Abstract
Nuclear medicine requires the use of medical radioisotopes for a wide variety of procedures, which have saved millions of lives. The most commonly used medical radioisotope is technetium 99 m, which is usually derived from the molybdenum created in medical research reactors fueled with above-average levels of enriched uranium. But the number of these reactors has declined to a handful of widely scattered facilities, at the same time as there is increasing concern over the risks of shipping large amounts of highly enriched uranium fuel—often enriched as high as 90 percent, a level considered “weapons grade”—over long distances. One solution tried in the past was to convert research reactors to use lower levels of enriched uranium. An alternative may be to use particle accelerators in place of reactors for producing molybdenum. Another possibility would be for medical practitioners to switch to some other, yet-to-be-determined, non-fission-produced isotopes. In either case, much more research will be needed, along with funding and support from the public and private sectors.
During recent negotiations, Iran tried to justify its nuclear program in part by arguing that it needed a supply of uranium that was enriched at a higher-than-usual level to produce medical nuclear radioisotopes. Such isotopes are used for medical diagnostics and therapy, and are produced in reactors using fuel enriched to a near 20 percent level. By this reasoning, Iran said that it needed enrichment well above that of the usual 5 percent level of enrichment used to operate a commercial nuclear reactor.
But critics charged that these assertions were just a smokescreen for quietly building up a stockpile of material at an enrichment level that is relatively easy to upgrade into the 90 percent version used in weapons-grade nuclear bombs—which Iran denied. After much back-and-forth, the speaker of the Iranian Parliament eventually said that Iran had more of this radioactive material than it needed for medical use, and the country ultimately agreed to limit its future enrichment to the 5 percent level needed for civilian nuclear power and to dilute its existing stockpile of 20 percent enriched material.
Complicating everything was the matter of terminology. Enrichment levels can be thought of as points on a continuous spectrum, with the cutoffs being somewhat arbitrary. In addition, researchers in a field such as nuclear medicine use “low-enriched uranium” to mean something different from those studying the control of weapons proliferation.
Nevertheless, there is general agreement that enrichment levels of 90 percent or higher are considered “weapons-grade,” e.g., ready to be loaded into a bomb. And enrichment levels of 4.5 percent—such as that found in civilian nuclear reactors—are considered “low.”
The disagreement about terminology is most acute between the two extremes, where medical research reactors are working with fuel enriched to a level of roughly 20 percent (the exact percentage may vary by one-half of a percentage point). In that region, the fuel is considered to still be possibly “weapons usable”—that is, a weapon could theoretically be built from it, but to do so would require hundreds of kilograms of material and would be extremely difficult, with the difficulty increasing as the enrichment level drops. But that does not mean that material enriched to a level of 20 percent or below could never be used in a nuclear weapon; it is just considered to be “not feasible,” according to a 1998 Oak Ridge National Laboratory report, “Definition of Weapons-Usable Uranium-233” (Forsberg et al., 1998).
What to call fuel enriched at the 20 percent level can be a conundrum, as its enrichment is far above that of civilian reactors yet well below the 90 percent, weapons-grade material. Yet it is still theoretically capable of being used in a bomb. For the purposes of this article, the term “low-enriched uranium,” or LEU, refers to levels in the region of 20 percent enrichment and below, as this is the level at which medical research reactors operate, and it is in keeping with the terminology of those working in the field of medical radioisotopes. Anything above that range is here called “highly enriched uranium,” or HEU.
The haze around Iran’s uranium stockpiles raises seldom-asked questions: Does the production of medical radioisotopes really require using uranium enriched to levels of 20 percent or even higher? Are there alternatives to fission-sourced medical isotopes, with practical solutions in reach that can minimize this pathway to nuclear proliferation, without sacrificing the huge medical benefits these substances have brought to humanity?
In tandem with the sea changes in the world nuclear industry that have occurred since the end of the Cold War, the infrastructure for producing and delivering the nuclear medicines of the 1970s has dwindled to the point where the supply chain for these drugs has become very fragile. Meanwhile, however, advances in nuclear physics and nuclear chemistry suggest it is possible to produce new and potentially better nuclear medicines down the road without uranium-fueled reactors at all. But there is a complicating factor: Medical practitioners are wedded to the older, familiar nuclear drugs, an attitude that is enhanced by economic and regulatory obstacles to innovation in the highly specialized arena of nuclear medicine.
The nuclear medicine complex
One-quarter of the patients admitted to hospitals in the United States receive nuclear medicine treatments such as cardiac scans (aka stress tests) and cancer diagnostics. About 28 million of these procedures are performed yearly, in more than 10,000 hospitals worldwide.
The workhorse of such nuclear medicine is technetium 99 m, first discovered in the late 1930s and in widespread medical use since the 1960s. Today, it is used in 80 percent of all nuclear medicine procedures, including those for the heart, bones, brain, thyroid, lungs, liver, spleen, kidney, tumors, and more. The supply of this material comes from the radioactive decay of the molybdenum 99 produced in nuclear reactors—often called “research reactors”—fueled by uranium enriched to a level in the region of 20 percent.
The use of technetium in medicine over the past four decades has improved the lives of hundreds of millions of people. At the same time, however, it does have drawbacks. The medical literature suggests (Toft et al., 1997) that a significant number of extremely common, technetium-assisted stress tests result in false positives. Such misleading results can lead to repeated testing and increased cost; in the case of false diagnosis, they can cause very costly and high-risk interventions that unnecessarily expose patients to greater medical jeopardy. In addition, patient radioactive exposure from technetium 99 m procedures, while small, is not trivial.
But technetium does have the advantage of being inexpensive, even though the US government recently imposed significant fees on doses. And considering the enormous volume of procedures involving this isotope of technetium, it is unlikely to be replaced wholesale anytime soon.
The deteriorating moly supply chain
There is a serious issue with reactor-manufactured radiopharmaceuticals: The supply chain for molybdenum 99 (often informally called “moly”) and its nuclear daughter, technetium 99 m, is very fragile. This is because the reactors making moly are aging—even leaking in some cases—and need costly and time-consuming repair. In fact, the United States has not produced moly since 1989, when the only domestic reactor making it was decommissioned because of radiation leaks. A handful of reactors in Canada, Belgium, the Netherlands, and South Africa, as pictured in Figure 1, now make about 95 percent of the world’s supply of moly.
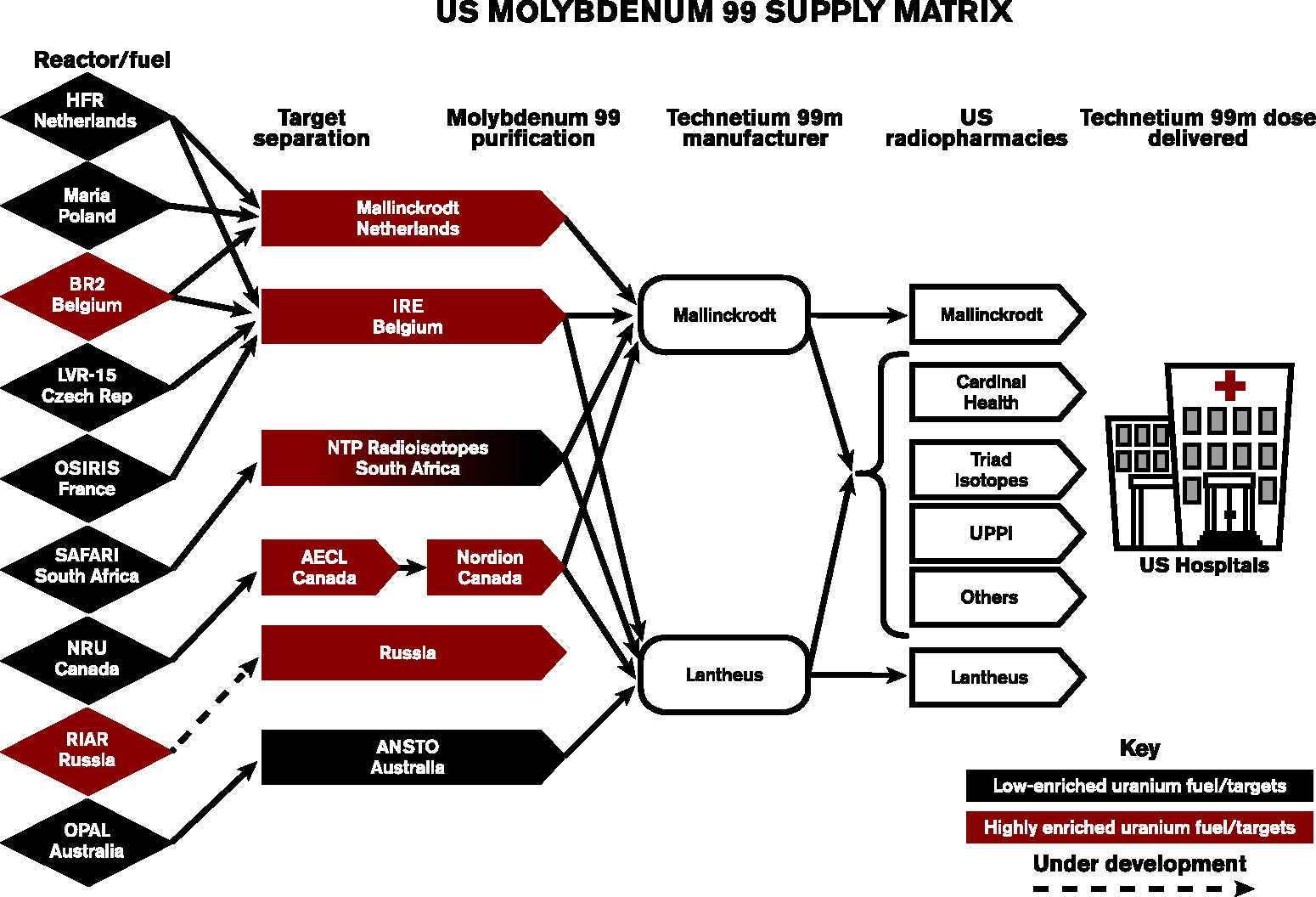
This relatively small number of producers means that whenever even one of these reactors goes down for maintenance or repair (which has happened in recent decades), the world price for nuclear medicine soars. Any crisis in the supply of moly has serious impacts on medical care globally.
For example, when two reactors—in Canada and the Netherlands—were shut down in 2009, the world’s supply of moly was reduced by 67 percent. As a result, the price of technetium 99 m spiked, leading to widespread international concern and major studies by the Nuclear Energy Agency of the Organisation for Economic Co-operation and Development, as well as by the International Atomic Energy Agency. When the reactors came back online, there was a drop in technetium prices.
And not only were prices affected; the technetium shortage also resulted in lengthy delays in diagnosis and treatment for patients with serious conditions.
Now this chain reaction may be about to occur again. In November 2013, a leak in South Africa’s research reactor caused it to be closed, resulting in an immediate global radioisotope shortage. That same month, Canada’s Chalk River reactor—which makes about one-third of the world’s supply of molybdenum 99—had to shut down temporarily. And an article in the December 12, 2013 issue of
In the short term, it is understandable that shipments of HEU were sent overseas to reactors that are still safe and functioning to forestall or offset a shortfall in the supply of technetium 99 m. In the long run, however, such HEU shipments do, in effect, increase the proliferation of nuclear material that is of weapons grade. This illustrates the fundamental tension in US policy on nuclear medicines: medical necessity versus nonproliferation imperatives. Despite the obvious immediate need, sending highly enriched uranium outside the United States flies in the face of national and international policy declarations to stem an important proliferation threat (Kuperman, 2006)
LEU-sourced isotopes: The near-term alternative
One of the obvious alternatives to using reactors fueled with 90-plus percent, weapons-grade HEU is to convert the production process to using fuel enriched with LEU. It is important to distinguish between two aspects of this “conversion” process—first, the enriched uranium that fuels the reactors themselves, and second, the composition of the targets that are bombarded and that actually generate the isotopes, such as molybdenum 99, that ultimately decay into usable medical isotopes like technetium 99 m.
In recent years, there has been significant progress in downgrading the fuel used in research reactors from HEU to LEU. According to the NNSA, with the exception of Russia’s Research Institute of Atomic Reactors facility, all but one of the world’s eight reactors that produce moly have been converted to LEU fuel.
The radioactive targets that actually generate the molybdenum 99 isotope, however, are still problematic, precisely because the potency of the fuel has been reduced from an enrichment level of 90 percent, weapons-grade material to the near-20 percent level of a medical reactor, or a factor of 4.5. Consequently, the fuel of such an LEU reactor would need 4.5 times as much uranium 235 as an HEU reactor, because the fuel is weaker by that ratio. However, to some extent, design changes such as the use of higher density uranium fuel, closer packing of fuel elements, and modified shapes can often compensate for the lower uranium 235 concentration in LEU. The same is true for the production target design.
But figuring out how exactly how to resolve all these variables is still an elusive technical problem. Under the American Medical Isotopes Production Act of 2012, the National Nuclear Security Administration is funding research at the Brookhaven and Los Alamos national laboratories that aims to address this key issue. Thus, while 95 percent of the targets used in isotope production have now been converted to LEU—an impressive accomplishment—significant technical challenges remain, and HEU-free production of molybdenum 99 and technetium 99 m will be more theoretical than real.
Apart from these technical obstacles, and more significantly in terms of global security, the substitution of LEU for HEU, either as fuel or in targets, in medical radioisotope production goes only so far in reducing proliferation. As anyone following the Iran discussion now knows, it is possible to enrich the LEU used in medical research relatively quickly to the 90 percent HEU material used to produce weapons, given the right equipment—which Iran managed to acquire through various means and then embed in fortified locations. There is also the less obvious but important problem of a decline in economic competitiveness, because potential US producers of LEU-fueled moly are at an inherent disadvantage compared with foreign vendors. This imbalance occurs because the United States does not subsidize this industry (which actually ceased producing moly at all in 1989), while foreign governments, wishing to maintain their stocks of HEU and near-HEU material, generally do so.
The problem of economic competitiveness was further compounded when the Centers for Medicare and Medicaid Services of the US Department of Health and Human Services imposed a $10 add-on fee to all radiopharmacological procedures using HEU-sourced technetium 99 m—in effect, asking US hospitals receiving Medicare and Medicaid reimbursements to pre-pay some of the costs of building the technology needed to make the conversion away from HEU and LEU on a large scale, and to another technology.
But what is this new technology?
Non-fission isotopes: The next generation
Fortunately, there is good nonproliferation news on the horizon, courtesy of basic scientific research. There is growing confidence that it is possible to produce effective radioisotopes—sometimes even more effective than those currently in use—without using nuclear reactors at all.
For example, rubidium 82, the nuclear daughter of strontium 82, is an accelerator-fueled isotope that is already being used in some cardiac positron emission tomography, or PET, scans. Though rubidium 82 has a very short half-life of only 1.27 minutes, it can be administered bedside with a generator in a manner similar to that already used with technetium 99 m. And rubidium 82 does not appear to entail as much radioactivity exposure as technetium 99 m. However, there are natural and technical limitations on the amounts of rubidium 82 that can be produced for medical use: This substance is produced by the fission of strontium 82, which yields much less rubidium 82 in comparison to the amount of technetium 99 harvested from a comparable amount of molybdenum 99. In addition, technetium 99 has a 6-hour half-life, which has made it ideal for the development of a large variety of technetium 99-based radiopharmaceuticals that are used in the imaging of different organs. Because rubidium 82 has a half-life of only 76 seconds, it cannot be used to duplicate the production of these radiopharmaceuticals. Thus it is exceedingly unlikely that rubidium 82 will ever replace the popular technetium 99 m.
However, there are many other radioisotopes that are candidates for medical use. A paper published by the American Association for the Advancement of Science lists dozens of accelerator-sourced isotopes and concludes: Accelerators can supplant reactors entirely in nuclear medicine. For technetium 99m, switching to accelerator production may reduce costs—at the very worst, it will not significantly increase them compared to procedure costs. And the necessary accelerator infrastructure, if diverted to fissile material production, would be able to produce only very small quantities of weapons-usable fissile material. Furthermore, this infrastructure would be spread out globally, so that in any one state only a fraction of the total amount of fissile material could be produced. This suggests, as discussed, that the proliferation risk of accelerators is very small when compared to the uranium enrichment and plutonium production inherent in operating reactors for medical isotope production. (Updegraff and Hoedl, 2013)
There may be added benefits—medical and economic—from switching to non-fission isotopes, in addition to their nonproliferation benefits. For example, non-fission radioisotopes may cause fewer false positive results than the current standby, technetium 99 m.
The economics of bringing this new generation of radioisotopes into use, however, are daunting. Much hinges on the right market conditions for developing non-fission radioisotopes. There will be capital construction costs for new accelerators and the expenses of bringing new production processes online. Aside from these direct costs, a key problem, especially in the United States, is regulatory. While important to public health, the Food and Drug Administration’s rules are time-consuming and expensive to comply with.
Another obstacle may be reluctance on the part of the US medical community to move away from technetium 99 m, which for decades has been the mainstay of millions of procedures. In the absence of compelling scientific evidence of the effectiveness of new drugs and the market pressures to use them, medical practitioners will stick with the known and resist the unknown. In the final analysis, all the arguments in the world for switching away from fission-based radiopharmaceuticals will not be as persuasive as these pragmatic factors.
The radiopharmaceutical path forward
In deciding how to change radiopharmaceutical production methods, the first consideration will be differentiating between short- and medium-term needs. In the short term, the world medical community will no doubt continue to rely on HEU-fueled fission reactors to produce adequate quantities of molybdenum 99 for current needs. That is the message of the NNSA’s augmented shipment of HEU to Europe for moly production. Also, there will continue to be a near-term need to continue using technetium 99 m and to investigate alternative, reactor-based methods to produce it in a proliferation-responsible way. But it is not clear that these approaches will have a game-changing effect on the proliferation threat posed by medical radioisotope production.
In the slightly longer term, the path forward would have to consist of a number of elements. Chief among them is the need for strong pressure from a key government entity possessing the mission and the power to bring together disparate agencies, with the common purpose of curbing the proliferation threat. It would seem that the White House would be the obvious candidate. Indeed, because there is already an interagency task force that focuses on the conversion of fission-fueled medical radioisotopes from HEU to LEU, as well as stabilizing the global molybdenum 99 supply chain, including not only the NNSA but also the Food and Drug Administration, the Department of Health and Human Services, and the Department of Veterans Affairs, then perhaps this group’s mission could be broadened to include encouraging the innovation and production of non-fission-fueled isotopes as well.
At the legislative end, congressional action is a more remote possibility, given today’s gridlock, but historically, nonproliferation has tended to be a bipartisan issue. There is legislative precedent for action on HEU-sourced medical isotopes, as in the 1992 Energy Policy Act and the 2012 American Medical Isotopes Production Act. Unfortunately, the handful of congressional actions taken to date fall short of the long-term goal of eliminating enriched uranium from the radioisotope fuel cycle and sometimes tend to be mutually contradictory.
Where Congress and the executive branch do have flexibility and power is in the support of scientific research. For the near term, the American Medical Isotopes Production Act of 2012 is already promoting conversion from HEU to LEU methods, and Congress may reauthorize it in the 2014 National Defense Authorization Act. In the longer perspective, the Energy Department, the National Institutes of Health, and possibly the National Science Foundation may have a role in targeting basic research in non-fission isotopes, particularly with a focus on small businesses through the Small Business Innovation Research and Small Business Technology Transfer programs. The Canadian government is already funding university research in this area, and American researchers might join forces with their colleagues to the north.
At the same time, regulatory relief may help. There may be a unique opportunity here now, since the current FDA Administrator, Margaret Hamburg, has a strong track record in nuclear nonproliferation as former senior scientist at the Nuclear Threat Initiative. Other regulatory action may also be brought to bear, as illustrated by the 2012 rule by the Centers for Medicare and Medicaid Services that imposed a surcharge on treatments involving HEU-sourced technetium 99 m—but the organization was very clear that its goal in this case was not to promote administration policy on nonproliferation, but instead to respond to it by equalizing payments to hospitals.
Finally, there are critical roles for the private sector. Professional societies concerned with nuclear medicine research and treatment, such as the Society of Nuclear Medicine and Molecular Imaging, could convene targeted studies and internal discussion on the merits and use of accelerator-based, non-fission isotopes in health care. They could also invite speakers from the national-security community to discuss the proliferation risks of perpetuating the status quo with HEU and LEU production. Industry can also review what nongovernmental regulations, such as industry-wide standards, might be put in place to promote work in this area. And industry must engage with government, and government with industry, to review the incentives and obstacles to developing accelerator-based radioisotopes for medical treatment and bringing them to market on a large scale. Industry should be at the table as the government devises practical actions to address this issue.
None of this will be inevitable or easy. Change in the United States will require high-level pressure from the security community, perhaps starting with the White House Office of Science and Technology Policy and the National Security Council. A high-priority interagency task force can set out to tweak the FDA rules and perhaps other regulations to streamline and reduce the cost of the obstacles confronting private companies that see major market potential in putting accelerator-based, non-fission isotopes on the market. Congress and executive branch agencies can take steps, both collectively and individually, to increase funding to accelerate the basic research to validate the underlying science. The private sector must do its share, and in order to do so, be present at the table.
Getting to a safer way of producing nuclear medicine will take resolve and long-term effort. But the time to start is now.
Footnotes
Acknowledgements
The author is deeply grateful to Stanley Satz, Chairman and Chief Scientific Officer of Bio-Nucleonics, LLC, for his generous contributions and guidance, and to Leonard Mausner of Brookhaven National Laboratory.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
