Abstract
In this study, the mineralogical compositions, concentrations and occurrence modes of the elements in the No.5 coal from the Yanzishan Mine were investigated, using X-ray powder diffraction (XRD), X-ray fluorescence spectrometry (XRF), inductively coupled plasma mass spectrometry (ICP-MS), and scanning electron microscopy equipped with an energy-dispersive X-ray spectrometer (SEM-EDS). The No. 5 coal is characterized by high ash yield content (mean 32.30%) and low sulfur content (mean 0.47%). The minerals are mainly composed of kaolinite, quartz and pyrite, with a small proportion of illite, apatite, siderite, sphalerite and Ti-oxide. Compared with the Chinese coals, the No. 5 coal has much higher percentages of SiO2 (mean 16.22%) and Al2O3 (mean 14.57%) than other major oxides. Compared with the world hard coals and Chinese coals, the No. 5 coal is enriched in some potentially valuable (Al2O3, Li, Ge, Zr, and Hf) and toxic (Be, As, Se, Cd, and Pb) elements. Lithium and Hf are mainly associated with clay minerals; Be, Se, and Zr mainly have both inorganically and organically affinities; As mainly occurs as a sulfide form in pyrite; Cd is incorporated in clay minerals; and Pb has an affinity for both clay minerals and organic sulfur. All the coal samples are characterized by weakly positive or no pronounced Eu anomalies, indicating that the rare earth elements and yttrium (REY) likewise originated from granite of the Yinshan Oldland together with injection of the low temperature hydrothermal fluids.
Introduction
Coal has played and still plays a leading role as primary energy source in China. As the “Statistical Review of World Energy (2017)” mentioned (B., 2017), although coal consumption decreased from 64% in 2015 to 62% in 2016, China is still the largest consumer of coal, accounting for 23% of the world’s annual coal consumption. Under some specific geological conditions, coals are enriched in some metal elements (Hower et al., 2013; Sun et al., 2016). This can provide geological information about depositional conditions of coals and regional tectonic history through analyzing elemental abundances, distributions, and modes of the occurrence (Arbuzov, et al., 2018; Dai, et al., 2012; Karayiğit et al., 2017). Some valuable elements, including Al, Li, Ga, Ge, In, Re, Nb, Ta, Zr, Hf, and U, etc., can be industrially utilized if their concentrations can be enriched to the levels exceeding usual economic grades (Scott et al., 2015; Sun, et al., 2012, 2013a, 2013b). The utilization of these elements can mitigate severe pressure of resource scarcity, and thus meet the growing demand for energy in China. Significant attention has been paid to the modes of occurrence of these valuable metals in coal (Qin, et al., 2015a; Sun, et al., 2016). For example, previous studies showed that the coals, located in the southern Yinshan Oldland, are enriched in Al, Li, Ga, and rare earth elements (REEs) which mainly occur in boehmite and kaolinite (Qin, et al., 2015a; Sun et al., 2016; Zhao et al., 2012). Moreover, Li has affinities for both organic and inorganic matter in coals (Finkleman, 1981; Sun et al.,2013a, 2013b; Swain, 1999). Gallium mainly occurs in clay minerals or hydroxide complexes, and is partly organically bound in coals (Bennett and Czechowsk., 1980; Dai et al., 2016a; Qin et al., 2015b; Zhao et al., 2017). Germanium is largely adsorbed by organic matter or clay minerals, and also occurs in inorganic carriers such as silicate, sulfide, and oxide minerals (Ren et al., 2006; Yudovic., 2003). With regard to the potentially toxic elements, Swaine and Goodarzi (1995) reported that As, Cr, Cd, Hg, and Se are the most toxic elements; B, Cl, F, Mn, Pb, Mo, and Ni are moderately toxic elements; Be, Cu, P, Th, U, V, and Zn are relatively less toxic elements; and Ba, Co, Sb, Sn, and Th are the least toxic elements. The extremely toxic elements in coals may impose potential adverse effects on the environment and human health during coal utilization (Singh et al., 2015b). The investigation of concentrations and occurrence modes of the potentially hazardous elements in coals is, and therefore, necessary to determine the possible adverse impacts on environment and human health during the exploration and utilization of coals (Finkelman and Tia., 2018; Kolker et al., 2017; Singh et al., 2016b; Tian et al., 2013).
The Datong Coalfield is one of the largest coal production bases of China and it is located in northern Shanxi Province, which is adjacent to Shaanxi Province, Hebei Province, Inner Mongolia (Figure 1). Many studies have been conducted on the geochemical characteristics of the Ordos Basin and the Ningwu Basin, which are adjacent to the Datong Coalfield (Do., 2013; Liu and Li., 2014; Liu et al., 2015). The Datong Coalfield is geographically similar to the Junger Coalfield and Ningwu Coalfield, which can provide ideas to discuss the occurrence modes of various elements in the coals from Datong Coalfield (Do., 2013; Liu et al., 2015). Datong Coalfield is enriched in a variety of both potentially valuable and toxic elements with relatively high concentrations. Noteworthy, only a few studies have been published about the concentrations and modes of occurrence of the potentially valuable and toxic elements in the coals from the Datong Coalfield.
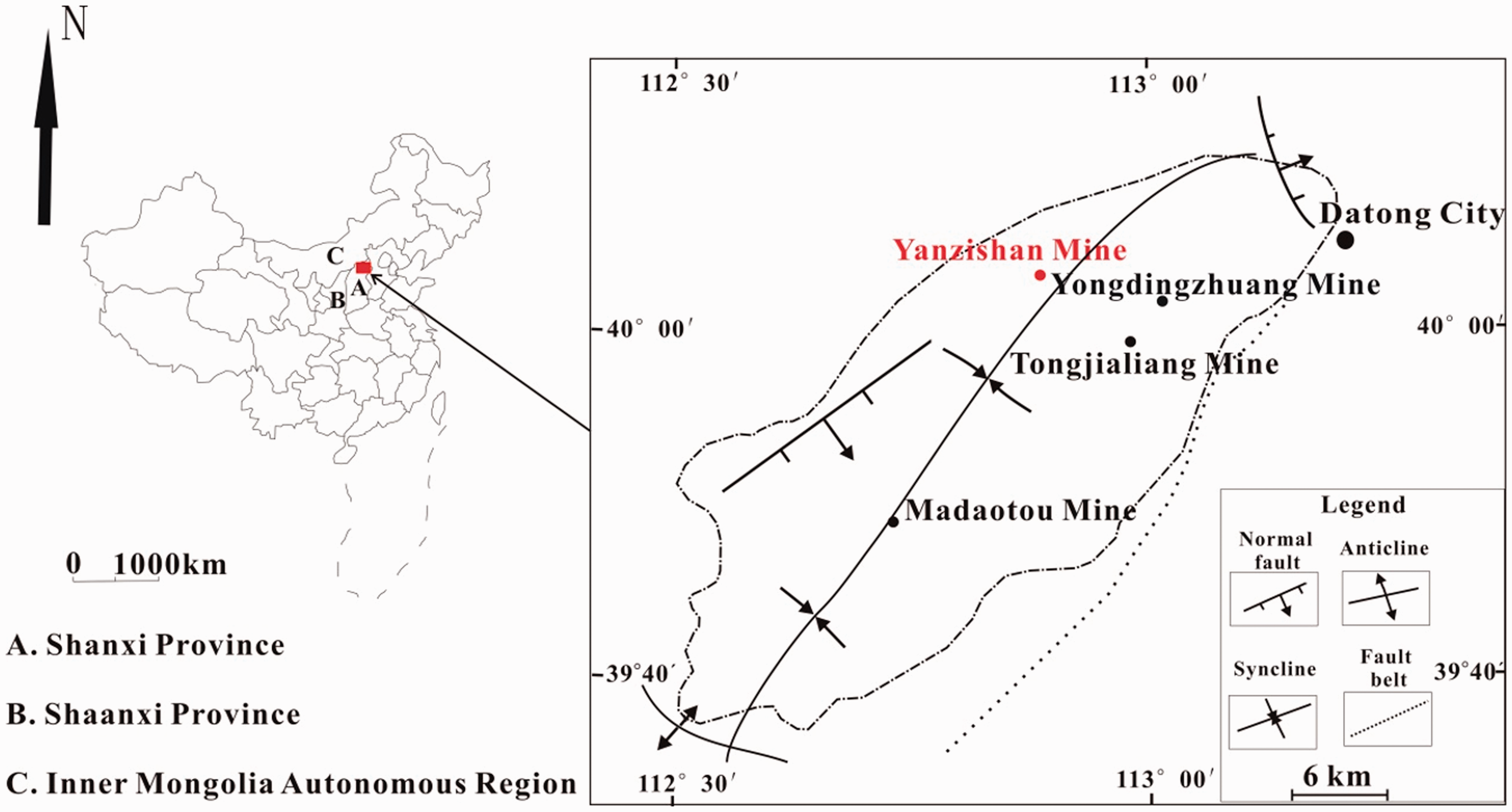
The location and geological map of the Datong Coalfield.
While the valuable elements in coals are being exploited and utilized to achieve economic benefits, the adverse effects which may be caused by the release of the toxic elements during exploitation and utilization of coals on humans and the environment should not be ignored. Thus, the main objective of this study was to determine the modes of occurrence of the potentially valuable and hazardous elements in coals from the Yanzishan Mine. This study is of significance for accurately evaluating the adverse impacts of the potentially toxic elements and the utilization of the potentially valuable elements.
Geological setting
The Datong Coalfield is located to the south of the Yinshan Oldland and stretches 50 km in length (N–S) and 30 km in width (W–E), covering a total area of 1900 km2 (Do., 2013; Liu et al., 2015). It is bound to the west by the Pingwang–Emaokou fault, to the east by the Lvliang mountain syncline, and to the north by the Hongtao mountain syncline. Owing to the Caledonian tectonic movement, the Ordovician Majiagou Formation has undergone extensive weathering, leading to erosion of the Upper Ordovician, the Silurian, Devonian, and the Lower Carboniferous strata. The coals in the Datong Coalfield began to accumulate during the Late Palaeozoic in part of the North China Craton. Consequently, the strata were assigned to the Benxi, Taiyuan, Shanxi, Shihezi, and Shiqianfeng Formations. The faults in the northern coalfield provided channels for magma intrusion. The lamprophyre intrusions, which occurred during the Indosinian epoch in the north of the Datong Coalfield, resulted in thermal contact metamorphism and silicification between coal seams (Do., 2013; Liu et al., 2015; Xi., 2013). The lamprophyre is widely distributed in the well field, with a gray–black color and grayish–yellow color after weathering, which has brown biotite and light-colored plagioclase phenocrysts. The basement consists of biotite, plagioclase, apatite, and siderite. The lamprophyre intruded into the upper Taiyuan Formation and had a destructive effect on all the No. 5 coal seams in Datong Coalfield (Do., 2013; Liu et al., 2015; Xi., 2013).
The Taiyuan Formation has a total thickness of 33–138 m and is mainly composed of conglomerate, gritstone, sandstone, siltstone, mudstone, and five coal seams identified as Nos. 2, 3, 4, 5, and 8 (Figure 2). The thickness of No. 5 coal seam varies from 0.3 to 20.4-m (6.9-m on average) (Do., 2013; Liu et al., 2015). The No. 5 coal seam can be mined in most parts of the Datong Coalfield, which are thicker in the northern and southern parts of the coalfield, with a slight decrease in the middle. According to the previous studies, the No. 5 coal from Taiyuan Formation was controlled in delta depositional environment (Do., 2013; Liu et al., 2015).
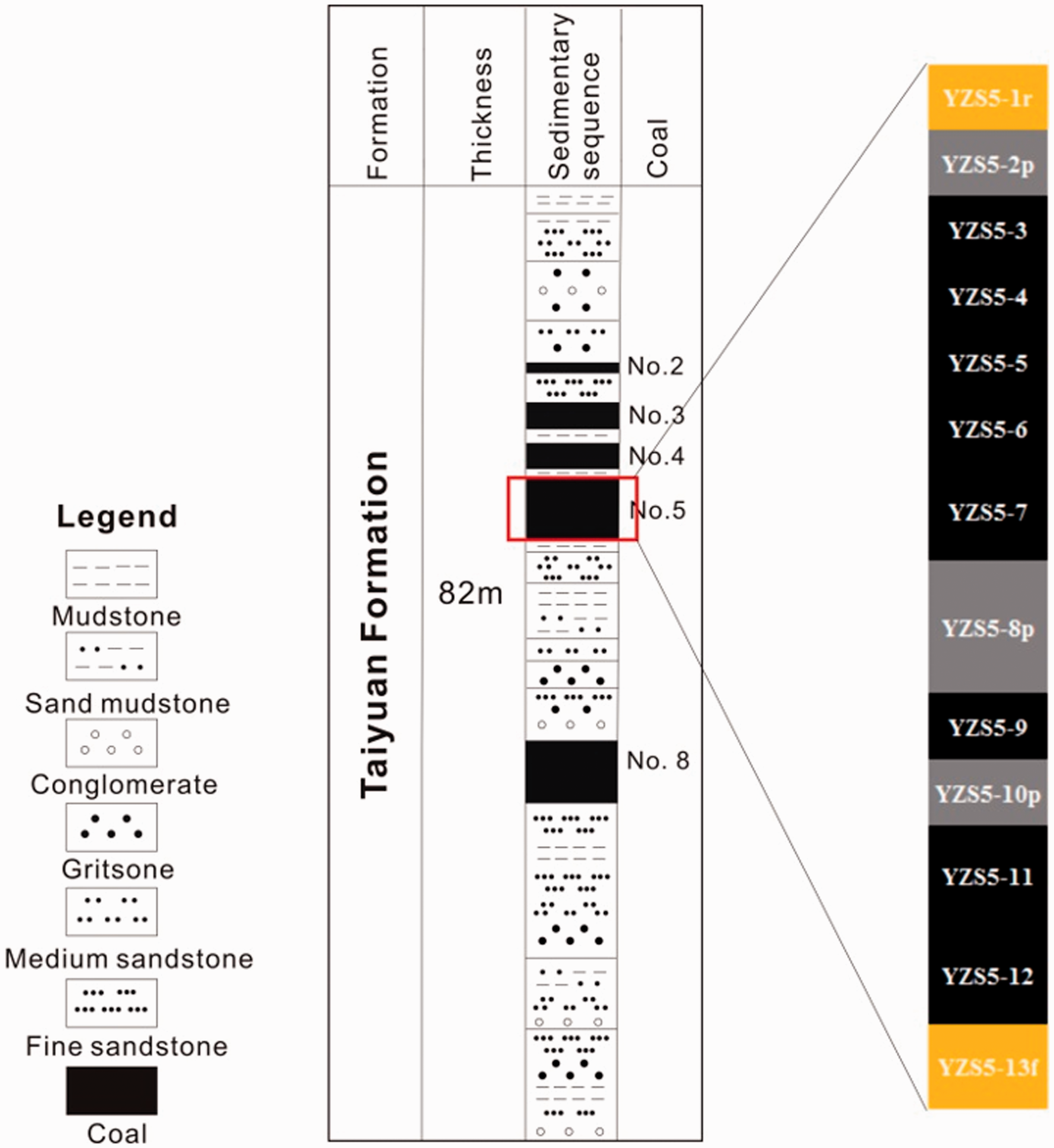
General stratigraphic sequence of the Taiyuan Formation of the Datong Coalfield.
Samples and analytical procedures
Thirteen samples, consisting of eight coal benches, three partings, one floor, and one roof samples with a total thickness of 3.2-m from top to bottom (Figure 2), were collected from the No. 5 coal in the Yanzishan mine. The samples were cut into 10-cm wide and 10-cm deep sections and stored in plastic bags to prevent contamination (GB/T 482–2008, 2008).
The proximate analyses, including moisture, ash yield, volatile matter, total sulfur, and forms of sulfur of the coals were based on ASTM Standards D3173-11, D3174-11, D3175-11, D3177-02, and D2492-02, respectively (ASTM, 2011a, 2011b, 2011c; 2002a, 2002b).
The mineralogical composition was determined by microscopy (Leica DM 2500P microscope by Leica Microsystems, Solms, Germany). Low-temperature ashing of coal was performed using a plasma asher (EMITECH K1050, Quorum, Ashford, UK), with the temperature maintained at below 200°C. X-ray diffraction (XRD) analysis on the low-temperature ashes was performed with Ni-filtered Cu-Kα radiation and a scintillation detector. The XRD patterns were recorded over a 2θ interval from 2.5°–70°, with a step size of 0.01°. In order to determine the compositions and contents of the minerals in coals from the X-ray diffractograms, the Siroquant™ commercial interpretation software developed by Taylor (1991), was used and further details of this technique were provided by Ward (2002) and Ruan and Ward (2002).
The 80-mesh size samples were used to analyze both the mineralogical composition and distribution. Scanning electron microscopy (SEM, HITACHI UHR FE-SEM, SU8220, HITACHI, Tokyo, Japan) coupled with energy-dispersive X-ray spectrometry (SEM-EDS) was used to study the mineral distribution, and the distribution patterns of several elements in the coal. The gold-plating coating was performed on the samples before the analysis of samples by scanning electron microscopy.
All of the 13 samples were crushed into 200-mesh size prior to elemental analysis. The 200 mesh samples were heated to 815°C to create high-temperature ashes. These ashes were then formed into tablets and measured via X-ray fluorescence spectrometry (XRF, (Thermo ADVANT XP+). All the residues were used to determine the contents of major-element oxides (SiO2, TiO2, Al2O3, Fe2O3, MgO, CaO, MnO, Na2O, K2O, and P2O5). Trace elements, except As, Se, Hg, and F, were determined by inductively coupled plasma mass spectrometry (ICP-MS). The ICP-MS analyses were performed on raw coal samples. Samples for ICP-MS were prepared by digesting using an UltraClave Microwave High Pressure Reactor (Milestone, Sorisole, Italy). The basic load for the digestion tank was composed of 330-mL distilled H2O, 30-mL 30% H2O2, and 2-mL 98% H2SO4. Initial pressure for nitrogen was set at 50 bars and the highest temperature was set at 240°C which lasted for 75 min. The reagents for the sample digestion (50 mg) were HF (5-mL, 40%), HNO3 (2-mL, 65%), and H2O2 (1-mL, 30%). Multi-element standards were used for calibration of trace element concentrations. Arsenic and Se were analyzed by ICP-MS with a collision/reaction cell technology (ICP-CCT-MS) (Li et al., 2014). Fluorine was determined via pyrohydrolysis combined with an ion selective electrode (ISE) based on the ASTM method (ASTM, 2002a, 2002b). Mercury was determined using an Hg analyzer (Milestone DMA-80, Milestone, Sorisole, Italy). Total organic carbon (TOC) was determined using a carbon-sulfur analyzer (LECO CS-230) according to GB/T19145-2003. The samples were ground to less than 200-mesh in size, weighed, and then diluted with 5% HCl at 80°C to dissolve any inorganic carbon within the carbonate. The samples were then rinsed, dried, and reweighed before combustion at high temperatures in the Leco CS230 (GB/T19145-200., 2003).
Results and discussion
Proximate analyses
Results of total sulfur and proximate analysis of 13 samples in the No. 5 coal from the Yanzishan mine are listed in Table 1. The No. 5 coal is characterized by high ash yield content (mean 32.30%) based on the Chinese Standards GB/T 15224.1–2010) (2011) and low sulfur content (mean 0.47%) based on the Chinese Standards GB/T 15224.2–2010) (2011). The low sulfur content indicated that the sulfur was primarily derived from parent plant materials (Cho., 2012). The total sulfur consists of inorganic sulfur (including pyritic sulfur and sulfate sulfur) and organic sulfur, and the pyritic sulfur is a dominant form in all the samples. The TOC values of coal samples range between 39.9% and 65%, and averages at 51.51%.
Proximate analysis of the No. 5 coal samples from the Yanzishan mine (%).
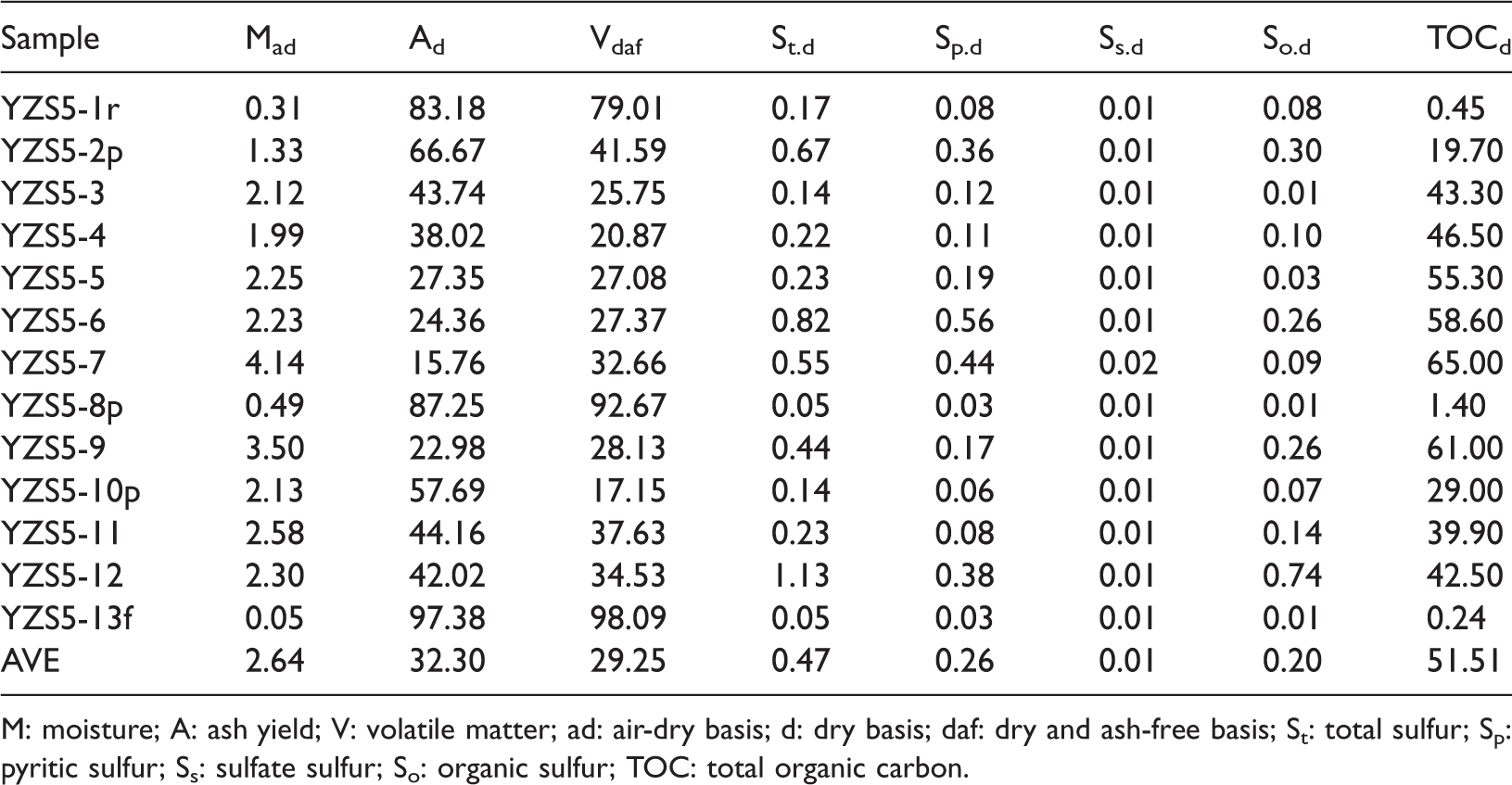
M: moisture; A: ash yield; V: volatile matter; ad: air-dry basis; d: dry basis; daf: dry and ash-free basis; St: total sulfur; Sp: pyritic sulfur; Ss: sulfate sulfur; So: organic sulfur; TOC: total organic carbon.
Mineralogy
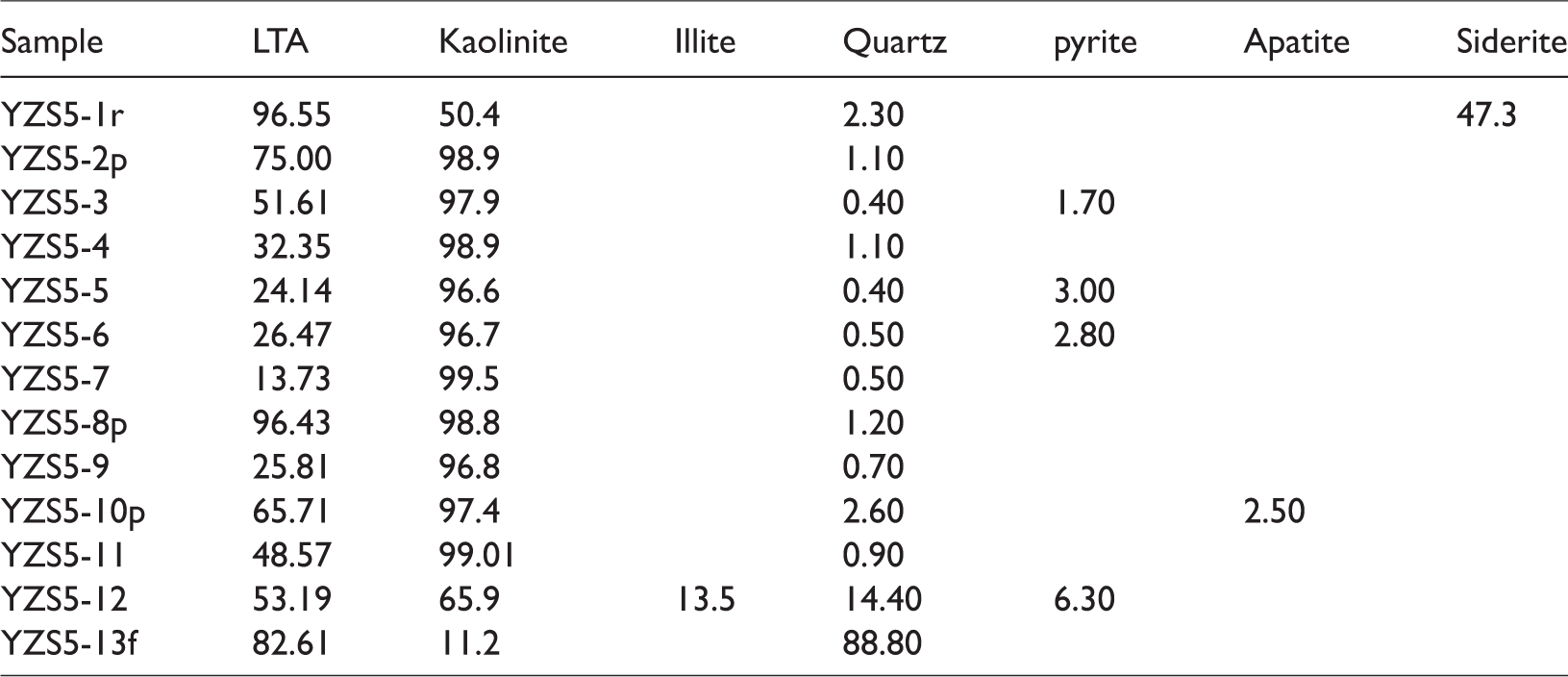
Table 2 lists that the No. 5 coal is mainly composed of kaolinite, quartz, and pyrite, and a small proportion of illite, apatite, and siderite, based on the LTA-XRD and Siroquant. Moreover, some small proportions of minerals (including sphalerite and Ti-oxide), present in some samples, were observed only by the SEM-EDS on account of the fact that the quantification was below the detection limit of the XRD system.
Concentrations of minerals in the No. 5 coal (%) (on ash basis).
Clay minerals
Kaolinite is the dominant mineral in the No. 5 coal, ranging from 96.8% to 99.5%. Kaolinite, identified by the optical microscopy and SEM-EDS, mainly occurs in infillings of cell-cavities in fusinite (Figure 3(a)) and disseminated fine particles in collodetrinite (Figure 3(b)), and telinite (Figure 3(c)) and in a few cases, as vermicular forms (Figure 3(d)). The kaolinite, found in the pore and cell-cavities of the macerals, indicates authigenic and probably syn-depositional origin (Ward,2002, 2016). Authigenic kaolinite may also occur as vermicular aggregates of individual crystals deposited within the precursor palaeomire (Permana et al., 2013; Ward,2002, 2016). Vermicular kaolinite is often considered to be derived from pyroclastic inputs, which corresponds to the report by Zhong et al. that synchronous volcanic activity may happen around the Datong coalfield during the peat accumulation of the No.5 coal (Burger et al., 2000; Zhong et al., 1995).

Clay minerals in the No.5 coal, (a) Kaolinite in fusinite (reflected light); (b) Kaolinite in collodetrinite (reflected light); (c) Kaolinite in cell lumens of tellinite (reflected light); (d) Kaolinite as vermicular (SEM, secondary electron image); (e) Illite (SEM, secondary electron image) and its energy dispersive X-ray spectrometry (EDS) (f).
The Siroquant and SEM-EDS analysis further indicates that illite is occasionally observed in YZS5-12 coal bench sample (13.5%) (Figure 3(e)). In general, illite is formed due to the weathering of feldspar and mica and could be also originated from clastic inputs from the sediment source areas (Siavalas et al., 2009; War., 2016). During the peat accumulation, the Datong Coalfield has been determined to originate from the Yinshan Oldland, located to the north of the studied area, which is mainly composed of granite and moyite. The illite most likely resulted from detrital materials of terrigenous origin.
Oxide minerals
Quartz is one of the common minerals present in coals, with the modes of occurrence as infillings in cell-cavities in macerals, veins, and disseminated particles in macerals (Sykes and Lindqvist, 1993). In the No. 5 coal, the proportion of quartz (ranging from 0.4% to 14.4%) is higher in lower samples near the floor than that in the upper samples. Moreover, some angular shape quartz (10–100 μm), with close combination with macerals and kaolinite matrix, indicates that quartz has a detrital origin and was transported over a short distance from the sediment source region (Figures 4(a) to (c)).

Oxide minerals in the No.5 coal, (a) Quartz disseminated in kaolinite (reflected light); (b) Quartz disseminated in kaolinite (SEM, secondary electron image); (c) Quartz disseminated in macerals and kaolinite matrix (reflected light); (d) Epigenetic Ti-oxide (SEM, secondary electron image.
Ti-oxide was occasionally observed under SEM-EDS. It exists as fracture-fillings (Figure 4(d)), indicating an epigenetic origin.
Sulfide minerals
Pyrite has contents varying from 1.7% to 6.3%, occasionally detected and observed in some coal bench samples. In the No. 5 coal, pyrite occurs mainly as infillings in the fractures of macerals and kaolinite (Figure 5(a) and (b)), indicating an epigenetic origin (Querol et al., 1989). Consequently, the epigenetic pyrite is considered to be derived from hydrothermal fluids (Diehl et al., 2012; Xiao et al., 2016).
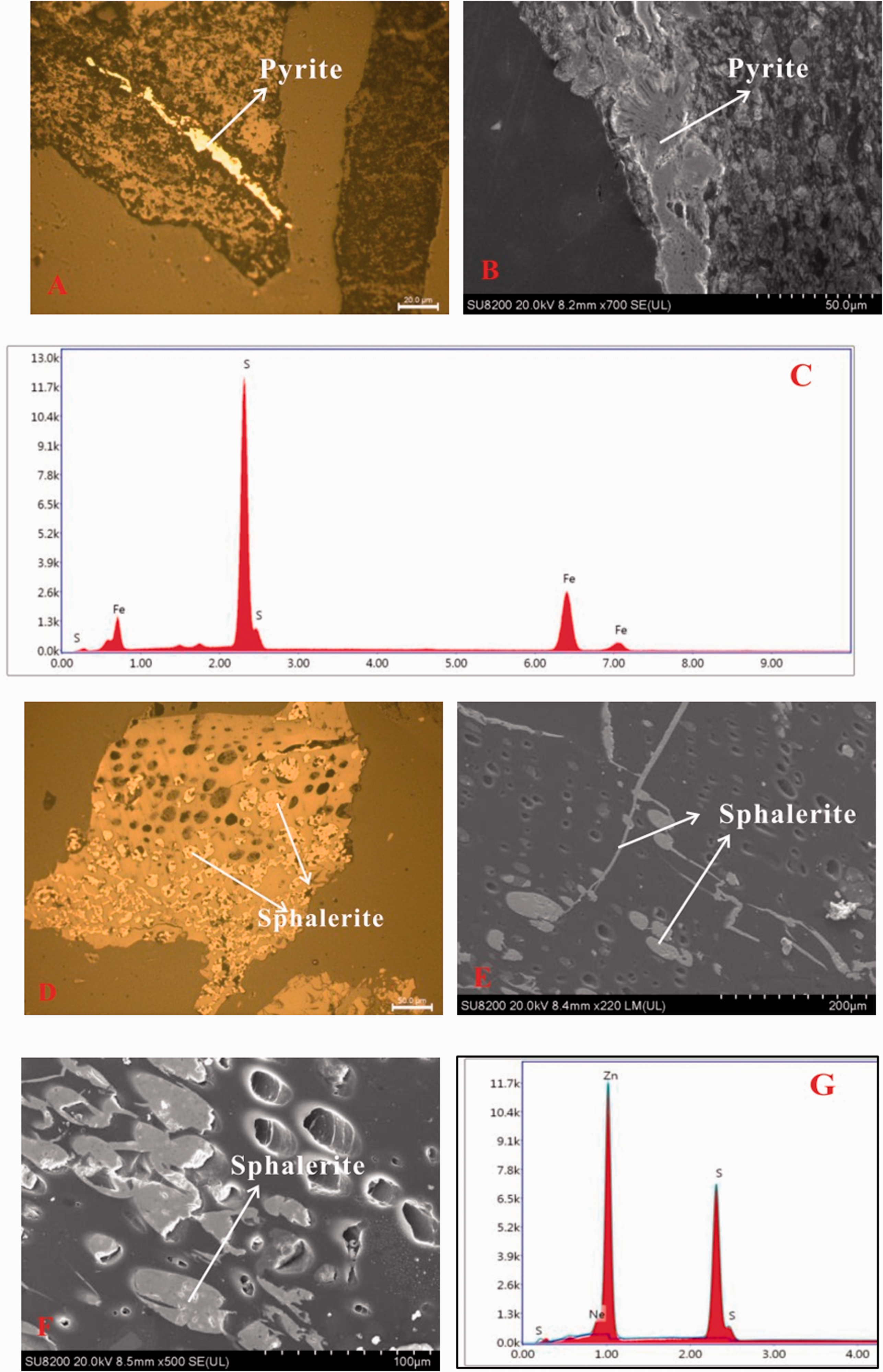
Sulfide minerals in the No.5 coal (a) Epigenetic pyrite in macerals; (b) Pyrite (SEM, secondary electron image) and its energy dispersive X-ray spectrometry (EDS) (c); (d) Sphalerite in fusinite; (e) Sphalerite (SEM, secondary electron image); (f) Sphalerite (SEM, secondary electron image) and its energy dispersive X-ray spectrometry (EDS) (g).
Sphalerite was not identified by XRD and Siroquant; however, it was observed by SEM-EDS in YZS5-8 sample. Sphalerite fills in maceral-cavities both by itself and together with kaolinite (Figures 5(d)–(f)). As described above, the epigenetic sphalerite was formed as a result of precipitation from circulating hydrothermal fluids infiltrating through microfractures into maceral-cavities (Figures 5(d) to (f)) (Karayiğit et al., 2017; Ward,2002, 2016).
Carbonate minerals
In the roof sample, the content of siderite is the highest (47.3%) among all the other samples. However, in the coal bench samples, the siderite can only be occasionally observed by SEM-EDS, rather than detected by XRD and Siroquant. In the No. 5 coal, siderite is present mainly as layers with some netted texture, which is combined with macerals and kaolinite, indicating a syngenetic origin (Figure 6(a) and (b)). Siderite may be formed by CO2 released from organic matter in coals together with Fe2+ in the basin (Gould and Smit., 1979; War., 2002, 2016). It could indicate that Fe and Ca-rich water injected into the palaeomires. Since pyrite was not detected by XRD in the YZS5-1r sample, the presence of siderite could also imply the reduction of sulfate concentrations in palaoemires.
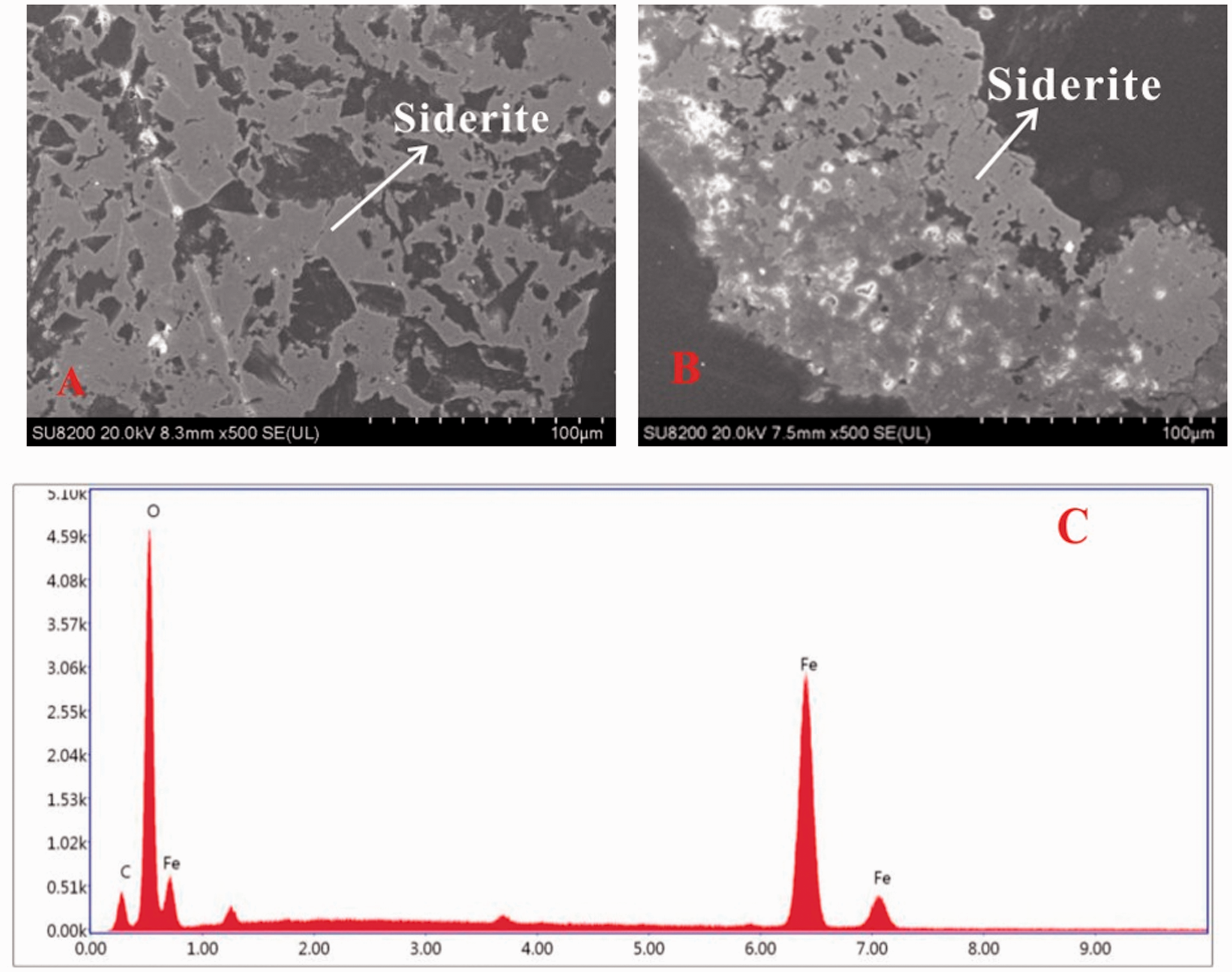
Siderite in the No.5 coal (a) Siderite (SEM, secondary electron image); (b) Siderite (SEM, secondary electron image) and its energy dispersive X-ray spectrometry (EDS) (c).
Elements in coals
Major elements
The major oxides in the No. 5 coal are listed in Table 3. Compared with the Chinese coals (Dai et al., 2012), the No. 5 coal has much higher proportion of SiO2 (mean 16.22%) and Al2O3 (mean 14.57%), to a lesser extent, TiO2 (mean 0.55%) and lower proportion of Fe2O3 (mean 0.60%), MgO (mean 0.05%), CaO (mean 0.06%), MnO (mean 0.02%), Na2O (mean 0.02%), K2O (mean 0.07%), and P2O5 (mean 0.05%), than the average concentrations of Chinese coals. The high concentrations of SiO2 and Al2O3 are consistent with predominately abundant kaolinite in coals.
Concentrations of major oxides in the No. 5 coal (%) (on whole-coal basis).
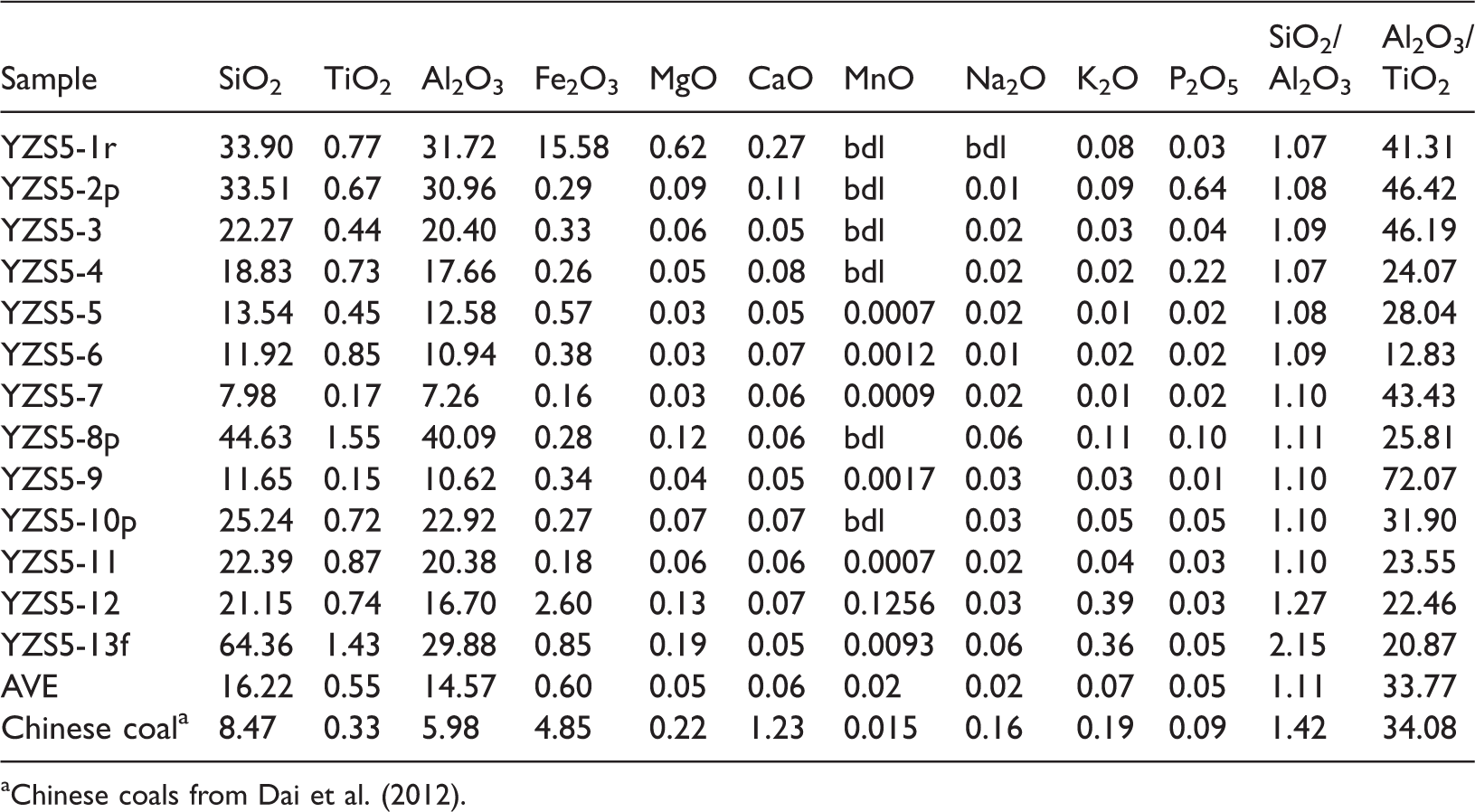
aChinese coals from Dai et al. (2012).
The SiO2/Al2O3 value (1.07–1.27, mean 1.11) is lower than that of the Chinese coals (1.42) (Dai et al., 2012) and relatively close to the theoretical value of kaolinite (1.18). Moreover, sample YZS5-13f has the highest SiO2/Al2O3 value (2.15), reflecting a high quartz content (88.8%). Aluminum and Ti are regarded as essentially immobile elements due to the low solubility of their oxides and hydroxides in low temperature aqueous solutions. Therefore, the ratios of Al2O3 and TiO2 should be close to the characteristics of their parent rocks (Prachiti et al., 2011). The ratios of Al2O3/TiO2 ranging from 3 to 8 indicate mafic source rocks, 8 to 21 represent intermediate source rocks, and 21 to 70 indicate felsic igneous rocks (He et al., 2010). The ratio of the No. 5 coal bench samples ranges from 12.83 to 72.07 (mean 34.08), indicating that the sediments of the parent rocks have felsic or intermediate geochemical characteristics.
Trace elements
Compared with the world hard coals (Ketris and Yudovic., 2009), a large number of trace elements are enriched in the No. 5 coal from the Yanzishan mine (Table 4).
Concentrations of trace elements in the No. 5 coal from the Yanzidshan mine (μg/g) (on whole-coal basis).
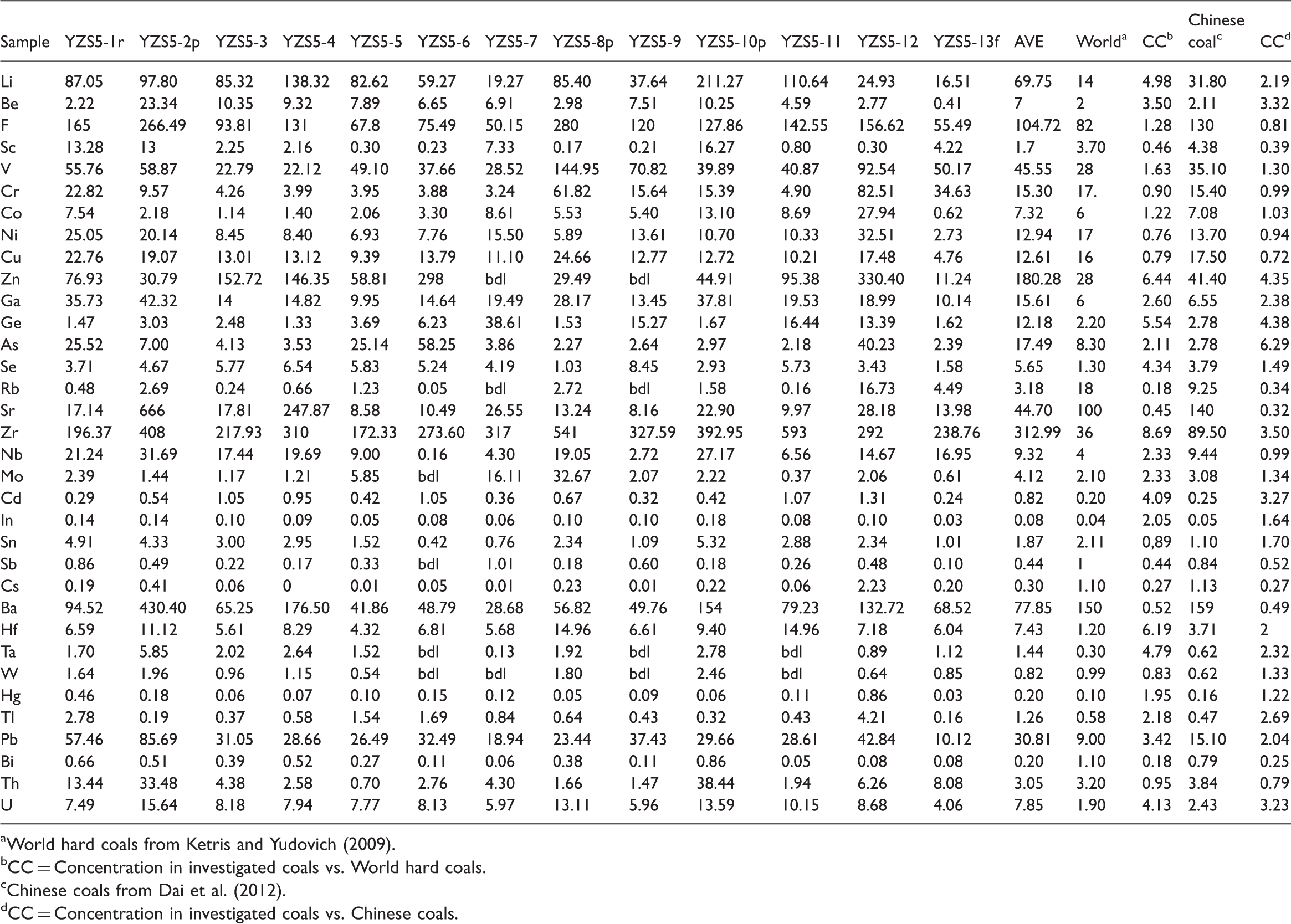
aWorld hard coals from Ketris and Yudovich (2009).
bCC = Concentration in investigated coals vs. World hard coals.
cChinese coals from Dai et al. (2012).
dCC = Concentration in investigated coals vs. Chinese coals.
Trace elements with a CC (CC = ratio of element concentration in the No.5 coal
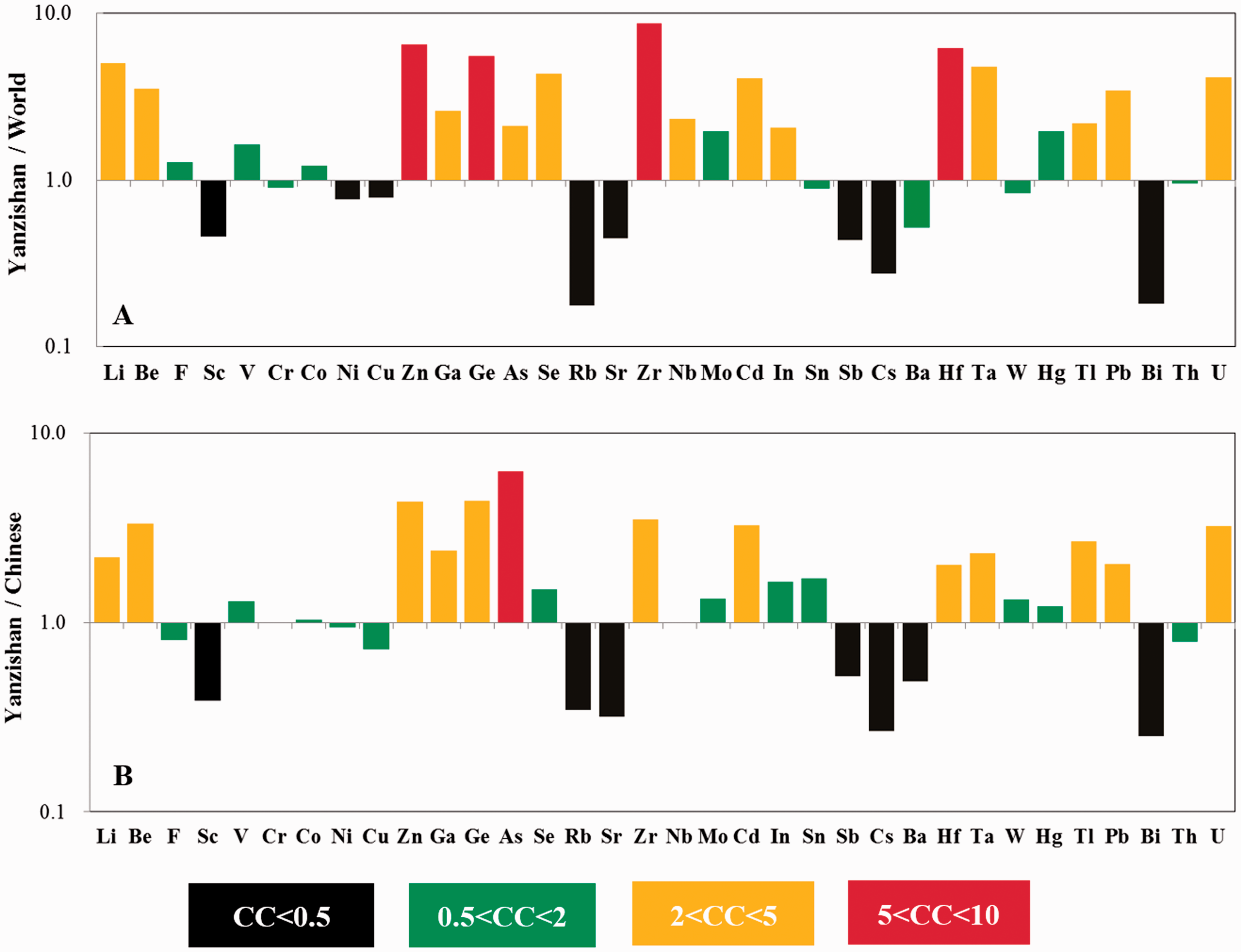
Concentration coefficients of trace elements in the No.5 coal, (a) normalized by average concentrations in the World hard coals; (b) normalized by average concentrations in the Chinese coals.
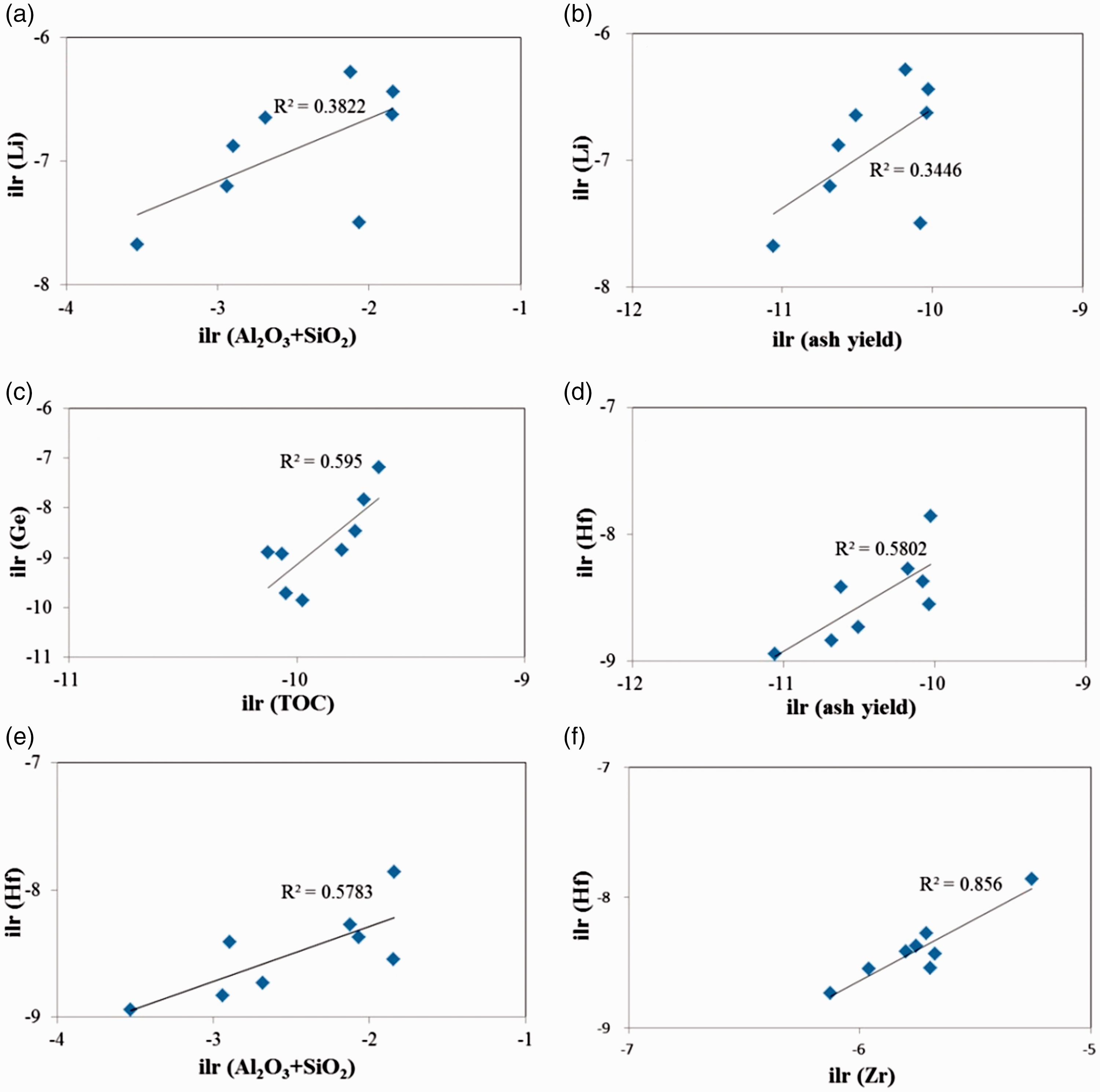
Correlation of (a) Li to Al2O3+SiO2; (b) Li to ash yield; (c) Ge to TOC; (d) Hf to ash yield; (e) Hf to Al2O3+SiO2; (f) Hf to Zr.
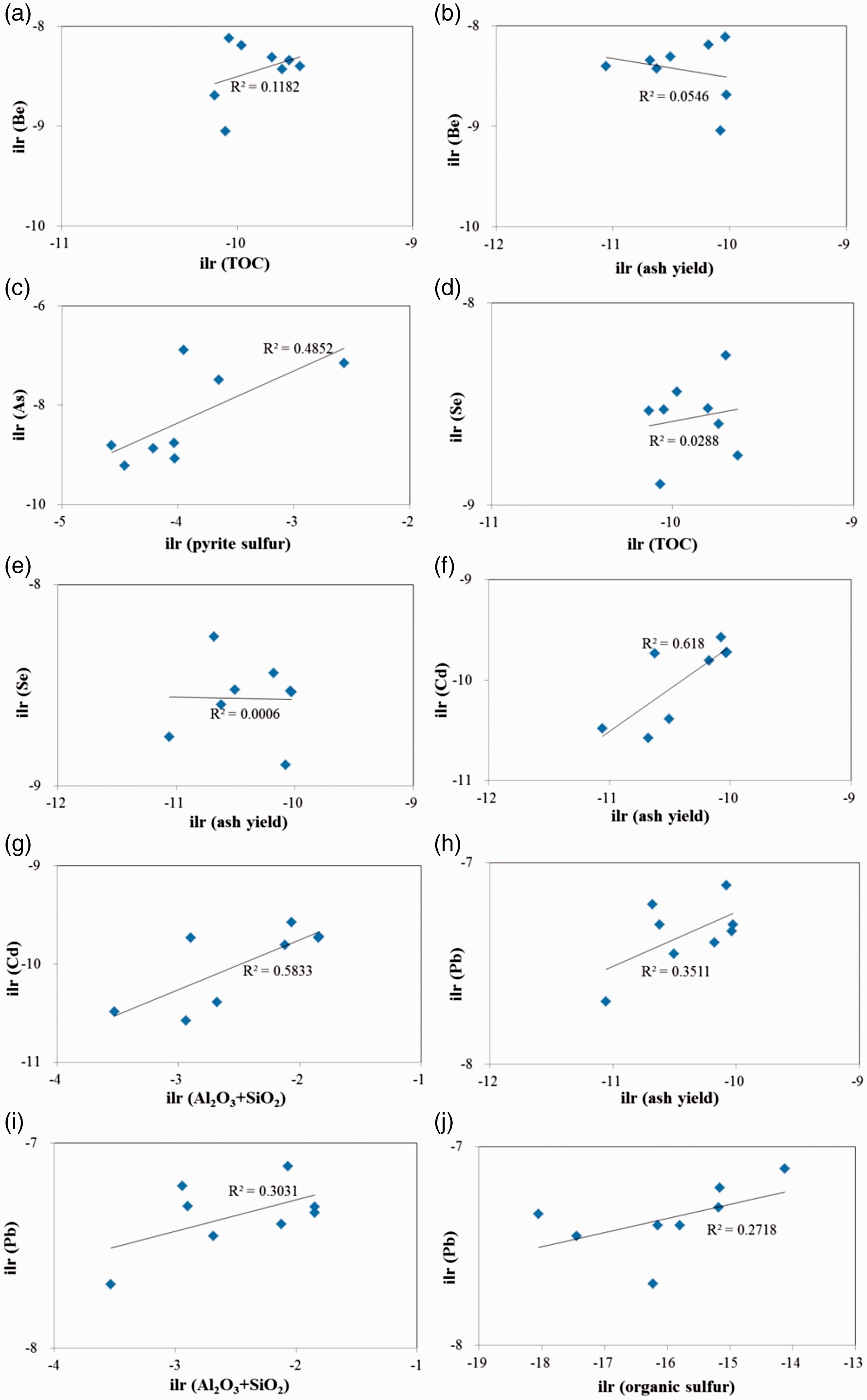
Correlation of (a) Be to TOC; (b) Be to ash yield; (c) As to pyritic sulfur; (d) Se to TOC; (e) Se to ash yield; (f) Cd to Cd to ash yield; (g); Cd to Al2O3+SiO2; (h) Pb to ash yield; (i) Pb to Al2O3+SiO2; (j) Pb to organic sulfur.
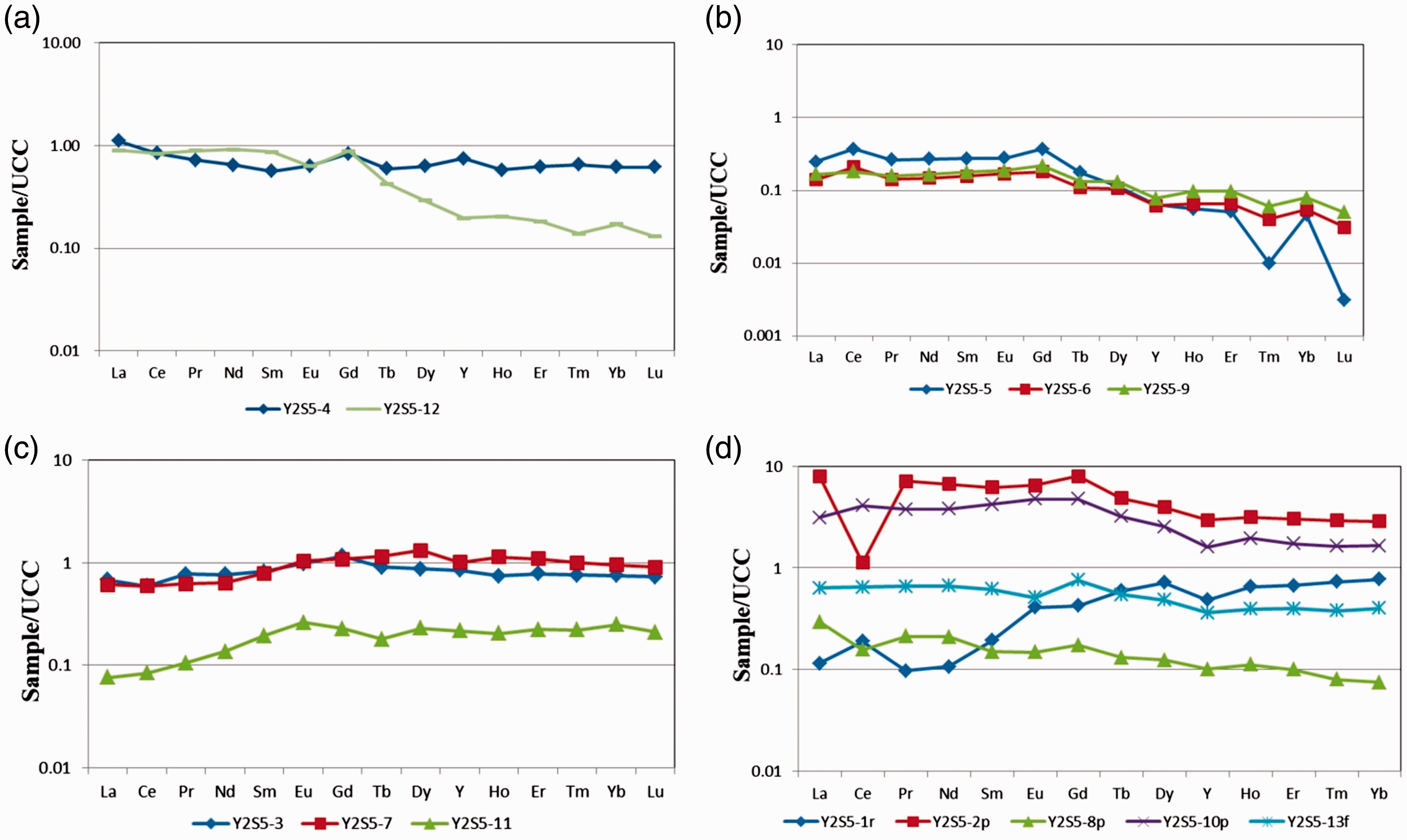
REY distribution patterns in the coal benches, roof, floor and partings, normalized to the upper continental crust (UCC); (a) pattern of L-type in coal benches; (b) pattern of L-M type in coal benches; (c) pattern of M-H type in coal benches; (d) pattern of REY in the roof, floor and parting samples.
Compared with the Chinese coals (Dai et al., 2012), the No. 5 coal is slightly enriched in Li, Be, Zn, Ga, Ge, Zr, Cd, Hf, Ta, Tl, Pb, and U, whereas Sc, Rb, Sr, Cs, Ba, and Bi are depleted. Only As is approximately 6.23 times the Chinese coals and enriched. Moreover, the remaining elements have similar concentrations to those of the Chinese coals.
Affinity of the elements
Affinity analyses among elements provide quantitative measurements of both the occurrence and correlation of elements (Eskenazy et al., 2010; Spears and Zhen., 1999). However, the data of the concentrations of the major and trace elements belongs to the “compositional data” which is a set of proportion and percentages, and therefore, spurious correlations between elements are often observed during the correlation coefficients analyses (Drew et al., 2008; Geboy et al., 2013). As a consequence, a fundamental part of working in compositional data is the use of the isometric logratio (ilr) transformations to process the data in order to eliminate the constraints of the “compositional data”. Thus, by processing data in this method, affinity analyses can be made more reliable to reflect the elemental modes of occurrence (Drew et al., 2008; Geboy et al., 2013).
In this study, all these elements were then divided into four groups according to their correlation coefficients with their ash yields (Table 5 and Figure 7).
Correlation coefficients between the each element in coal and ash yield or selected elements.

Li: Li-TiO2 (0.56), Li-Se (0.53), Li-U (0.58).
Be: Be-Se (0.65), Be-Bi (0.68).
Ge: Ge-Co (0.81), Ge-Zn (0.66), Ge-W (0.62).
As: As-Fe2O3 (0.70), As-Mo (0.64), As-Sb (0.60), As-Hg (0.61).
Cd: Cd-Ba (0.69), Cd-U (0.84).
Zr: Zr-Ga (0.74), Zr-Hf (0.93, Zr-W (0.61).
Hf: Hf-F (0.65), Hf-Ta (0.53), Hf-U (0.58).
Pb: Pb-Fe2O3 (0.71).
Group 1 exhibits a high positive correlation coefficient with ash yield (0.7–1, including MgO, SiO2, Al2O3, TiO2, F, Sn, Cd, Ba, and U), indicating inorganic affinity. The SiO2 and Al2O3 are major constituents of the ash-forming minerals kaolinite, which is attributed to the high correlation coefficients of ash-SiO2 and ash-Al2O3 corresponding to 1.00 and 0.98, respectively. Furthermore, the other trace elements have relatively high correlation coefficients with both Al2O3 and SiO2, indicating an aluminosilicate affinity, mainly in kaolinite. Due to the high correlation coefficients with CaO (r = 0.79) and P2O5 (r = 0.81), it is an indicator that Ba could also be incorporated in apatite.
Group 2 consists of P2O5, K2O, MnO, Li, Nb, In, Hf, and Pb with a lesser, but still relatively high inorganic affinity. Furthermore, TiO2, MnO, Li, Nb, and In have relatively high correlations with SiO2 and Al2O3, indicating that these elements mainly occur in aluminosilicate minerals (kaolinite and illite).
Group 3 includes Na2O, CaO, Fe3O4, Cr, Rb, Sr, and Bi which have correlation coefficients with ash yield varying from 0.2 to 0.39. Strontium is positively correlated with P2O5 (r = 0.92) and CaO (r = 0.75), indicating an apatite affinity. Furthermore, Cr, Rb, and Bi have mixed affinites (inorganic and organic affinities). However, Rb mainly occurs in organic matter, which is due to the relatively high correlation with TOC (r = 0.64).
Group 4 includes Sc, V, Co, Ni, Ga, As, Se, Zr, Ta, Hg, Tl, and Th, which has correlation coefficients in the range of 0.2 to 0.19, indicating the mixed affinities (inorganic and organic affinities). Vanadium, Co, Ni, Ga, and Hg are associated with organic sulfur. Arsenic, Hg and Tl have affinity for sulfide minerals (mainly in pyrite).
Group 5 consists of Be, Zn, Ge, Mo, Sb, and W. These elements have significantly negative correlation with ash yield, probably indicating their organic association. Moreover, Mo, and Sb are also indicative of an affinity for sulfide and sulfate minerals. Correlation coefficient of Zn with TOC (
Potentially valuable elements
Al2O3
Based on the standard DZ/T, 0203–2002 (2003), when the content of Al2O3 in coal ash is more than 40%, it is considered to be economic grade Al2O3 deposits in coal ash, indicating that this coal has attained the status of an Al2O3 deposit. In studied area, the content of Al2O3 in coal ash ranges from 39.75% to 46.65%, with an average of 45.27%. Thus the No. 5 coal can be considered as a high-Al deposit. Previous studies showed that the Yinshan Oldland was the main sedimentary region of the Datong Coalfield during the Late Carboniferous and Middle Permian.80 The strong peraluminous granite and moyite from the Yinshan Oldland may be the main source of Al and rare metal elements in the coals (Peng et al., 2010; Sun et al., 2016). The minerals in the No. 5 coal are mainly represented by kaolinite, which is thought to be the main carrier of Al.
Lithium
The average concentrations of lithium in the common Chinese coals and world hard coals are 31.80 μg/g and 14 μg/g, respectively (Dai et al., 2012; Ketris and Yudovic., 2009). Lithium in the No. 5 coal is slightly enriched, which is due to that the Li concentrations range from 19.27μg/g to 138.32 μg/g (mean 69.75 μg/g). Sun et al. studied Li concentrations in many Chinese coals and reported that it was reasonable to take 80 μg/g Li as the minimum mining grade and 120 μg/g as the economic grade or industrial grade for potentially economic Li beneficiation from Chinese coals (Sun et al., 2012, 2013a, 2013b, 2016). Thus, the majority of No. 5 coal benches have Li concentrations up to the minimum mining grade and the economic grade or industrial grade.
It has also been proposed that Li is in the clay minerals and partly in mica and tourmaline (Finkelma., 1981; Sun et al.,2013a, 2013b; Swain, 1999). In the No. 5 coal, the correlation coefficients of Li-ash, Li-Al2O3, and Li-SiO2 are 0.55, 0.66, and 0.53 (Figures 8(a) and (b)), respectively, indicating that Li largely has an affinity for clay minerals (kaolinite and illite).
Germanium
In some previous studies, although the concentration of Ge in coal ranges from 10 to 100 μg/g, it can be considered that it has reached the industrial scale (DZ/T, 0203–2002, 2003). Germanium concentrations in present study range from 1.33 μg/g to 38.61 μg/g (mean 12.18 μg/g), which are much higher than those of the Chinese coals (2.78 μg/g) and world hard coals (2.0 μg/g). This indicates that Ge in some No. 5 coal benches has commercial value (Dai et al., 2016a; Ketris and Yudovic., 2009).
The low correlation coefficient with ash yields
Zirconium and hafnium
The concentrations of Zr (mean 312.99 μg/g) and Hf (mean 7.43 μg/g) are significantly higher than those in the common Chinese coals and world hard coals (Dai et al., 2012; Ketris and Yudovic., 2009).
The correlation coefficients of Hf-ash (0.76), Hf-Al2O3 (
Potentially toxic elements
When the concentrations of potentially toxic elements exceed the allowable concentration limit (ACL) and maximum allowable concentration limit (MACL), it can be reasonable to consider these elements as imposing the adverse effects on the environment and human health (Singh et al.,2015a, 2016a; Wang et al., 2005). As discussed above, compared with the world hard coals and Chinese coals, the potentially toxic elements, including Be, Zn, As, Se, Cd, Tl, Pb, and U, are relatively enriched in the No. 5 coal. Moreover, in the studied area, the concentrations of Be, As, Se, and Pb exceed the potentially harmful level and should be of the greatest concern (Table 6). Although the average Cd concentration is below the ACL, most of the coal benches have higher concentrations than the ACL. Therefore, it can be believed that Be, As, Se, Cd, and Pb are potentially environmentally threatening elements in the No. 5 coal.
Environmental standard limits of the potentially toxic elements in the No.5 coal (μg/g).
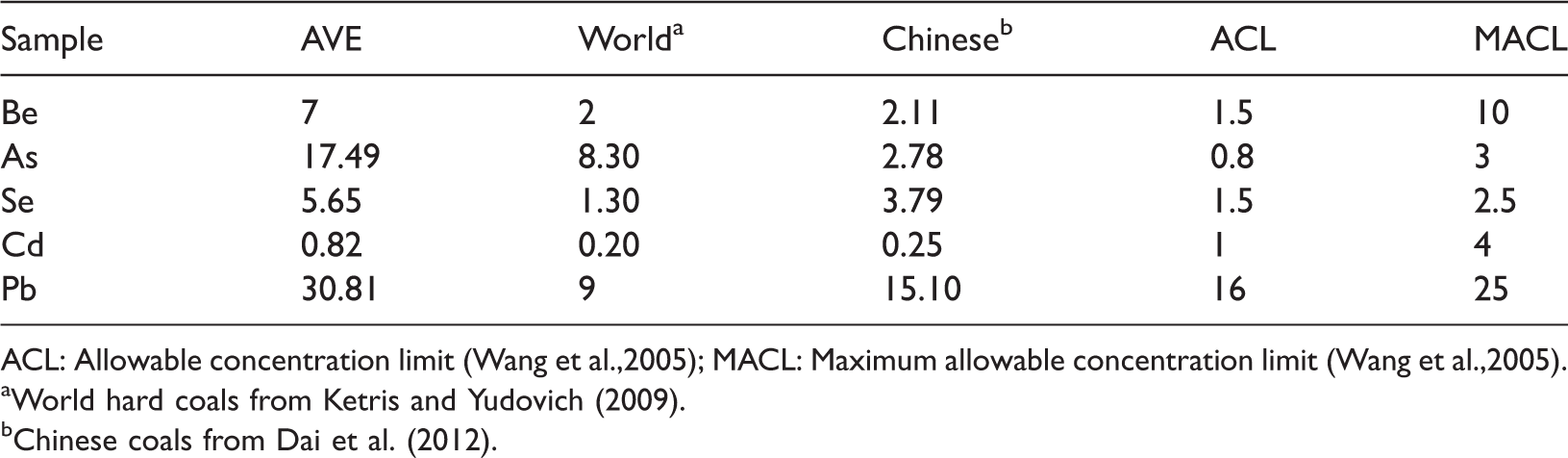
ACL: Allowable concentration limit (Wang et al.,2005); MACL: Maximum allowable concentration limit (Wang et al.,2005).
aWorld hard coals from Ketris and Yudovich (2009).
bChinese coals from Dai et al. (2012).
Beryllium
The Be concentration varies from 2.77 to 10.35 μg/g, with an average of 7 μg/g, which is slightly higher than the averages of common Chinese and world hard coals (2 and 2.11 μg/g, respectively) (Dai et al., 2012; Ketris and Yudovic., 2009).
According to literature, if the Be concentration in coal is high, it is predominantly organically bound; whereas the inorganic form prevails in coal when the Be concentration approximates the Clarke values. Considering the weak correlations among Be with TOC and ash yield (Figures 9(a) and (b)), a mixed affinity could be considered for Be in the No.5 coal.
Arsenic
The average concentrations of As in common Chinese and world hard coals are 8.30 μg/g and 2.78 μg/g, respectively (Dai et al., 2012; Ketris and Yudovic., 2009). In studied area, As is slightly concentrated (mean 17.49 μg/g) in coal benches. Owing to the high toxicity of As and its compounds, it is considered as a potentially toxic element in the No. 5 coal.
Some studies reported that pyrite, sphalerite, galenite, and chalcopyrite can be the most common form of inorganic As (Diehl et al., 2012; Karayiğit et al., 2017; Qin et al., 2018). Moreover, As in association with organic matter has also been observed (Karayiğit et al., 2017; Swain., 1990). In the present study, the correlation coefficient between As and pyritic sulfur (
Selenium
The concentration of Se in the No. 5 coal shows a variation ranging from 3.43 μg/g to 8.45 μg/g, with an average of 5.65 μg/g, which is higher than that of the world hard coals (1.30 μg/g) and Chinese coals (3.79 μg/g) (Dai et al., 2012; Ketris and Yudovic., 2009).
Se is known to have affinities toward pyrite and clausthalite; however, it can also have organic associations (Diehl et al., 2013; Qin et al., 2018; Tian et al., 2013; Wagner and Hlatshway., 2005 ). In the No. 5 coal, Se displays no significant correlation with ash yield (
Cadmium
Compared with the average concentration of Cd in Chinese coals (0.25 μg/g) and world hard coals (0.20 μg/g) (Dai et al., 2012; Ketris and Yudovic., 2009), Cd in the No. 5 coal has higher concentration (mean 0.82 μg/g).
This study indicates that Cd is predominately associated with clay minerals, because of positive correlation coefficients with ash yield (
Lead
The concentration of Pb (mean 30.81 μg/g) is enriched, compared with Chinese coals (9 μg/g) and world hard coals (15.1 μg/g) (Dai et al., 2012; Ketris and Yudovic., 2009).
Previous reports indicate that Pb in coals mainly occurs in galena or it is associated with other sulfide minerals (Finkelman,1981, 1995; Tian et al., 2013) Lead is positively correlated with ash yield (
The rare earth elements and yttrium
Geochemistry of rare earth elements and yttrium
In this study, a threefold geochemical classification of rare earth elements and yttrium (REY) was used. Based on Seredin and Dai’s classification (Seredin and Da., 2012), the REYs were divided into three types: light (L-REY including La, La, Ce, Pr, Nd, and Sm), medium (M-REY including, Eu, Gd, Tb, Dy, and Y), and heavy (H-REY including, Ho, Er, Tm, Yb, and Lu).
The concentration of REY ranges from 20.94 μg/g to 138.08 μg/g, with an average of 77.59 μg/g, which is lower than the average of Chinese coals (135.89μg/g) and closed to that of the world hard coals (68.27μg/g) in the coal benches samples (Dai et al., 2012; Ketris and Yudovic., 2009). The REY contents in YZS5-2p (698.84μg/g) and YZS5-8p (577.87μg/g) are more concentrated than that in the roof, floor and coal benches samples. The content of LREE (12.79–114.45 μg/g, mean 61.76 μg/g) is much higher than that of MREE (2.70–27.36 μg/g, mean 11.69 μg/g) and HREE (0.78–11.47 μg/g, mean 4.14 μg/g) (Table 7).
Concentration and geochemical parameters of REY in the No.5 coal (μg/g) (on whole-coal basis).
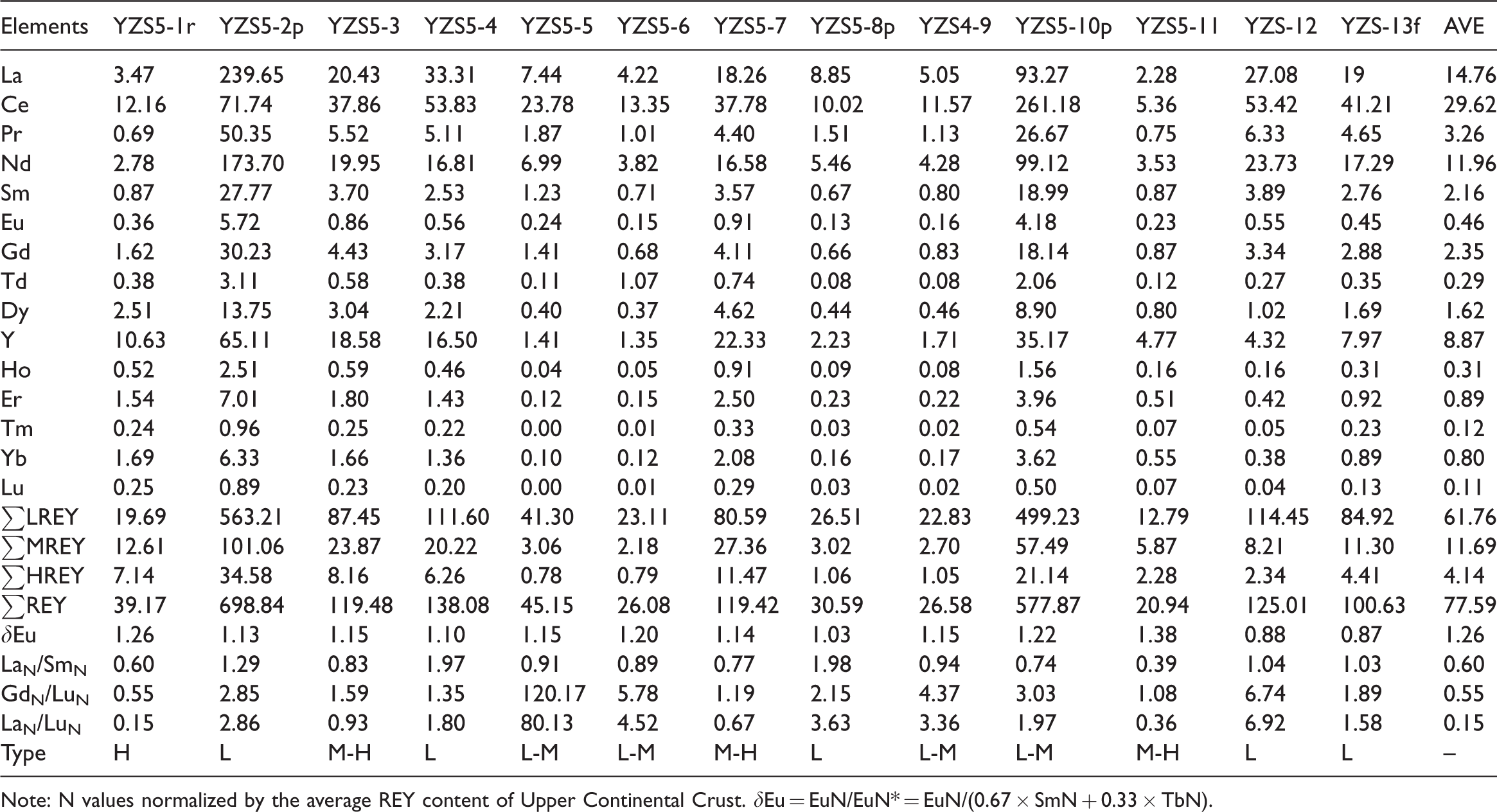
Note: N values normalized by the average REY content of Upper Continental Crust. δEu = EuN/EuN* = EuN/(0.67 × SmN + 0.33 × TbN).
Pattern of rare earth elements and yttrium
Compared to the Upper Continental Crust (UCC) (Taylor and McLenna., 1985), three types of coal benches were identified as: L-type (L-REY; LaN/LuN > 1), M-type (MREY; LaN/SmN < 1, GdN/LuN > 1), and H-type (H-REY; LaN/LuN < 1) (Dai et al., 2016b). All the coal samples are characterized by weakly positive Eu anomalies (δEu = EuN/EuN* ranging from 0.88 to 1.20, average of 1.14).
The REY enrichment patterns can be characterized as M–H type in most coal benches (YZS5-3, YZS5-7, and YZS5-11) (Figure 10(c)). The samples (YZS5-5, YZS5-6, and YZS5-9) are characterized as L–M type enrichment as well as one parting (YZS5-10p) (Figures 10(b) and (d)). The L-type enrichment pattern includes two coal bench samples (YZS5-4 and YZS5-12), two partings (YZS5-2p and YZS5-8p) (Figures 10(a) and (d)), and one floor sample (YZS5-13f). Only one roof sample (YZS5-1r) belongs to H-type enrichment pattern. The L-type enrichment generally reflects a terrigenous origin (Seredin and Da., 2012). The H-type enrichment is probably due to the injection of hydrothermal solutions, while the M-type is possibly related to natural waters (Seredin and Da., 2012).
Yan et al. showed that positive Eu anomalies could be caused by over estimation of Eu during the ICP–MS analysis due to interference from BaO or BaOH (Yan et al., 2018). If Ba/Eu is >1000, the interfered Eu is highly elevated. This indicates that when samples contain Ba/Eu less than 1000, the interference of Ba on Eu can be ignored (Yan et al., 2018). However, no significant correlation between Ba and Eu concentrations were recorded and lower Ba/Eu values (226.1 on an average) in the investigated samples indicate that the positive Eu anomalies in these samples are not caused by the interference of Ba.
The ratio of Al2O3/TiO2 (mean 34.08) indicates that the sediment-source region of the No. 5 coal is characterized by rocks with felsic or intermediate-felsic detrital materials. Coals with aforesaid input usually display distinct negative Eu anomalies; however, all the studied samples are characterized by no pronounced and weakly positive Eu anomalies. Dai et al. showed that the detrital input of alkali mafic rocks and the injection of high-temperature hydrothermal solutions coupled with an extremely reducing environment could cause positive Eu anomalies in coals. The coal bench samples are characterized by L-type and L–M type enrichment with the weak negative Eu anomalies and weak poaitive Eu anomalies, indicating the main origin from felsic or felsic-intermediate detrital materials and to a lesser extent, from mafic detrital materials. Dai et al. reported that the strong positive Eu anomaly was expected to be due to the injection of high-temperature hydrothermal solutions (>200°C) coupled with an extremely reducing environment (Dai et al., 2016b). The strong positive Eu anomaly would be due to the transformation from Eu3+ to Eu2+, which not only requires extremely reducing conditions but also high temperature. However, the samples with the H-type and M-H type enrichment patterns showed no pronounced and weak positive Eu anomalies, indicating that the temperature of the hydrothermal fluids circulating in the basin was relatively low (<200°C) (Dai et al., 2016b; Seredin and Da., 2012). Thus, in present study, the samples with M- and M-H type enrichment are characterized by the weak positive Eu anomalies, which may be attributed to the injection of low temperature hydrothermal solutions (Seredin and Da., 2012).
Overall, it can be inferred that the REY might have originated from sediment source region (Yinshan Oldland) (Sun et al., 2016) and low temperature hydrothermal solutions that might have circulated in coal basin.
Conclusion
In this study, the mineralogical compositions, concentrations, and occurrence modes of elements in the No. 5 coal in Yanzishan mine were investigated by various techniques. The results indicated that the No. 5 coal is characterized by high ash yield content (mean 32.30%) and low sulfur content (mean 0.47%). The pyritic sulfur is a dominant form in all the samples. Total organic carbon (TOC) was determined to be in the range from 39.9% to 65% (mean 51.51%).
The suite of minerals found in coal includes kaolinite, quartz, and pyrite, with a small proportion of illite, apatite, siderite, sphalerite, and anatase. The kaolinite, which occurs in infillings of cell-cavities of macerals with a well crystallized or vermicular shape, indicates syngenetic and authigenic origins. The quartz mainly has a detrital origin. The epigenetic pyrite and sphalerite were considered to be derived from the low temperature hydrothermal fluids.
Some potentially valuable and hazardous elements with elevated concentrations in the No. 5 coal include Al2O3, Li, Be, Ge, As, Se, Zr, Hf, Cd, and Pb. Potentially valuable elements (Al2O3, Li, Ge, Zr, and Hf) might be recovered as value-added byproducts from coal ash. Toxic elements (Be, As, Se, Cd, and Pb) may impose potential adverse effects on the environment and human health during coal processing. Lithium and Hf are mainly associated with clay minerals; Be, Se, and Zr mainly have both inorganically and organically bound; As occurs mainly as a sulfide form in pyrite; Cd is incorporated in clay minerals; and Pb largely occurs in clay minerals, followed by organic sulfur.
The concentration of REY ranges from 20.94 μg/g to 138.08 μg/g, with an average of 77.59 μg/g. All the coal samples were characterized by weak positive or no pronounced Eu anomalies, indicating that the REY likewise originated from granite of the Yinshan Oldland together with injection of the low temperature hydrothermal fluids.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/orpublication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Program of the National Natural Science Foundation of China (No. 41330317).
