Abstract
In the present research, the effect of shear on the crystallization behavior of polypropylene (PP) nanocomposites loaded with carbon nanotube (CNT) and carbon black (CB) has been evaluated. Rheological properties, crystallization under shear, and electrical conductivity under shear of the bi-filler PP/CNT/CB nanocomposites have been evaluated and compared with those of the mono-filler PP/CNT and PP/CB nanocomposites. According to the rheological analyses, in the comparison between mono-filler and bi-filler nanocomposites with the same filler content, the storage modulus, complex viscosity, non-terminal behavior, and solid-like behavior of the bi-filler nanocomposites were higher than those of the mono-filler nanocomposites. Whereas, the storage modulus and complex viscosity of the bi-filler nanocomposites with half filler content were intermediate of the mono-filler nanocomposites. The zero viscosity of the bi-filler nanocomposite with the same filler content was about 6 times that of the mono-filler nanocomposites. Whereas, the zero viscosity values of the bi-filler nanocomposites with half filler content were 1.1-3.4 times those of the CB-loaded and 0.3-0.9 those of the CNT-loaded nanocomposites. According to the non-isothermal crystallization test, in the comparison between mono-filler and bi-filler nanocomposites with the same filler content, the crystallization temperature (Tc) of the bi-filler nanocomposites was 2.7 °C–3.5 °C higher than that of the mono-filler nanocomposites. Whereas, the Tc of the bi-filler nanocomposites with half filler content were 1.4 °C–3.7 °C higher than those of the CB-loaded and 0.8 °C–1.5 °C lower than those of the CNT-loaded nanocomposites. According to the isothermal crystallization test, applying shear and increasing the shear rate and time accelerated the crystallization of the mono-filler and bi-filler nanocomposites, however to different extents. Under shear, the crystallization rate of mono-filler and bi-filler nanocomposites increased by 2 and 5 times, respectively. Electrical conductivity measurement revealed that shearing reduced the conductivity of the mono-filler and bi-filler nanocomposites by 120 and 200 times, respectively.
Introduction
Polypropylene, a common thermoplastic polymer, has been widely used in various industries including automotive, 1 construction, 2 packaging, 3 textile, 4 adhesive, 5 etc. The widespread attention to PP arises from its outstanding merits like high heat deflection temperature, high strength, excellent processability, and reasonable price. In addition to pure PP, PP composites6–8 and nanocomposites9–11 have also received much attention. Various fillers have been added to PP to improve its mechanical, electrical, thermal, and functional properties and make it appropriate for particular applications.12–14 Especially, electrically conductive nanocomposites have attracted attention in various application including packaging, automotive, energy, biomedical engineering, etc. due to their brilliant advantages like reasonable price, tunable properties, light weight, and processability. In the field of packaging, conductive nanocomposites can be used to create antistatic coatings to prevent the accumulation of static charge on surfaces and reduce its damage, as well as to create electromagnetic interference (EMI) shielding to reduce the penetration of electromagnetic waves into electronic devices and mitigate their interference effects. In the automotive field, conductive composites can be utilized for the fabrication of antistatic and EMI coatings, seat heaters, sensors for various parts, soft actuators, and so on. In the energy field, they are widely used to make electrodes for batteries and supercapacitor, as well as plates for fuel cells. In the biomedical engineering field, conductive nanocomposite can be used to fabricate tissue engineering scaffolds to enhance the regeneration of electrically responsive tissue like bone, nerve and muscle, as well as to fabricate wearable strain sensors to detect a wide range of physiological activities.15–18
Carbon nanotube (CNT) is one the most attractive fillers for the fabrication of electrically conductive nanocomposites.19,20 Having extremely high aspect ratio, high electrical and thermal conductivity, and remarkable stiffness, CNT is one the best choices for enhancing the properties of thermoplastic polymers and expanding their applications.21–23 However, the concerns raised about CNT, namely poor dispersion in polymer matrix and high price, have limited its industrial application to some extent.24–26
To surmount these imitations, the idea of using a second filler has been proposed. Adding a second filler not only can decrease the interaction between CNTs and improve their dispersion in the polymer matrix, but also can reduce the amount of CNT required to achieve the desired electrical and mechanical properties. A great number of researchers have used a second filler together with CNT and developed bi-filler thermoplastic nanocomposites. In this regard, Yousefi et al. 27 prepared bi-filler nanocomposites consisting of CNT and nanosilica. They showed that the tensile and flexural strengths of bi-filler nanocomposites were higher than those of CNT-loaded nanocomposites, although this trend was observed for nanocomposites with more than 2 wt% nanosilica. Moreover, they proved that bi-filler nanocomposites had higher storage modulus and complex viscosity than CNT-loaded nanocomposites. Ju et al. 28 synthesized bi-filler nanocomposite foams by combing CNT and carbon black. The bi-filler foams represented higher electrical conductivity than CNT-loaded foams. The electrical percolation threshold in bi-filler nanocomposites was as low as 0.016 vol% of the total filler. Al-Saleh et al. 29 combined organically modified clay and CNT to make a bi-filler nanocomposite. The addition of clay decreased the electrical percolation threshold from 1 wt% in the CNT-loaded nanocomposites to 0.5 wt% in the bi-filler nanocomposite. Moreover, adding clay improved the dispersion of CNTs in the polymer matrix and decreased the torque applied to CNT during nanocomposite fabrication. Reducing the torque applied to CNT can reduce the probability of CNT degradation and maintain its high aspect ratio. Prashantha et al. 30 fabricated bi-filler nanocomposites based on PP, CNT and clay nanoparticles. The tensile strength, tensile modulus and impact strength of the bi-filler nanocomposites were higher than those of the mono-filler nanocomposites. Furthermore, in the presence of clay nanoparticles, the dispersion of CNT in the polymer matrix was improved.
The addition of fillers to the polymer matrix not only influences the electrical and mechanical properties of thermoplastic polymers, but also affects their crystallization behavior.31,32 Nanofillers can create heterogeneous nuclei and facilitate the crystallization, at the same time they can make steric hindrance and reduce crystal growth.33–35 The shear stress applied during the fabrication process of nanocomposites can also affect polymer crystallization, and the presence of nanoparticles can change the response of the polymer to shear.36–38 Therefore, it is necessary to evaluate the simultaneous effects of adding nanoparticles and applying shear on polymer crystallization.
Various researches have studied the simultaneous effect of nanoparticle and shear on the crystallization of PP. For instance, Sangroniz et al. 39 evaluated the effect of shear on the crystallization behavior of a nanocomposite made of PP/polyethylene terephthalate (PET) and CNT or TiO2 nanoparticles. They reported that applying shear in the presence of nanoparticles raises the crystallization temperature of the polymer, and CNT has a more remarkable effect on the crystallization temperature than TiO2. Wang et al. 38 revealed that the presence of nanoparticle and the application of shear increased the crystallization kinetics. In the absence of shear, the crystallization kinetics did not change after the CNT content reached 0.1 wt%. However, in the presence of shear, increasing the CNT beyond 0.1 wt% also raised the crystallization kinetics. In the previous research conducted by our group, 40 the concurrent effect of CNT and shearing on the crystallization of PP was evaluated. The evaluation results corroborated that the addition of CNT significantly increased the crystallization temperature and kinetics, while the application of shear mainly affected the crystallization kinetics of polymer.
In view of the above, various studies have investigated the effect of shear on the crystallization behavior of PP nanocomposites. However, there are rare studies that have evaluated the effect of shear on the crystallization of bi-filler nanocomposites. In the present research, the effect of shear on the crystallization behavior of PP nanocomposites loaded with CNT and CB has been evaluated. In this research, CNT was selected as the main filler and CB was selected as the second filler to fabricate a nanocomposite with improved rheological, crystallization, and electrical properties. CB has many advantages compared to other fillers. For example, it has better processability, lower cost, and higher environmental stability compared to metal oxide nanoparticles. Besides, it possesses higher electrical and thermal conductivity compared to nanoclays, leading to the production of bi-filler nanocomposites with better overall properties. In addition, CB has much better processability and is much cheaper compared to other carbon fillers such as graphene and its derivatives. Finally, CB has low density, high reinforcing effect, and high UV protection, leading to the production of lightweight and strong composites with excellent UV protection. Overall, CB has been selected as a second filler due to its high electrical and thermal conductivity, high reinforcing effect, high UV protection, high processability, and high stability, as well as its low price, low weight, and low degradation. The combination of CNT and CB can integrate the high electrical conductivity and high elasticity of CNT with the high processability and reasonable price of CB.
In this research, mono-filler nanocomposites, PP/CNT and PP/CB, as well as bi-filler nanocomposites, PP/CNT/CB, with different weight percentages of CNT and CB were prepared and their rheological properties were fully evaluated. Then, the non-isothermal and isothermal crystallization of the mono-filler and bi-filler nanocomposites under shear were evaluated. And finally, the electrical conductivity of the samples in two states of quiescent and sheared were measured.
Materials and Methods
Materials
Polypropylene (PP, Jampilen HP525 J, MFI = 3 g/10 min at 230 °C) and polypropylene-graft-maleic anhydride (PP-g-MA, MFI>100 g/10 min at 230 °C) were purchased from Jam Polypropylene Comapny (Iran) and Honam Petrochemical Corporation (Korea), respectively. Multi-walled carbon nanotubes (MWCNTs, NC7000, average diameter = 9.5 nm, average length = 1.5 µm were obtained from Nanocyl (Belgium). Carbon black (CB, CAS 1333-86-4, particle size 24 nm) was purchased from Sigma-Aldrich.
Preparation of Nanocomposites
Codes and corresponding compositions of mono-filler and bi-filler nanocomposites.

Characterization of Nanocomposites
Rheometrics mechanical spectrometer (RMS)
A dynamic rheometer (UDS200, Anton Paar Physica, Austria) with a parallel plate geometry (plate diameter=25 mm and plate distance=1 mm) was used to study the rheological properties and crystallization of the nanocomposites. To study rheological properties, frequency sweep analysis was performed in the frequency range of 0.01-1000 rad/s (temperature of 190 °C). To evaluate the crystallization behavior, time sweep and temperature sweep analyses were done. For the temperature sweep analysis, the nanocomposite melts were cooled at a rate of 5 °C/min and the storage modulus changes with temperature were evaluated. For the time sweep analysis, the nanocomposite melts were cooled to the crystallization temperature at a rate of 15 °C/min and the storage modulus changes with time were evaluated. To evaluate the effect of shear on the crystallization behavior, in the temperature sweep analysis, a pre-shear was applied to the nanocomposite melt. In the time sweep analysis, a pre-shear was applied to the nanocomposite melt at the crystallization temperature. In the temperature sweep and time sweep analyses, the strain amplitude and frequency were 2% and 2 rad/s, respectively. The test atmosphere was N2.
Differential scanning calorimetry
A differential scanning calorimeter (DSC822e, Mettler-Toledo, USA) was employed to study the thermal properties and crystallization of the nanocomposites. The samples were heated rapidly to 180 °C and held at this temperature for 5 min to remove the thermal history, then cooled at a cooling rate of 5 °C/min to obtain the cooling thermograms, then heated at a heating rate of 5 °C/min to obtain the second heating thermograms. The crystallinity of nanocomposites was calculated using equation (1):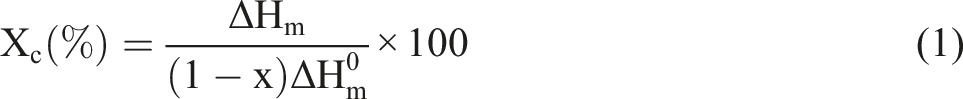
The crystal thickness of the nanocomposites was calculated by Gibbs-Thomson equation:
Conductivity measurement
An electrometer (6517B Electrometer/Keithley Instrument, USA) was used to measure the electrical conductivity of the nanocomposites.
Statistical analysis
Statistical analysis of quantitative results was performed using one-way analysis of variance and Tukey’s post hoc analysis with SPSS software. The significance level was considered to be
Results and Discussions
Rheology Results
The complex viscosity versus frequency curves for pure PP, PP/CNT, PP/CB, and PP/CNT/CB nanocomposites are shown in Figure 1(a). According to the Figure, the viscosity increased, when CNT, CB, or both were added to the PP matrix. However, the viscosity values of the nanocomposites with low amount of nanoparticles, i.e. PP/CNT0.1, PP/CB3, and PP/CNT0.05/CB1.5, were close to those of the pure polymer. Nanocomposite with 4% CNT had the highest viscosity in the low frequency range. Moreover, this nanocomposite showed a very sharp viscosity upturn at low frequencies. The high viscosity values and the sharp viscosity upturn at low frequencies confirm the formation of a three-dimensional network of CNTs in this nanocomposite, which confines the polymer chains and slows down their motions. Rheological properties of the mono-filler and bi-filler nanocomposites. Viscosity (a), storage modulus (b), loss modulus (c), and 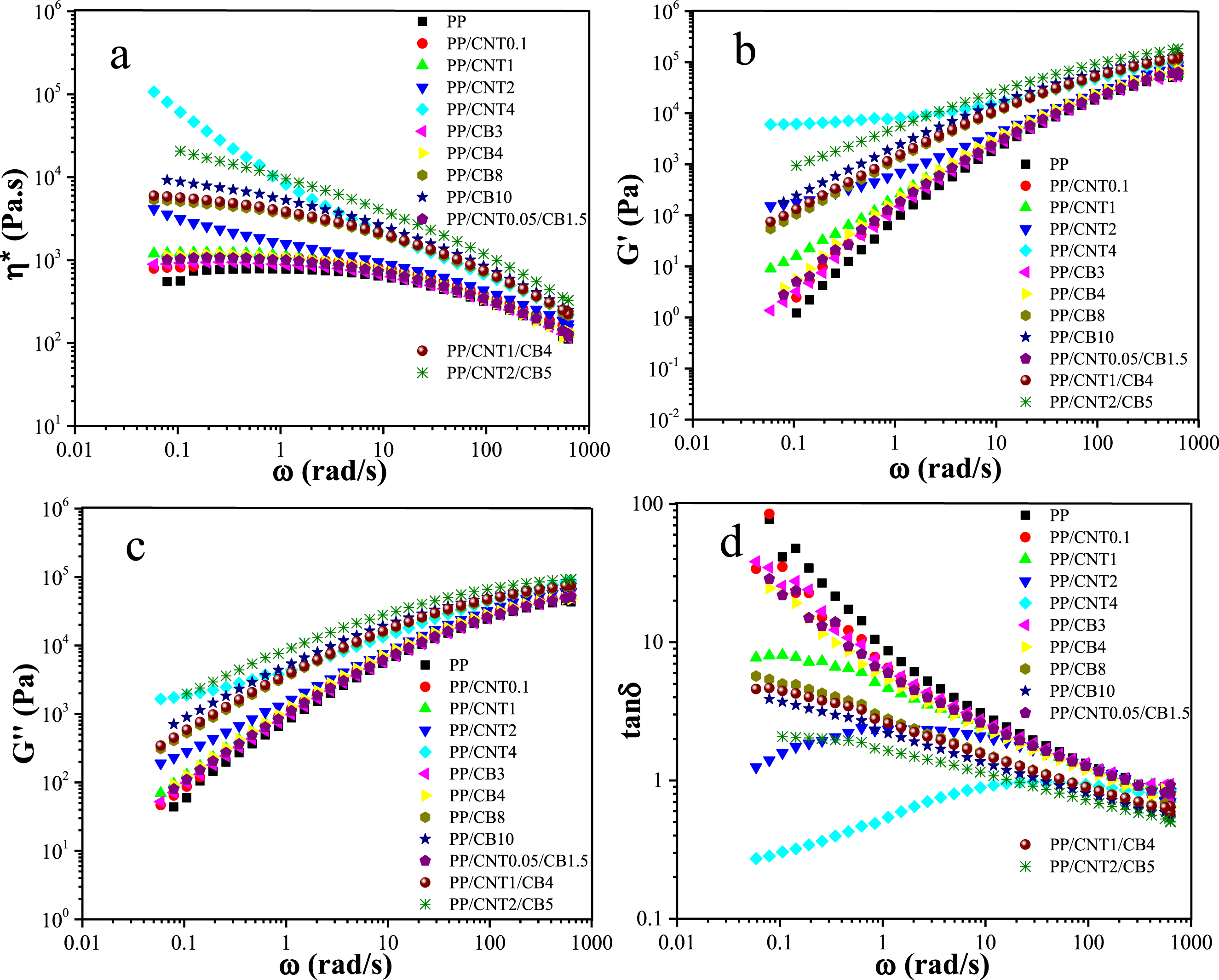
The bi-filler nanocomposite with 2% CNT and 5% CB had the highest viscosity in the high frequency range and also the second highest viscosity in the low frequency range, corroborating that the CNTs and CBs formed a continues network in the polymer matrix. In PP/CNT, PP/CB, and PP/CNT/CB nanocomposites, the complex viscosity increased proportionally to the amount of nanoparticles, and the increase in viscosity was more evident at lower frequencies, because the nanoparticles mostly affect the dynamics of the long polymer chains.
In the comparison between mono-filler nanocomposites and bi-filler nanocomposite with the same weight fractions of fillers, i.e. PP/CNT1, PP/CB4, and PP/CNT1/CB4, it can be seen that the inclusion of the second nanoparticle significantly increases the complex viscosity. CNT and CB have different structures and dimensions. CNT is a one-dimensional cylindrical nanostructure, while CB is a zero-dimensional spherical nanostructure. Therefore, the concurrent presence of CNT and CB causes a synergistic effect and facilitates the formation of a continuous and hierarchical network of nanofillers, which imposes a greater restricting effect on the chain dynamics. Moreover, CB not only can improve the dispersion of CNTs in the polymer matrix and prevent their agglomeration, but also can help to connect isolated CNTs and boost the formation of a more robust and interconnected network.43,44 Other researches have shown that the presence of a second filler that is spherical can improve the CNT dispersion in the polymer matrix.45,46
The only interaction between PP and carbon fillers, CNT and CB, is weak van der Waals forces, which are usually not sufficient for the good dispersion of fillers in the polymer matrix. In PP/CNT composites, strong entanglements of CNTs are often formed, and in PP/CB composites, large aggregates of CB are frequently created. CNT and CB are carbon fillers with different structural properties. CB is a spherical filler and CNT is a cylindrical filler with high aspect ratio. These structural differences allow CB particles to fill the gaps between CNTs and facilitate the formation of a rheological percolated network. Furthermore, CNTs can prevent the aggregation of CB particles by creating an interconnected network.
The curves of storage and loss modulus versus frequency for pure PP, PP/CNT, PP/CB, and PP/CNT/CB nanocomposites are shown in Figure 1(b) and (c). According to the curves, the storage modulus increased when nanoparticles were added to the PP matrix. The increase in storage modulus was due to the reduction of polymer dynamics. In the low frequency range, PP/CNT4 had higher moduli than PP/CNT2/CB5. However, in a high frequency range, the moduli of PP/CNT2/CB5 were higher than those of PP/CNT4, most likely due to the destruction of CNT network at high frequencies.
Storage modulus-angular frequency and loss modulus-angular frequency slopes at low frequencies.

The rheological behavior of the mono-filler nanocomposite and bi-filler nanocomposite with the lower content of each filler, namely PP/CNT0.1-PP/CB3-PP/CNT0.05/CB1.5, PP/CNT2-PP/CB8-PP/CNT1/CB4, and PP/CNT4-PP/CB10-PP/CNT2/CB5, was compared. In the case of PP/CNT0.1, PP/CB3 and PP/CNT0.05/CB1.5, the bi-filler nanocomposite had higher storage modulus and more non-terminal behavior than PP/CNT0.1 and PP/CB3. In the case of PP/CNT2, PP/CB8 and PP/CNT1/CB4, the
The
The curves of loss modulus versus storage modulus for pure PP, PP/CNT, PP/CB, and PP/CNT/CB nanocomposites are shown in Figure 2(a). These curves allow for the easy comparison of the viscous and elastic behavior of the samples. For better comparison, the line Rheological properties of the mono-filler and bi-filler nanocomposites. Loss modulus versus storage modulus changes (a), imaginary viscosity versus real viscosity changes (b) and phase angle versus complex modulus changes (c).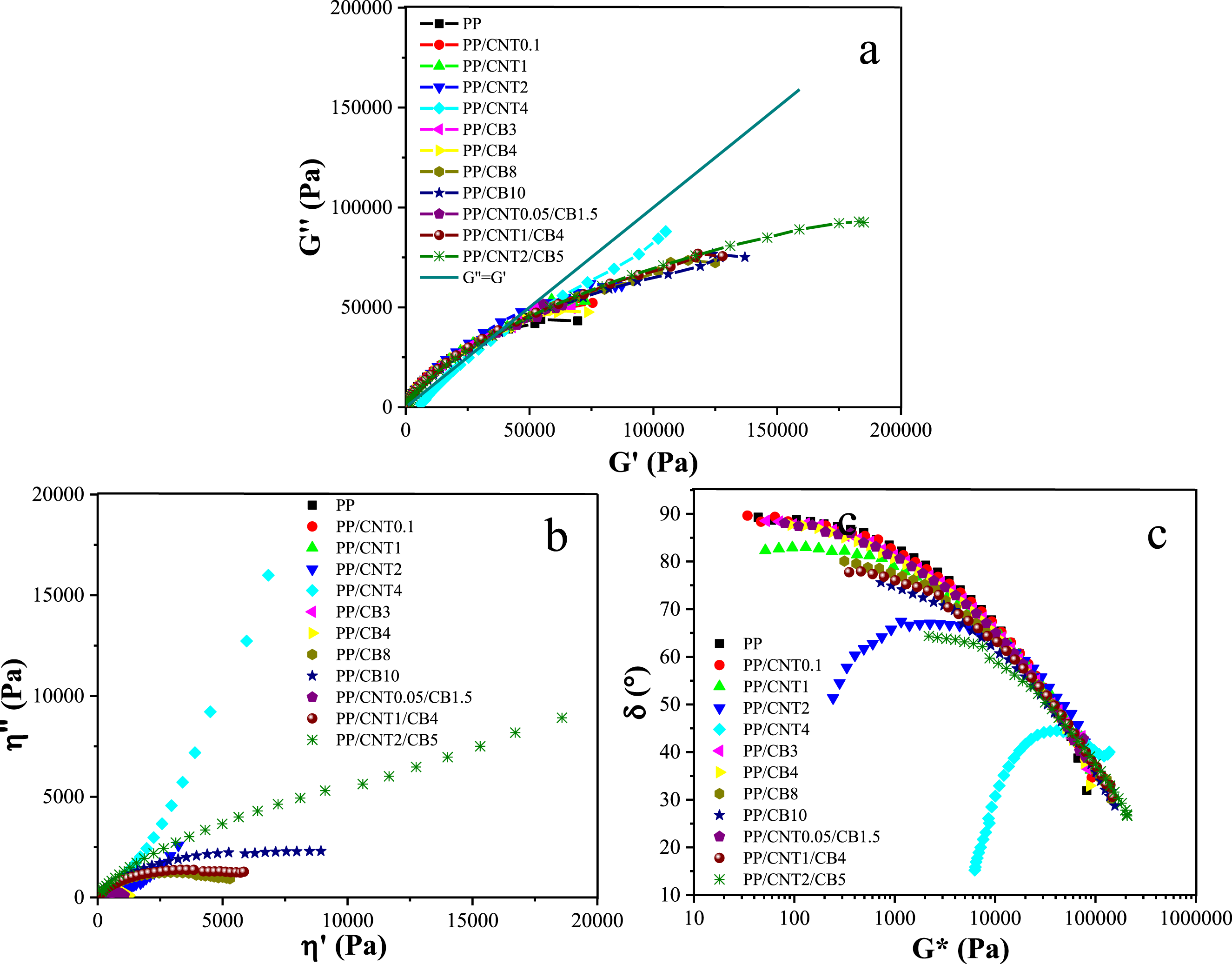
According to the Figure 2(a), the PP/CNT4 curve lies below the
The curves of imaginary viscosity versus real viscosity for pure PP, PP/CNT, PP/CB, and PP/CNT/CB nanocomposites are shown in Figure 2(b). Pure PP, PP/CNT0.1, PP/CNT1, PP/CB3, PP/CB4, and PP/CNT0.05/CB1.5 represented semicircular curves because the amount of nanoparticles was not sufficient to form an interconnected network. For the other nanocomposites, a deviation from the semi-circular form was observed, which can be attributed to the formation of a 3D network of nanoparticles. The highest deviation was observed for PP/CNT4 and PP/CNT2/CB5 nanocomposites, confirming the formation of a stronger network of nanoparticles in these compositions.
The curves of phase angle versus complex modulus (van Gurp-Palmen plot) for pure PP, PP/CNT, PP/CB, and PP/CNT/CB nanocomposites are shown in Figure 2(c). According to the curves, PP/CNT4, PP/CNT2/CB5 and PP/CNT2 have the lowest phase angles, respectively. The lower the phase angle, the higher the solid-like behavior. The shift of the van Gurp-Palmen plot to lower angles indicates the formation of a 3D network of nanoparticles in these compositions. In the comparison between mono-filler nanocomposite and bi-filler nanocomposite with the same weight fractions of fillers, i.e. PP/CNT1-PP/CNT1/CB4 and PP/CNT2-PP/CNT2/CB5, it can be seen that the bi-filler nanocomposite had lower phase angles compared to the mono-filler nanocomposites, confirming the formation of a stronger network in the presence of the second filler.
Non-isothermal Crystallization Results
Non-isothermal crystallization of the samples was studied by temperature sweep analysis. This analysis was performed under two conditions; in the first condition, the samples were analyzed without shear and in the second condition, the samples were analyzed after exposure to shear. The samples were gradually cooled from the molten sate to 100 °C and the changes in storage modulus versus temperature were plotted. In the temperature sweep plot (Figure 3(a)), the temperature at which storage modulus increases substantially is considered as the crystallization temperature (Tc) (Figure 3(b)). Storage modulus changes with temperature (a) and crystallization temperature (b) of mono-filler and bi-filler nanocomposites.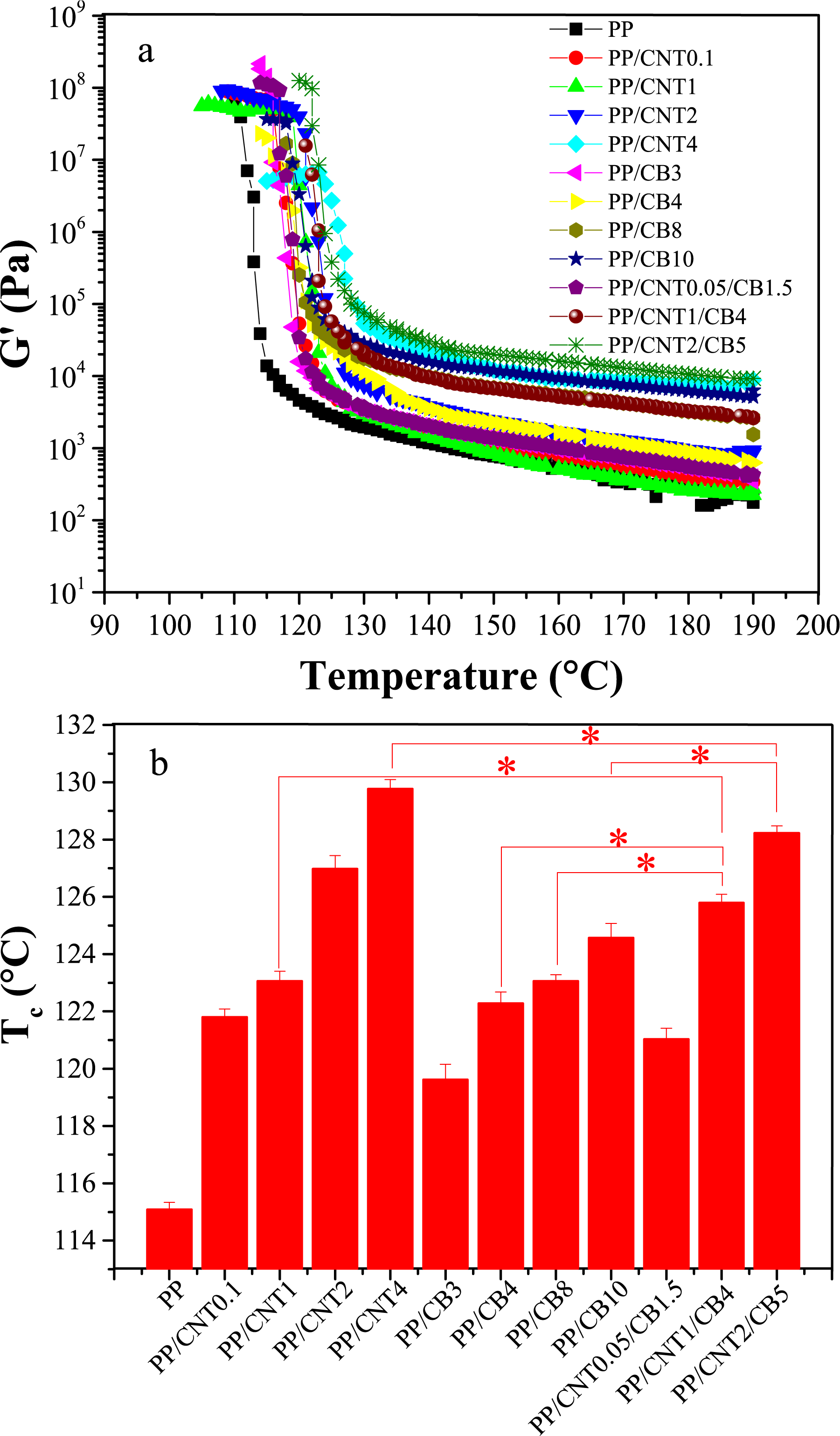
The Tc of the pure polymer was 115.08 °C. The Tc of PP/CNT0.1, PP/CNT1, PP/CNT2 and PP/CNT4 was 121.80, 123.06, 126.97 and 129.77 °C, respectively. The Tc of PP/CB3, PP/CB4, PP/CB8 and PP/CB10 samples was 119.62, 122.28, 123.06 and 124.56 °C, respectively. The Tc of PP/CNT0.05/CB1.5, PP/CNT1/CB4 and PP/CNT2/CB5 samples was 121.03, 125.79 and 128.22 °C, respectively.
According to the obtained results, the addition of CB and CNT nanoparticles to PP increased the crystallization temperature of the polymer, but with the addition of CNT compared to CB nanoparticles, this increase was significantly higher. This difference can be attributed to the strong nucleating effect of CNT as well as its high aspect ratio. Carbon black also can act as a nucleating agent, however, it lacks the well-organized structure and high surface area of CNT and cannot play the nucleation role as much as CNT. Moreover, CNT has a higher thermal conductivity than CB, which causes more uniform heat distribution during cooling and thus better crystallization. The highest Tc was for PP/CNT4 followed by PP/CNT1/CB4 nanocomposites. The Tc of PP/CNT4 and PP/CNT1/CB4 increased by 14.96 and 13.40 °C compared to pure PP, respectively.
In the comparison between mono-filler and bi-filler nanocomposites with the same weight fractions of fillers, i.e. PP/CNT1, PP/CB4, and PP/CNT1/CB4, the increase in Tc of the bi-filler nanocomposite was higher than that of the mono-filler nanocomposites (
For evaluating the effect of shear on non-isothermal crystallization, mono-filler nanocomposite, PP/CNT2, and bi-filler nanocomposites, PP/CNT0.05/CB1.5 and PP/CNT1/CB4, were selected. These samples were sheared in the molten state and then were cooled from the molten state to 100 °C. The changes in storage modulus with temperature were recorded (Figure 4(a)). According to the obtained results, in the mono-filler nanocomposite, shearing increased the crystallization temperature (Figure 4(b)). The Tc of PP/CNT2 in the quiescent condition was 126.97 °C and increased to 128.07 °C after shearing, however this increase was not statistically significant ( Storage modulus changes with temperature (a) and crystallization temperature (b) of mono-filler and bi-filler nanocomposites in quiescent and sheared conditions.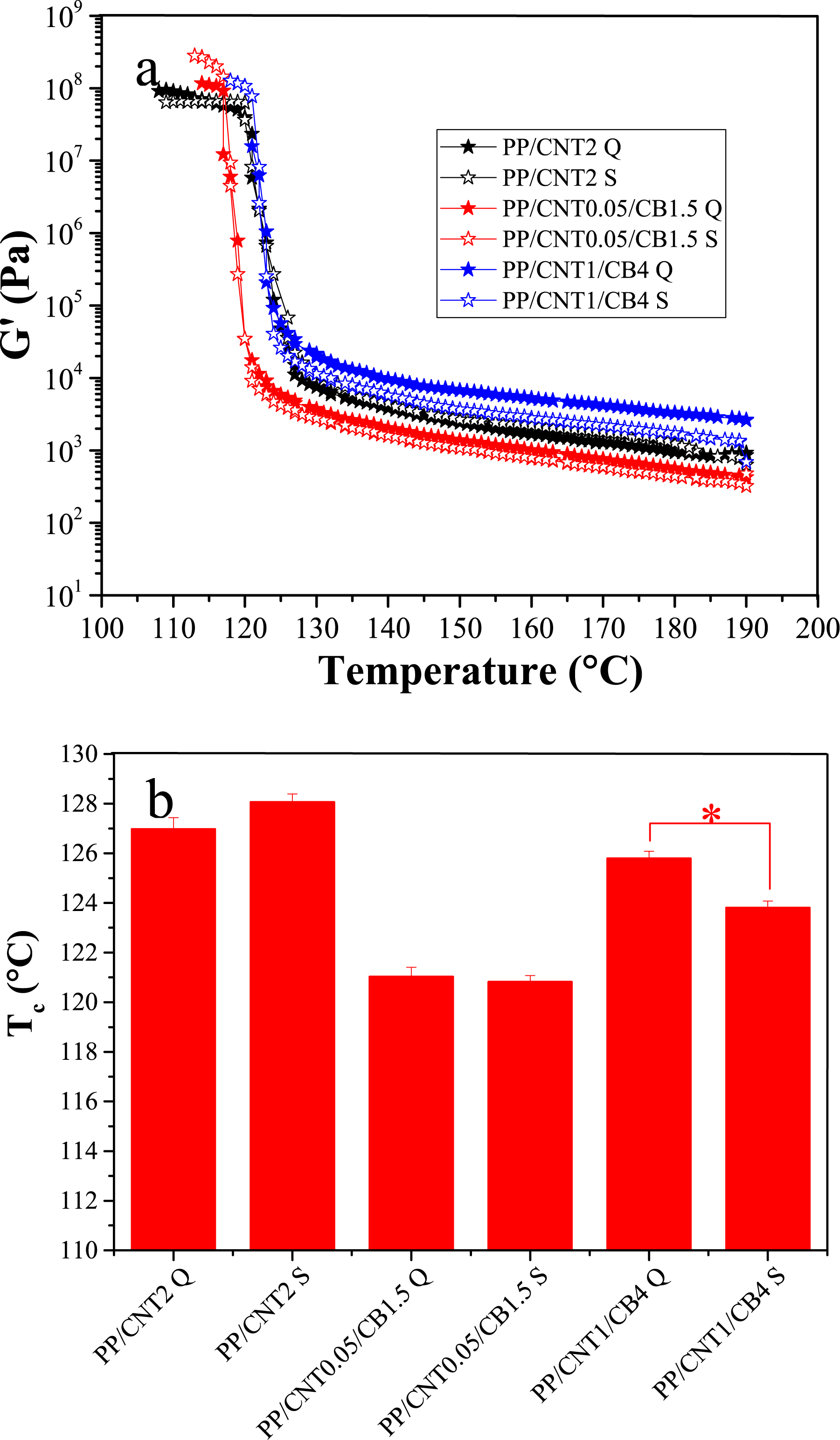
Shearing can increase the polymer chain alignment and improve the CNT dispersion. Indeed, shearing increases both homogeneous and heterogeneous nucleation, both of which facilitate PP crystallization. However, in the bi-filler nanocomposites, shearing reduced the crystallization temperature of PP. The Tc of PP/CNT0.05/CB1.5 and PP/CNT1/CB4 in the quiescent condition was 121.03 and 125.79 °C, respectively. After shearing, their Tc decreased to 120.82 (
The increase in crystallization temperature with shearing in PP/CNT nanocomposites can be explained by the dispersion of CNTs in the polymer matrix and the increase in the available surface area for heterogeneous nucleation. Furthermore, the application of shear orients the polymer chains and increases the probability of homogeneous nucleation. In bi-filler nanocomposites, applying shear can rearrange the filler network and reduce their contribution to heterogeneous nucleation. Moreover, the rearranged filler network may impose further constraints on chain dynamics, preventing the movement of polymer chains and reducing their role in crystallization. Besides, shearing can even cause the aggregation of filler particles and reduce their surface area available for nucleation.
The effect of shear on the arrangement of polymer chains and carbon fillers is schematically illustrated in Figure 5. In both mono-filler and bi-filler nanocomposites, shearing causes orientation of polymer chains and CNTs and enhances the formation of heterogeneous and homogeneous nuclei for crystallization. However, in bi-filler nanocomposites, shearing results in the rearrangement of the filler network, which can lead to a reduction in the total surface area available for crystallization. In the mono-filler nanocomposite with pre-shearing, the ultimate modulus was lower than that of the nanocomposite without shearing. Because in this sample, the CNT network is destructed and the CNTs are dispersed in the polymer matrix. Destruction of the nanoparticle network can lead to a decrease in the ultimate modulus. Schematic representation of the effect of shear on the arrangement of PP chains and carbon fillers in PP/CNT (a) and PP/CNT/CB (b) nanocomposites.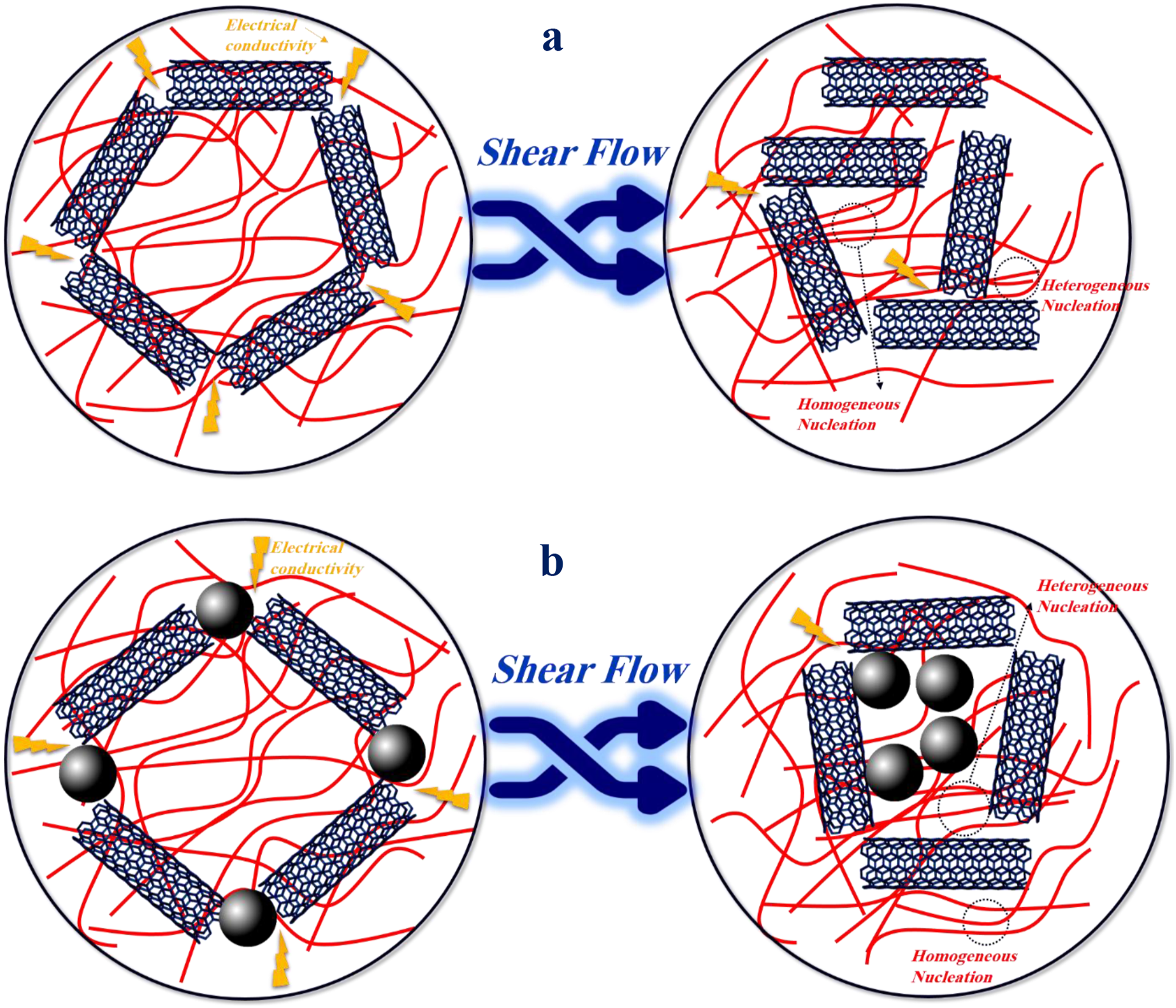
Isothermal Crystallization
Isothermal crystallization of the samples was studied by time sweep analysis. Time sweep was done at crystallization temperature of the samples in quiescent and sheared conditions, and the changes in storage modulus with time were evaluated (Figure 6). Storage modulus changes with time of mono-filler and bi-filler nanocomposites.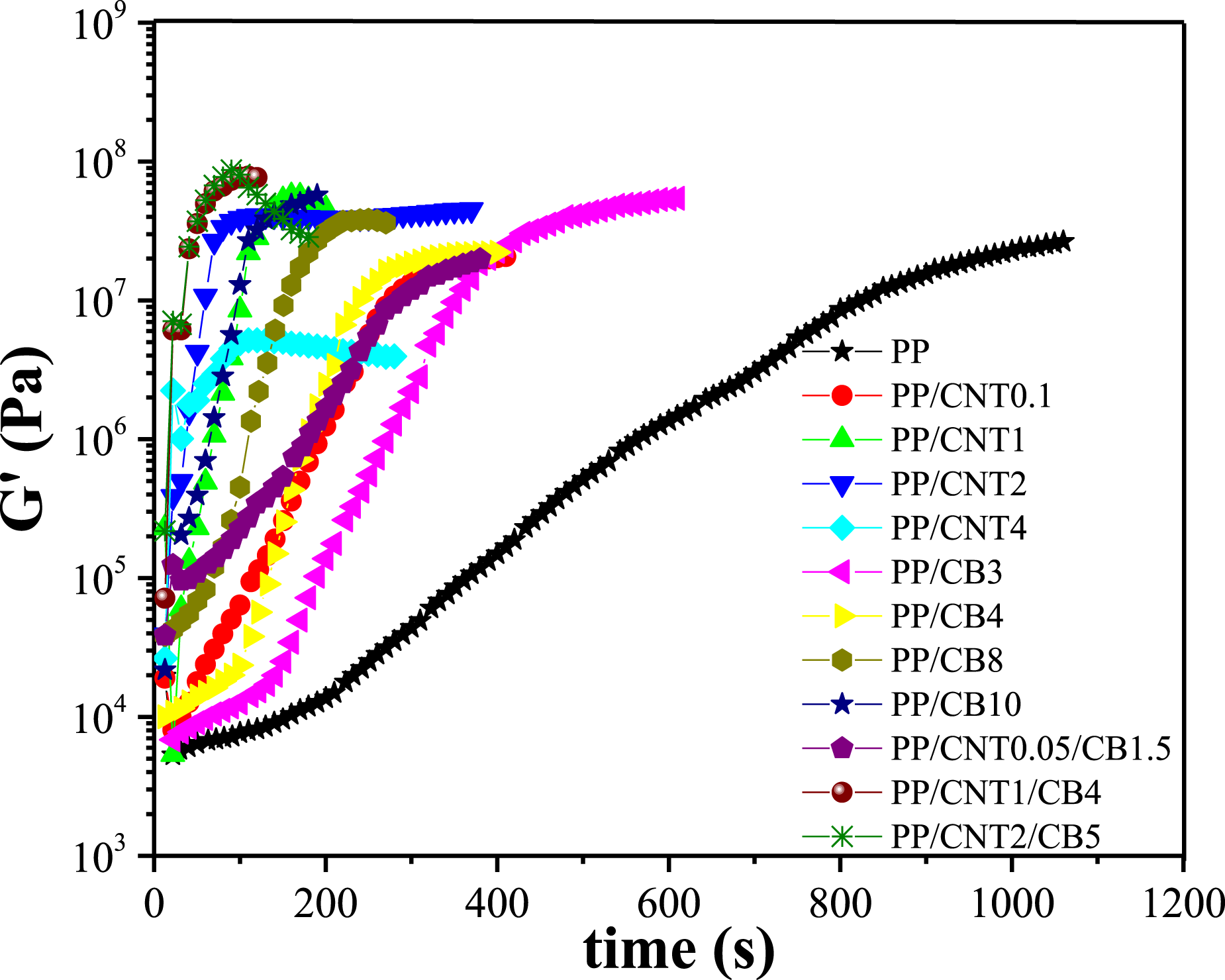
In the time sweep curves, the time that modulus increases sharply, was considered as the crystallization onset time. According to the curves, with the addition of nanoparticles, the crystallization started sooner, suggesting the accelerating role of nanoparticles in crystallization. This effect can be attributed to the role of nanoparticles as heterogeneous nucleating agents. The higher the nanoparticle content, the sooner the crystallization. CNT nanoparticles have higher effect on crystallization time of PP compared to CB nanoparticles due to their stronger nucleation effect and their higher aspect ratio.
In the comparison between mono-filler nanocomposites and bi-filler nanocomposite with the same weight fractions of fillers, i.e. PP/CNT1, PP/CB4, and PP/CNT1/CB4, the crystallization of bi-filler nanocomposite was faster than that of mono-filler nanocomposites. This is because the presence of the second filler can improve the dispersion of the first nanoparticle and create more nucleation sites. In the comparison between mono-filler nanocomposites and bi-filler nanocomposite with the lower content of each filler, namely PP/CNT0.1-PP/CB3-PP/CNT0.05/CB1.5, PP/CNT2-PP/CB8-PP/CNT1/CB4 and PP/CNT4-PP/CB10-PP/CNT2/CB5, the bi-filler nanocomposites had faster crystallization than the CB-loaded nanocomposites and a crystallization rate almost equivalent to that of the CNT-containing nanocomposites.
Among the bi-filler nanocomposites, PP/CNT1/CB4 had the fastest crystallization. Because for the nucleation role of nanoparticles on polymer matrix, there is an optimal content at which nucleation is saturated and after that increasing the amount of nanoparticles reduces the polymer dynamics and hence the crystallization rate.
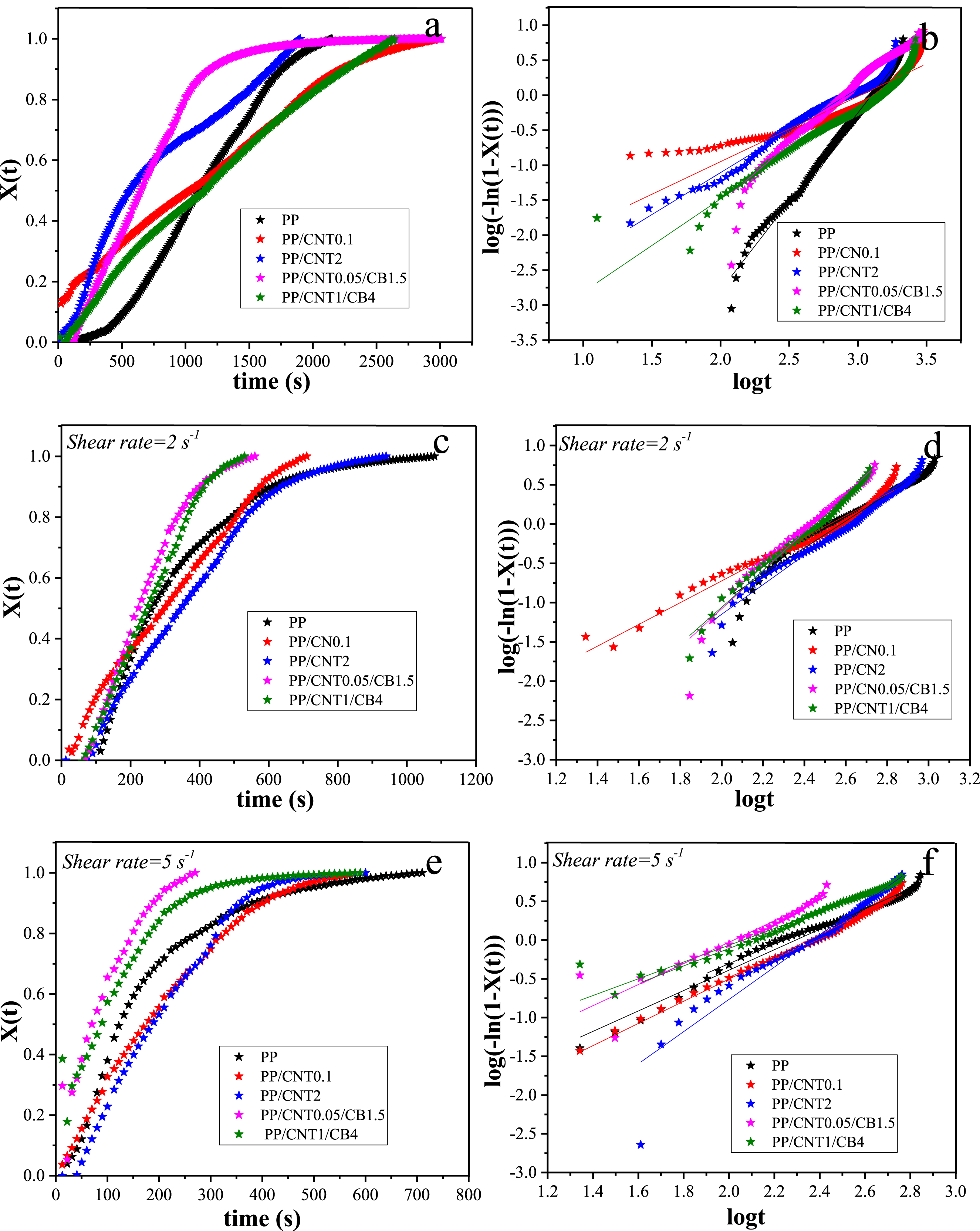
In the following, the effect of shear on the isothermal crystallization of samples was evaluated. For this purpose, the molten samples were sheared with different shear rates including 2 and 5 s−1 and different shear times including 5, 10, 20, 60 and 90 s. The changes in storage modulus with time for the sheared samples are shown in Figure 7. Storage modulus changes with time for mono-filler and bi-filler nanocomposites in quiescent and sheared conditions with different shear rates and times. PP (a), PP/CNT0.1 (b), PP/CNT2 (c), PP/CNT0.05/CB1.5 (d), PP/CNT1/CB4 (e).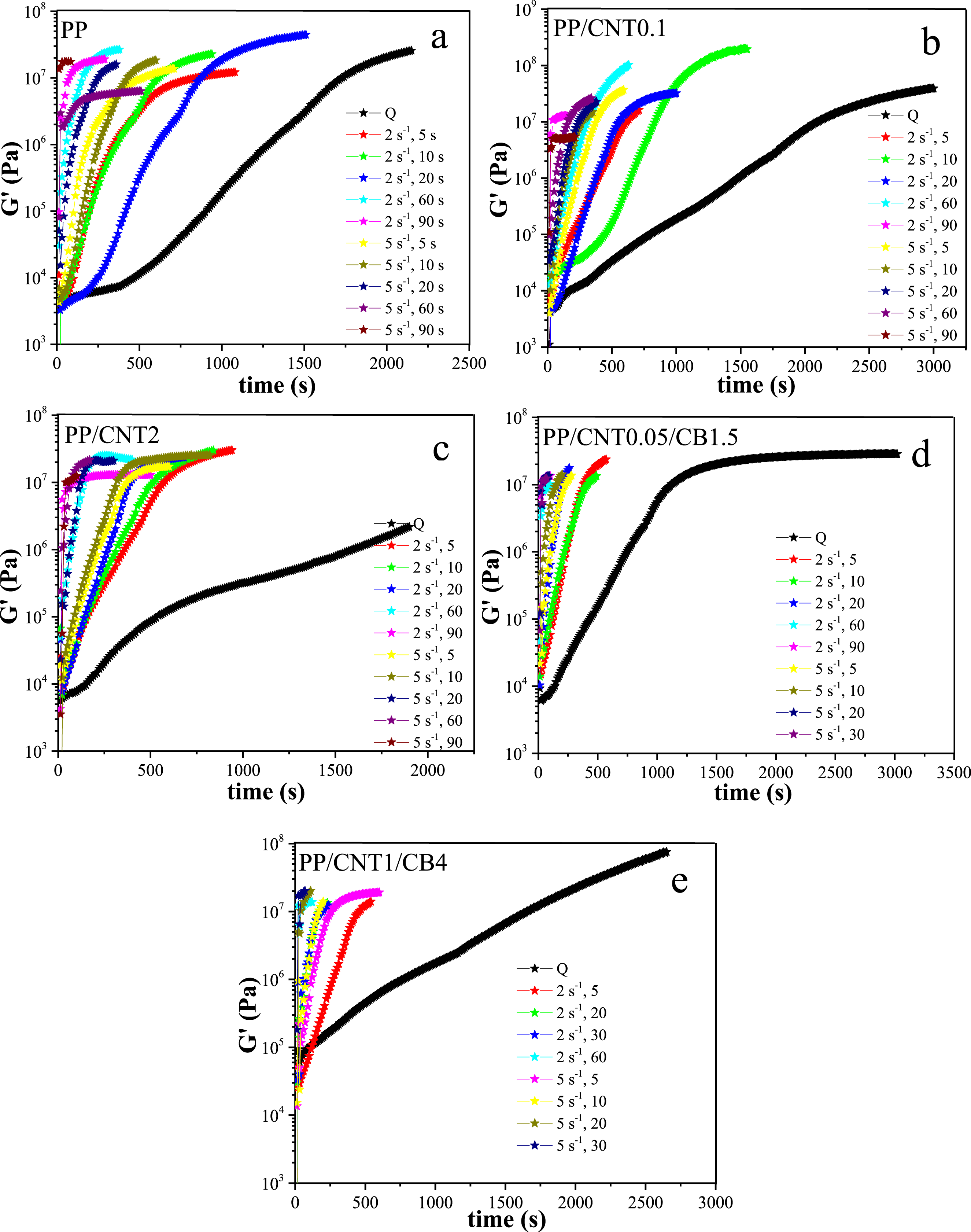
According to the obtained results, increasing the shear rate and time decreased the crystallization time and enhanced the crystallization kinetics. The slope of modulus curves increased significantly in nanocomposites compared to the pristine polymer. However, changing the shear rate and time did not remarkably change the slope of the curves. Because, the slope of the curve is related to the mechanism and the type of the nucleation as well as the geometry of the crystal growth. Therefore, the slope of the curves did not change with shear time and rate, and only the crystallization onset time changed. It has to be noted that in bi-filler nanocomposites, the final modulus in the sheared samples was lower than that in the quiescent samples, most likely due to the slow crystal growth and not due to less crystallization. Because, as a result of shearing, highly ordered crystalline structures are formed which require more time to grow.
To provide quantitative data of isothermal crystallization, Avrami equation was fitted to the time sweep curves.47,48 For this purpose, quiescent and pre-sheared samples with shear rates of 2 and 5 s−1 and shear time of 5 s were selected (Figure 8). Fitting the isothermal crystallization data with Avrami equation for samples without shear (a and b), sheared at 2 s−1 (c and d) and 5 s−1 (e and f).
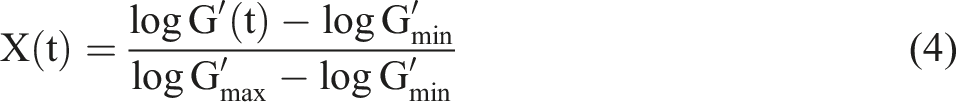
Half-time of crystallization was estimated by
49
:
To better compare the crystallization rare and to exclude the effect of Avrami exponent on K, another parameter in form of
Quantitative data of isothermal crystallization of PP, mono-filler and bi-filler nanocomposites obtained from Avrami equation including Avrami exponent, crystallization rate and crystallization half-time.
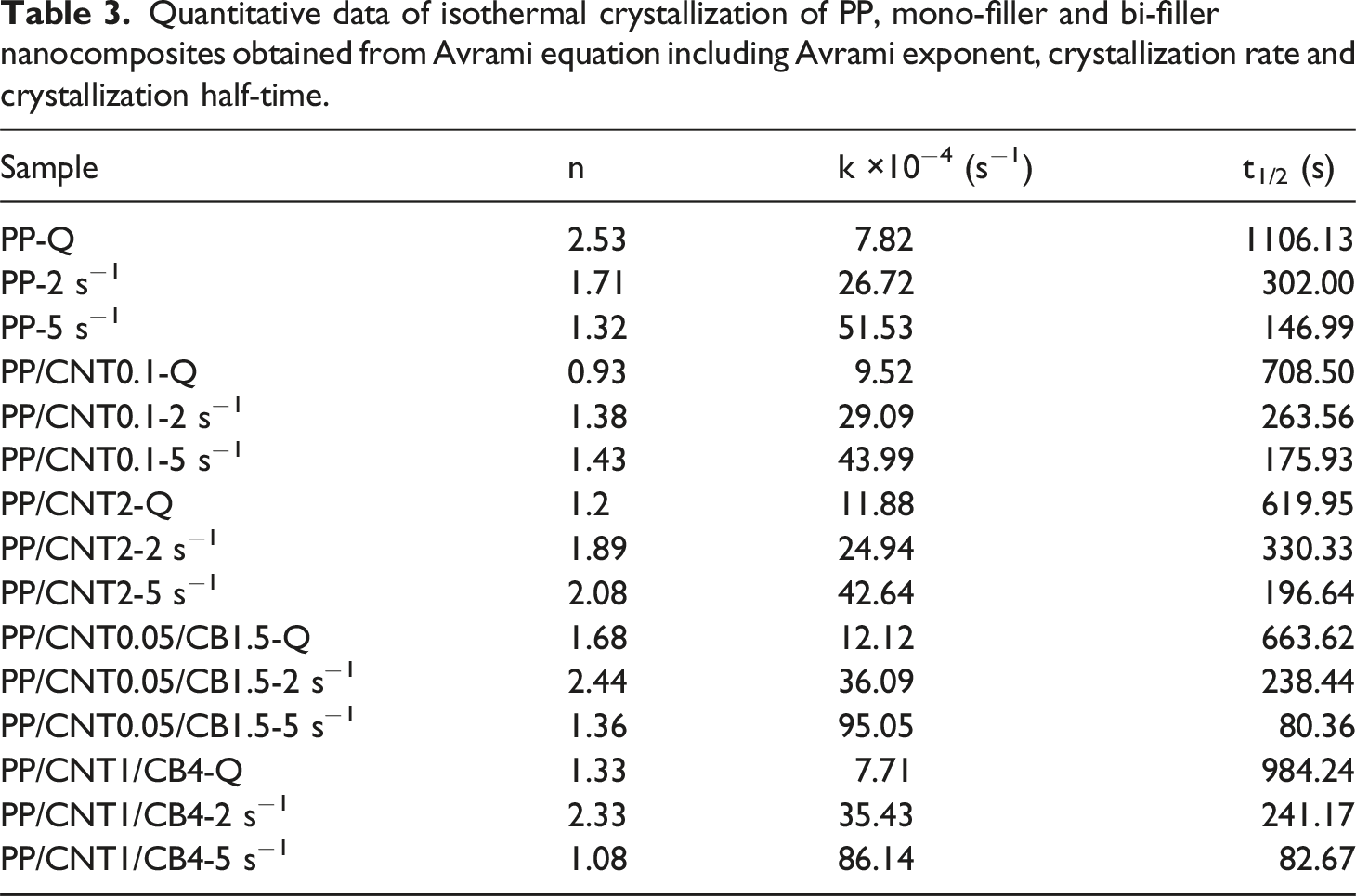
Applying shear to pure PP decreased Avrami exponent. Because applying shear can transform the spherulitic structure into shish-kebab structure. However, in nanocomposites, applying shear increased the Avrami exponent. Because applying shear causes the nanoparticles to disperse uniformly in the polymer matrix and reduces the limiting role of nanoparticles on crystal growth. Furthermore, applying shear can change the nucleation from instantaneous type to sporadic type, the nanoparticles can gradually orient under shear flow and crystal nuclei can gradually form.
Adding nanoparticles or applying shear increased
In bi-filler nanocomposites, with increasing filler content, the crystallization kinetics became more sensitive to shear. In bi-filler nanocomposite with lower filler content, applying shear caused a 3-fold increase in k value, while in bi-filler nanocomposite with high filler content, applying shear caused a 4.6-fold increase in k value. This is because the presence of a high amount of nanoparticles and the formation of a 3D network between them can restrict the chain mobility and reduce the crystal growth. Applying shear can counteract these effects, destroy the nanoparticle network, improve the nanoparticle dispersion and promote the crystallization kinetics. In bi-filler nanocomposite, applying shear with low shear rate increased Avrami exponent, while with high shear rate decreased it. Because at low shear rate, applying shear causes the destruction of nanoparticle network and the dispersion of nanoparticles in the polymer matrix, thus decreasing the restriction imposed by nanoparticles on crystal growth. However, when the shear rate increases, the polymer chains become highly oriented and the crystal growth becomes anisotropic.
In the comparison between bi-filler nanocomposites, PP/CNT0.05/CB1.5 and PP/CNT1/CB4, and mono-filler nanocomposites, PP/CNT0.1 and PP/CNT2, the bi-filler nanocomposite had a higher crystallization rate than its mono-filler counterpart, due to the lower amount of CNT and less restriction on crystal growth.
To evaluate whether the crystallization of the samples is strain-controlled or shear rate-controlled, the time sweep analysis at various shear rates and strains was performed. This analysis was done on PP/CNT0.1, PP/CNT2, PP/CNT0.05/CB1.5 and PP/CNT1/CB4 nanocomposites and the results are shown in Figure 9. According to the obtained results, in mono-filler nanocomposite with 0.1 wt% CNT, the crystallization rate was completely in line with the shear rate, and increasing the shear rate increased the crystallization rate. The shear rate-controlled trend is suggestive of liquid-like behavior of the nanocomposite melt. In mono-filler nanocomposite with 2 wt% CNT, the crystallization rate became less dependent on shear rate and was somewhat sensitive to strain. This behavior can be explained by the higher amount of CNT in this nanocomposite and the formation of a weak network of CNTs. However, the behavior was still not completely strain-controlled. Time sweep analysis of mono-filler and bi-filler nanocomposites at various shear rates and strains. PP/CNT0.1 (a), PP/CNT2 (b), PP/CNT0.05/CB1.5 (c), PP/CNT1/CB4 (d).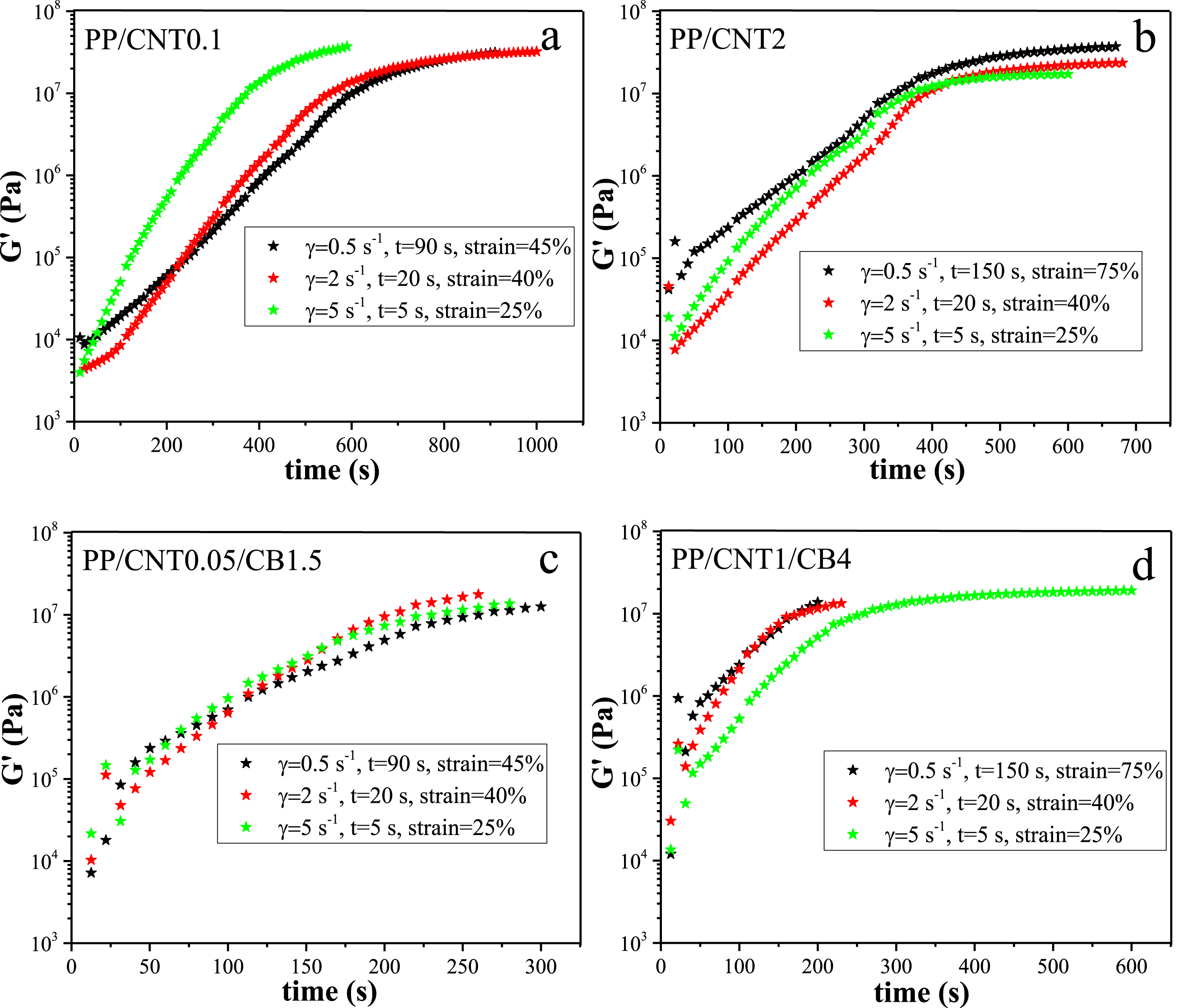
In the bi-filler nanocomposites, the crystallization rate was mainly strain-dependent, most likely due to the formation of a 3D network of CB and CNT nanoparticles. In the presence of two nanoparticles, the 3D network is formed at their lower concentrations, and the formation of 3D network of nanoparticles changes the behavior from shear rate-controlled to strain-controlled. In this case, the applied strain should be large enough to first break the 3D network of nanoparticles and then orient the nanoparticles and polymer chains. The strain-dependent behavior was clearly evident in bi-filler nanocomposites with higher filler content (PP/CNT1/CB4), due to the formation of a stronger network of nanoparticles. In this nanocomposite, the crystallization rate increased proportionally to the strain.
DSC Results
DSC cooling and second heating curves of pure PP and nanocomposites are shown in Figure 10. From the curves, crystallization temperature, melting temperature, crystallinity percentage and crystal thickness were obtained (Table 4). Crystallinity was calculated by normalizing the melting enthalpy of the sample to that of the fully crystalline PP, and the crystal thickness was calculated by Gibbs-Thomson equation. According to the obtained results, when nanoparticles were added to PP, the crystallization temperature increased due to the nucleating effect of nanoparticles, while melting temperature and crystal thickness decreased. This is because nanoparticles have two opposing effects on the nucleation and growth of polymer crystals. They promote crystal nucleation by providing appropriate surface for the arrangement of polymer chains, while reducing the crystal growth by inhibiting the dynamics of polymer chains and reducing the available space for crystal thickening. DSC cooling (a) and second heating (b) curves of mono-filler and bi-filler nanocomposites. Quantitative data of DSC curves for PP, mono-filler and bi-filler nanocomposites including crystallization temperature, melting temperature, crystallinity percentage and crystal thickness.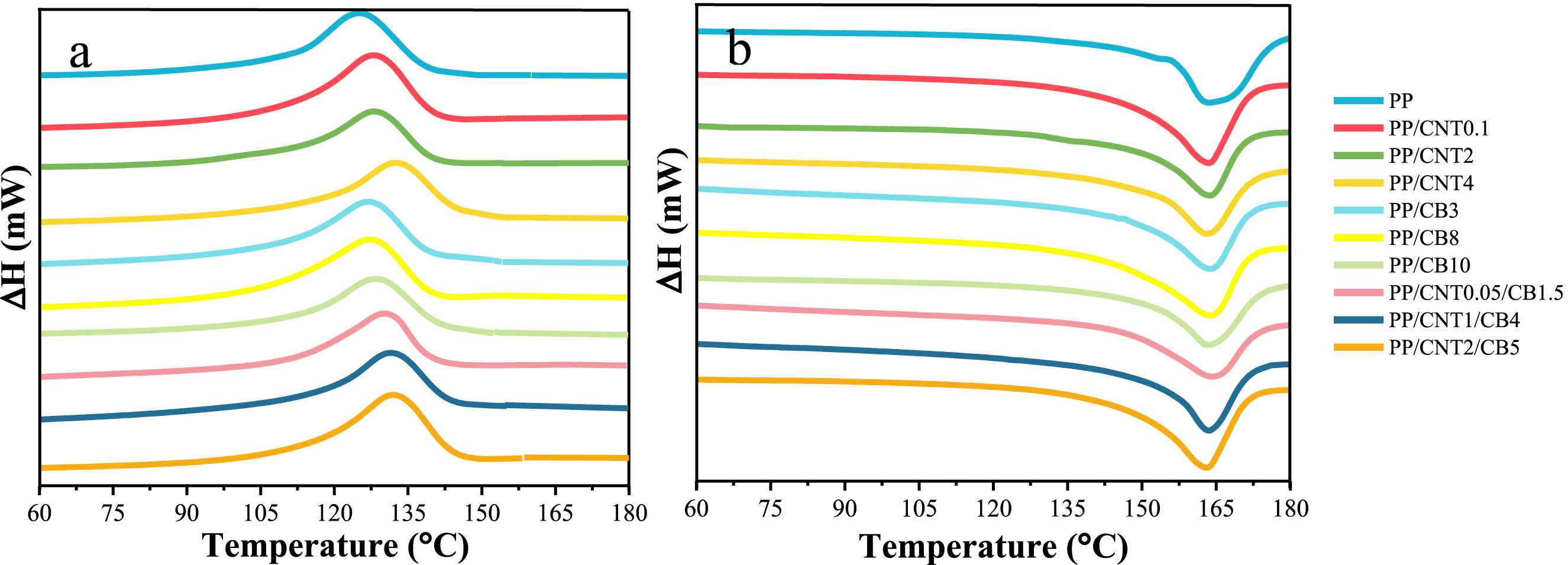
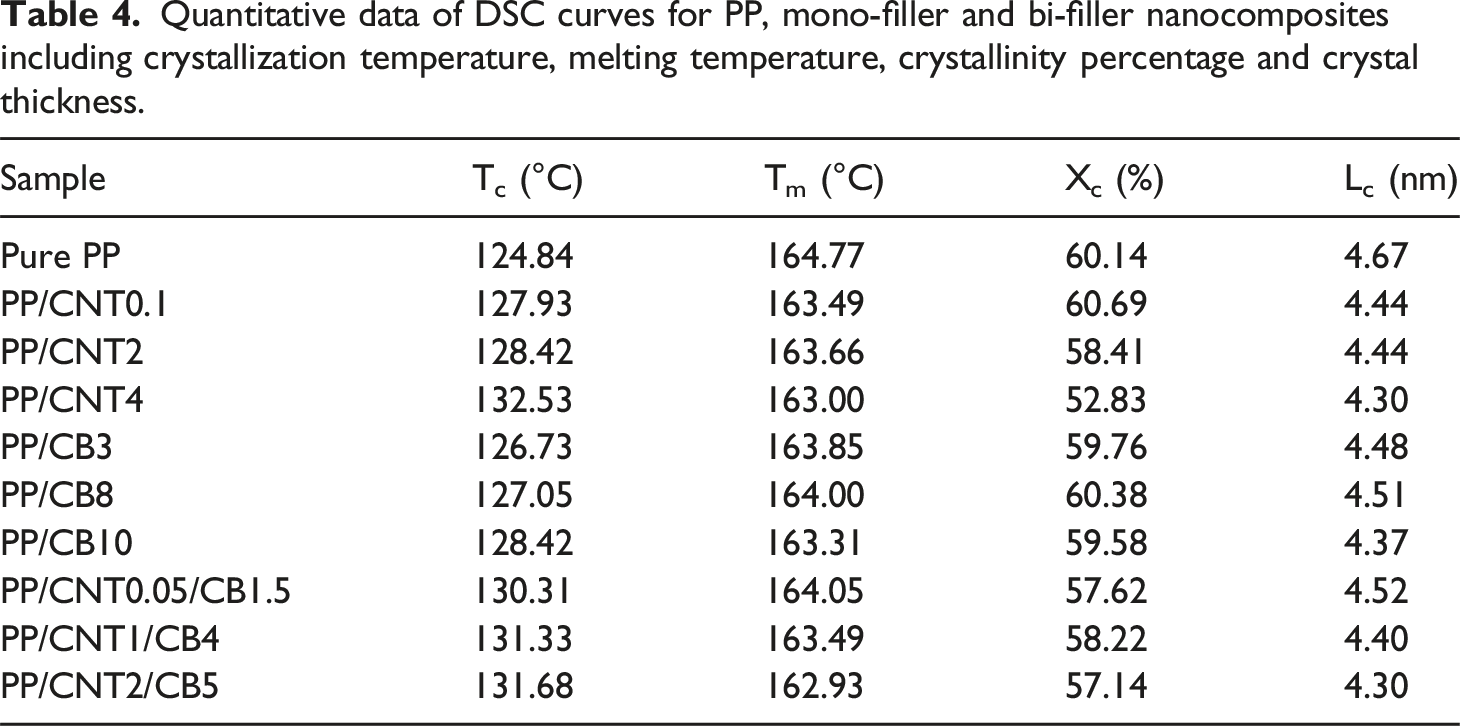
The crystallinity of the nanocomposites and the pure polymer was almost the same and no statistically significant difference was observed between them (
The greatest influence of nanoparticles on thermal transition and crystallization was related to nanocomposites with 4% CNT. In this nanocomposite, the CNTs are enough to form a continuous network and significantly inhibit the crystallization of PP. This effect is known as confined crystallization, in which the presence of a large amount of nanoparticles prevents the formation of crystalline structures in the polymer. In addition, in bi-filler nanocomposite with the high CNT and CB content, namely PP/CNT2/CB5, the decrease in Tm, crystal thickness and crystallinity percentage was relatively pronounced. However, the decrease in crystallinity was as not as high as PP/CNT4.
Electrical Conductivity Results
The electrical conductivity of the mono-filler and bi-filler nanocomposites are represented in Figure 11(a). As expected, the electrical conductivity increases proportionally with the amount of nanoparticles. The increase in electrical conductivity in PP/CNT nanocomposites was much more than that in PP/CB nanocomposites. When CNT increased from 0.1 to 4%, the electrical conductivity was raised by more than nine orders of magnitude ( Electrical conductivity of the mono-filler and bi-filler nanocomposites (a), electrical conductivity of the mono-filler and bi-filler nanocomposites in quiescent and shear conditions (b).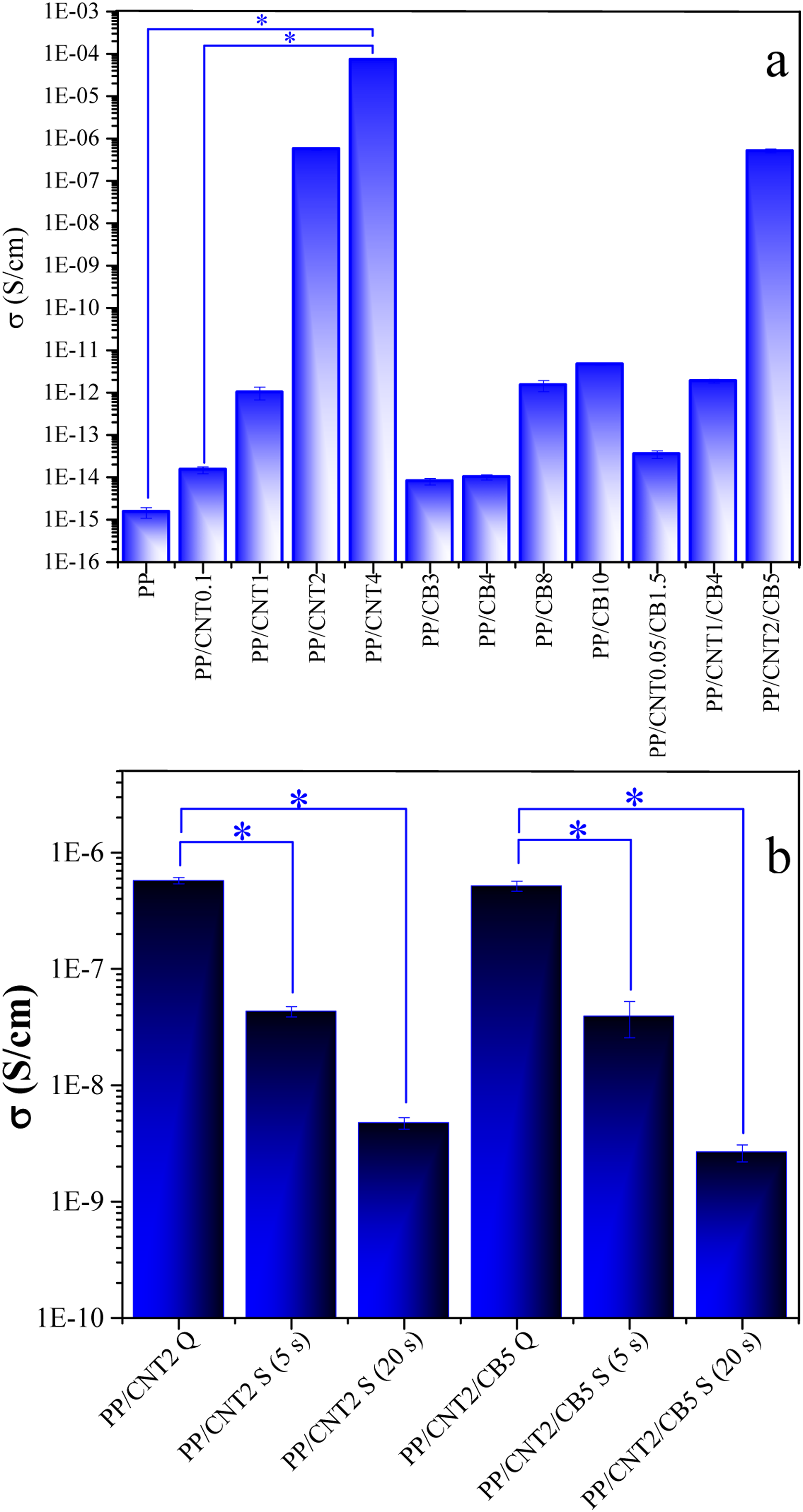
The electrical conductivity of mono-filler nanocomposites and bi-filler nanocomposite with the same weight fractions of fillers, i.e. PP/CNT1-PP/CNT1/CB4 and PP/CNT2-PP/CNT2/CB5, was compared. The conductivity of PP/CNT1 and PP/CNT1/CB4 was 1.01 × 10−12 and 1.87 × 10−12 S/cm, respectively. The conductivity of PP/CNT2 and PP/CNT2/CB5 was 5.75 × 10−7 and 5.17 × 10−7 S/cm, respectively. Therefore, it can be said that at low CNT content, the addition of CB to CNT-loaded nanocomposite improved the electrical conductivity, but at high CNT content, the addition of CB did not change the electrical conductivity.
Moreover, the electrical conductivity of mono-filler nanocomposites and bi-filler nanocomposite with the lower content of each filler, namely PP/CNT0.1-PP/CNT0.05/CB1.5, PP/CNT2-PP/CNT1/CB4 and PP/CNT4-PP/CNT2/CB5, was compared. The conductivity of PP/CNT0.1 and PP/CNT0.05/CB1.5 was 1.5 × 10−14 and 3.5 × 10−14 S/cm, respectively. The conductivity of PP/CNT2 and PP/CNT1/CB4 was 5.75 × 10−7 and 1.87 × 10−12 S/cm, respectively. The conductivity of PP/CNT4 and PP/CNT2/CB5 was 7.42 × 10−5 and 5.17 × 10−7 S/cm, respectively. Therefore, it can be implied that the electrical conductivity of bi-filler nanocomposite is mainly dependent on the CNT content.
To study the effect of shear on the electrical conductivity of the nanocomposites, the samples sheared before isothermal crystallization were analyzed (Figure 11(b)). According to the obtained results, the conductivity values of the pre-sheared samples were lower than those of the quiescent samples (
By applying shear to PP/CNT2 for 2 and 5 s, the electrical conductivity decreased from 5.75 × 10−7 S/cm to 4.31 × 10−8 S/cm and 4.73 × 10−9 S/cm, respectively. That is, by applying shear, in the mono-filler nanocomposite, the conductivity decreased by 13 and 120 times, respectively and reached 4.73 × 10−9 S/cm after 5 s of shearing. Also, in PP/CNT1/CB4, by applying shear for 2 and 5 s, the electrical conductivity decreased from 5.17 × 10−7 to 3.9 × 10−8 and 2.64 × 10−9, respectively. Namely, by applying shear, in the bi-filler nanocomposite, the conductivity decreased by 13 and 200 times, respectively and reached 2.64 × 10−9 S/cm after 5 s of shearing. Although the electrical conductivity of the nanocomposites after shearing is below the conductivity range required for applications such as supercapacitors and electromagnetic interference (EMI) shielding, it should be paid attention that in the present research, our goal was only to understand whether mono-filler PP/CNT and bi-filler PP/CNT/CB nanocomposites respond differently to shear. It was found that the response of the nanocomposites to shear is completely dependent on their percolation threshold. Reaching the percolation threshold reduces the sensitivity of the nanocomposites to shear. Another noteworthy point is that the evaluation here was performed on bi-filler nanocomposites with filler content below the percolation threshold. By increasing the amount of the second filler, the electrical conductivity of the nanocomposites can be significantly improved and the conductivity loss during shearing can be prevented.
The decrease in electrical conductivity by shearing can be explained by the alignment of both fillers and polymer chains post shearing. When the shear is applied to nanocomposites, the network formed by the conductive fillers may be disrupted. This disruption can increase the distance between the fillers and even completely separate them from each other. Therefore, the contact between the conductive fillers is reduced and the possibility of charge transfer decreases. Moreover, under shearing, the polymer chains become oriented and this orientation can lead to further alignment of CNTs and further reduction of their contact.
The effect of shear on the arrangement of polymer chains and carbon fillers is schematically illustrated in Figure 5. In both mono-filler and bi-filler nanocomposites, shear causes the orientation of polymer chains and CNTs and reduces the contact between fillers, leading to decreased charge transfer.
The electrical conductivity of the nanocomposites was reduced by about one and two orders of magnitude after 5 s and 20 s shearing, respectively. However, the decrease in electrical conductivity of the bi-filler nanocomposite was slightly greater than that of the mono-filler nanocomposite. Since the bi-filler nanocomposite had a lower CNT content and a weaker conductive network. Thus, the CNT network was easily affected by shear and the CNTs were oriented. Besides, in this sample, the amount of CB was not sufficient to form a percolated network and contribute significantly to the electrical conductivity, and the CNT was the main component determining the electrical conductivity.
To determine the electrical percolation threshold, the electrical conductivity of CNT and CB- loaded nanocomposites with increasing nanoparticle content was measured. According to the obtained results (Figure 12(a)), the CNT and CB percolation was found to be 1.75 wt% and 17.5 wt%, respectively. The critical exponent of the percolation model was estimated using equation (6): Percolation threshold of PP/CNT and PP/CB nanocomposites (a). Estimation of critical exponent of PP/CNT and PP/CB nanocomposites (b).
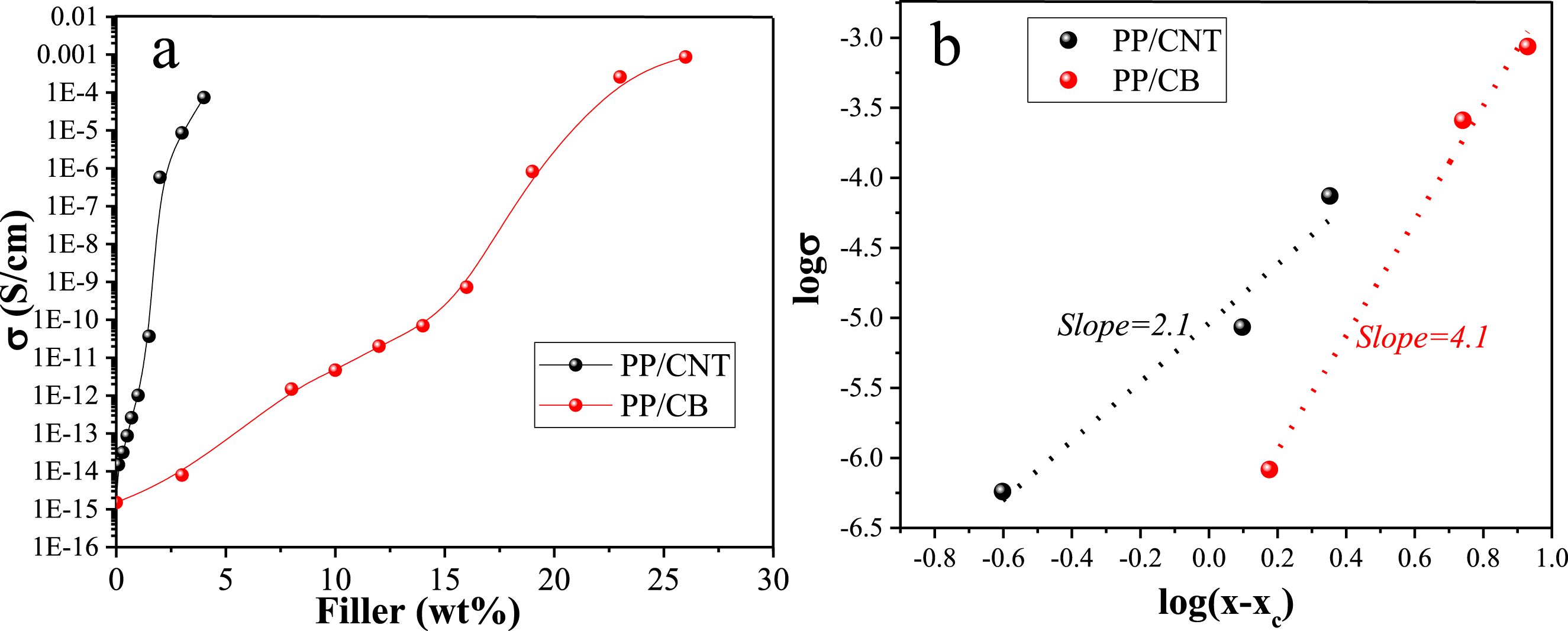
The critical exponent for PP/CNT and PP/CB nanocomposites was 2.1 and 4.1, respectively (Figure 12(b)). A critical exponent higher than two indicates that the conductivity is mediated by the electron tunneling rather than the direct contact of the conductive fillers.
Moreover, to assess whether the addition of higher CB content could have a synergistic effect on the electrical conductivity of CNT-loaded nanocomposites, the changes in electrical conductivity of bi-filler nanocomposites with CNT contents were evaluated (Figure 13). The obtained curve revealed that CNT percolation threshold in bi-filler nanocomposite was 0.85 wt%, corroborating the positive role of the second filler in reducing the amount of CNT required for the nanocomposite to became conductive. However, this synergistic effect was feasible when a large amount of CB was added to the nanocomposite. Percolation threshold of PP/CNT/CB nanocomposites.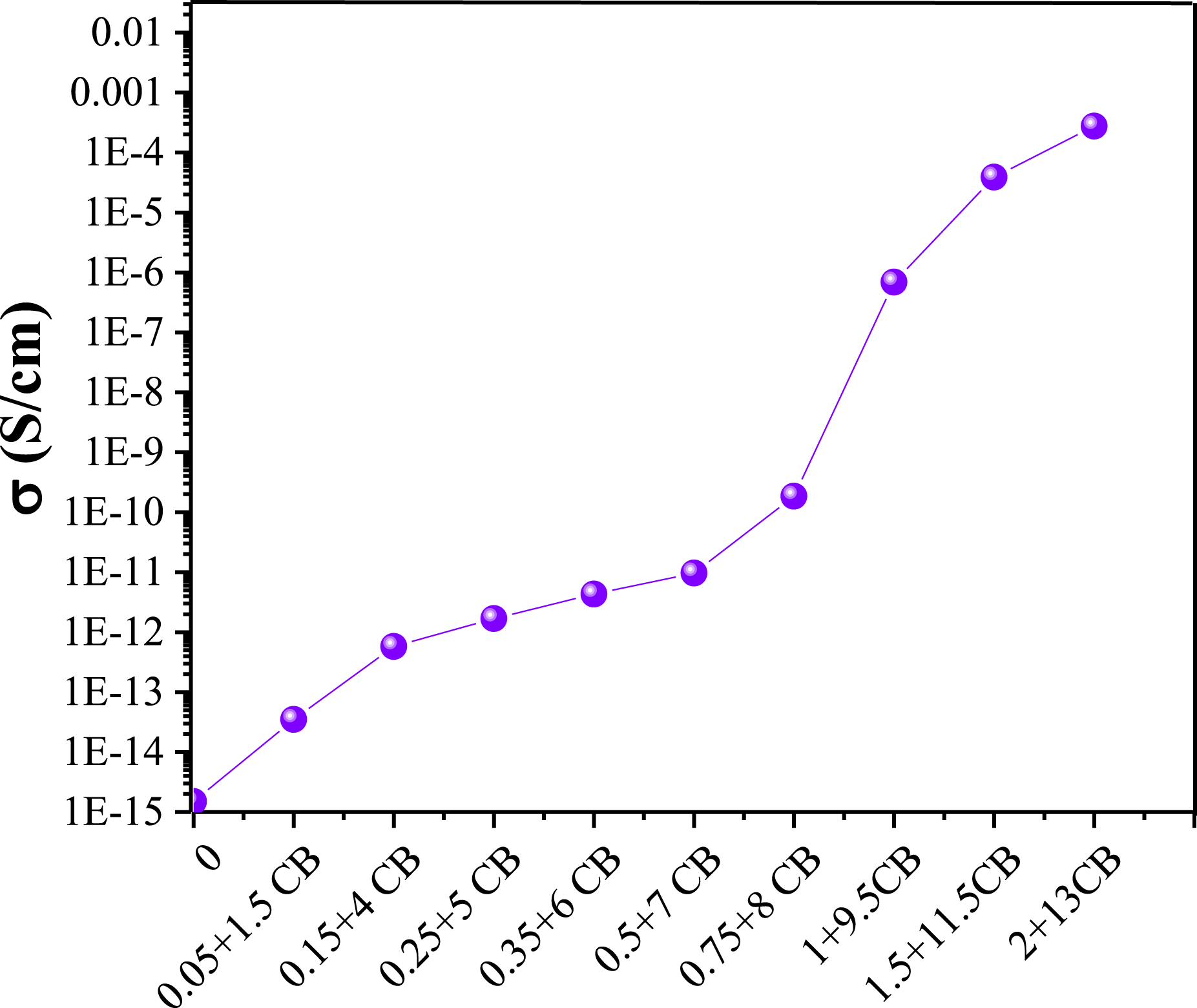
CNT is a 1D nanofiller that can provide a significant electrical conductivity along its length, while CB is a 0D nanofiller that is interspersed between CNTs and helps to form a 3D network and provide electrical conductivity throughout the nanocomposite structure. CBs can improve CNT dispersion and prevent CNT agglomeration, leading to sooner formation of a percolated network. Besides, the presence of CB reduces the distance between the conductive nanofillers and polymer matrix, increasing the possibility of electron tunneling. Indeed, the presence of CB not only can improve the contact conduction mechanism, but also can enhance the tunneling conduction mechanism. In the first mechanism, CB increases the possibility of physical contact between the conductive fillers, and in the second mechanism, CB reduces the gap between the conductive filler and the non-conductive polymer layer.
In the following, the effect of aging on the electrical conductivity and non-isothermal crystallization of the bi-filler nanocomposites was studied. For, this purpose, the samples were kept at 120 °C for 120 h and then their electrical conductivity was measured by an electrometer. In addition, the non-isothermal crystallization of the aged samples was studied by a rheometer.
The changes in storage modulus with temperature for bi-filler nanocomposites before and after aging are shown in Figure 14(a). Besides, the Tc of nanocomposites are represented in Figure 14(b). As can be seen, aging for 120 h at 120 °C increased the crystallization temperature of nanocomposites. The Tc of PP/CNT0.05/CB1.5, PP/CNT1/CB4 and PP/CNT2/CB5 was 121.03, 125.79 and 128.22 °C, respectively. After aging, their Tc increased to 124.94, 126.97 and 129.92 °C, respectively. The increase in crystallization temperature with aging can be elucidated by the rearrangement of polymer chains and the decrease in the free volume of the polymer. The increase in Tc was statistically significant only for PP/CNT0.05/CB1.5 ( Non-isothermal crystallization (a), crystallization temperature (b), and electrical conductivity (c) of bi-filler nanocomposites before and after aging.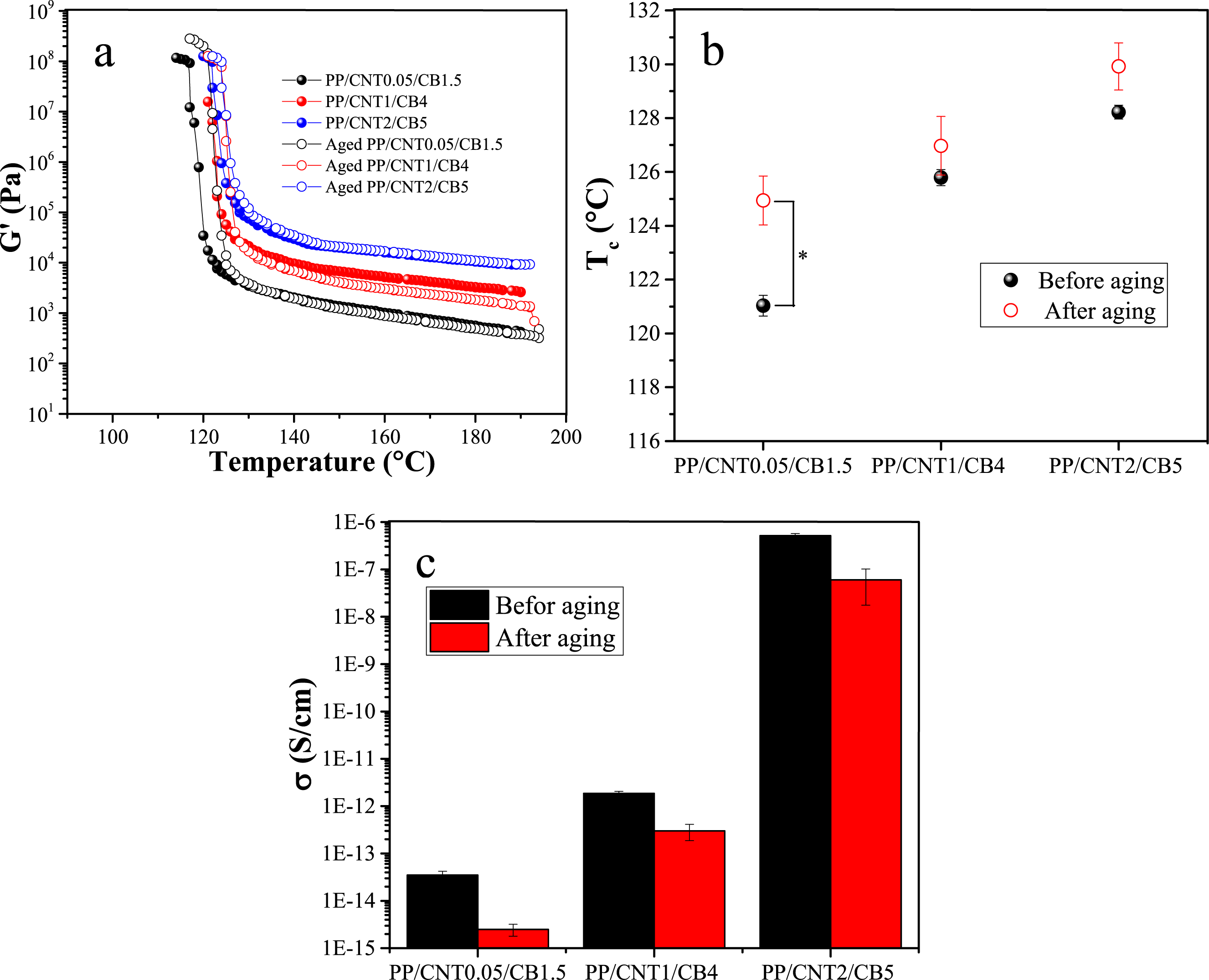
The changes in electrical conductivity of bi-filler nanocomposites before and after aging are shown in Figure 14(c). According to the obtained results, aging caused a decrease in the electrical conductivity of nanocomposites. The electrical conductivity of PP/CNT0.05/CB1.5, PP/CNT1/CB4 and PP/CNT2/CB5 was 3.5 × 10−14, 1.87 × 10−12 and 5.17 × 10−7 S/cm, respectively. After aging, their conductivity decreased to 2.5 × 10−15, 3.0 × 10−13 and 6.0 × 10−8 S/cm, respectively. The decrease in electrical conductivity with aging can be explained by changes in the polymer arrangement, nanofiller properties, and polymer-filler interface. Thermal degradation can cause the scission and crosslinking of PP chains and disrupt the connectivity of the conductive fillers. Moreover, as a result of polymer scission or crosslinking, the polymer-filler interface may be damaged, leading to less efficient charge transfer.
Different properties of the bi-filler nanocomposites versus mono-filler nanocomposites with the same or half filler content.

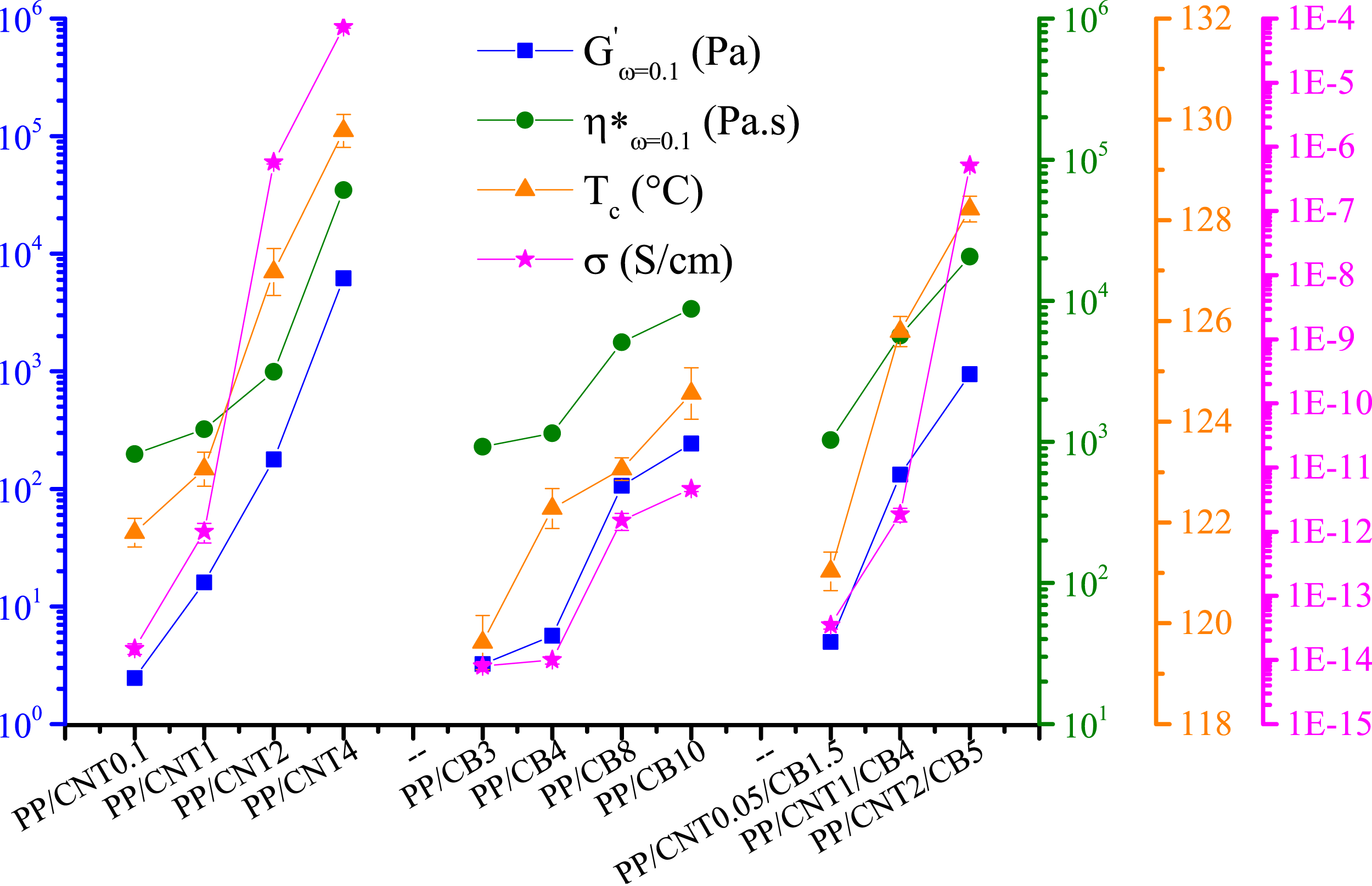
Illustration of various properties of mono-filler and bi-filler nanocomposites including storage modulus at a frequency of 0.1 rad/s, complex viscosity at a frequency of 0.1 rad/s, crystallization temperature and electrical conductivity.
However, it has to be noted that for the practical application of these nanocomposites in industry, the evaluation of their mechanical properties, thermal stability and environmental resistance is necessary. PP is a well-known polymer with many applications in various industries. PP has a tensile strength in the range of 31-38 MPa, elastic modulus in the range 1-1.5 GPa, and impact strength in the range of 0.8-2.5 kJ/m2. 52 Moreover, its heat deflection temperature (HDT) reaches 100 °C and it can be safely used up to this temperature. 45 It has been documented that the addition of nanofillers like CB and CNT can further improve the mechanical properties and thermal stability of PP.53–55 Moreover, PP is highly resistant to moisture and chemical components, however, it is susceptible to oxidation and UV degradation. It has been reported that adding nanofillers can improve PP resistance to oxidation and UV irradiation by providing barrier effect and quenching free radicals. 56 The simultaneous presence of two fillers can change the extent of their influence on the mechanical, thermal and environmental stability of PP. Therefore, it will be essential to evaluate the effect of the simultaneous presence of CNT and CB on these properties. The main goal in our future work is to fully evaluate the mechanical properties, thermal stability and environmental resistance of PP/CNT/CB nanocomposites to achieve a composition that can be used in industries needing conductive PP nanocomposites.
Moreover, it should be mentioned that in real-world applications, PP/CNT/CB nanocomposites are frequently exposed to thermal, mechanical, electrical and other environmental damages. Very high or very low temperature, high humidity environment, cyclic loading and electric and electromagnetic fields can change the viscoelastic, electrical and functional properties of PP/CNT/CB nanocomposites. Very low or very high temperatures can rearrange the polymer chains and break the nanofiller network, leading to a decrease in the elasticity and electrical conductivity of the nanocomposites. High humidity causes water molecules to penetrate the polymer structure and weaken the polymer-CNT interface, leading to lower load and charge transfers. Cyclic loading can cause the gradual separation of CNTs from the polymer network and create structural defects, significantly changing the viscoelastic and electrical properties of the nanocomposites. Finally, exposure to electric and electromagnetic fields for a long time can damage the nanofiller structure and give rise to inferior electrical and mechanical properties. Therefore, the structure and properties of the PP/CNT/CB nanocomposites should be optimized according to the requirements of each specific application.
It is worth mentioning that although in the present research, the rheological and electrical properties of bi-filler PP/CNT/CB nanocomposites under quiescent and shear conditions have been fully evaluated, the influence of aging on these properties has not been fully studied. Since, aging can drastically change the structure and properties of polymeric nanocomposites, it is essential to evaluate the changes in nanocomposite properties with aging. Aging of PP/CNT/CB nanocomposites can occur as a result of different parameters including mechanical stress, heat, moisture, oxygen and other oxidizing agents, electric and magnetic files, and ionizing or non-ionizing radiations. 57 Therefore, for practical applications, it is essential to evaluate the effect of these parameters on the physicochemical properties of PP/CNT/CB nanocomposites.
Moreover, it should be mentioned that in the present research, the effect of shear on the rheological, electrical and crystallization properties of nanocomposites with polypropylene matrix has been investigated. Changing the polymer matrix from nonpolar to polar, such as polyesters and polyamides, can strongly change the effect of shear on the properties of nanocomposites. This is because nanofillers disperse better and interact more with polar matrices than nonpolar matrices. Therefore, the nanocomposite becomes more resistant to shear and its properties change less. And even if the properties change, they are quickly recovered. The effect of shear on the crystallization and electrical properties of nanocomposites with a polar matrix will be evaluated in our future work.
Conclusion
Bi-filler nanocomposites can overcome the common limitations of CNT-loaded nanocomposites, namely the CNT expensiveness and poor dispersion in polymer matrix. With this in mind, in the present research, the bi-filler nanocomposites were prepared by adding carbon black as a second filler to PP/CNT nanocomposites. The rheological and electrical properties as well as the crystallization behavior of mono-filler and bi-filler nanocomposites under quiescent and shear conditions were fully evaluated. The results of rheological tests revealed that the addition of the second filler facilitated the formation of a 3D network of nanoparticles and increased the storage modulus and complex viscosity. The results of crystallization assay confirmed that adding CB increased the crystallization temperature and rate, and applying shear further augmented these effects. The results of conductivity measurement corroborated that the addition of the second filler at high content improved the electrical conductivity of the nanocomposite, while the use of shear decreased the electrical conductivity. To sum up, in the comparison between mono-filler and bi-filler nanocomposites with the equivalent filler content, the bi-filler nanocomposites exhibited better rheological and electrical properties as well as superior crystallization temperature and kinetics, whereas in the comparison between mono-filler and bi-filler nanocomposites with half filler content, the bi-filler nanocomposites represented the rheological, electrical and crystallization properties intermediate of CB and CNT-loaded nanocomposites.
Footnotes
Authors’ contribution
Younes Alimoradi is the main researcher who conducted the experiments and wrote the article. Elnaz Ghadyani is the assistant researcher who participated in conducting the experiments and analyzing the test results. Hossein Nazockdast is the supervisor of the research work who provided the scientific and financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be available on reasonable request.
