Abstract
Fibrous textiles readily absorb and desorb ambient odors. However, information on the composition and dynamic analysis of tobacco smoke and hot pot odors on fabrics during desorption is limited. This study used gas chromatography–mass spectrometry to analyze the desorption components of cotton, wool, and polyester fabrics exposed to these two odors, respectively. Then, a dynamic diffusion fabric structure model demonstrated the effect of airflow velocity and fabric porosity on nicotine desorption. Furthermore, we proposed mass diffusion coefficients with different molecular weights. The results showed that cotton fabrics with tobacco smoke released many low molecular weight compounds, while with wool fabrics significantly fewer compounds were detected than for the other two fabrics. Notably, 3-ethenylpyridine, a marker of tobacco smoke, was not detected in wool fabrics. For hot pot odor, cotton fabrics released more hexanal, nonanal, and anethole than wool and polyester, while wool fabrics released many β-pinenes. The numerical results of the dynamic model showed that the air inlet velocity significantly affects the nicotine concentration in the fabric. Meanwhile, the concentration of nicotine in fabrics with lower porosity decreased faster. A lower mass diffusion coefficient will cause odors to remain in the fabric. This study aimed at the composition and the dynamics of odor in fabrics and offers essential information and simple models for reducing unnecessary washing of textiles and odor resistance textile design.
Keywords
Introduction
Tobacco smoke and hot pot odor are the two common annoying ambient odors that can be easily absorbed and desorbed by fabrics. Tobacco smoke desorbed by textiles is also defined as “thirdhand smoke.” It can cause health problems for non-smokers. 1 The textile is a carrier for tobacco smoke to human skin during the process.2,3 Hot pot, a popular Chinese food, often retains odors in fabrics for a long time. In addition to causing people to be uncomfortable, it can lead to unscheduled washing behaviors, resulting in additional carbon and chemical emissions. Harmful compounds in odors can accumulate in textiles, be slowly released, and continue to cause harm to the human body. 4 Therefore, there is a need for a simple and low-cost deodorization technology to remove tobacco smoke and hot pot odor from textiles. The study of tobacco smoke and hot pot odor from various fabrics has made some progress in analyzing odor components and the difference between fabrics.5–8 Nonetheless, the fabric odor desorption research is minimal. The desorption process is also a dynamic process influenced by odorous molecules, fiber properties, fabric structure, and the external environment. Information on the composition and dynamics of tobacco smoke and hot pot odors from fabrics during desorption is still lacking.9,10
The ability of a fabric to absorb volatile compounds is related to the physical and chemical properties of the fibers that make it up.3,10,11 Natural fibers absorb ambient odors more capably than synthetic fibers and take longer to desorb.7,11,12 At the same time, the adsorption behavior of volatile compounds by fabrics is related to the compounds’ content, structure, and characteristics, such as polarity and aromaticity.6,13,14 McQueen has successively investigated the body odor intensity of cotton, wool, and polyester. The results showed that polyester has the highest odor intensity. It is concluded that the fiber moisture absorption is negatively correlated to the body odor strength from fabric. The lipophilicity of polyester increases the adsorption of volatile compounds on its surface.15,16
Furthermore, the adsorption and desorption of volatile compounds by fabrics are related to the mass, volume, surface area of the fabric, 17 and air velocity, temperature, and humidity.9,17,18 Petrick studied the nicotine adsorption and desorption kinetics in cotton, nylon, polyester, and other materials. They found that nicotine desorption in the fabric is influenced by relative humidity (RH), air exchange rate, and fabric type. 14
Existing models of desorption dynamics suggested that desorption time and fabric volume fraction (i.e. porosity) affect odor deposition and release from textiles. Some studies suggested that porous materials with larger pore sizes may have higher adsorption rates for tobacco smoke. 19 Besides, external boundary conditions (airflow velocity, ventilation time, etc.) can also significantly affect the odor concentration in the fabric. 18 The fabric has a porous structure, and the airflow speeds up the desorption of odorous compounds by the Johannes Diderik van der Waals force. That viscosity significantly affects the airflow rate, and time will affect the effect of air velocity. 18 Although a few studies have been performed on the initial and boundary conditions of the fibrous porous material, there are still some general porous material discussions.20–22 All the results show that the boundary and initial conditions significantly impact the mass transfer inside the porous media. Zhu has Engineered Water Nanostructures (EWNS) that take advantage of the Reactive Oxygen Species (ROS, which are made up of hydroxyl and superoxide radicals), to remove odors produced by smoking, cooking, and sweating. 23 Current studies have not systematically considered the effects of these factors on the desorption of odor compounds from fabrics. 14
The literature review showed that an odor deposition and desorption dynamic model and analysis of the fibrous porous media are still unavailable. The fiber and fabric structure properties, such as fiber diameter and volume fraction effect, are unclear. The tobacco and hot pot components vary depending on the sample preparation methods.
In this study, we first detected the desorption components of cotton, wool, and polyester fabrics after exposure to tobacco smoke and hot pot odor. Then, we analyzed the concentration changes of nicotine, a typical substance in tobacco smoke, based on a fiber density function and a dynamic model of diffusion in porous materials. The model also discussed the effects of boundary conditions, fabric fiber volume fraction, and odorous molecular diffusivity.
Experimental and dynamic models
Materials
Table 1 shows the detailed parameters of fabrics used in this study. Cotton, wool, and polyester specimens were purchased from the Grace Group, Hangzhou, China. To enrich as many odor molecules as possible in the fabric, terry towel fabrics were used. Hongshuangxi cigarette (the tobacco tar 11 mg, sourced from Shanghai Tobacco Group Co., Ltd., Shanghai, China) and Haidilao hot pot soup base (the net content is 150 g per bag, sourced from Yihai Food Co., Ltd., Shanghai, China) were utilized for the experiments of adsorption.
Experimental fabric information.

Samples preparation and testing
Fabrics were prewashed and dried three times using program 5A in an Electrolux Wascator FOM71 CLS standard washing machine (Electrolux Laundry Systems, Ljungby, Sweden) according to ISO 6330-2021. 24 The fabrics were cut to a preset size, and then the STIK CTH1-150B constant temperature and humidity box (STIK Co., Ltd., Shanghai, China) was used, kept for 24 h in a standard environment (20 ± 2 °C, 65 ± 5% RH) to balance humidity and temperature. In order to eliminate interference from other factors, all samples were tested three times, and the median value was taken as the experimental result.
Tobacco odor adsorption and analytical protocol
As Figure 1 shows, three pieces of fabric were hung equidistantly at the top of a self-made closed suspended adsorption chamber (50 cm × 50 cm × 50 cm). Three cigarettes were lit simultaneously at the bottom. After the cigarettes had burnt entirely, the samples were left for 1 h to absorb the tobacco smoke fully. After completion, the tester quickly put the middle part (4 cm × 7 cm) of the samples into the headspace bottles. The fume hood was the device where the experiments of adsorption were performed. Agilent 5977B gas chromatography–mass spectrometry (GC-MS; Agilent Technologies Co., Ltd., Shanghai, China) was utilized for analysis, and the conditions were as follows: the injector temperature was 250 °C and the initial temperature of the oven was 40 °C, held for 2 min, rising to 280 °C at the rate of 15 °C/min, held for 45 min, and then the operating temperature was consistent with the initial temperature. The carrier gas was helium (purity >99.999%) at a constant flow rate of 1.0 mL/min.
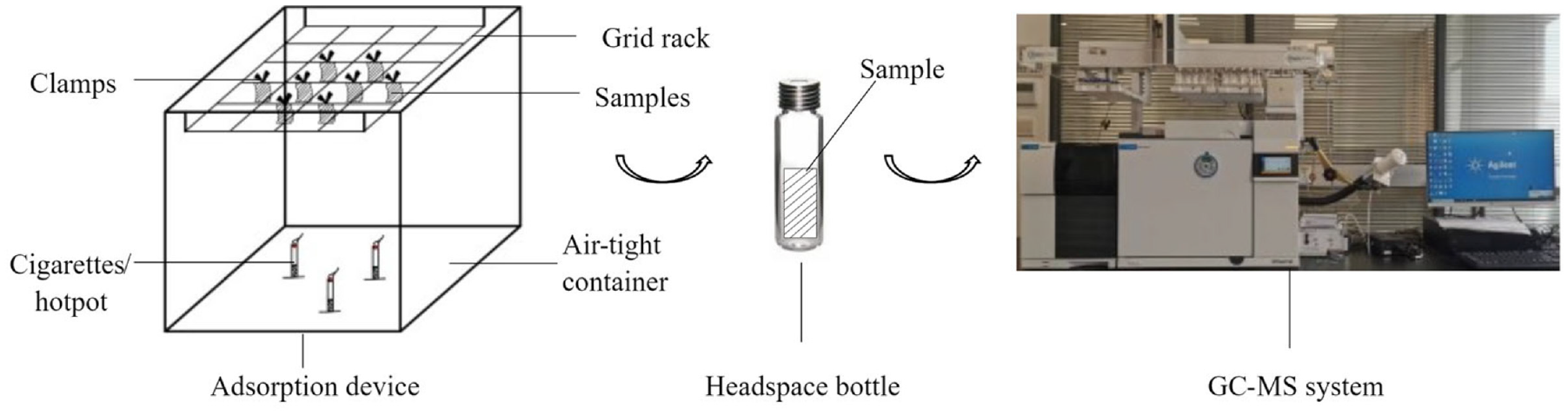
Schematic of the experiment.
The separations were completed using a DB-WAX column (30 m × 0.25 mm, 0.25 μm), and the mass spectrometer conditions were as follows: ionization mode, electron-impact (EI), MSD transmission line temperature keeping consistent with the final temperature (280 °C), scanning mode for a full scan, and the range of 25–400 m/z.
Hot pot odor adsorption and analytical protocol
Add a bag of hot pot soup base and 750 mL of purified water to the heating device simultaneously at the bottom of the adsorption chamber. After heating the hot pot to boiling, the samples were left for 1 h to fully absorb the hot pot odor. The subsequent operation was the same as described above. The conditions of gas chromatography–mass spectrometry (GC-MS) were as follows: the injector temperature was 250 °C and the initial temperature of the oven was 35 °C, held for 3 min, rising to 100 °C at the rate of 3 °C/min held for 3 min, then rising to 160 °C at the rate of 3 °C/min held for 3 min; after rising to 270 °C at the rate of 12 °C/min, the operating temperature was consistent with the initial temperature.
The same column was used for the separations. The mass spectrometer conditions were as follows: ionization mode, EI, MSD transmission line temperature keeping consistent with the final temperature (270 °C), scanning mode for a full scan, and the range of 33–450 m/z.
Dynamic model of odors in fabric
After the fabric adsorbs, the human body or the environment odor is not caused by a single component of the odor emission. 25 The fabric absorbs tobacco smoke, emitting highly complex tobacco odor components, and in order to simplify the model, we selected nicotine, one of the markers of tobacco smoke, for model calculations.
The adsorption and diffusion of odorous molecules in fabrics are related to fabric structure, airflow velocity, distribution coefficient between fabric and air, temperature, and humidity.9,17,18,26 The adsorption and diffusion processes of fabrics in a one-dimensional thickness direction can be described by equation (1): 27
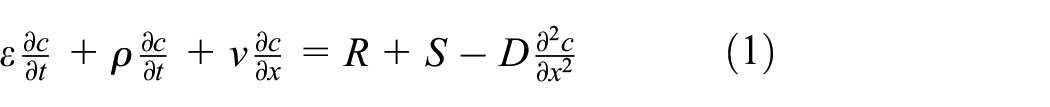
In the equation,

where

The above equation shows that the average flow radius in a fabric depends on the fiber volume fraction
In addition, the porosity
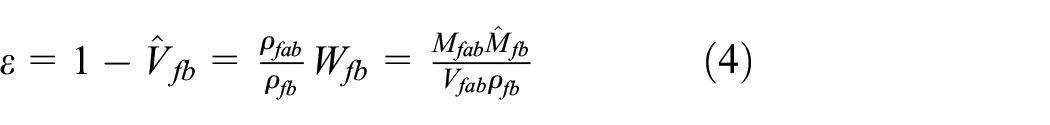
When the air quality is negligible,
Equation (1) is solved in the one-dimensional thickness direction of the cotton fabric, as shown in Figure 2. The initial substance concentration in the fabric is 100%, and the inlet velocity is

where
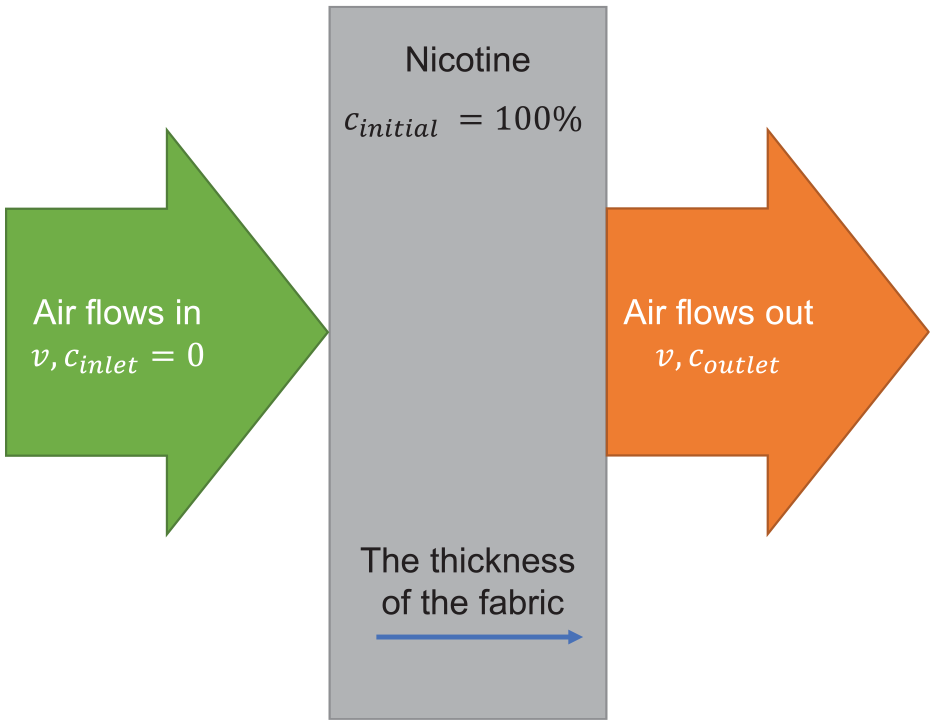
Schematic of the effect of airflow on nicotine concentration in fabric.
Parameters for the calculation. a
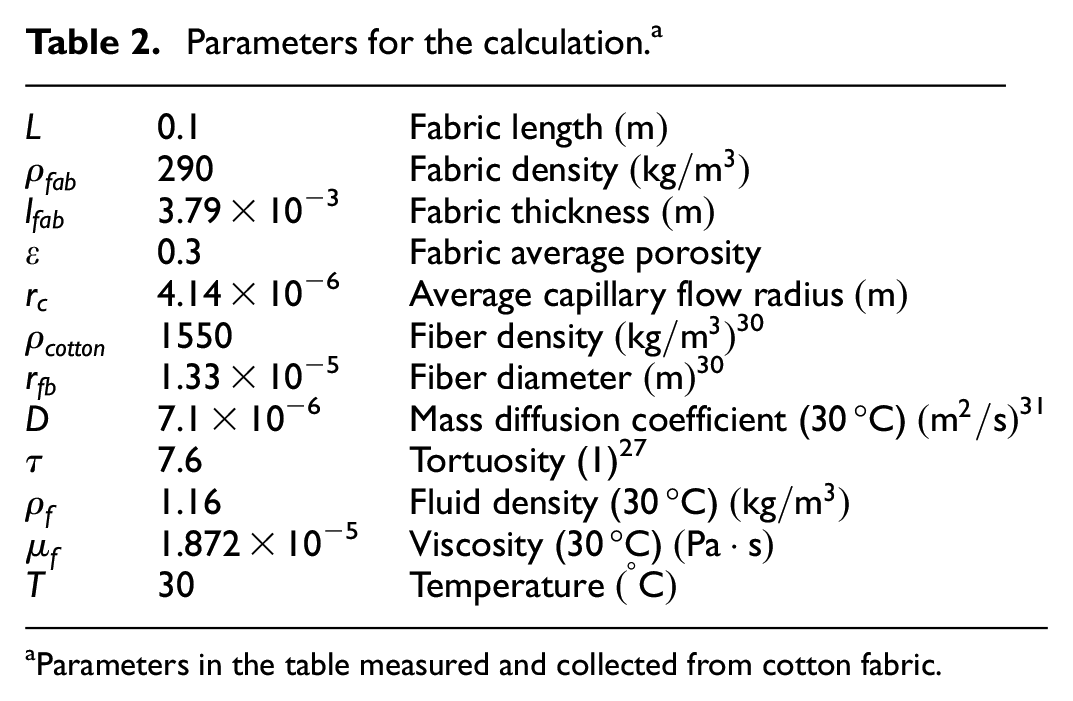
Parameters in the table measured and collected from cotton fabric.
Results and discussion
Desorption components of tobacco odor and influencing factors
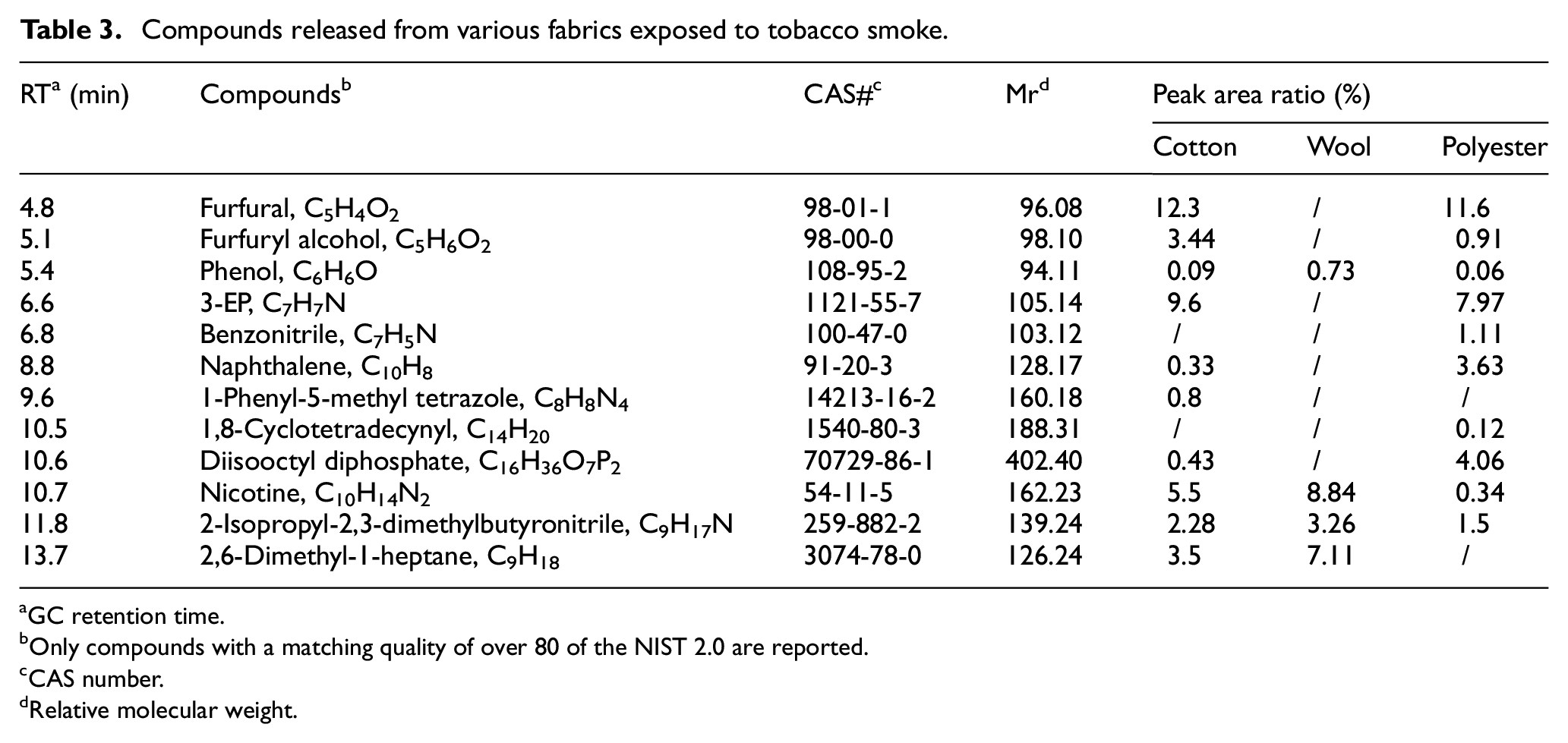
Figure 3 and Table 3 show the desorption components of fabrics exposed to tobacco smoke. The results showed that all three fabrics released nicotine, phenol, and 2-isopropyl-2,3-dimethylbutyronitrile. However, the types of compounds detected in the wool fabric were significantly lower than in the other two. Cotton and polyester fabrics released many be harmful or irritating substances, such as furfural, 3-ethenylpyridine (3-EP) and dioctyl diphosphate. Cotton fabrics desorbed many low molecular weight compounds with high polarity and volatility, such as furfural, 3-EP, and benzonitrile, which is consistent with the previous studies.8,32 At the same time, consistent with the previous research, the odorous molecules released by wool fabrics are more extensive and less diverse.33–35 Nicotine and 3-EP are often considered the marker compounds of thirdhand smoke, 36 but 3-EP was not detected in wool fabrics.
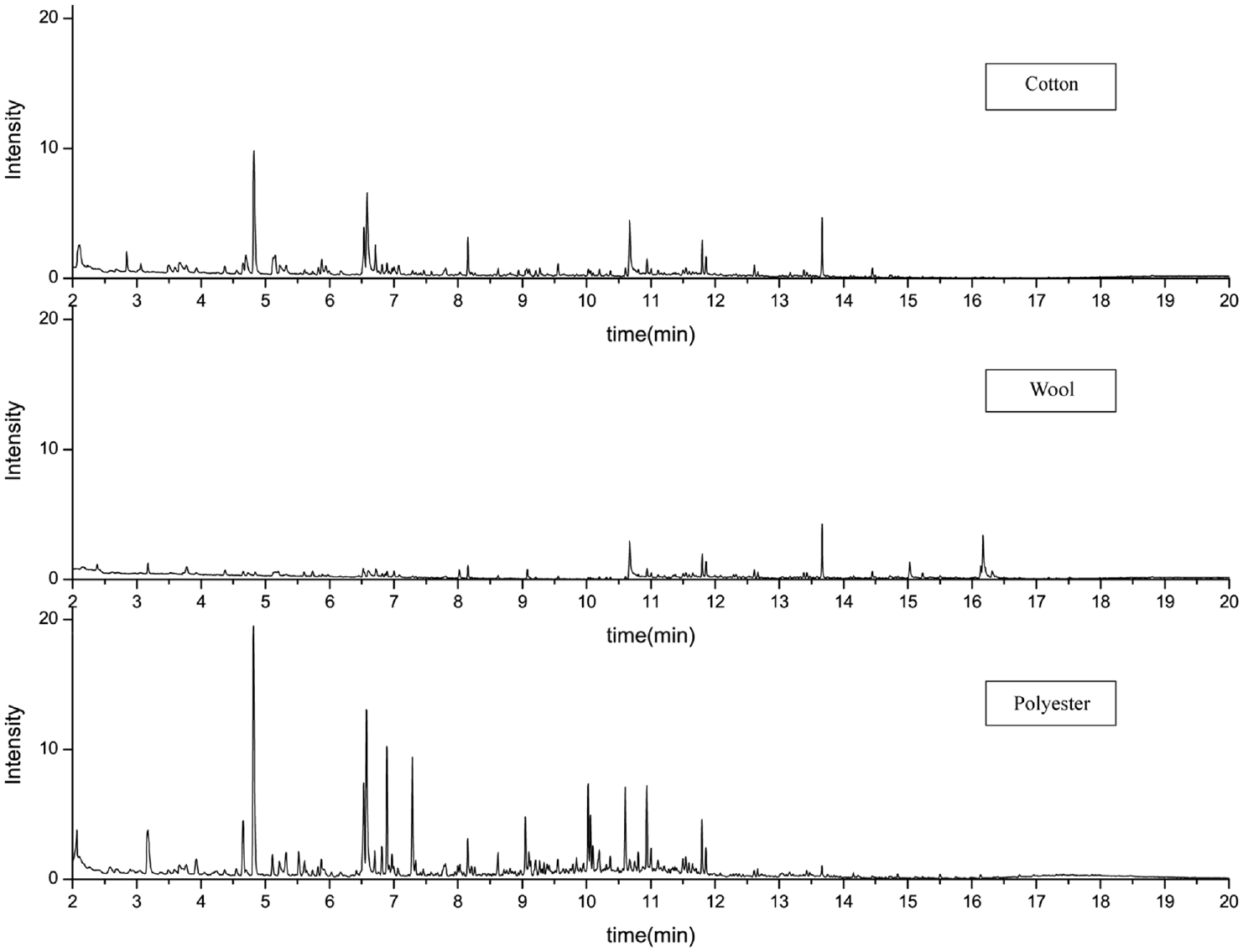
Chromatogram results of fabrics exposed to tobacco smoke.
Compounds released from various fabrics exposed to tobacco smoke.
GC retention time.
Only compounds with a matching quality of over 80 of the NIST 2.0 are reported.
CAS number.
Relative molecular weight.
The moisture content of fabrics influences the tobacco smoke absorbing ability. 37 The hygroscopicity of fibers is related to the strength of polar groups in the molecules that make up the fibers, the crystallinity of the aggregate structure of the fiber, and the bonding strength of polar groups and water. 38 It is concluded that the free hydroxyl groups in cotton fibers can be bonded with nicotine and its derivatives through hydrogen bonds, making it easier for cotton fibers to adsorb polar compounds. 39 The lower odor release ability of wool fibers may be related to the potential ionic interactions between odor compounds and functional groups such as amino and carboxyl groups. The high odor release ability of polyester fabric may be related to its low odor absorption ability and lack of functional groups that can interact with odor compound ions. 40 Compared to polyester, wool and cotton fibers are more hygroscopic, so they absorb more polar compounds.38,41,42 In contrast, non-polar compounds like naphthalene bind more easily to non-polar substrates such as polyester. For example, the study of Yao on cotton, polyester, and wool found that wool has a higher absorption rate of decanal and cyclohexanone, but a lower release rate. This may be due to the interaction of a large number of amino acids with various oily substances through hydrogen bonding, sulfur–sulfur interaction, COOH–NH2 chain, and phosphorus–hydrogen interaction. The high adsorption of ethylbenzene and methyl butyrate on polyesters may be due to the non-covalent interaction of these compounds through moderate strength (dipole–dipole force, hydrogen–sulfur bond). At the same time, cotton has a lower ability to absorb and release odor, which may be due to the effect of hydrogen bonds. 43 The presence of acidic groups and amino groups in wool provides additional adsorption sites for odor molecules, acidic and peptide sites can form bonds with odors, odor emission is reduced, and adding 20% wool to the polyester fabric can significantly reduce the odor emission intensity of the polyester fabric. Polar compounds such as furfural, 3-EP, and benzonitrile are more desorbed from cotton and wool fabrics. At the same time, polyester emits more non-polar compounds such as naphthalene. This is consistent with the above conclusion. 40 Polar compounds such as furfural, 3-EP, and benzonitrile are more desorbed from cotton and wool fabrics. At the same time, polyester emits more non-polar compounds such as naphthalene. This is consistent with the above conclusion.
Fabric structure, usually through the thickness and density of odor adsorption on the fabric, has an impact. 8 For example, Noble 37 found that the weight gain of the different structures of wool fiber was different, and the heavier structure of Gabardine gained the most weight. In this study, the result of the chromatogram shows that the wool fabric has fewer peaks than the other two fabrics, which may be influenced by fabric structure.
Desorption components of hot pot odor and influencing factor
Figure 4 and Table 4 present the desorption components of the fabric exposed to hot pot odor. The results showed that all three fabrics released hexanal, myrcene, β-pinene, nonanal, and anethole. However, the linalyl ester, (−)-4-terpineol, the typical compound of hot pot odor, was not found in cotton fabrics. 7 Cotton fabrics released more hexanal, nonanal, and anethole than wool and polyester. Wool fabrics released much more β-pinene than the other two fabrics.
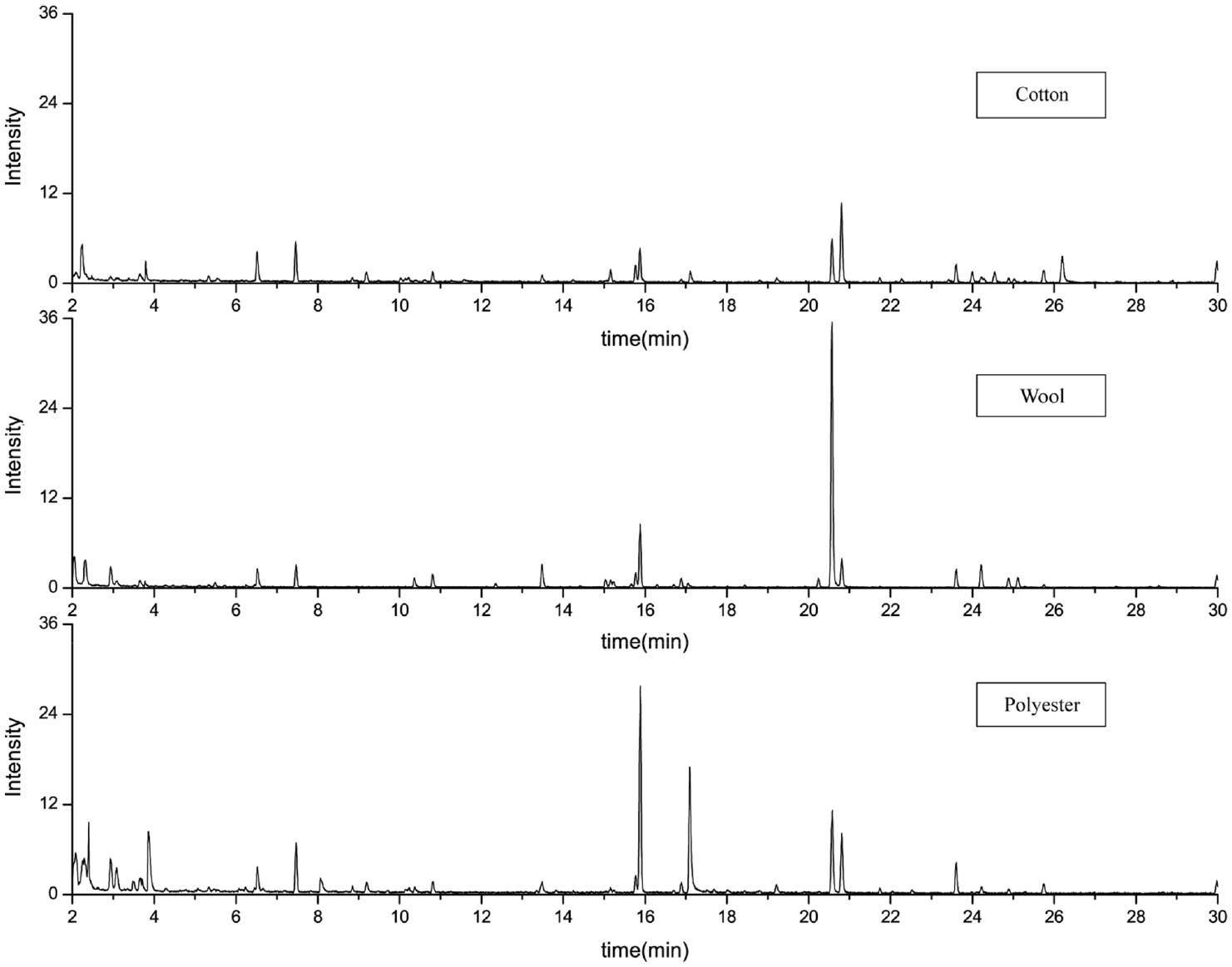
Chromatogram results of fabrics exposed to hot pot odor.
Compounds released from various fabrics exposed to hot pot odor.

aGC retention time.
Only compounds with a matching quality of over 80 of the NIST 2.0 are reported.
CAS number.
Relative molecular weight.
The adsorption and desorption of hot pot odor on the fabric’s surface are a process affected by many factors. First, polar compounds tend to bind to reactive polar sites. Polar compounds are thereby more easily adsorbed on the fiber with polar groups. In addition, the nanoscale water film formed on the fiber surface also dissolves odorous molecules.8,14,23,36 Assuming that a large amount of water vapor condenses on the surface of the fiber during the above process, it may hinder the adsorption of polar compounds on the surface of the fibers with more non-polar groups.7,44
The hydroxyl groups (–OH) in the long cellulose chain of cotton fiber have a strong polarity and can bind to odorous molecules through weak interactions. 45 Wool fibers can form hydrogen bonds with nitrogen atoms in nicotine and 3-EP through carboxyl and amino groups. 38
In addition, the physical properties of fibers can also affect the results of odor adsorption. 19 There are many micropores and lumens in cotton fibers and the scale layer of wool fibers also increases the surface area for adsorbing odorous molecules. The surface of polyester fibers is smooth, long, and hydrophobic, and the chemical adhesion of odorous molecules on its surface is low.34,46
Numerical simulation of the dynamic model
Figure 5 shows the percentage distribution of nicotine concentration over time at the midpoint of the fabric thickness. The nicotine concentration in the fabric gradually decreases with time. After 1800 s of ventilation, when
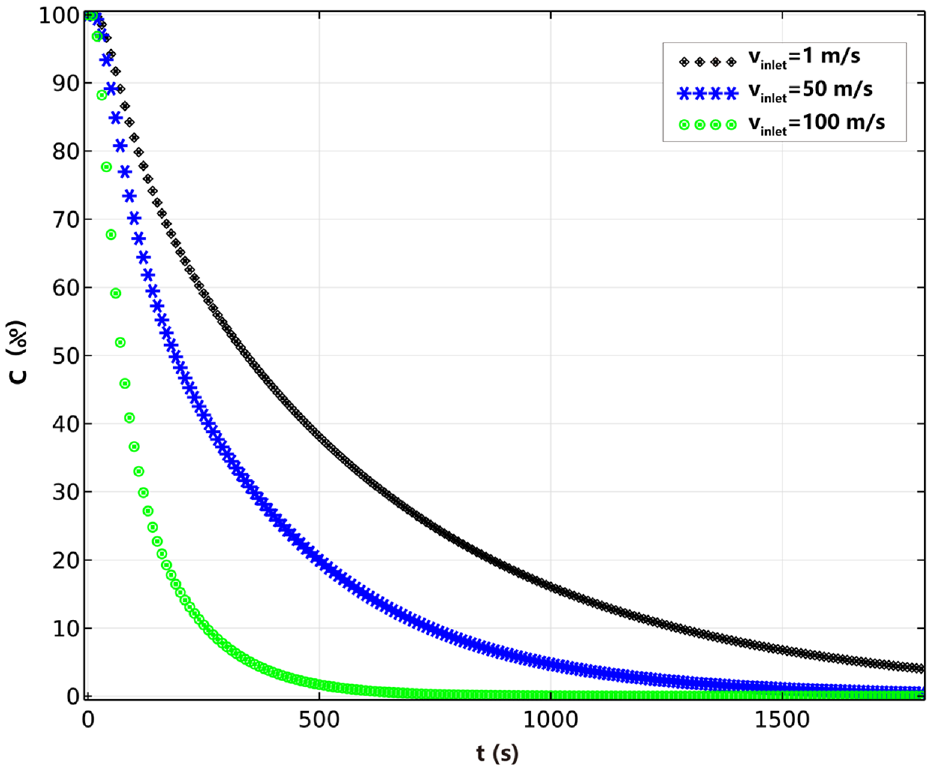
Effect of inlet velocity on the concentration of nicotine over time in the thickness direction.
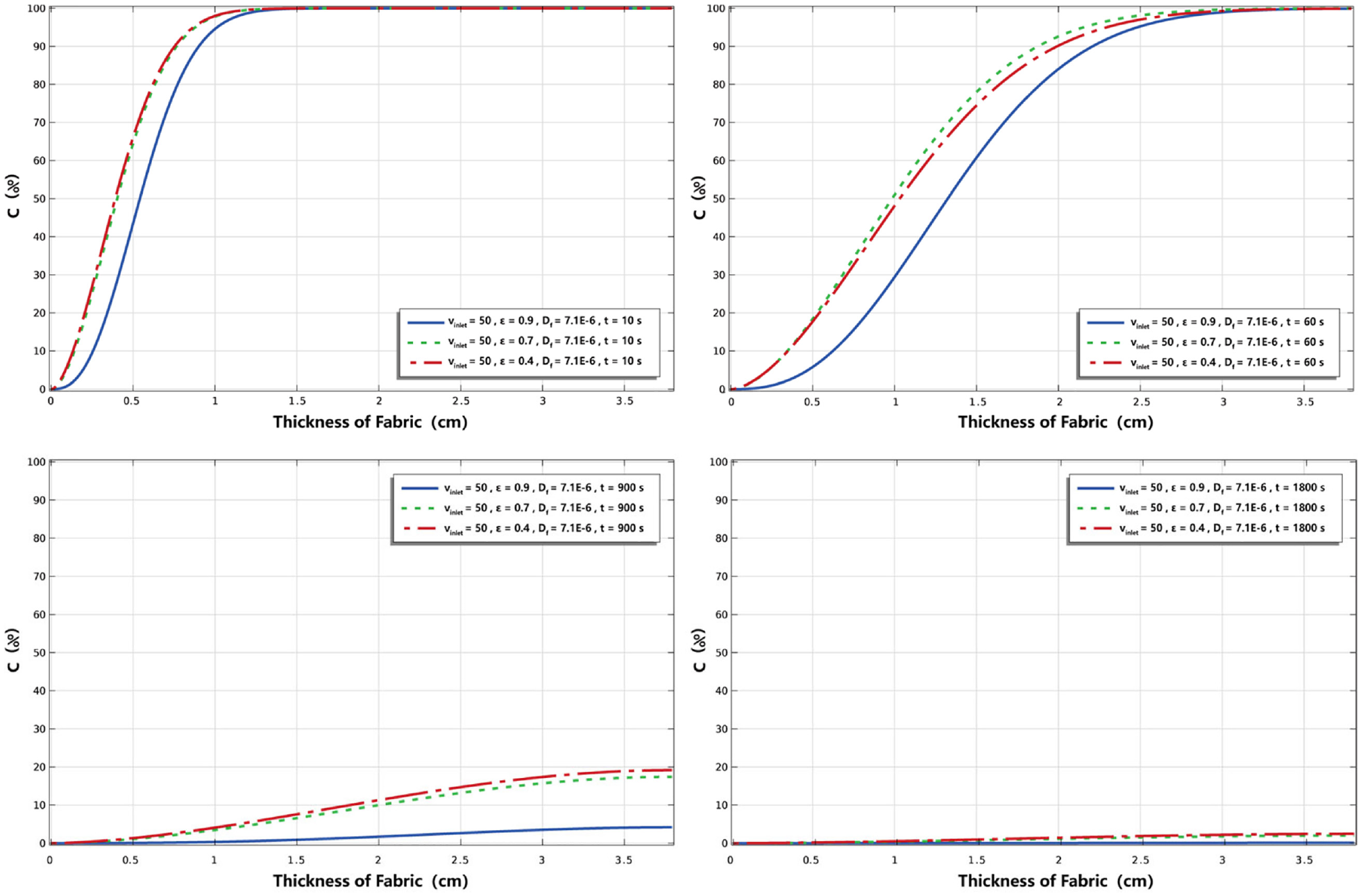
Figure 6 shows the percentage distribution of nicotine concentration in the direction of fabric thickness at 10, 60, 900, and 1800 s, respectively. At the situation of
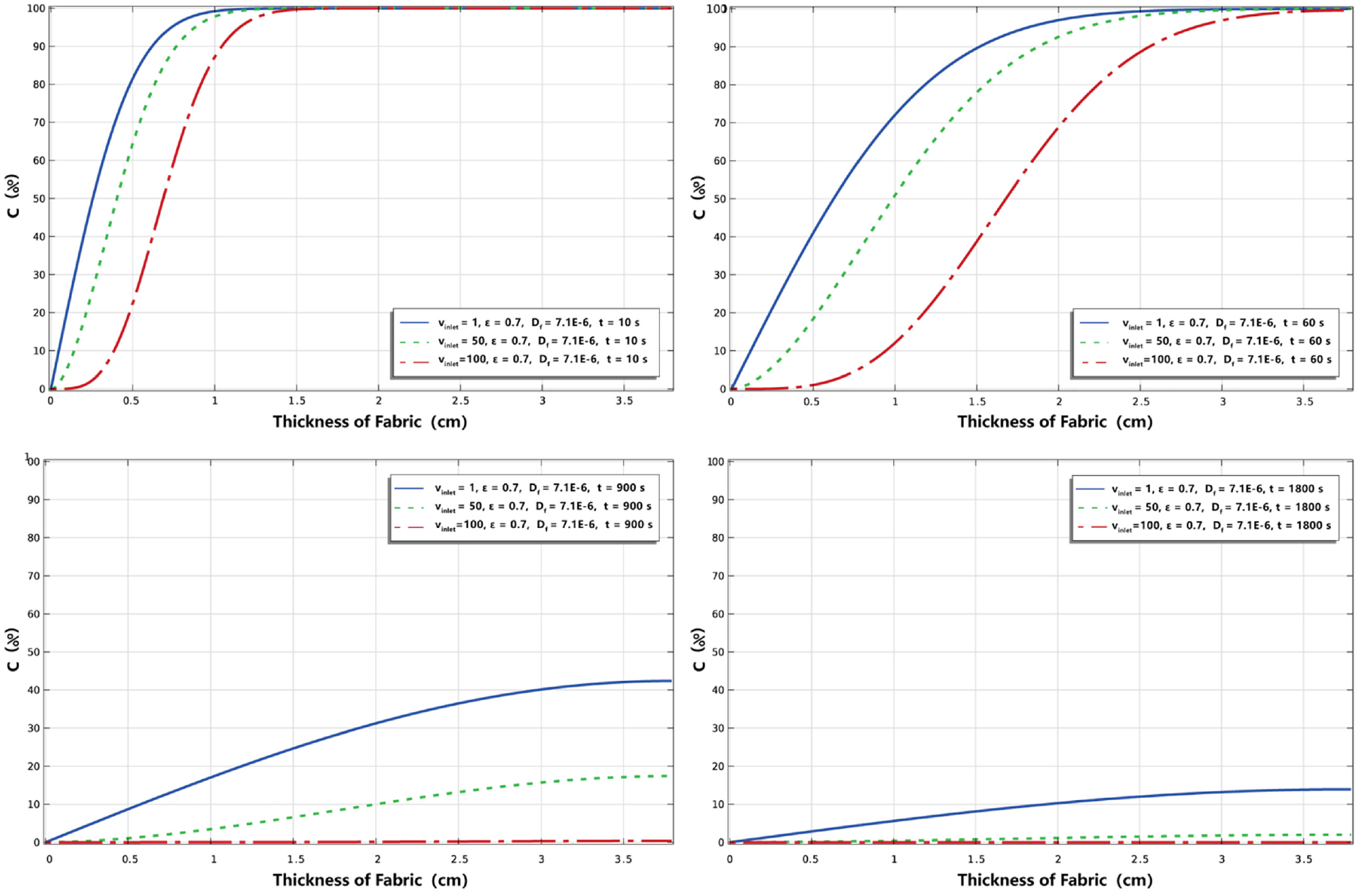
Effect of inlet velocity on the percentage distribution of nicotine concentration in the thickness direction.
The odor molecule mass diffusivity coefficient depends on the molecular structure, molecular mass, and molecular diameter.
47
Figure 7 shows the estimated results when the mass diffusivity coefficient of the odor molecule is larger and smaller than the nicotine used in this study by one order of magnitude, including
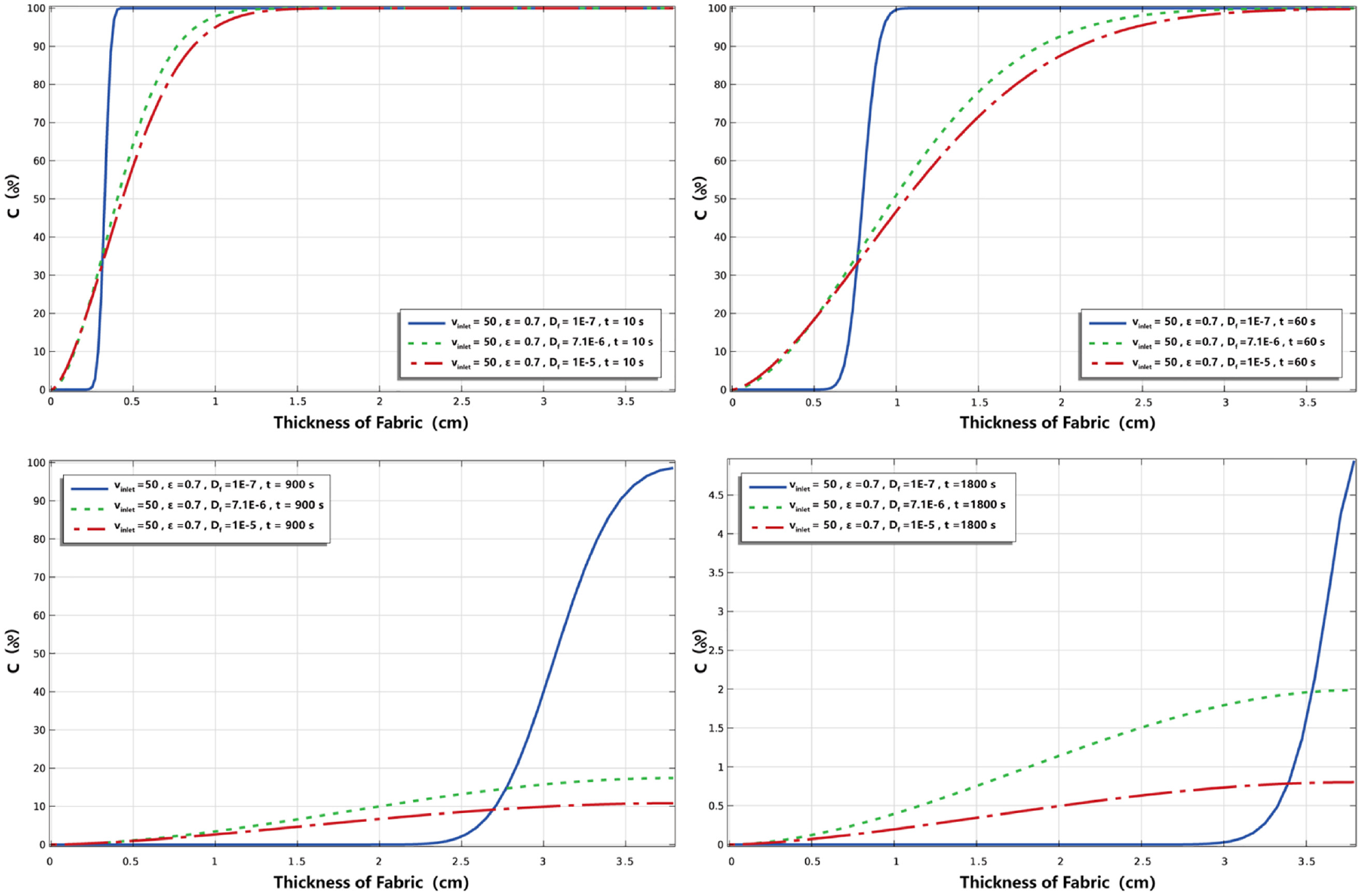
The numerical results under different diffusion coefficients.
Table 5 shows the paired t-test of nicotine concentration percentage with different mass diffusion coefficients at t = 1800 s, where
Paired t-test of nicotine concentration percentage with different mass diffusion coefficients at t = 1800 s.
Figure 8 shows the effect of porosity
Effect of porosity on nicotine concentration in fabric.
t-Test of nicotine concentration percentage with different fabric porosity at

The fabric-specific surface area (Assa, 1/m, equation (6)) is a helpful tool to partly understand how the fabric structure and fiber properties influence the desorption process:

where
Conclusion
In this study, the desorption components of cotton, wool, and polyester fabrics after absorbing tobacco smoke and hot pot odors were examined, respectively, using GC-MS and other instruments. Subsequently, the effects of different boundary conditions and fiber volume fractions on the odor concentration in the fabrics were investigated based on a dynamic diffusion model with nicotine as an example. The numerical results of the diffusion coefficients were also discussed. The following conclusions can be drawn:
For tobacco smoke, cotton released many low molecular weight compounds such as furfural, 3-EP, and benzonitrile. Compared to cotton and polyester, wool fabrics released larger molecular weight and significantly fewer types of odors. 3-EP, considered a signature compound for thirdhand smoke, was not detected in wool fabrics.
For hot pot odor, cotton fabrics released more hexanal, nonanal, and anethole than wool and polyester. In contrast, wool fabrics released much more β-pinene than the other two. The result may be related to the fact that the large amount of water vapor generated during heating dramatically affects the adsorption and desorption of the fabrics.
The dynamic model’s numerical results showed that the nicotine concentration in the inner part of the fabric decreased faster in fabrics with lower fiber volume fractions. A lower mass diffusion coefficient will result in a concentration residual in the second half of the fabric. The airflow’s inlet velocity greatly influences nicotine concentration in the fabric. When the fabric’s porosity was similar, the nicotine concentration in the fabric was also similar.
This study aimed at the composition and the dynamics of odor in fabrics offers essential information and simple models for prediction to reduce unnecessary washing of textiles and odor resistance textile design. In addition, the reaction rate and other key parameters can be determined based on the transient numerical calculation results of odor and the kinetic model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors acknowledge the support of Shanghai Engineering Research Center for Clean Production of Textile Chemistry. The authors also would like to acknowledge the illuminating discussions with the engineer Mr Guan Xuewei from Wuxi Little Swan Company Limited.
