang et al., 2008, 2015). According to the above analysis, during mechanical vibration, the work done by the vibration excitation system is transferred to three parts: the kinetic energy of the coal sample tank and the coal sample; the energy needed to overcome system resistance; and the energy transferred into the coal sample and the gas internal energy through continuous friction collisions between the coal particles and the gas molecule layers. At the same amplitude, the higher the vibration frequency is, the greater the power provided by the vibration excitation system, and the more work done on the coal sample tank, coal sample and gas. Because the system resistance was constant, the higher the vibration frequency was, the greater the internal energy of coal particles and gas molecules, and the more intense the thermal movement of gas molecules. Therefore, gas molecules in the adsorbed state are more likely to obtain energy higher than the adsorption potential and then desorb from the coal matrix surface. The energy exchange during the vibration process provided energy for gas desorption and promoted the gas desorption process in coal.
Conclusions
In this paper, coal samples with particle sizes of 0.25–0.5 mm and 1–3 mm were used for the study, and desorption-diffusion experiments of coal samples under different frequency vibration conditions were carried out. A dynamic diffusion coefficient model was used to quantitatively analyze the parameters of coal gas diffusion. Additionally, the mechanism of vibration affecting gas desorption and diffusion was discussed. The conclusions are as follows:
As the vibration frequency increased, the gas desorption percentage of the coal sample initially increased and then decreased. The gas diffusion coefficient of vibrating coal samples was larger than that of nonvibrated coal samples. Mechanical vibration was beneficial to gas desorption and diffusion in coal particles. The initial gas diffusion coefficient increased with increasing vibration frequency, but the attenuation coefficient of the diffusion coefficient showed a quadratic polynomial relationship that initially decreased and then increased with vibration frequency. The influence of vibrations on gas desorption and diffusion in the coal body was as follows: the “thrown effect” of vibrations on gas molecules in the initial stages of desorption, energy conversion during mechanical vibration, and changes to pore structures. There exists a frequency that causes coal resonance, which results in the most dramatic effect of vibration on gas desorption and diffusion in coal. Mechanical vibration can promote gas desorption and diffusion in coal, which may lead to increased gas concentrations in local coal seams, which can induce gas accidents.
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
Footnotes
Acknowledgments
We sincerely thank the anonymous reviewers for improving the quality of article. We would also like to thank National Natural Science Foundation of China and the Fundamental Research Funds for the Central Universities for providing funding.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant no. 51574124) and the Fundamental Research Funds for the Central Universities (grant no. 3142015020).
