Abstract
Electrically conductive nanocomposites based on polypropylene (PP) were prepared using carbon black (CB) nanoparticles via melt blending in the presence of glycerol and castor oil as hydroxyl functional dispersing aids and maleic anhydride–grafted polypropylene (MAgPP) as a compatibilizer. Initially, electrical conductivity of PP/CB nanocomposites with different dosages of both CB nanoparticles and dispersing aids were investigated through electrical analysis. Results indicated that castor oil had no beneficial effects on electrical conductivity. However, the efficiency of 3 wt% loading of glycerol was verified through measurements of electrical, rheological, thermal and mechanical properties as well as by morphological studies. The volume electrical conductivity value of PP nanocomposites containing 12 wt% CB significantly enhanced to 1.2 × 10−4 S/cm by adding 3 wt% glycerol in compared with nanocomposite without glycerol with the volume conductivity value of 1.8 × 10−6 S/cm. Incorporation of optimum amount of glycerol favoured the dispersion of nanofiller in the matrix which was confirmed by scanning electron microscopy observations, leading to higher conductivity due to better formation of conductive pathways within the matrix. Additionally, at the same concentration of CB nanofiller, samples treated with 3 wt% glycerol showed higher melting temperatures, storage modulus (Gˊ), complex viscosity, tensile modulus and elongation at break. These findings indicate that adding an optimum amount of an efficient hydroxyl functional component like glycerol into PP/CB nanocomposite can be a promising way to enhance the electrically conductive performance while other properties could be maintained well or even improved.
Introduction
Electrically conductive polymer composites have received great attention owing to their promising applications in many fields,1–3 including antistatic materials, 4 electromagnetic interference shielding,5,6 self-regulated heating materials, 7 and sensors. 8 Binary conductive polymer systems consisting of conducting fillers such as carbon black (CB),9,10 graphite,11,12 multi-walled carbon nanotubes (MWCNTs),13–15 and carbon fibers16,17 in a polymer matrix can create polymer composites that are tough and flexible with appropriate electrical conductivity based on their applications.
Carbon black is one of the widely used nanofillers, which is cheap and has permanent electrical conductivity. The strong interaction between polymer and CB nanoparticles can improve the mechanical properties of the nanocomposites.18–20 However, a considerable amount of CB is needed to achieve high levels of electrical conductivity. Therefore, the incorporation of high contents of CB nanofiller can adversely influence the viscosity and mechanical properties, leading to challenges and limitations for industrial manufacturing.
Attempts to reduce filler content have been made by various means such as combining two kinds of electrically conductive fillers or creating two-component polymer matrix. There are a considerable number of reports on the electrical properties of polymer systems containing more than one conductive filler.21–23 Recently, Haghgoo et al. investigated the electrical conductivity of carbon nanotube/carbon black-filled polymer matrix hybrid nanocomposites by both model predictions and experimental data. Results indicated an improvement in conductivity by the incorporation of hybrid CB/CNT filler. 24 However, dispersion difficulties and high costs of combining two or more kinds of electrically conductive fillers in the polymer matrix can be serious drawbacks.
On the other hand, a number of studies have been reported that electrically conducting composite materials based on polymer blends can achieve high electrical conductivities with lower filler contents than a single-component polymer.25,26 This is caused by creating double percolation because the conductive filler is heterogeneously distributed within one phase of the blended matrix or at the interface between polymer blends. Recently, Lencar et al. designed a novel two-step mixing approach to pre-localize MWCNTs within the polyethylene phase, and subsequently allow them to migrate into the thermodynamically favored poly(vinylidene fluoride) phase. Results exhibited great electromagnetic interference shielding effectiveness, suggesting the formation of more interconnected MWCNT networks over time. 27 However, these methods are seldom used in actual manufacturing owing to their complicated preparation.
From a practical point of view, the development of conductive polymer composites with low content of a conductive filler like CB promises an improvement in the processability and the decline of manufacturing costs. It is, therefore, significant to enhance the electrical characteristics of conductive polymer composites while reducing their conductive filler contents. One strategy can be the use of a low-cost and widely available component in order to favour the dispersion of a conductive filler in the polymer matrix during melt mixing.
In this study, polypropylene (PP), which is a general polymer, was used as the matrix. Glycerol and castor oil were used due to their hydroxyl functionalities to compare their impact on the dispersion of CB nanoparticles in a single-component PP matrix. As it is known, glycerol has usually been used as a plasticizer to improve starch melt blending with other polymers like PP in order to enhance the processing of starch and produce thermoplastic starch (TPS). 28 To the best of our knowledge, there have been no reports on the electrical properties of PP containing CB nanoparticles with the contribution of glycerol or castor oil.
To achieve the mentioned purposes, PP-based nanocomposites containing CB in the presence of hydroxyl functional dispersing aids like glycerol and castor oil were prepared by means of melt mixing. The effects of CB content and dispersing aid dosage on the electrical, morphological, rheological, thermal and mechanical properties of nanocomposites were comprehensively investigated.
Experimental section
Materials
Polypropylene with the trade name of JAMPILEN EP440L, purchased from Jam Petrochemical Company in Iran with a melt flow index (MFI) of 6 g/10 min (230°C, 2.16 kg) and density of 0.9 g/cm3, was used as the polymeric matrix. Nanocarbon black powder was purchased from Research Institute of Petroleum Industry, Iran.
Maleic anhydride–grafted polypropylene (MAgPP), model FUSABOND P613, from Dow Chemical, Irganox 1010 and Irgafos 168 from BASF, and calcium stearate from Sinwon Chemical were used. Glycerol and castor oil were purchased from Sigma-Aldrich.
Preparation of nanocomposites
PP/CB nanocomposites were prepared via melt blending in a co-rotating twin screw extruder (ZSK 25, 2002, Coperion, Germany), with screw diameter of 25 mm, L = 40D, screw speed of 200 min−1 and throughput of 70 kg/h. The temperature profile was 180 to 210°C from hopper to the die. The extruded samples were pelletized at the die exit, dried and compression molded to thin films using a hot press (Mini Test Press, Toyo Seiki, 2002, Japan).
Compositions of PP/CB nanocomposites.
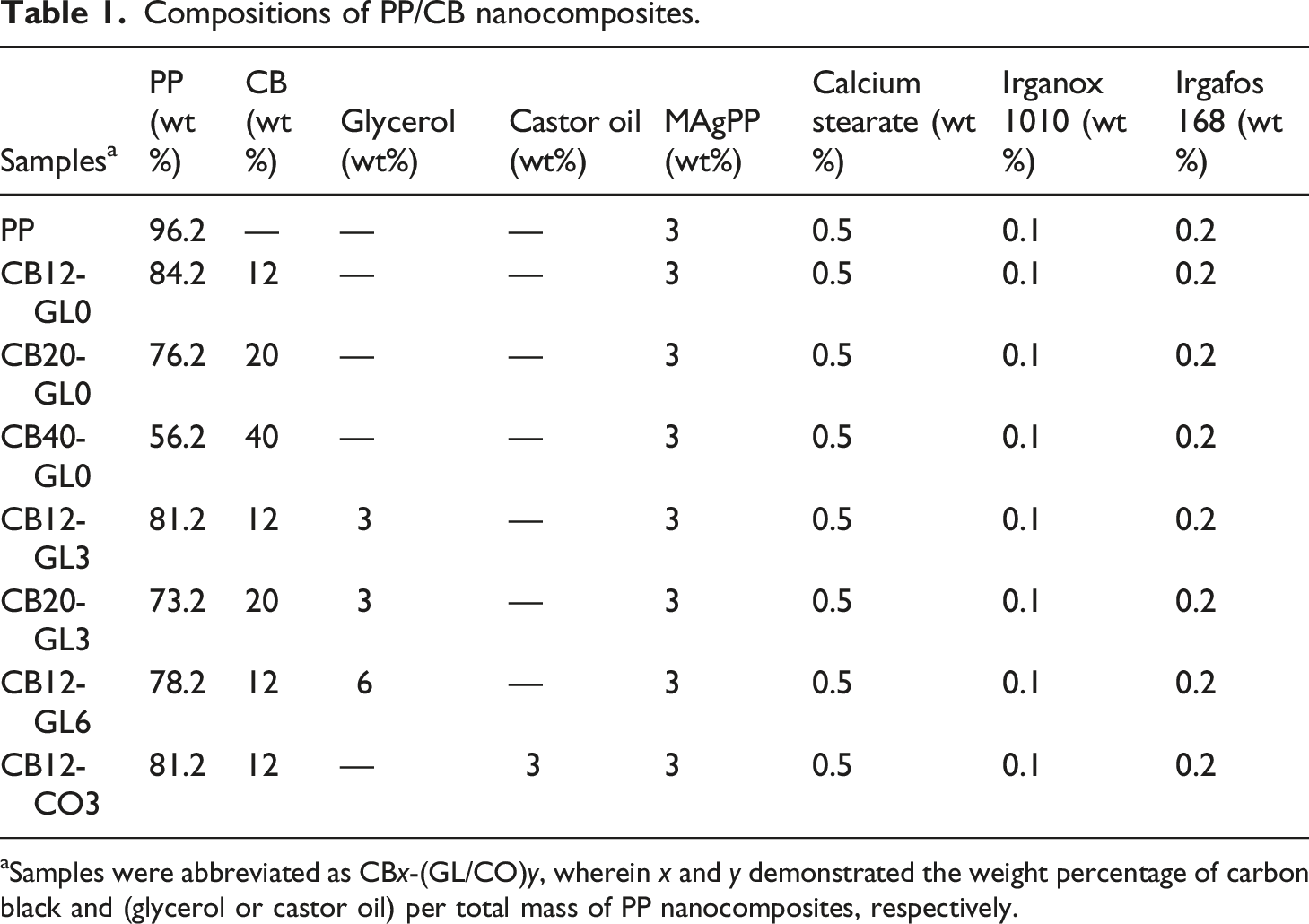
aSamples were abbreviated as CBx-(GL/CO)y, wherein x and y demonstrated the weight percentage of carbon black and (glycerol or castor oil) per total mass of PP nanocomposites, respectively.
Characterizations
The chemical structure of nanocarbon black was investigated by FTIR spectroscopy (Tensor II, Bruker, Germany). Samples were mixed with KBr powder and pressed to disk. The analysis was done in the range of 4000–400 cm−1 wavenumbers at room temperature.
A digital multimeter (HIOKI DT4282, Japan) was used to perform room-temperature resistivity measurements on molded sheets with 2 mm thickness. The voltage of 500 V was applied and the volume electrical resistivity was measured according to ASTM D 257 (2014).
A Perkin-Elmer Pyris one differential scanning calorimeter (DSC) was used. Samples of 11 ± 1 mg were used in the tests. They were sealed in aluminum pans and heated up to 200°C at a rate of 10°C/min to record the melting behaviour. Samples were held at mentioned temperature for 5 min in order to erase any thermal history.
The dynamic thermomechanical properties of the composites were measured with Mettler Toledo-model DMA1. The bending method was used at a frequency of 1 Hz in a temperature range of −50°C to 140°C. The heating rate was 3°C/min. Testing was performed on rectangular bars measuring approximately 25 mm × 10 mm × 1 mm. Samples were prepared with a hydraulic press, at a temperature of 200°C and a pressure of 100 bar, for a time period of 5 min.
The rheological behavior was investigated by Anton Paar (Model MCR501, Austria). The size of measured samples was 25 mm in diameter, with a gap of 1.0 mm. Frequency sweeping was performed at 180°C at a frequency from 0.01 to 100 s−1 in the nitrogen environment, with a strain of 1% in order to make the materials be in the linear viscosity range. The storage modulus describes the capacity to accumulate the energy in the elastic part which is evaluated by the frequency sweep measurements of the polymer, and the complex viscosity explains the resistance to flow.
A MIRA3 TESCAN scanning electron microscope (SEM) was employed to study the distribution and the morphology of the PP/CB composites. Samples were broken in liquid nitrogen and were gold-sputtered, and then the cross-section was observed in the SEM at an accelerating voltage of 15 kV.
Tensile properties of composites were measured using Sanaf Tensile Tester-STT 50 KN with a cross-head speed of 50 mm/min. Five tensile specimens were tested for each reported value according to ASTM D638 test method.
Results and discussion
Electrical properties
Volume electrical conductivity (EC) results and DSC data of PP/CB composites.
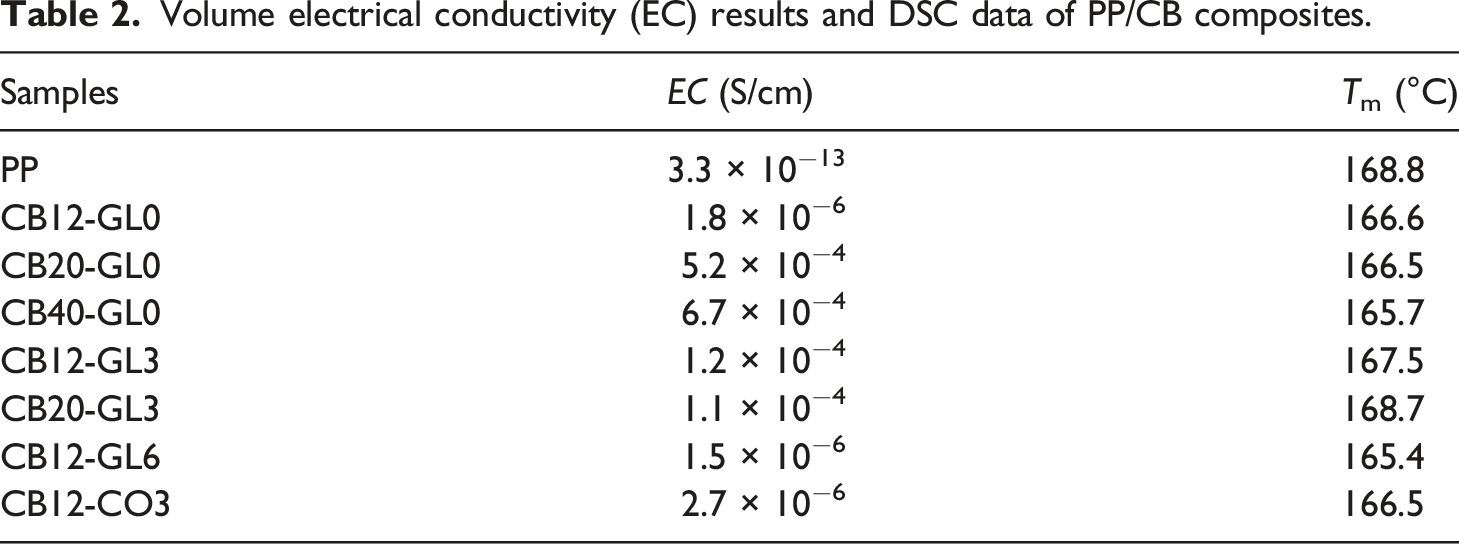
Moreover, the incorporation of 3 wt% glycerol into PP containing 12 wt% CB led to a great improvement in the volume electrical conductivity result, reaching the value of 1.2 × 10−4 S/cm as seen in Table 2. This result meant that the same electrical conductivity was obtained with lower filler content for composite mixed with 12 wt% CB and 3 wt% glycerol than for composite mixed with only 20 wt% CB. In other words, less CB filler added into the composites with a good conductive property, not only reduced the cost of the materials, but also could enhance the processability and the mechanical properties.
According to the literature, during the manufacturing process of carbon black, oxygen-containing functional groups can be formed on the surface to varying degrees. The presence of these functional groups depends on the specific manufacturing process and the conditions.
29
Figure 1 (inset) depicts the FTIR spectrum of nanocarbon black. In this graph, the peak shown in wavenumber range of 3650–3200 cm−1 was related to stretch vibrations of OH groups.
30
Consequently, an improvement in the electrical conductivity of CB12-GL3 nanocomposite containing 3 wt% glycerol could be related to the following fact. On one side, glycerol played a key role in reacting with maleic anhydride, and on the other side, glycerol’s unreacted hydroxyl groups could provide extra sites to make hydrogen bonds with the hydroxyl groups on the surface of CB nanoparticles. In other words, although hydroxyl groups of CB nanoparticles could make hydrogen bonds with maleic anhydride, glycerol resulted in an increase in the total interactions, leading to better dispersion of CB in the polymer matrix. Figure 1 depicts the proposed mechanism of glycerol’s role in PP/CB nanocomposite. Therefore, the addition of 3 wt% glycerol in PP containing 12 wt% CB nanofiller could help the effective interactions between polymer and the conductive nanofiller. As a result, the conductive paths increased and the conductivity improved due to more bridges formed by CB in the PP matrix. Proposed mechanism of glycerol’s role in PP/CB nanocomposite, inset of which is FTIR spectrum of nanocarbon black.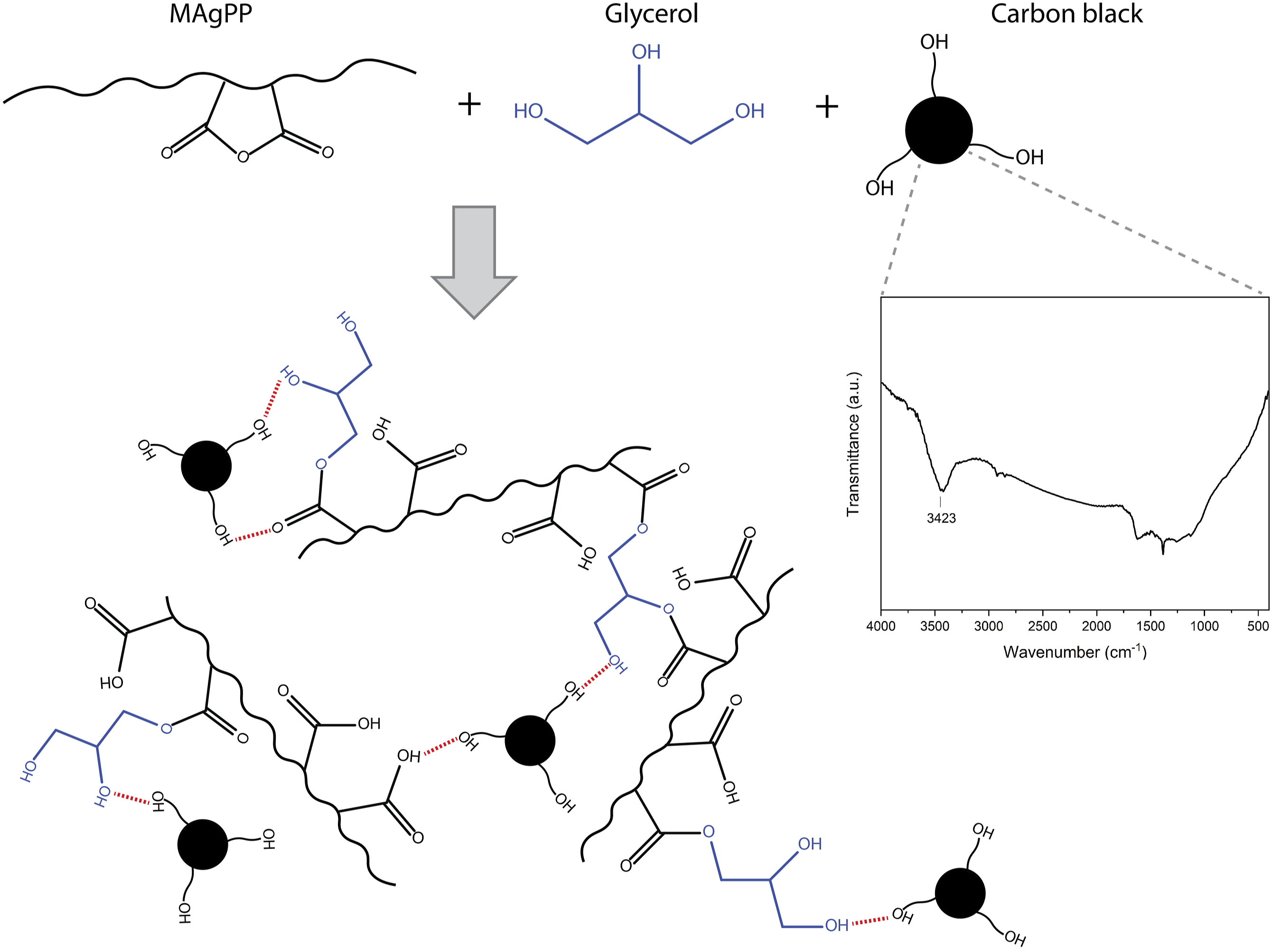
However, a rise in the loading of glycerol to 6 wt% had no effect on the electrical conductivity of PP containing 12 wt% CB nanofiller which will be further discussed by SEM observations. Furthermore, it was seen that the same amount of castor oil could not be effective which might be related to its less reactive hydroxyl groups in comparison with glycerol. To sum up, the amount of glycerol should be optimized in the polymeric system depending on the content of a conductive filler and the best electrical conductivity result was achieved by the nanocomposite containing 12 wt% CB and 3 wt% glycerol (CB12-GL3).
In order to comprehensively investigate the role of glycerol in PP/CB nanocomposites, other properties including morphological, thermal, rheological, and mechanical measurements have also been conducted.
Morphological studies
To visually observe CB dispersion in the PP matrix with and without glycerol, SEM micrographs of the fracture surface of PP/CB nanocomposites after embrittlement with liquid nitrogen were shown. As depicted in Figures 2 and 3, the estimated size of the CB nanoparticles was in the range of 30-70 nm. Dispersion of CB nanofiller in the PP matrix containing 12 wt% CB in the presence of 3 wt% glycerol (Figure 2(b) and (e)) was obviously more uniform than PP/CB nanocomposite without glycerol (Figure 2(a) and (d)). It has been proved that the distribution of nanoparticles has a significant impact on the electrical properties of nanocomposite. Consequently, the main reason for the best electrical conductivity for the sample containing 12 wt% CB with the incorporation of 3 wt% glycerol could be related to the best formation of conducting networks, owing to the most uniform distribution of CB particles in the PP matrix. SEM micrographs (200 nm and 5 μm) of the cross-section of PP composites containing 12 wt% CB without glycerol (a and d), and with 3 wt% (b and e) and 6 wt% (c and f) glycerol. SEM micrographs (200 nm and 5 μm) of the cross-section of PP composites containing 20 wt% CB without glycerol (a and c) and with 3 wt% glycerol (b and d).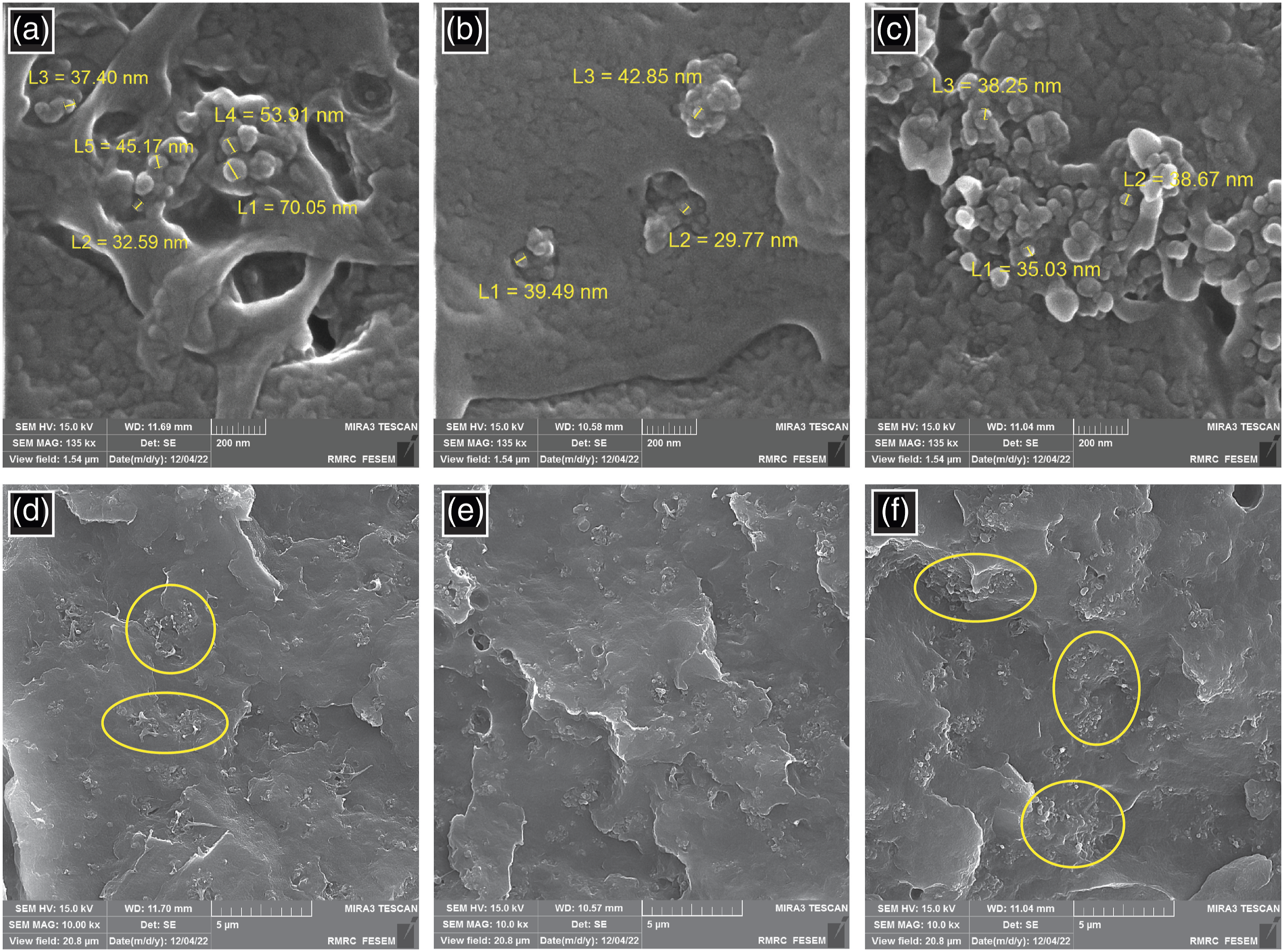

However, as glycerol content increased in Figure 2(c) and (f), more CB aggregates appeared which adversely affected the distribution of nanoparticles in the PP matrix. In other words, a rise in the loading of glycerol up to 6 wt% caused an increase in nanoparticles’ tendency to agglomerate probably because of an incompatibility due to an increase in the polarity of the system. Therefore, the reason for no effect of increased glycerol content on electrical conductivity results could be related to appearing more aggregations. Moreover, as seen in Figure 3, adding 3 wt% glycerol in the PP matrix containing 20 wt% CB had negligible effect on the distribution of nanoparticles which had been previously confirmed by electrical conductivity results.
Thermal properties
The melting behavior of PP/CB nanocomposites was investigated using a differential scanning calorimetry. As shown in Table 2 and Figure 4, melting peaks of PP containing carbon black filler slightly shifted to lower temperatures.
31
However, with the addition of 3 wt% glycerol (CB12-GL3 and CB20-GL3), the melting temperatures (Tm) increased, indicating better interactions between CB particles and the polymer in the presence of glycerol. On the other hand, higher concentration of glycerol (CB12-GL6) had an opposite impact on Tm which had been also confirmed by electrical conductivity and SEM results. Moreover, the incorporation of castor oil (CB12-CO3) had almost no effect on the value of Tm. Second heating runs of DSC thermograms for PP/CB nanocomposites.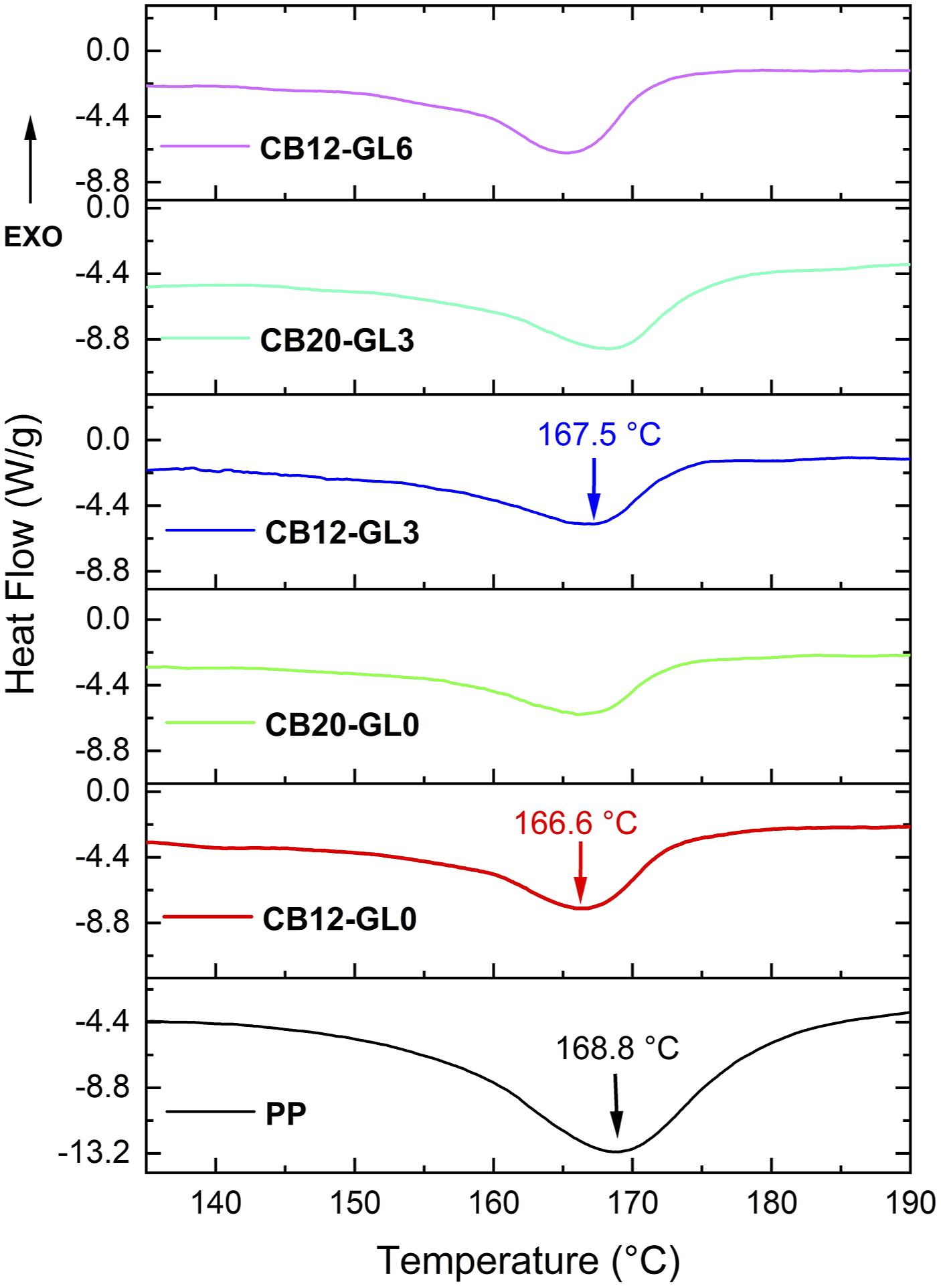
The viscoelastic behavior of PP/CB nanocomposites were studied by dynamic mechanical thermal analysis (DMTA). Curves indicated the variations of the storage modulus (E') and the damping factor (tan δ) as a function of temperature which are given in Figures 5 and 6, respectively. As seen in Figure 5, the presence of 12 wt% CB filler (CB12-GL0) plus 3 wt% glycerol (CB12-GL3) revealed a great improvement in the storage modulus values for PP nanocomposites in the glassy region. However, the rise in the amounts of CB filler and glycerol adversely affected the stiffness of polymers. It was proved that E' decreased by rising temperature, owing to the increased segmental motion above the glass transition temperature (Tg). According to Figure 5, there was a small difference between the storage modulus values for PP nanocomposites in the rubbery region. Storage modulus curves from dynamic mechanical relaxation behavior of PP/CB nanocomposites. Tan δ curves from dynamic mechanical relaxation behavior of PP/CB nanocomposites.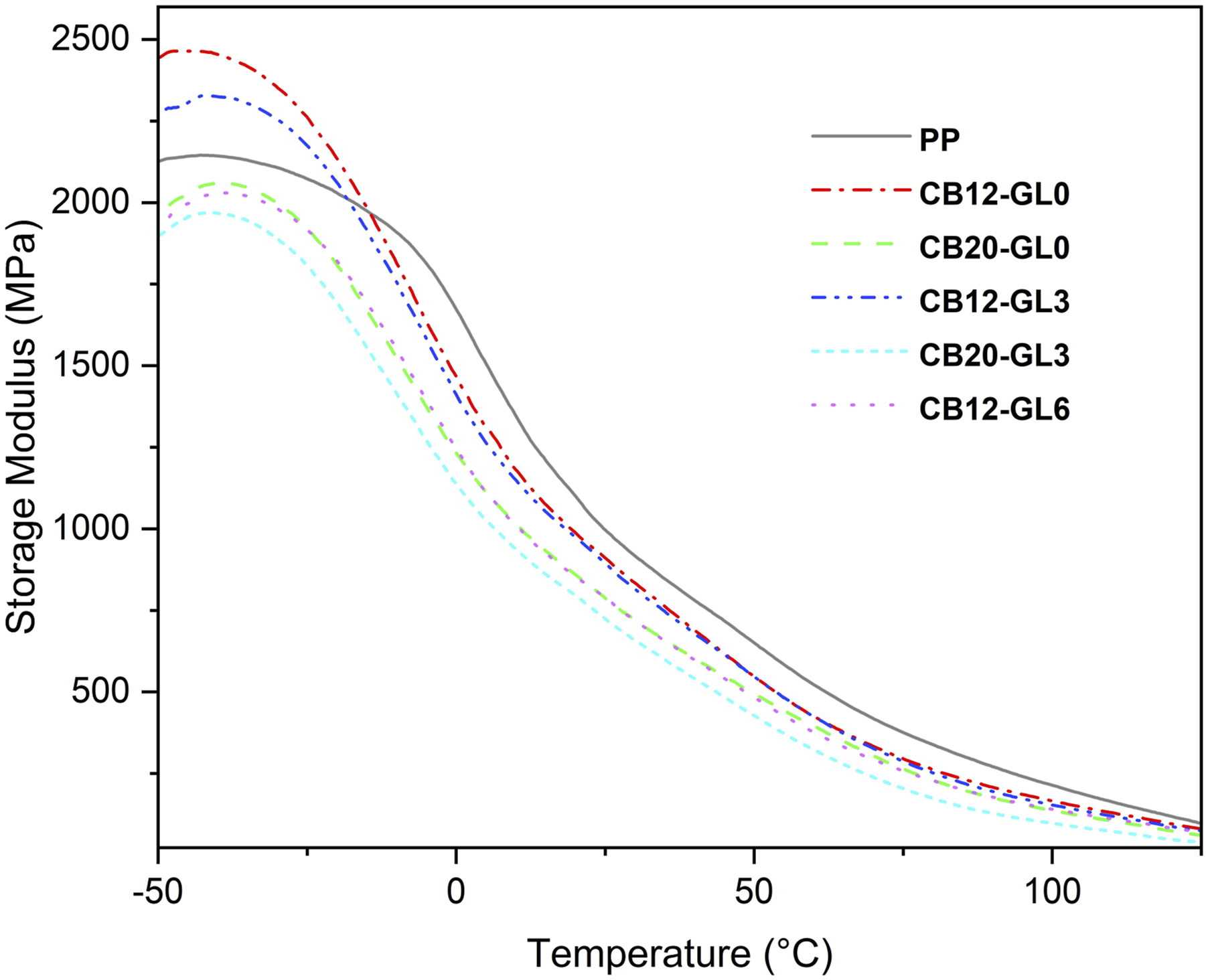
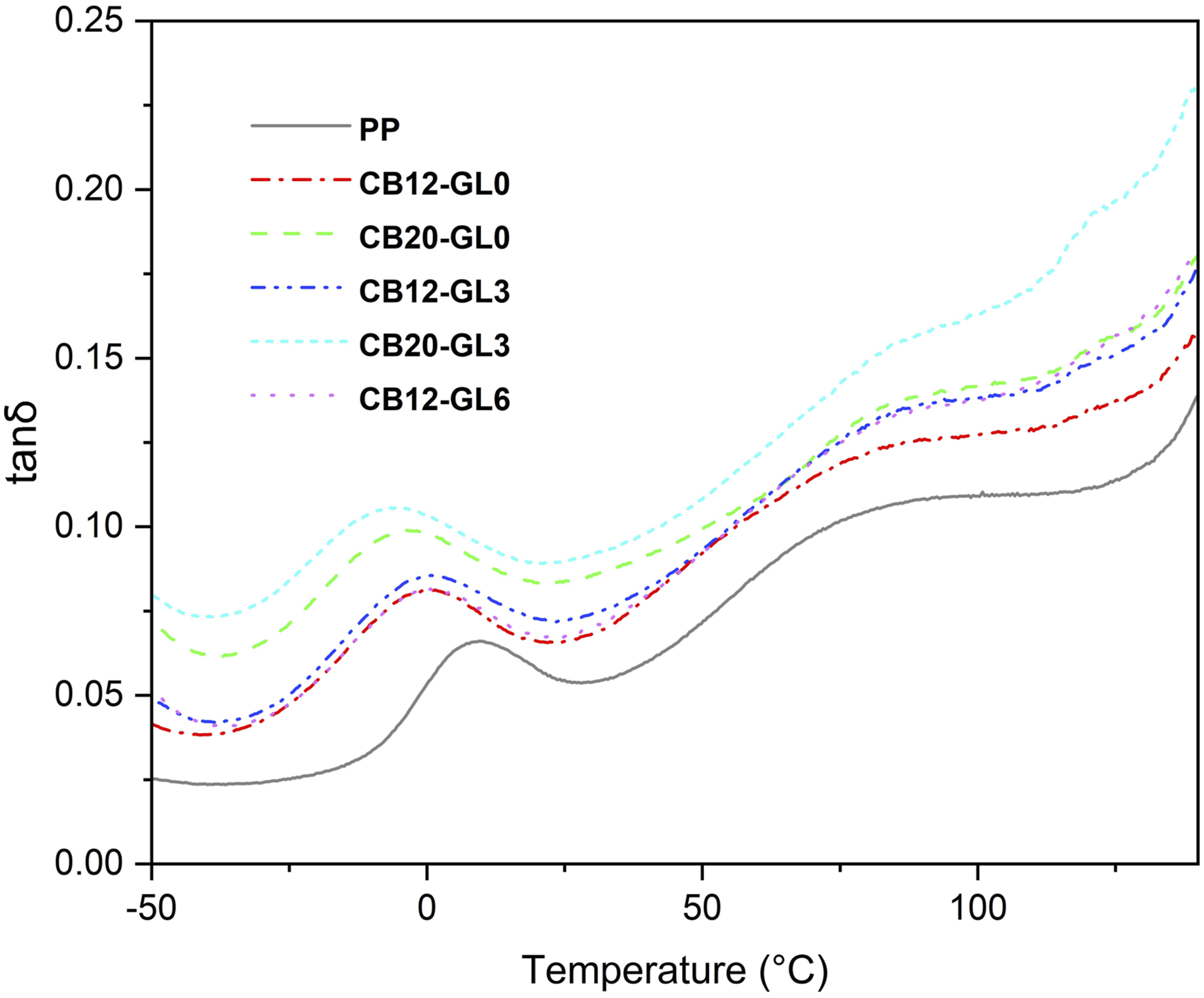
As shown in Figure 6, tan δ curve of PP depicted two relaxations recorded at 80 °C–100°C (α) and -5–15°C (β), respectively. The β-relaxation represents the Tg of PP and the α-relaxation is related to the crystalline–amorphous polymer regions and the softening point of PP. Usually, the Tg of a polymer matrix tends to increase with the addition of nanoparticles, owing to the reduction of the macromolecular chain mobility at the interface around nanoparticles. However, it was shown that Tg sometimes remained unaffected or decreased by adding nanoparticles. 32 As seen in Figure 6, the Tg of PP/CB nanocomposites indicated a slight shift towards lower temperatures with increase in CB contents. It means that, as the CB content increased, the nanoparticles experienced stronger van der Waals attractions, resulting in the aggregation of CB particles. This caused a reduction in surface area and disruption in the CB network structure. Consequently, the restriction of CB on PP molecular chains was weakened, leading to a slight decrease in the Tg of the PP/CB composite. Therefore, according to the literature, a change in the Tg by the incorporation of nanoparticles is not consistent, but depends on the microstructure/amount of nanoparticles, as well as the interactions taking place between the polymer and the nanofiller. 33
Rheological behavior
Rheology has been proved to be a great tool to investigate the internal microstructure of nanocomposites, especially the formation of the network between nanofillers and the polymer matrix. The storage modulus (Gˊ) and the complex viscosity (|η*|) for PP/CB nanocomposites as a function of frequency (ω) are depicted in Figures 7 and 8, respectively. As expected, the addition of CB nanoparticles resulted in a considerable increase in the matrix polymer viscosity and storage modulus at the whole frequencies due to the physical interactions between the polymer matrix and nanoparticles. It has been reported that the Gˊ curves exhibited a distinct plateau at the low frequencies when the CB loadings were higher than 5 wt% which indicated a transition from a liquid state to a solid state, accompanying by the formation of a mechanically stable network structure. This response resulted from the formation of a percolation network in the nanocomposites due to the confinement effect of nanofillers on the motion of polymer chains.
34
Storage modulus of pristine PP and PP/CB nanocomposites as a function of frequency. Complex viscosity of pristine PP and PP/CB nanocomposites as a function of frequency.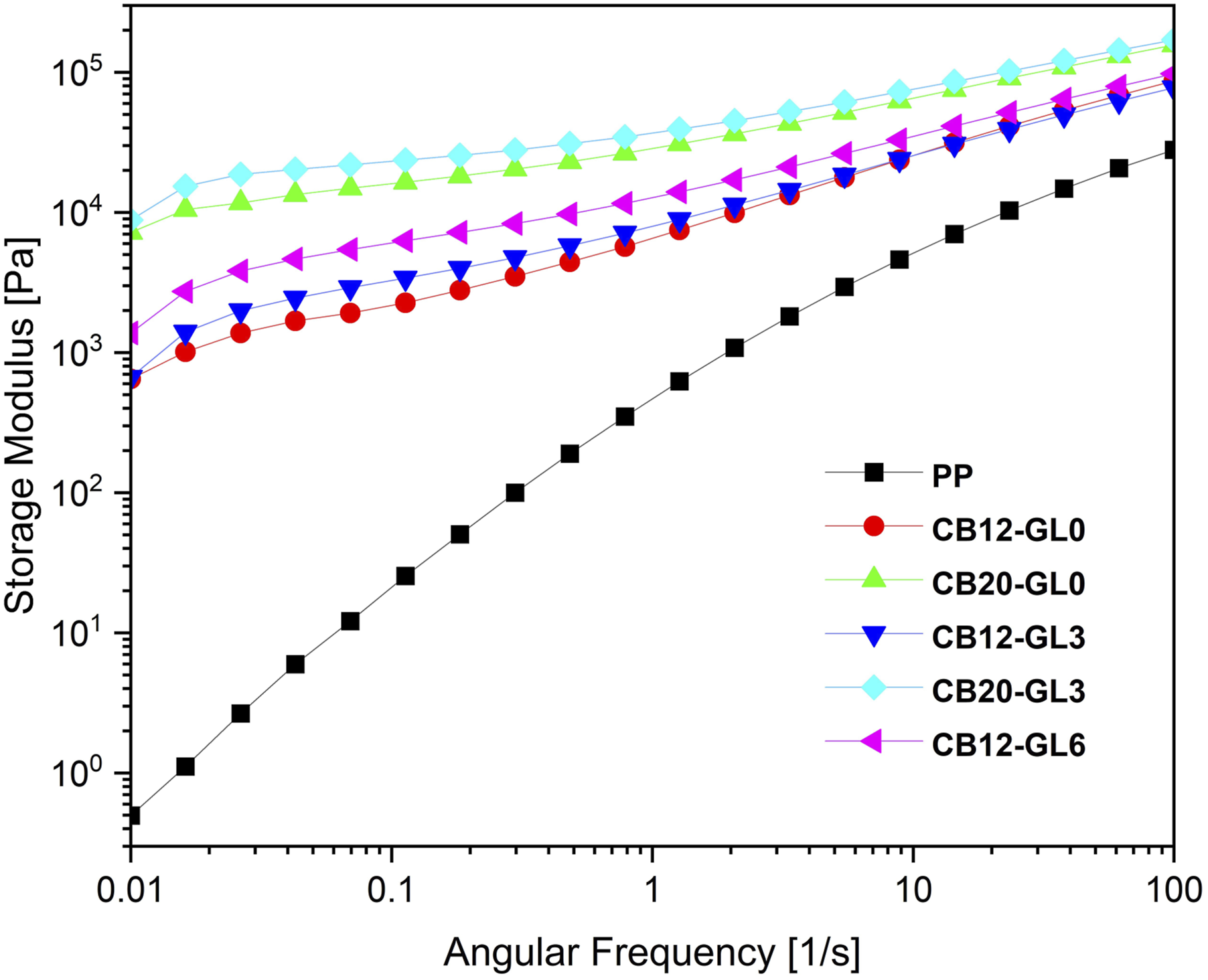
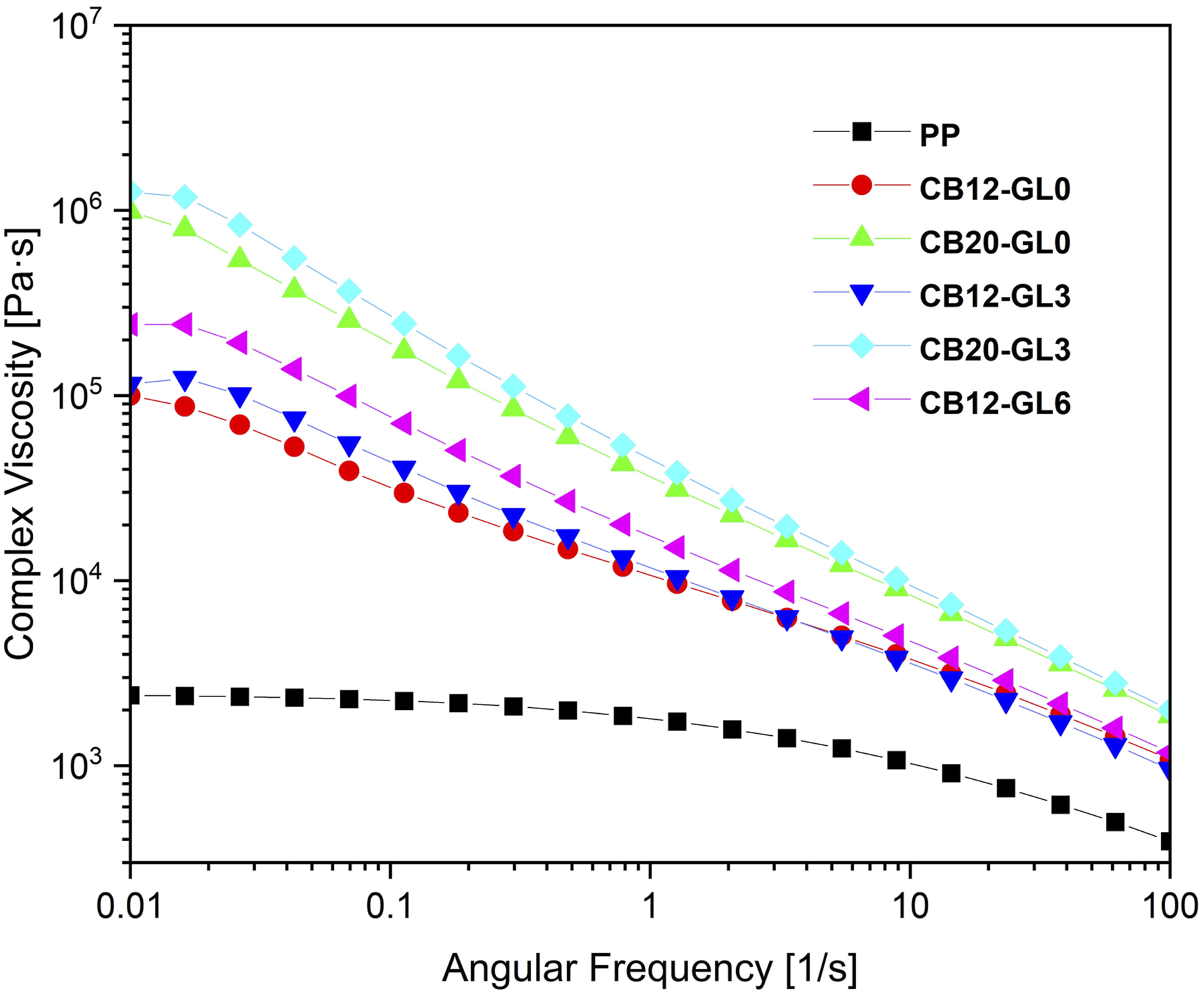
As seen in Figure 8, pristine PP displayed a pseudo-Newtonian behavior at low frequency range, whereas PP/CB nanocomposites showed higher complex viscosity and more pronounced shear thinning behavior with the increase in CB loadings. Moreover, at the same concentration of CB nanofiller, samples which were treated with glycerol showed higher values of Gˊ and |η*|, especially at low frequencies. This could be attributed to better dispersion of CB nanofiller in the PP matrix and also better polymer-filler and filler-filler interactions with the incorporation of glycerol. In summary, according to Figures 7 and 8, values of Gˊ increase with increasing the amounts of CB filler and glycerol, mainly in the low frequency region, following the same tendency related to the |η*| behavior. 35
Mechanical properties
Stress-strain data of PP/CB nanocomposites.
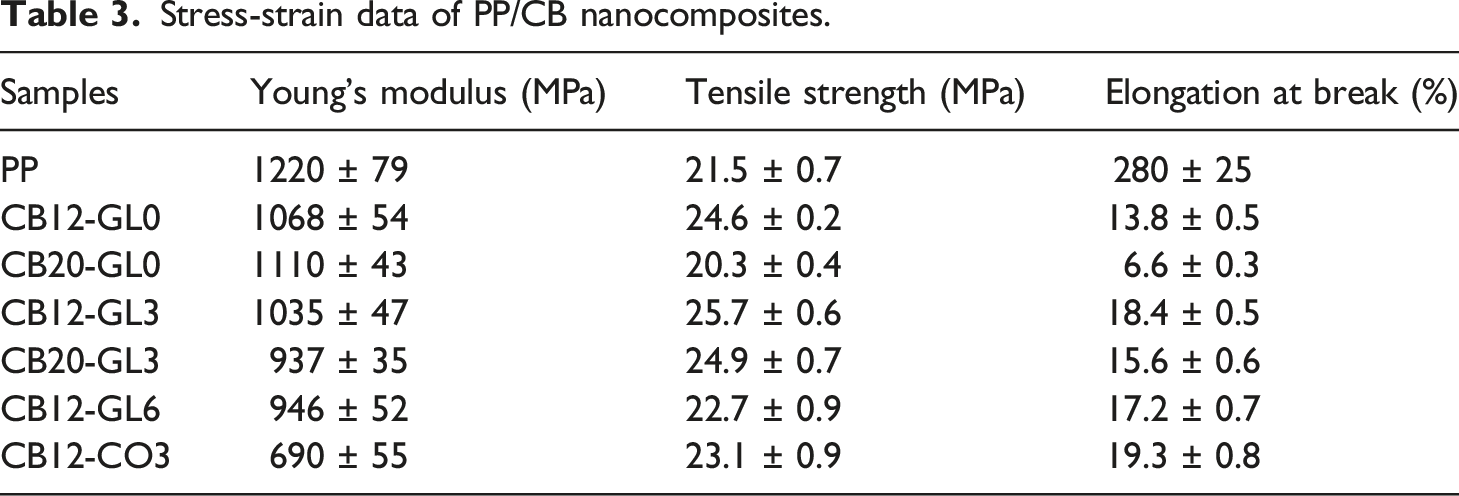
Due to nanoparticles’ large surface energy, they have a strong inclination to agglomerate. This results in a decrease in the effective contact area between the nanoparticles and the polymeric matrix, which, in turn, reduces the effectiveness of the nanoparticles in improving the mechanical properties. Therefore, as seen in Table 3, adding CB nanofiller caused a slight decrease in the Young’s modulus of the nanocomposite due to CB agglomerations. However, the distribution of nanoparticles within the polymeric matrix and the formation of nanostructures has a significant impact on the mechanical properties. Therefore, in PP/CB nanocomposites containing 3 wt% glycerol, the increment of tensile strength and simultaneously elongation at break (Table 3) may be attributed to the uniform distribution of nanoparticles with minimum formation of large agglomerates in the polymeric matrix which was previously confirmed by SEM. In other words, the primary reason for the improvement in tensile properties with the incorporation of 3 wt% glycerol could be related to the better interaction between the filler and matrix, which enhanced the transfer of stress within the interphase of the polymer and carbon black. However, as seen in Table 3, the presence of 6 wt% glycerol adversely affects the mechanical properties mainly because of poor dispersion of CB particles in PP matrix which was previously confirmed by electrical conductivity results and SEM. On the other hand, there was a sharp drop in the Young’s modulus of the PP/CB composite with the addition of castor oil in the system, indicating its negative effect on the mechanical properties of PP/CB nanocomposites. This result can be related to the fact that castor oil can function as a plasticizer in a polymer matrix by interacting with the polymer chains and reducing the intermolecular forces between them. 36
Combined with the previous results, improved electrical conductivity with well-maintained other properties or even enhanced ones were achieved by using an optimum amount of glycerol in PP reinforced with nanocarbon black. This innovative and simple design concept for conductive composites can also be applied to other electrically conductive fillers with different polymer matrices.
Conclusions
PP/CB conducting nanocomposites were prepared by means of the melt processing method and the impacts of CB content and hydroxyl functional dispersing aids including glycerol and castor oil were investigated. It was observed that the electrical conductivity of PP/CB nanocomposites improved with the increase in CB concentration up to 20 wt%. As for the effect of glycerol, several important results were obtained, while castor oil had almost no effectiveness on the electrical property. To begin with, the Tm slightly increased with the addition of 3 wt% glycerol, indicating better interactions between CB particles and the polymer. Secondly, the presence of 12 wt% CB filler as well as 3 wt% glycerol revealed a significant improvement in the storage modulus values for PP nanocomposites in the glassy region. Furthermore, at the same concentration of CB nanofiller, samples which were treated with glycerol showed higher values of Gˊ and |η*|, especially at low frequencies. Most importantly, the incorporation of 3 wt% glycerol into PP containing 12 wt% CB nanofiller made a great contribution to the dispersion of CB nanoparticles in the PP matrix by interacting with both MAgPP and CB nanoparticles, leading to a considerable enhancement in electrical conductivity. Finally, enhancement in tensile properties of PP/CB samples containing 3 wt% glycerol could be related to the better interaction between the filler and matrix, which enhanced the transfer of stress within the interphase of the polymer and carbon black. In summary, glycerol played a key role in reducing conductive filler content, meaning that the cost of the materials can be reduced, as well as the processability and mechanical properties can improve. Therefore, the strategy of using a small amount of a cheap and easily accessible hydroxyl functional component like glycerol in the composition can pave the way for making PP/CB nanocomposite very promising in the large-scale fabrication of lightweight and cost-effective electrically conductive polymer composites.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
