Abstract
Modified latex is prepared via the soap-free emulsion polymerization of ethyl acetate (VAc) and vinyl versatate (VeoVa10). DFHMA is used as modified monomers. The composite surfactants of SR-10 and ANPEO-10 are used as the composite emulsifiers. The polymerization is initiated by potassium sulfate (KPS). The structure of latex film was characterized by Fourier transform infrared spectroscopy (FTIR). The latex films were tested by thermogravimetric analysis (TGA), differential scanning calorimetry (DSC) and contact angle (CA) determinator. The particle size of the latex emulsion particle size is determined by the Zetatrac dynamic light scatter. Results show that the optimum condition of preparing the modified latex can be obtained, i.e. the amount of emulsifier is 4.0%; the mass ratio of SR-10 to ANPEO-10 is 1:1; the amount of initiator is 0.5%; the mass ratio ofVAc to VeoVa10 is 1:1; the amount of DFHMA is 5.0%. The appearance of the latex is blue and translucent with a small particle size. The conversion rate is more than 98%. Compared with the conventional latex, the hydrophobicity and heat resistance of the film have been improved. Moreover, the latex has good chemical stability and mechanical stability.
Introduction
Tertiary vinyl carbonate has a similar reactivity rate with vinyl acetate, which makes the chemical reaction of emulsion copolymerization between tertiary vinyl carbonate and vinyl acetate easier.1–5 Since tertiary vinyl carbonate can improve the water resistance of vinyl acetate emulsions, there are some reports on tertiary vinegar copolymer latex, which is prepared with the efficient and environmentally friendly emulsifiers and new functional monomers.6–11 Soap-free emulsion polymerization can be used to obtain emulsion particles with clean surface and uniform size. The way of bonding is combined with the polymer chain. This strong bonding makes the emulsifier molecules not migrate and desorb when the polymer is stored and used.12–18 As a reactive emulsifier, SR-10 not only has environmental protection properties but also has the excellent emulsification ability, which do not reduce various physical properties (water resistance, adhesiveness, weather resistance and so on) imparted to the polymer film.
Fluorine-modified polymer latex refers to the addition of fluorine-containing monomers in the emulsion polymerization stage to participate in the emulsion polymerization together with the main reaction monomer, thereby introducing fluorine into the macromolecular chain of the polymer latex, which can protect other molecules and structures on the main carbon chain from the interference of external factors.19–24 Shi et al reported that chlorotrifluoroethylene, vinyl acetate and vinyl tertiary carbonate were used as co-monomers, potassium persulfate was used as initiator, ammonium perfluorooctanoate was used as emulsifier and the deionized water was used as the reaction medium to prepare a series of stable fluoropolymer latexes with a solid content of about 33% and a latex particle size of 151–205 nm.
25
However, there are few reports on the modified VAc-VeoVa10 latex prepared by soap-free emulsion polymerization. In this work, the modified VAc-VeoVa10 latex prepared by soap-free emulsion polymerization, which dodecafluoroheptyl methacrylate (DFMA) is used as a modified monomer. The emphasis was put in the present work on factors which have on the properties of the resultant latex and the soap free emulsion polymerization. The specific synthetic pathway is shown in Scheme 1. Synthetic pathway of modified latex.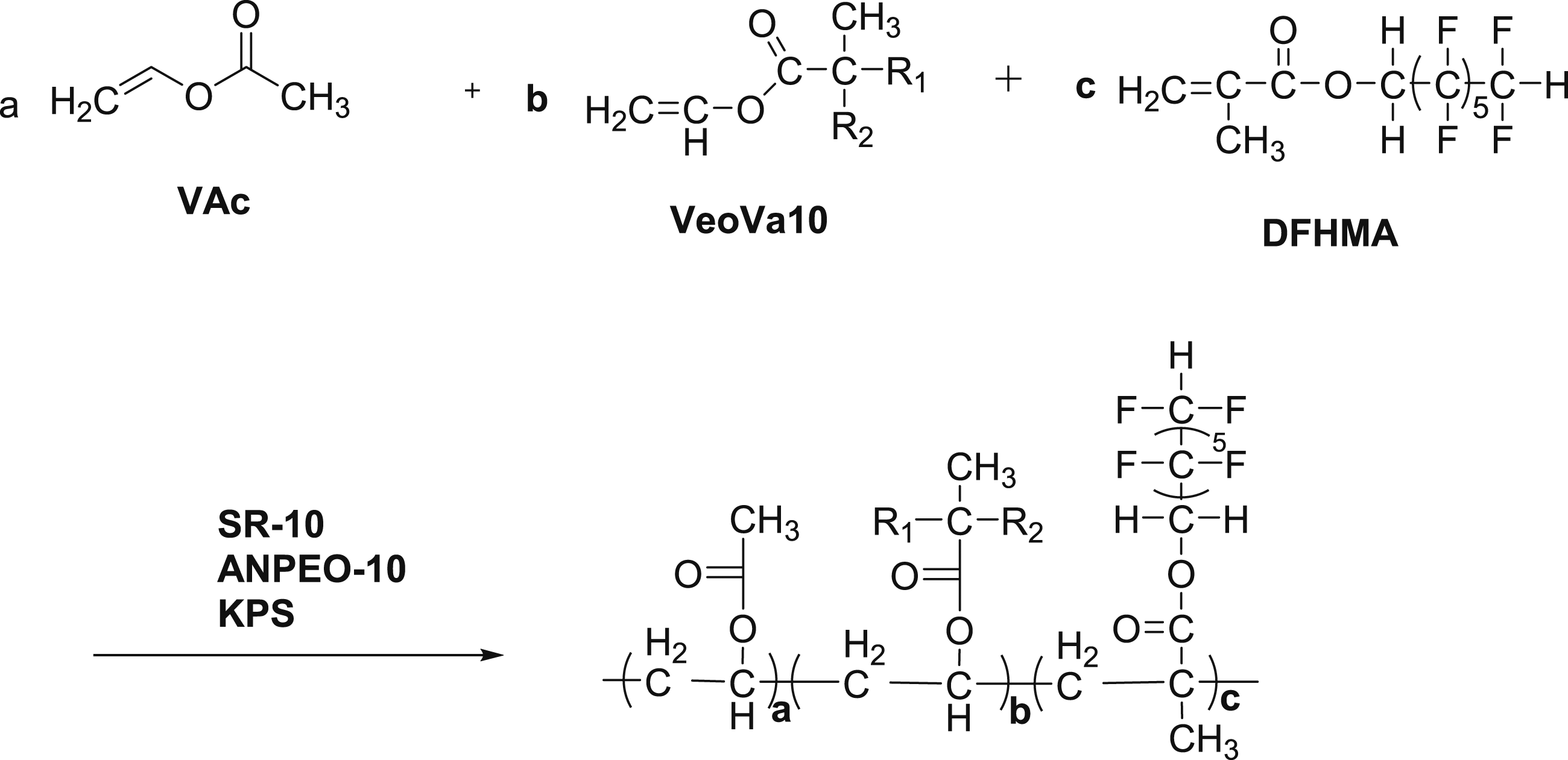
Experimental
Materials
VAc and Veova10, which were chemically pure, were obtained from Aladdin reagents and Guangdong Wengjiang Reagent Co. Ltd (China), respectively. DFMA was purchased from Aladdin reagent. SR-10 and ANPEO-10, which were the industrial grade, were from Shenzhen Xiangrun New Materials Co. Ltd and Shanghai Macin Biochemical Technology Co. Ltd (China), respectively. KPS, which was chemically pure, was supplied by Shanghai United Company (China). The deionized water was distilled in our laboratory.
Preparation of modified latex
Recipe of preparing modified latex.

Characterizations
The molecular structure of the polymer was characterized by Fourier transform infrared spectroscopy (FTIR, Thermo-Nicolet-infrared AVATAR370,USA). The thermal stability of the polymer was performed from 40 to 60°C with a heating rate of 10°C/min under a nitrogen atmosphere with a thermogravimetric analyzer (TGA, Q50, and USA). The glass transition temperature of the polymer was carried out by differential scanning calorimetry (DSC, Q100, and USA). The contact angle (CA) of latex film was detected by the solid drop method on the data physical contact angle instrument (oca-20, Germany). The average particle size of the latex was tested by NanoBrook Omni (Brookhaven Instruments Corporation, USA). The water absorption was calculated according to formula (1):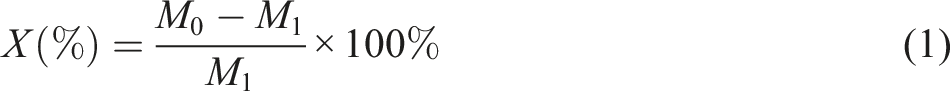
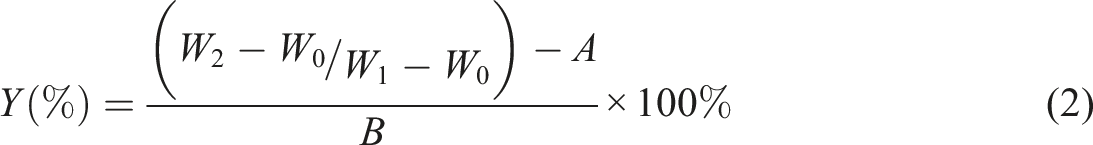

Results and discussion
FTIR and DSC of latex film
FTIR of the latex film is shown in Figure 1. 2963.5 cm−1and 2875.9 cm−1 are the characteristic stretching peaks of C-H (CH3, CH2). 1737.4 cm−1 is the stretching vibration of C=O. The absorption peak at 1463.8 cm−1 indicates that it is a characteristic peak of CH3-CO-O-. The characteristic peak at 1370.6 cm−1 is the C-C bending vibration peak. 1128.1 cm−1 is the symmetrical absorption peak of C-O-C. The characteristic peak at 1020.3 cm−1 is C-O vibration peak and the characteristic peak at 944.9 cm−1 is the absorption peak of the C-F absorbing peak. Furthermore, there is no stretching vibration in the range of C=C of 1500-1700 cm−1, which indicates that all the monomers are involved in the emulsion polymerization. FTIR of latex film.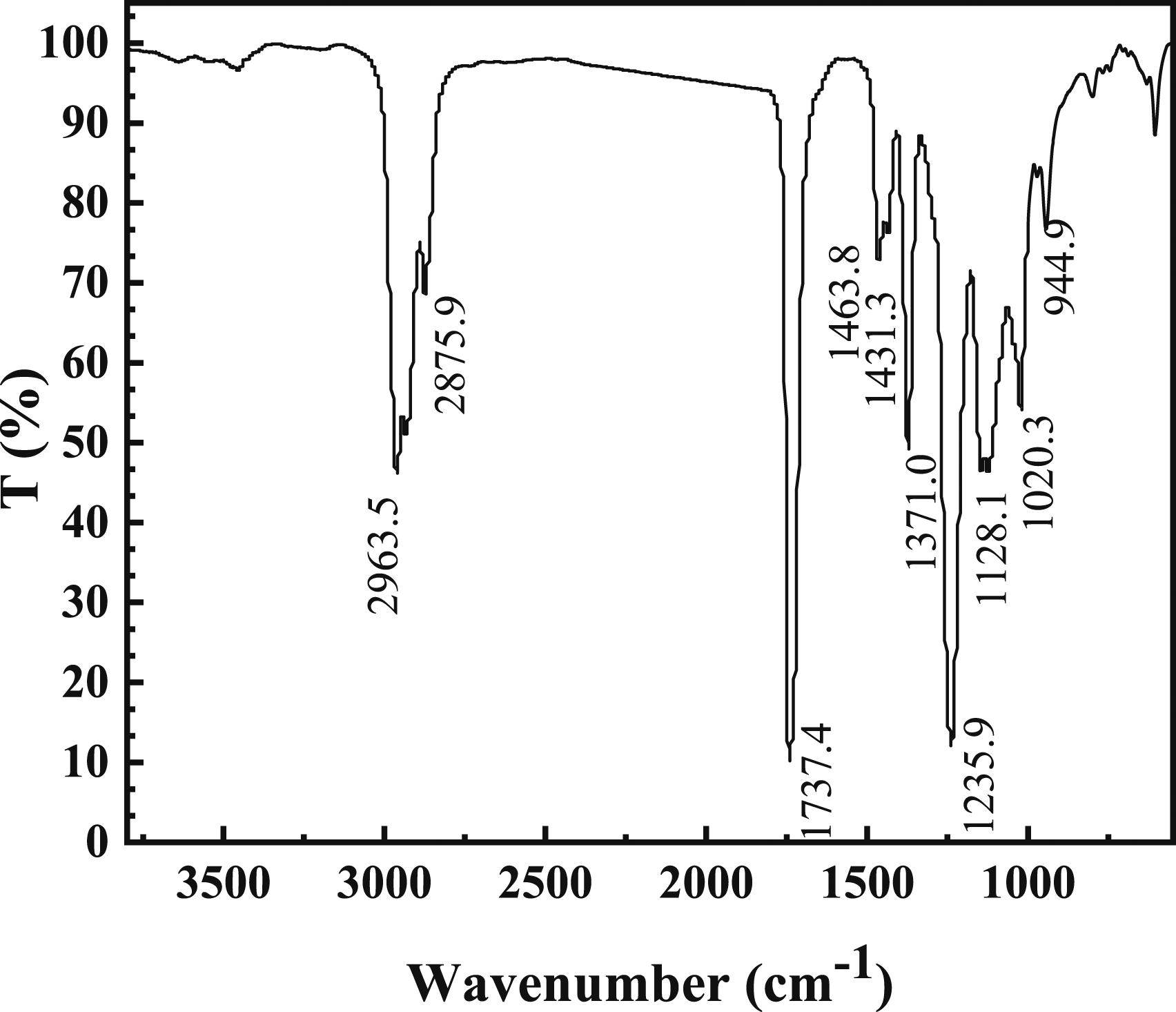
Tg is the minimum temperature that the molecular chain segment in the polymer can move. Through the morphological change trend of polymer at different temperatures, the state of polymer within different temperature range is obtained, and the heat analysis curve is drawn based on the measured data. This temperature is the critical temperature of polymer from glass state to high-elastic state, and it is also the minimum temperature for free movement of non-crystal polymer molecular segments. The DSC curves of the resultant latex film were shown in Figure 2. From Figure 2, it can be found that the Tg of the polymer latex film is 8.79°C, which is different from those of VAc homopolymer (28–40°C) and VeoVa10 homopolymer (−3°C).
26
In addition, only one Tg is detected, which means that all monomers are introduced into the polymerization and the latex is the random copolymer. DSC of latex film.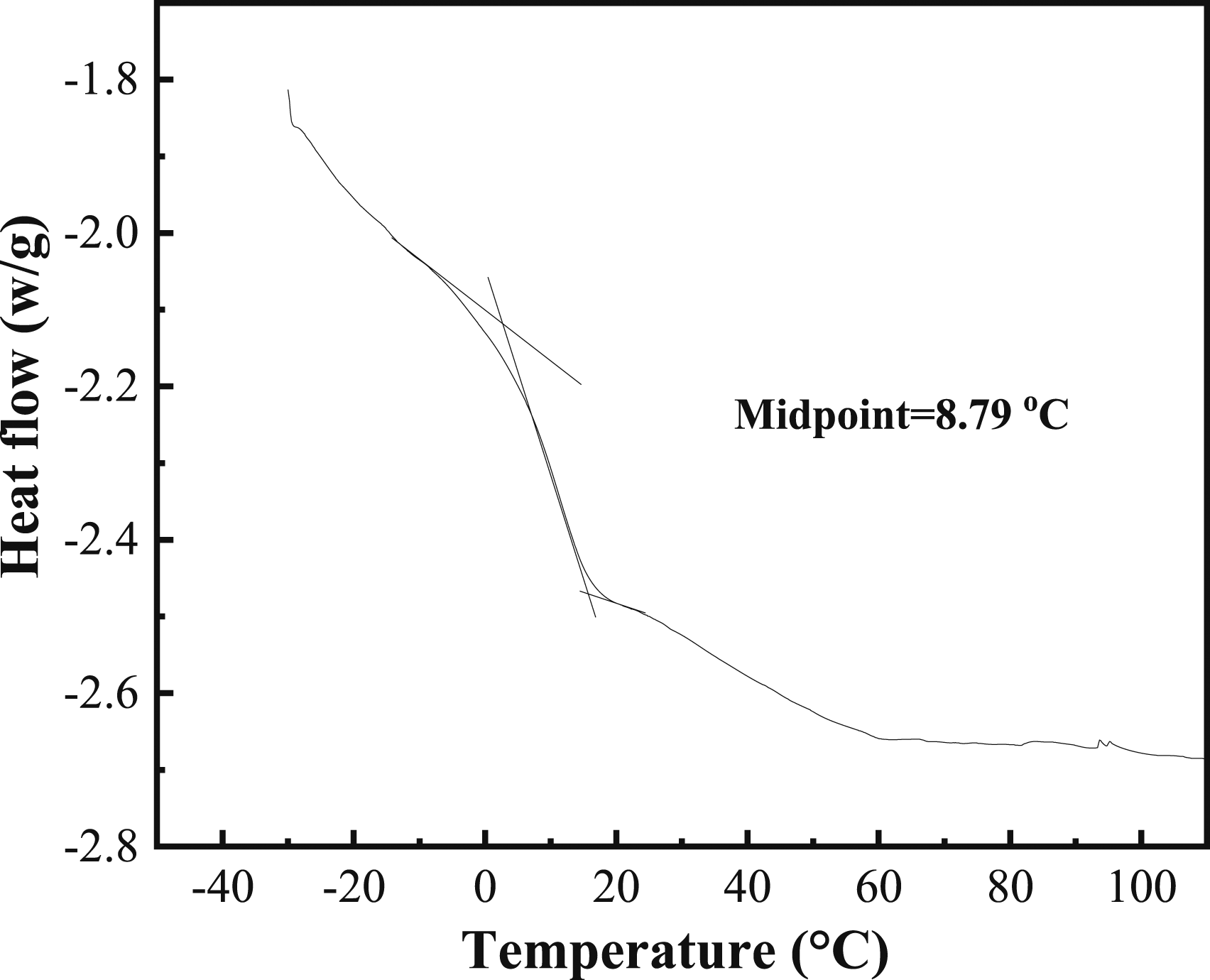
Effect of emulsifier on emulsion polymerization and latex
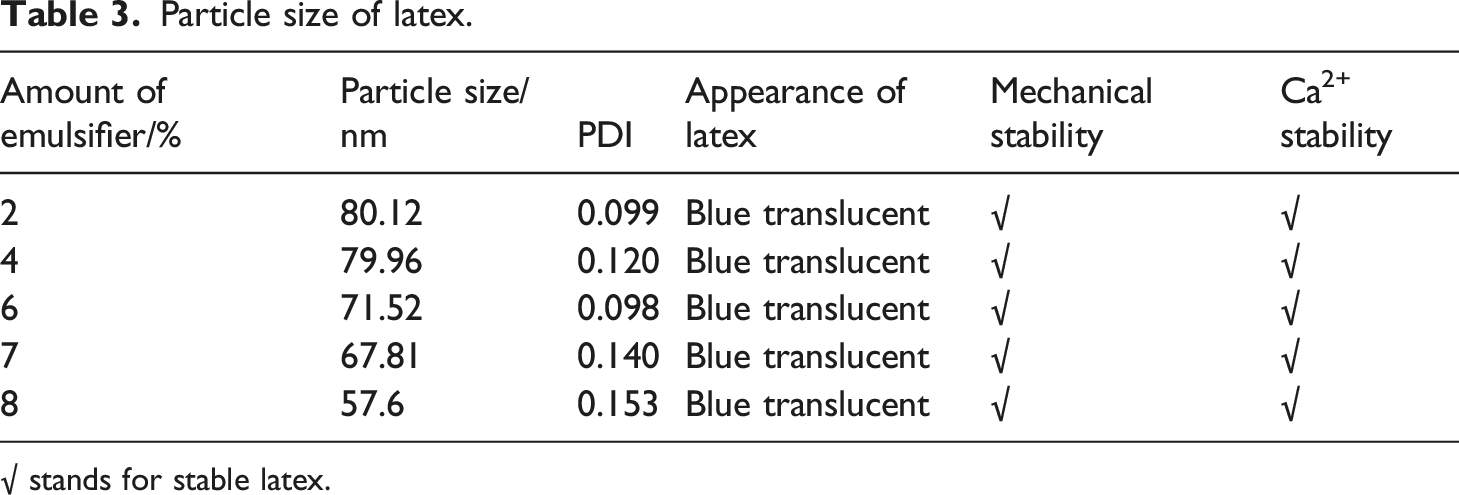
In this study, the emulsification system for preparing latex is based on ANPEO-10 and SR-10. The influence of the emulsified system on emulsion polymerization and latex is studied. From Figure 3, it can be found that the amount of composite emulsifier has an effect on the conversion percentage and coagulation percentage. The latex is translucent and blue, which indicates that the composite emulsifier has a good emulsified capacity. With the increased amount of the emulsifier, the monomer conversion rate is increased first and then decreased. This may be caused by the fact that the increased amount of the emulsifier content will form more micelles, which provide more reaction centers. Thus, the conversion rate of mixed monomers continues to increase. When the amount of emulsifier is more than 4%, the coagulation percentage increased first and then decreased slightly but the monomer conversion rate is decreased. When the amount of emulsifier reaches a certain level, the emulsifier will cover the latex particles, which results in low monomer conversion and more coagulation. Thus, the optimum amount of emulsifier is 4%. Furthermore, influences of different mass ratios of emulsifiers on the conversion rate and coagulation percentage is presented in Table 2. Table 2 shows that the monomer conversion rate is the maximum and the gel rate is correspondingly lower when the mass ratio of SR-10 to ANPEO-10 is 1:1. The average particle size and particle size distribution of the latex are given in Table 3. The results indicate that the particle size range of the emulsion is between 57.6 nm and 80.12 nm. As the amount of emulsifier is increased, the average particle size of the latex is decreased. This is mainly caused by the fact that the reaction site of emulsion polymerization is micelles. When the amount of emulsifier is increased, more micelles are formed, which is equivalent to increasing the reaction site. The number of monomers, which takes part in the reaction in the system, is certain. Thus, the number of monomers that can be obtained for each micelle is reduced. Hence, the length of the polymer chain becomes shorter and the particle size of the polymer becomes smaller. The PDI value indicates that the particle size distribution is correspondingly uniform. Effect of amount of emulsifier on conversion percentage and coagulation percentage. Influence of mass ratio of emulsifier on emulsion polymerization. Particle size of latex. √ stands for stable latex.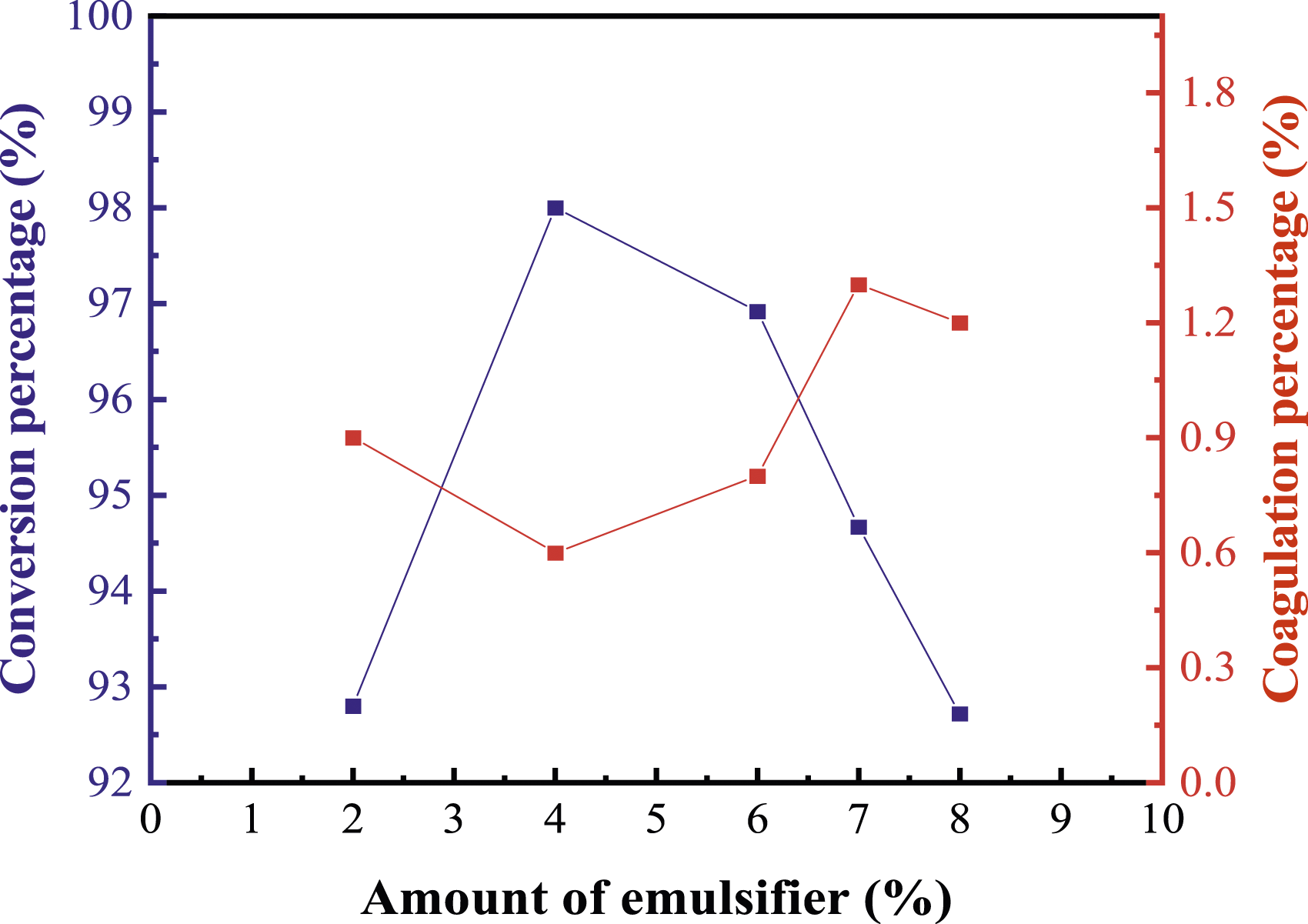

Influence of amount of initiator on conversion percentage and coagulation percentage
The influence of initiator amount on conversion rate and coagulation percentage is given in Figure 4. In the early stage of polymerization, the amount of free radicals is few, the polymerization reaction is slow and the monomer conversion is low. With the increased amount of KPS, the conversion rate rises and the gel rate declines. When the amount of KPS is more than 0.5%, the conversion rate is gradually decreased and the coagulation rate is gently increased. The reasonable explanation is that when the initiator concentration is small, the number of free radicals entering the colloidal particles is relatively reduced. The monomer polymerization rate is gentle. The reaction is incomplete and the coagulation is large. With the continuous addition of the initiator, the number of active free radicals dissolved in water rises. The probability of entering the micelles for initiation improves, thus causing the conversion rate to improve. Too many initiators will lead to too many active centers and release more reaction heat in the system. The heat of reaction is not easy to eliminate, which makes the polymerization system be difficult to control and the stability of the emulsion decreases. At the same time, a small number of monomers outside the micelles will also initiate polymerization, which will easily collide with each other to produce coagulations and the conversion rate will also decrease. Furthermore, KPS is also an electrolyte in water solution. With the increase in electrolyte concentration, the thickness of double layer of emulsion particles decreases due to the salt effect of electrolyte, and the cohesion rate of the system increases. Therefore, the optimal amount of initiator in this study is 0.5%. Effect of amount of initiator on conversion percentage and coagulation percentage.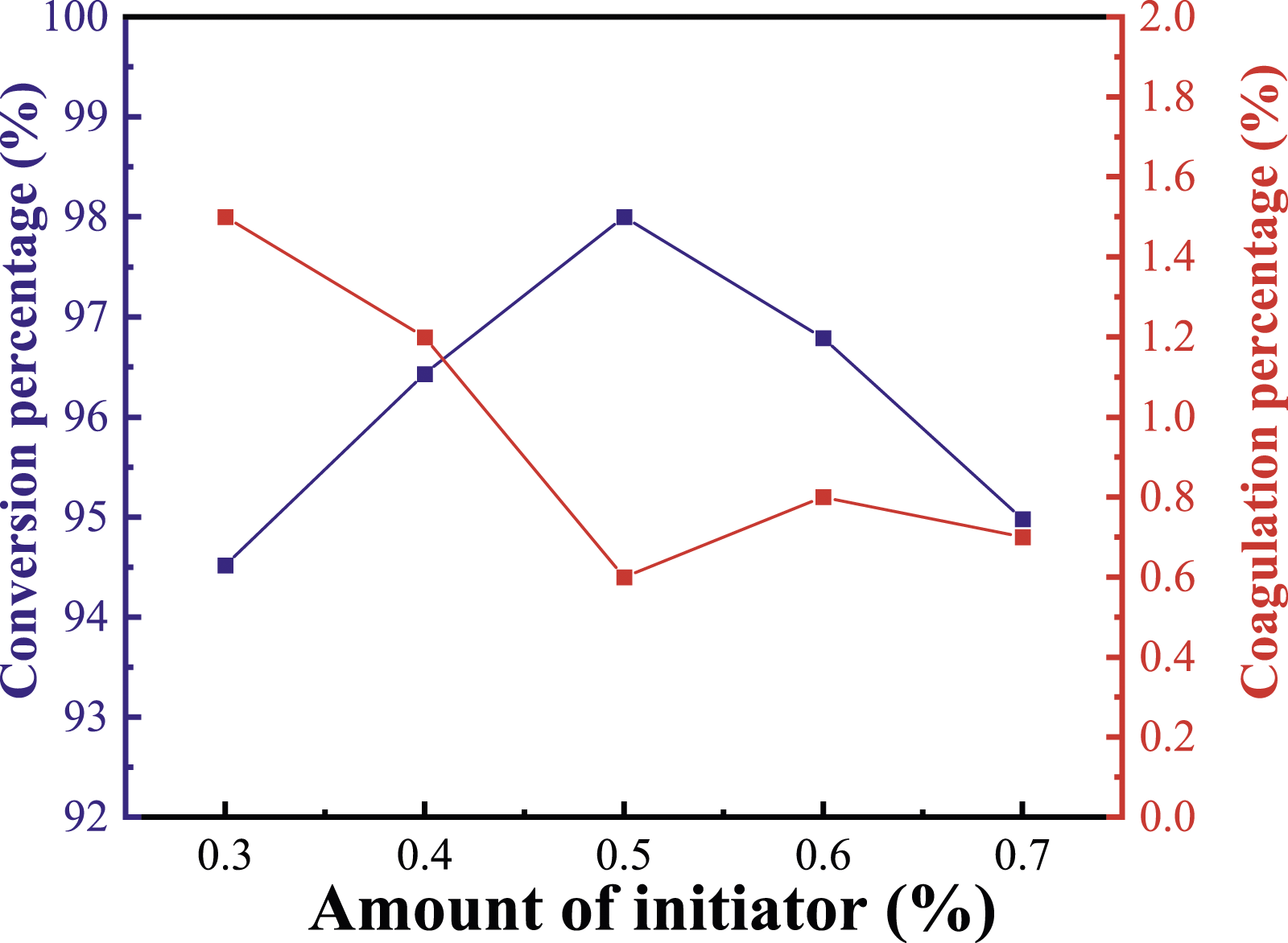
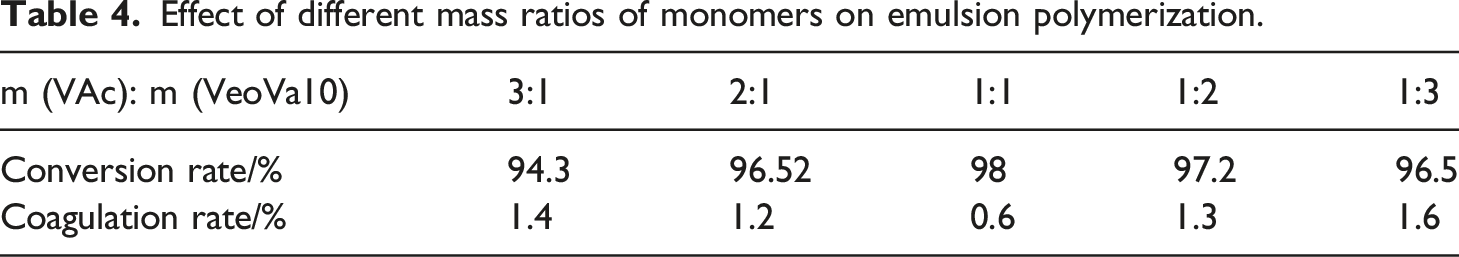
Determination of mass ratios of monomer
Effect of different mass ratios of monomers on emulsion polymerization.
Contact angle of film
The contact angle of the liquid on the solid surface is an important parameter to evaluate the surface of hydrophobicity. The larger the contact angle is and the greater the hydrophobicity of the film is. The effect of the amount of DFMA on the contact angle of film is presented in Figure 5. Figure 5 shows that the contact angle is increased with the increased amount of DFMA when it is less than 5%. With the addition of DFMA, the contact angle of the latex film is increased, which is attributed to the introduction of fluoride monomer, the fluorinated groups of acrylate preferentially migrate and accumulate on the surface during the course of film forming. Thus, the surface energy of film is decreased, which leads to the increase of the hydrophobicity.
27
However, the contact angle is decreased with the increased amount of DFMA when it is more than 5%. This may be explained by the following fact. The stability of the latex will be destroyed by the excessive DFMA, which leads to less fluorine in the polymer. Fewer fluorinated groups of acrylate preferentially migrate and accumulate on the surface during the course of film forming, thus causing the contact angle to be decreased. Influence of amount of functional monomer on contact angle of film.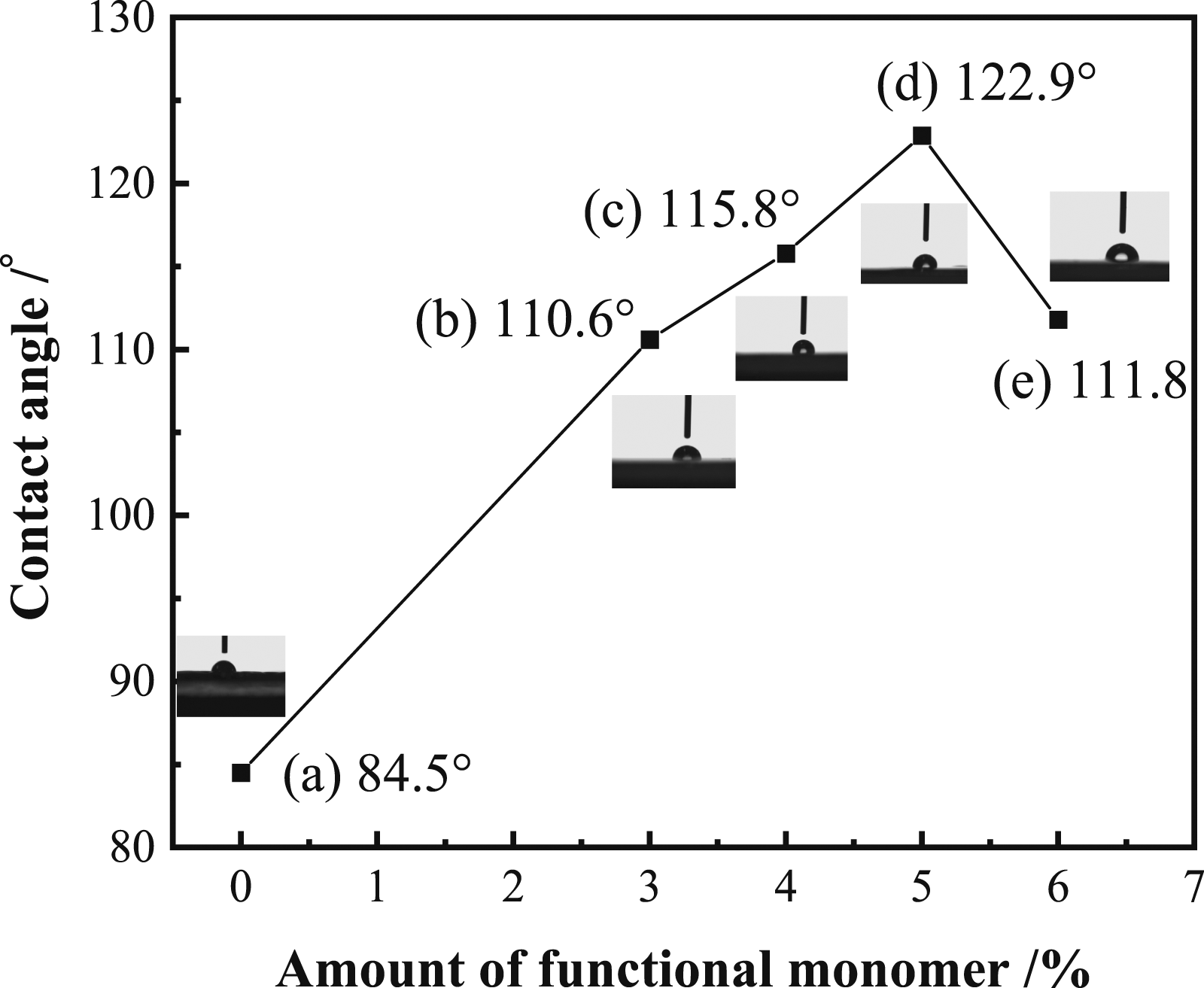
Thermal properties of latex film
The comparison of the thermal properties between the conventional latex and modified latex is given in the Figure 6. The thermal decomposition of the modified polymer latex film begins at 302.25 C, which is higher than that of the latex film without fluorine (294.81 C), and the heat resistance is improved. It demonstrates that when the fluorinated monomer is polymerized to the macromolecular chain, the thermal decomposition temperature of the latex film is improved owing to the existence of the C-F bond with high bond energy. TGA of latex film (A: conventional latex without fluorine; B: modified latex).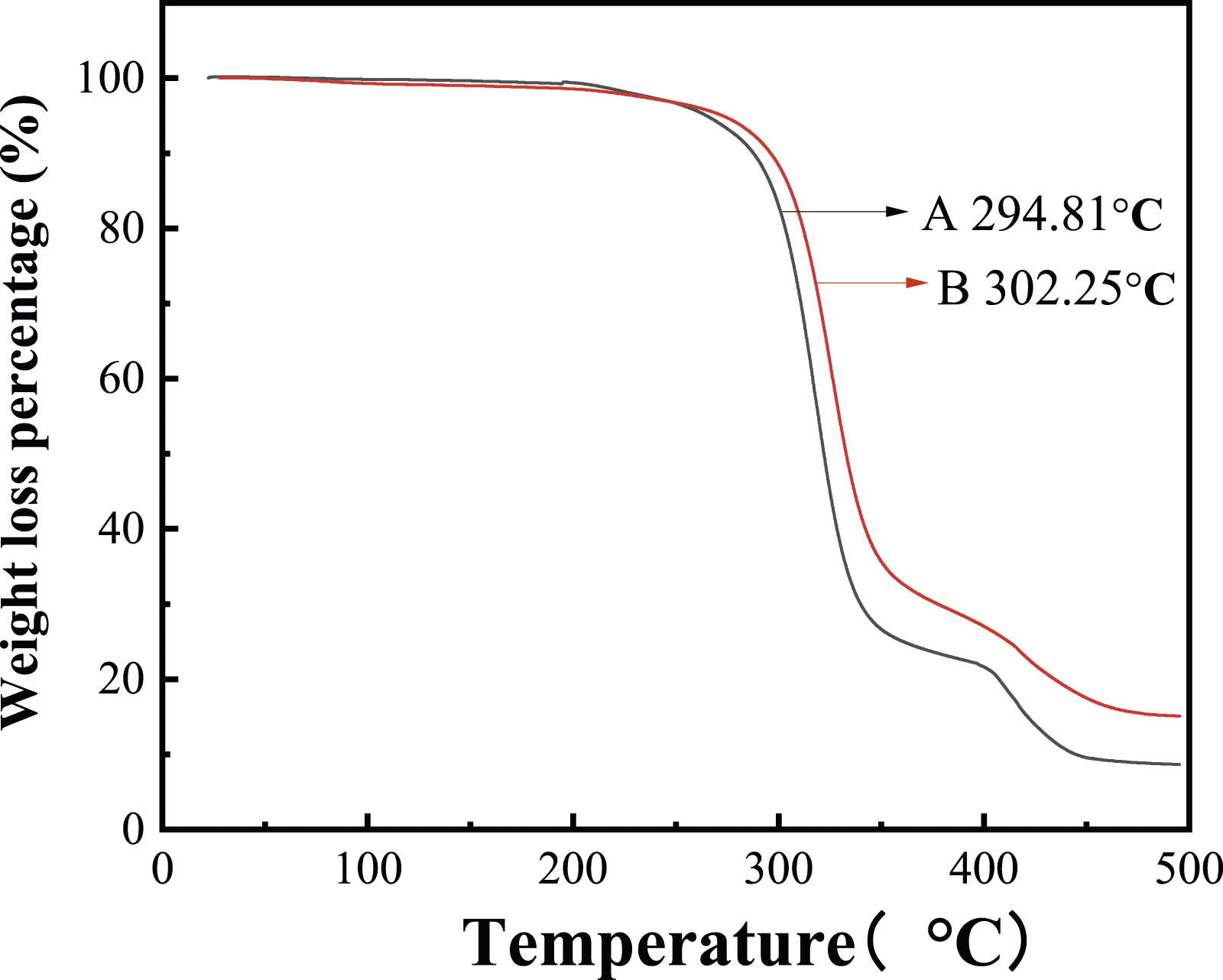
Conclusions
The modified latex is synthesized by soap free emulsion polymerization, which VAc and VeoVa10 are used as the main monomers and DFMA is used as modified monomers. The composite surfactants of SR-10 and ANPEO-10 are used as the composite emulsifiers and KPS was used as the initiator. FTIR confirms that DFMA has been successfully introduced into the modified latex. The optimum synthetic conditions are as follows: the amount of emulsifier is 4.0%; the mass ratio of SR-10 to ANPEO-10 is 1:1; the amount of initiator is 0.5%; the mass ratio of VAc to VeoVa10 is 1:1; the amount of DFMA is 5.0%. The appearance of the latex is blue and translucent with a small particle size. The conversion rate is more than 98%. Compared with the conventional latex, the hydrophobicity and heat resistance of the film have been improved. Moreover, the latex has good chemical stability and mechanical stability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
