Abstract
Aedes (Stegomyia) albopictus has been identified for the first time in the municipality of La Tebaida (Quindio department of Colombia), an area with active transmission of dengue fever by Ae. (Stegomyia) aegypti. Specimens of Ae. albopictus were detected in a tire used as an ovitrap in a remnant of bamboo plantation in a rural area of the municipality of La Tebaida; Ae. aegypti presented co-occurrence and both species were molecularly typed using cytochrome oxidase I (DNA barcode region). The first DNA barcode sequences were recorded for 13 Colombian specimens of Ae. albopictus, identifying an asiatic origin (Singapore) and demonstrating the utility of this method for molecular identification. These sequences can be used to identify genetic flow with other populations in Colombia, in ecological studies, and in studies of vector incrimination in outbreaks of emerging and re-emerging arbovirus in Colombia.
Introduction
Aedes albopictus (Skuse, 1894) is a prominent native mosquito originating from Southeast Asia with anthropophilic eating habits that is considered to be a vector-bridge between the enzootic cycle of West Nile virus and susceptible humans (Turell 2001; Sardelis 2002). Additionally, it is a vector of 22 arboviruses: Flavivirus (dengue, West Nile virus, yellow fever), Alphavirus (chikungunya, Eastern Equine Encephalitis virus), Orthobunyavirus (Tensaw virus, Potosi virus, Cachey Valley virus, LaCrosse virus), and nematodes [Dirofilaria immitis (Leidy, 1856), Dirofilaria repens Railliet & Henry, 1911] (Cancrini et al. 2003; Gratz 2004; Tilston et al. 2009). The range of geographic expansion of this species has increased to Europe, Africa, and America (Kraemer 2015) through the sale of used tires (Reiter 1998) and bamboo (Demeulemeester et al. 2014) and the carriage of adults in commercial aircraft (Gratz 2000). In Colombia, it was first recorded in Leticia-Amazonas in a collection using human bait (Vélez et al. 1998) and was subsequently identified in Buenaventura (Suárez 2001), Cali (Cuellar et al. 2007), Barrancabermeja (Gutiérrez et al. 2010), and Medellin (Rúa et al. 2011). The presence of this species in geographically distinct locations is probably the result of land transport, human passive dispersal, and the failure of entomological surveillance programs associated with dengue in Colombia (Rúa-Uribe et al. 2012). The proven vector competence of Ae. albopictus (Gratz, 2004); the circulation of Flavivirus, Alphavirus and Orthobunyavirus in Colombia (Groot 1964; Rivas et al. 1995; Groot et al. 1996; Mattar et al. 2005; Hoyos et al. 2012; Muñoz and Navarro 2012); the recent introduction of the Chikungunya virus in 22 departments of the country (Mattar and González 2015); and favorable ecological conditions for the establishment of the insect populations in both rural and urban areas could imply a long-term vectorial role and serious epidemiological implications for human health. During dengue epidemiological surveillance activities, Ae. albopictus was identified in the municipality of La Tebaida (Quindio, Colombia) and the first sequences cytochrome oxidase I (DNA barcode) were recorded to identify possible phylogeographic origins and permit the tasks of entomological surveillance and vector incrimination.
Materials and methods
Following information from the vector control program of the secretary of health of the Quindio department about the identification of Ae. albopictus in a larvae trap in the municipality of La Tebaida, an entomological surveillance outing was coordinated in February 2015 in the Sector Las Brisas - Vereda La Palmita, of the rural jurisdiction of the municipality of La Tebaida (4°25'50.61"N, 75°51'37.24"W), Quindio department in order to collect immature mosquitoes. Sampling was done following Belkin et al. (1969). Immatures were collected in both artificial and natural breeding places associated with human domicile using a plastic pipette. Samples were placed in containers previously encoded for transport to the Center for Biomedical Research (CIBM) of the University of Quindio for observation until adulthood and later identification considering external morphological characteristics and dichotomous keys (Lane 1953; Forattini 2002; Rueda 2004; González and Carrejo 2007). Legs from specimens identified as Ae. albopictus were removed and DNA extracted utilizing the potassium acetate (AcK) method modified by Rosero et al. (2010). DNA extracts were used to amplify the DNA fragment barcode (barcode) of ~ 700 nt of mitochondrial gene cytochrome oxidase I with the MTNF / MTNR (Hebert et al. 2003; Kumar et al. 2007) oligonucleotides. Each PCR mixture contained 1x NHSO

Tire-ovitrap in the guadua remanent (rural area from municipality of La Tebaida, Quindío Department) where were found Ae. albopictus and Ae. aegypti.
Results
A total of 55 Aedes immature in stages, from L
Polymorphic sites between close COI-haplotypes of Singapore and Ae. albopictus sequences from La Tebaida (Quindio).
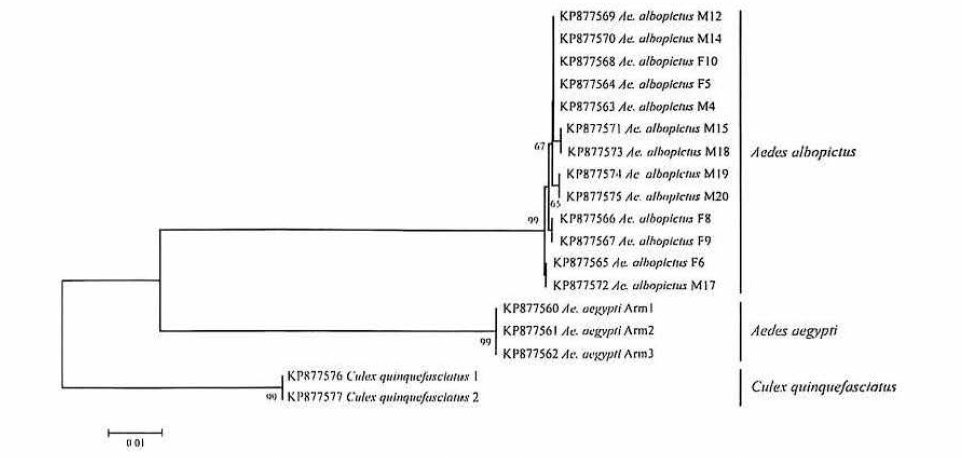
Neighbor-joining dendrogram estimated with sequences of cytochrome oxidase I obtained of Aedes albopictus adults, Ae. aegypti and Cx. quinquefasciatus (Kimura-2-parameter, bootstrap = 1000 replicates). The branch values indicate the bootstrapp clusters in same MOTU (values > 50). The final alignment was of 692 nucleotides and GenBank numbers accessions are in front of every specimen typing.
Discussion
Differentiation and taxonomic identification of Culicidae is a priority in vector incrimination and disease control (Besansky et al. 2003), however, the high morphological similarity in diagnostic features among vector and non-vector species, species complexes, and cryptic diversity prevents biodiversity and epidemiological studies (Cywinska et al. 2006). Significant efforts in the characterization of molecular markers in order to resolve these taxonomic problems and provide for the rapid recognition of vectors have been made. As was shown our results, cytochrome oxidase I - barcode fragment (Hebert et al. 2003) is highly reliable for the identification of a wide range of mosquitoes, the split of species complexes (Cywinska et al. 2006; Kumar et al. 2007), and confirmation of invasive mosquitoes (Golding et al. 2012). In our case, the sequences of COI-barcode reported belonging to Ae. albopictus allowed differentiate to species with which it shares habitats and geographical areas in Colombia, thus this tool can help clarify important ecological questions about the occupation and/or segregation of habitats in rural and urban areas (Olano and Tinker 1993; Silva et al. 2006; Valentini et al. 2008), identification of immatures stages (Dhananjeyan et al. 2010), prediction of niche (Medley 2010), ecological competition (Murrell and Juliano 2008), taxonomic confirmation (Oter et al. 2013) and arboviruses transmission (Cook et al. 2005). Another relevant interest about DNA barcode methodology to level-species is that marker should be indicating population aspects about structure, gene flow and phylogeography (Hajibabaei et al. 2007), in a context for invasive species as Ae. albopictus in Colombia. The phylogeographic origin of the La Tebaida population related to Singapore specimens, similar results find Zhong et al. (2013) for specimens collected from 2001 in Los Angeles - California, this information is complementary to Asiatic route for introduction of Ae. albopictus in Colombia, where Navarro et al. (2013) related Colombian haplotypes with Hawaii populations using ND5, suggesting as most probably hypothesis the introduction from Hawaii or directly through the trade exchange from Africa through the Pacific port of Buenaventura, location with presence of haplotypes related to Asian populations. Interestingly, there is a significant molecular differentiation between haplotypes La Tebaida and Singapore, in this sense, multiple introductions and adaptation to Colombian ecosystems may involve new variability in COI, reflecting population evolution. The COI DNA barcode characterization for populations of Ae. albopictus in Medellin, Buenaventura, Leticia and Barrancabermeja could help to identify phylogeographic origins, colonization and dispersion routes taking account the limited information by other mitochondrial regions (ND5, COI, CytB) and increasing the geographical sampling in American countries with reported Ae. albopictus (Argentina, Cuba, Mexico) and others African/Asiatic populations (Birungi et al. 2002; Mousson et al. 2005; Navarro et al. 2013). By the way, this is the major advantage of COI-DNA barcode: connectivity and common language of DNA sequences for different research groups working in locations inside geographic range of target species insect (Hoyos et al. 2012), allowing typing more sequences of different sites and taking advantage of the high genetic variability of COI for studies in flow and structure populations (Cook et al. 2005).
The emergence and re-emergence of pathogenic microorganisms depends on the convergence of ecological and evolutionary factors that allow the disease in susceptible human hosts (Hoyos et al. 2012); the recent introduction of chikungunya and its epidemic outbreak in Colombia, the presence of arboviruses of epidemiological importance, and the geographical records of Ae. albopictus are indicative of risk for the emergence of new pathogens and consequent outbreaks in human populations. On this regard, the vectorial role of Ae. albopictus in outbreaks of dengue/chikungunya and the role it plays in communities of competent vectors on an ecological level in the occupation of natural and artificial habitats is important. Molecular characterization of Ae. albopictus with COI DNA barcode region should contribute to knowledge about the flow and genetic structure of Colombian populations of this species and identify the phylogeographic origin of recent populations detected in Medellin, for identify possible points of entry into Colombia and to increase entomological surveillance in these locations for the purpose of intercepting new sources of foreign mosquito introduction (Oter et al. 2013; Demeulemeester et al. 2014).
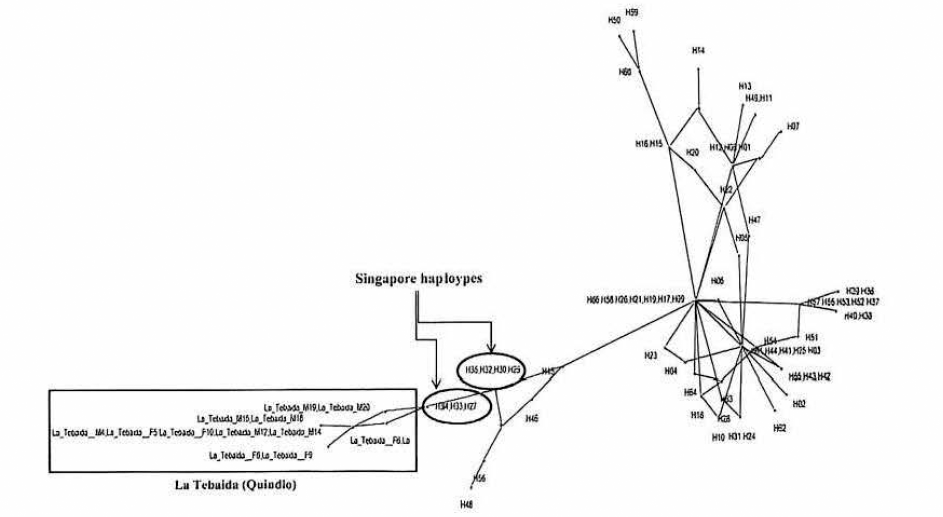
Phylogenetic Network estimated with haplotypes registered by Zhong et al. (2013) and sequences characterized for Ae. albopictus from La Tebaida (Quindío). The haplotypes enclosed in red belonging to Singapore.
Footnotes
Acknowledgements
To the National Doctoral Program - Colciencias for the scholarship granted for Richard Lopez Hoyos (number 528). The authors wish to thank to Jesus Arias and Carlos Salazar of Secretary Health of Quindio's Department, for their technical assistance in the entomological sampling.
