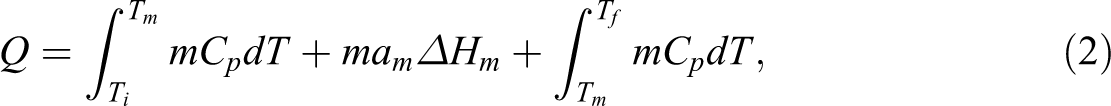Abstract
Phase change materials are utilized in heat storage applications, as they have high latent heat during the phase transition. In addition, high thermal conductivity is required from the heat storage materials to achieve high energy efficiency. In this study, the effect of carbon nanotubes (CNTs) and nanodiamonds (NDs) on the thermal conductivity and the heat storage capacity of the paraffin–natural rubber composites was studied. It was found that the CNTs work better than NDs in such composites. They increase thermal conductivity significantly and thus improve the heat transfer rate of the composite. They were also found to prevent the migration of paraffin out of the rubber during the vulcanization process, which increases the lifetime of the composite.
Keywords
Introduction
Increased environmental awareness has roused researchers to search for the methods to decrease energy consumption and hence, the use of fossil fuels, which still are the main sources of energy. An efficient way to reduce the energy consumption is to store thermal energy. The so-called phase change materials (PCMs) have been found to be useful materials in thermal energy storage applications, as they are able to absorb or release high amount of thermal energy, that is, they have a high latent heat. PCMs store energy during the first-order phase transitions (melting and crystallization processes), while their temperature remains constant and thus they are able to control the temperature of environment. 1 At the melting temperature region, the amount of latent heat is calculated by the following equation. 2
where Q 1 is the amount of stored latent heat, m is the mass of the PCM, am is the fraction melted, and ΔHm is the latent heat of fusion for the PCM. Outside the melting region of the PCM, the heat is stored as a sensible heat, that is, the temperature of the material increases. Then, the total amount of the stored energy can be determined by the following equation. 2
where Q is the amount of stored heat, Ti , Tm , and Tf are initial temperature, melting temperature, and final temperature of the PCM, respectively. Cp is the specific heat capacity of the material. However, the high latent heat materials rise above the sensible heat materials in terms of heat storage ability. For example, in the case of water, the heat can be stored about 80 times more as latent heat than as sensible heat. 3
Most commonly used PCMs are paraffin, fatty acids, and salt hydrates from which paraffins are preferred in polymer composites due to the high latent heat, low supercooling as well as low price, versatility, and commercial availability. 4,5 In addition, their compatibility with organic polymers is good compared to inorganic salt hydrates. As the phase change of PCMs is commonly based on the melting, they need to be shape-stabilized to prevent leaking. A common way to do this is to use a polymer matrix. Thermoplastic has been widely investigated as matrix materials 6 –9 but also elastomers have been studied, 10 –14 and for example, ethylene propylene diene rubber has been found to prevent the leakage of PCM better than polyethylene (PE). 12
A recent study showed that 50 parts per hundred rubber (phr) paraffin can be included in natural rubber (NR) compound without a loss of rubbery characteristics and thus the NR composite having ability to store thermal energy is achieved. 15 However, both paraffin and NR, like most of polymers, are thermal insulators that decreases the energy storing efficiency and response time of PCM-polymer composites. This drawback can be overcome by increasing thermal conductivity of the NR composites by incorporating thermally conducting fillers.
Several studies show the efficiency of carbon-based nanofillers in the enhancement of the thermal conductivity of pure paraffin. 16,17 Furthermore, the increment of thermal conductivity by expanded graphite (EG) in PCM-PE composites has been studied. 10,18 EG has been found to increase heat transfer rate in both melting and solidification processes significantly: over 200% enhancement in thermal conductivity was reported with 10% EG content in a paraffin-PE composite. 18 This is said to be higher than the thermal conductivity achieved with carbon nanotubes (CNTs), 19 although CNTs have the highest thermal bulk conductivity (approximately 3000 W/mK for multiwalled CNTs) among the different carbon allotropes. 20 CNTs have high surface energy, hence it tends to form agglomerates 21 especially at high filler loadings and filler dispersion remains poor. Thus, the formation of filler network, which is critical for the high thermal conductivity, is hindered.
A novel approach in the increment of thermal conductivity is the use of nanodiamonds (NDs). This carbon-based filler have not been studied in heat storage applications, although the thermal conductivity of the diamond is the same level with graphite (approximately 2000 W/mK). 20 Further, NDs have a special characteristics among the carbon-based fillers: they are dielectrics, which could be a benefit in several heat storage applications.
In the earlier studies, the incorporation of the nanofillers have been mainly done into the molten polymer. Due to that, the shear forces in the mixing process remain low and a good dispersion and distribution of nanofillers is difficult to achieve. 19 In the case of rubbers, the fillers are incorporated into solid polymer. This requires high shear forces enabling the breakage of large filler agglomerates into smaller aggregates. Thus, the real benefits of nanofillers can be achieved. It has been shown that good CNT dispersion in NR compound can be achieved by solid mixing up to 5 phr filler concentration. 21,22 However, higher filler concentrations are often required for high thermal conductivity.
In the current study, the efforts are put into the improvement of the thermal conductivity of NR composite to be used in heat storage applications. CNTs and NDs are used as thermally conducting fillers. The effect of CNTs and NDs on morphology, thermal conductivity, and heat storage ability is discussed.
Experiment
Materials
NR (standard Malaysian rubber, SMR10) was used as a matrix material and paraffin (RT64HC from Rubitherm GmbH, Germany) with a melting point of 63–65°C and a heat storage capacity of 250 kJ/kg (±7.5%) was used as PCM.
Thermal conductivity was improved by multiwall CNTs and NDs. As CNTs, Baytubes C 150P from Bayer (Germany) with a diameter of 13–16 nm and a length of 1–10 µm were used. NDs studied were carboxylated NDs (uDiamond® Vox P) from Carbodeon Ltd Oy (Finland) with a particle diameter of 2–6 nm.
Preparation of PCM-NR composites
The recipes of the rubber compounds investigated are presented in Table 1. The compound contained the above-mentioned NR, PCM, CNT, or ND and a curing package, that is, 1% oil extended sulfur from Solvay Barium Strontium GmbH (Germany) as curing agent, zinc oxide from Grillo Zinkoxid GmbH (Germany) and stearic acid from Oleon NV (Belgium) as activators, and N-cyclohexyl-2-benzothiazole sulfonamide (CBS) from Lanxess (Germany) as accelerator.
The rubber formulation used in the study and the mixing sequence of the ingredients.

CBS: N-cyclohexyl-2-benzothiazole sulfonamide; PCM: phase change material; NR: natural rubber.
The compounds were mixed in a Brabender (Germany) 350 E mixer with tangential rotors. The starting temperature was 70°C and the rotor speed was 60 r/min. The mixing sequence is presented in Table 1. The total mixing time was 13 min. The samples were vulcanized to 2 mm thick sheets in a heating press (MPD40E from Tarkmet Oy, Finland) at 160°C for their respective vulcanization time determined for each mixture separately. The vulcanization times of the PCM-NR composites were determined by Advanced Polymer Analyzer (APA 2000; Alpha Technologies, United States). The measurements were performed at 160°C for 30 min.
Characterization
The phase change temperatures and the latent heat of PCM-NR composites were studied by differential scanning calorimeter (DSC; Netzsch, Germany, DSC 204 F1) with a temperature range from 0°C to 100°C and a heating rate of 10 K/min.
The thermal conductivity of the composites was measured by laser flash analysis (LFA; Netzsch, Germany, LFA 467 HyperFlash) at 25°C. Circular samples with a diameter of 10 mm were coated with a thin layer of electrically conductive graphite to guarantee an even heat pulse. Five parallel measurements were performed and the average values were calculated.
Thermogravimetric analysis was performed for unvulcanized and vulcanized PCM-NR composites by Netzsch (Germany) TG 209 Tarsus. Measurements were conducted in nitrogen atmosphere from 25°C to 900°C with a heating rate of 10 K/min.
Pure PCMs and the fracture surfaces of the PCM-NR composites after tensile tests were studied by field emission scanning electron microscope (FESEM; Zeiss ULTRA plus, Germany). The PCMs and the fracture surfaces of the composites were attached to the aluminum SEM stub by carbon clue or tape. Prior to FESEM studies, the samples were carbon- or gold-coated to avoid sample charging during the characterization.
The Payne effect measurements were performed using Advanced Polymer Analyzer (APA 2000; Alpha Technologies) in a strain sweep mode from 0.28% to 100% at 100°C.
Results and discussion
The curing time analysis showed that the addition of CNTs accelerated the curing process of NR but NDs retarded it (Figure 1). The similar effect of CNTs was found in earlier studies. 22 This is due to the increased thermal conductivity of the PCM-NR composite. Surprisingly, NDs increase the curing time although they also improve the thermal conductivity of the composite as seen in Figure 2. Reason for that is the carboxylated surface chemistry that adsorbs accelerator as was proposed by Shanmugharaj et al. in the case of CNTs. 23 Altogether, the effect of surface chemistry is stronger than the expedited thermal conductivity.
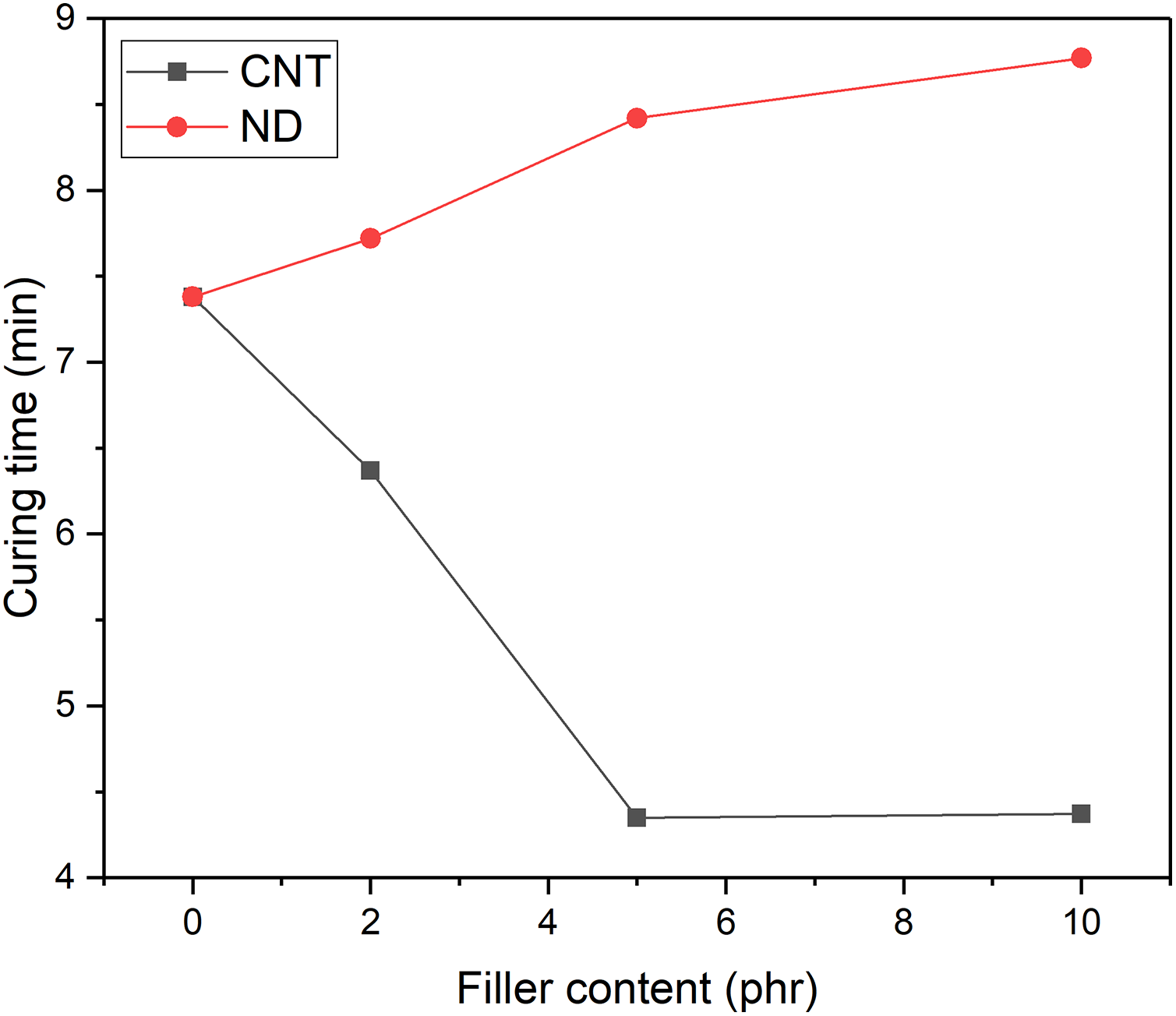
The effect of CNTs and NDs on vulcanization of rubber. CNT: carbon nanotube; ND: nanodiamond.
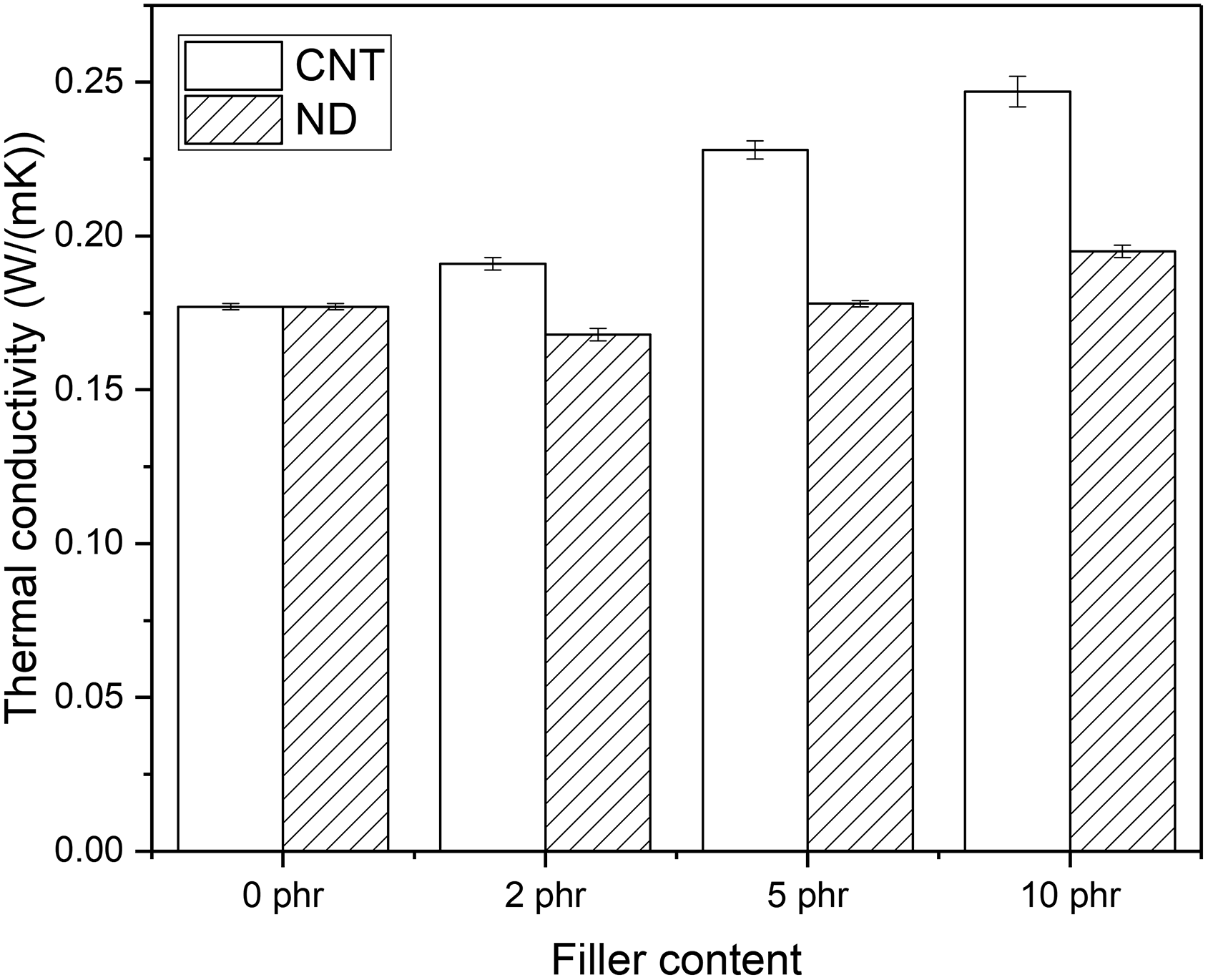
The effect of CNTs and NDs on the thermal conductivity of PCM-NR composite. CNT: carbon nanotube; ND: nanodiamond; PCM: phase change material; NR: natural rubber.
Figure 2 shows that the thermal conductivity of the NR composite increases when the CNT content increases as supposed. In the current case it means that the energy storing efficiency of the NR composites increases and it responses faster to the outer temperature. However, the increase in thermal conductivity was surprisingly low after the addition of NDs. The different effect of these fillers can be explained by the volume fraction, aspect ratio as well as the dispersion and distribution of fillers. According to the suppliers’ data sheets, the bulk densities of CNTs and NDs are 0.12–0.17 g/cm3 and 0.5 g/cm3, respectively. Thus, the same weight fraction of fillers leads to fourfold higher volume fraction of CNTs. This enables the formation of filler network at lower filler concentrations. In addition, NDs are spherical particles, which means that their aspect ratio is only one whereas the aspect ratio of CNTs can vary between 60 and 750. Therefore, NDs require higher concentration for percolation threshold, that is, to form a filler network than CNTs.
The state of dispersion and the formation of filler network was proven by the Payne effect measurements and FESEM analysis. The Payne effect is a common method to analyze filler–filler interactions in rubbers and it describes the difference in the complex or storage modulus at low strain and high strain. The Payne effect is based on the strain dependency of the filler–filler interaction. When strain amplitude increases, the filler agglomerates are diverged and the filler network breaks down, which decreases the modulus. Thus, the higher the Payne effect value, the higher the filler–filler interaction. The Payne effect is a widely used method in silica technology, where low Payne effect is considered to mean better filler dispersion. Nevertheless, in the case of nanofillers, the opposite effect has been proven by several studies. 24,25 The Payne effect increases when the filler–filler network is formed, that is, the dispersion of nanofillers is good.
The Payne effect results of the studied composites are shown in Figure 3. It is evident that the Payne effect increases when CNT or ND content increases. At lower filler concentrations (2 phr and 5 phr), the Payne effect is similar in both cases but at 10 phr, CNTs have much higher Payne effect than NDs. Therefore, CNTs form better network than NDs due to the above-mentioned reasons: volume fraction and aspect ratio.
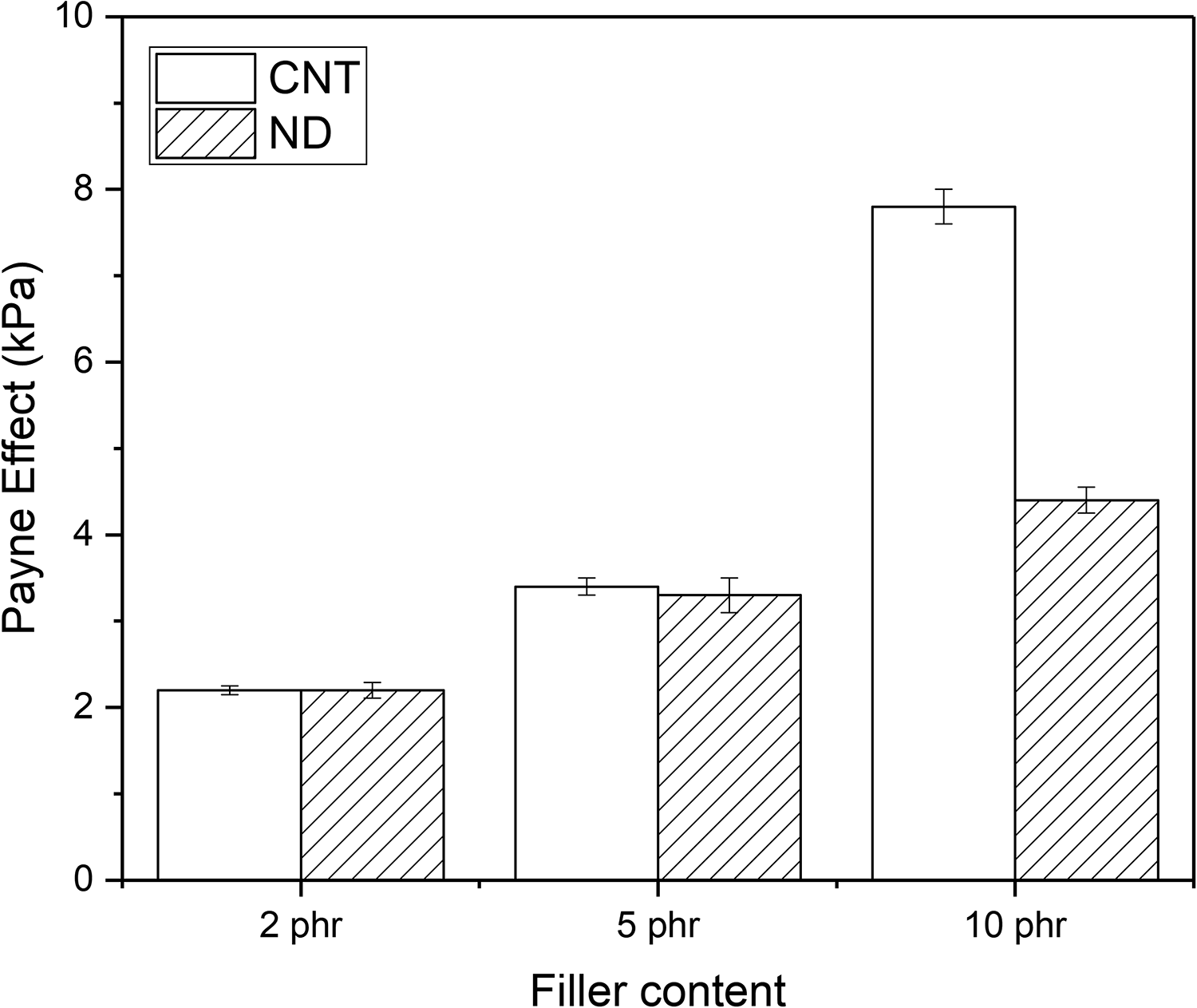
The effect of CNTs and NDs on the Payne effect of PCM-NR composite. CNT: carbon nanotube; ND: nanodiamond; PCM: phase change material; NR: natural rubber.
The FESEM study was conducted to analyze the dispersion of nanofillers in a PCM-NR composite. The pictures from the fracture surfaces (the tensile test) of the PCM-NR composites are shown in Figure 4, where the paraffin can be seen as circular clusters. In the reference compound (no fillers), the average diameter of the clusters is over 20 µm. The addition of CNTs decreases their size. With 2 phr and 5 phr CNT concentrations, the average size of the clusters is about 10 µm and with 10 phr CNTs, the clusters have disappeared. Therefore, the addition of CNTs improves the dispersion and distribution of paraffin. NDs do not improve dispersion of paraffin as much as CNTs do. The size of the paraffin clusters is large and they are not evenly distributed.

Fracture surfaces (the tensile test) of the PCM-NR composites: (a) No filler, (b) 2 phr CNT, (c) 5 phr CNT, (d) 10 phr CNT, (e) 2 phr ND, (f) 5 phr ND, and (g) 10 phr ND. CNT: carbon nanotube; ND: nanodiamond; PCM: phase change material; NR: natural rubber.
The PCM-NR-nanofiller composites’ ability to store thermal energy was investigated by DSC and the results are shown in Figures 5 and 6. It can be seen that the amount of latent heat (enthalpy of fusion) decreases slightly after the addition of nanofillers, which is due to the decreased relative amount of the PCM in the composite. According to the rule of the mixtures, the latent heat of the composites containing 2 phr, 5 phr, and 10 phr nanofillers should have the latent heats of 77.2 J/g, 75.8 J/g, and 73.3 J/g, respectively. Figure 6 shows that the compounds containing CNTs have slightly higher latent heat than the theoretical values show. Therefore, it seems that the paraffin loss observed in earlier studies 12,14 can be prevented by the addition of CNTs. NDs do not have such an effect. The latent heats are lower than the theoretical values.
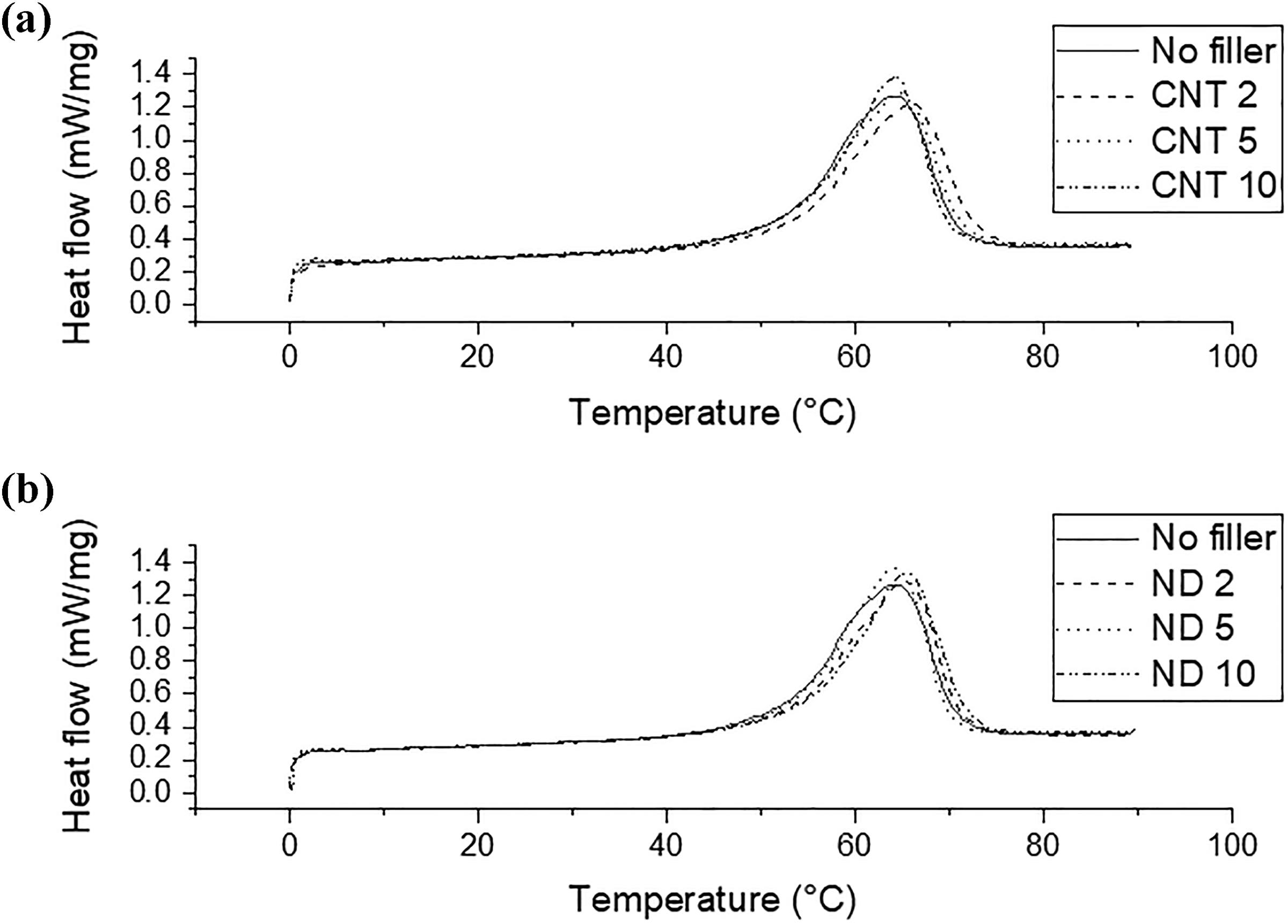
DSC curves for (a) CNT compounds and (b) ND compounds. DSC: differential scanning calorimetry; CNT: carbon nanotube; ND: nanodiamond.
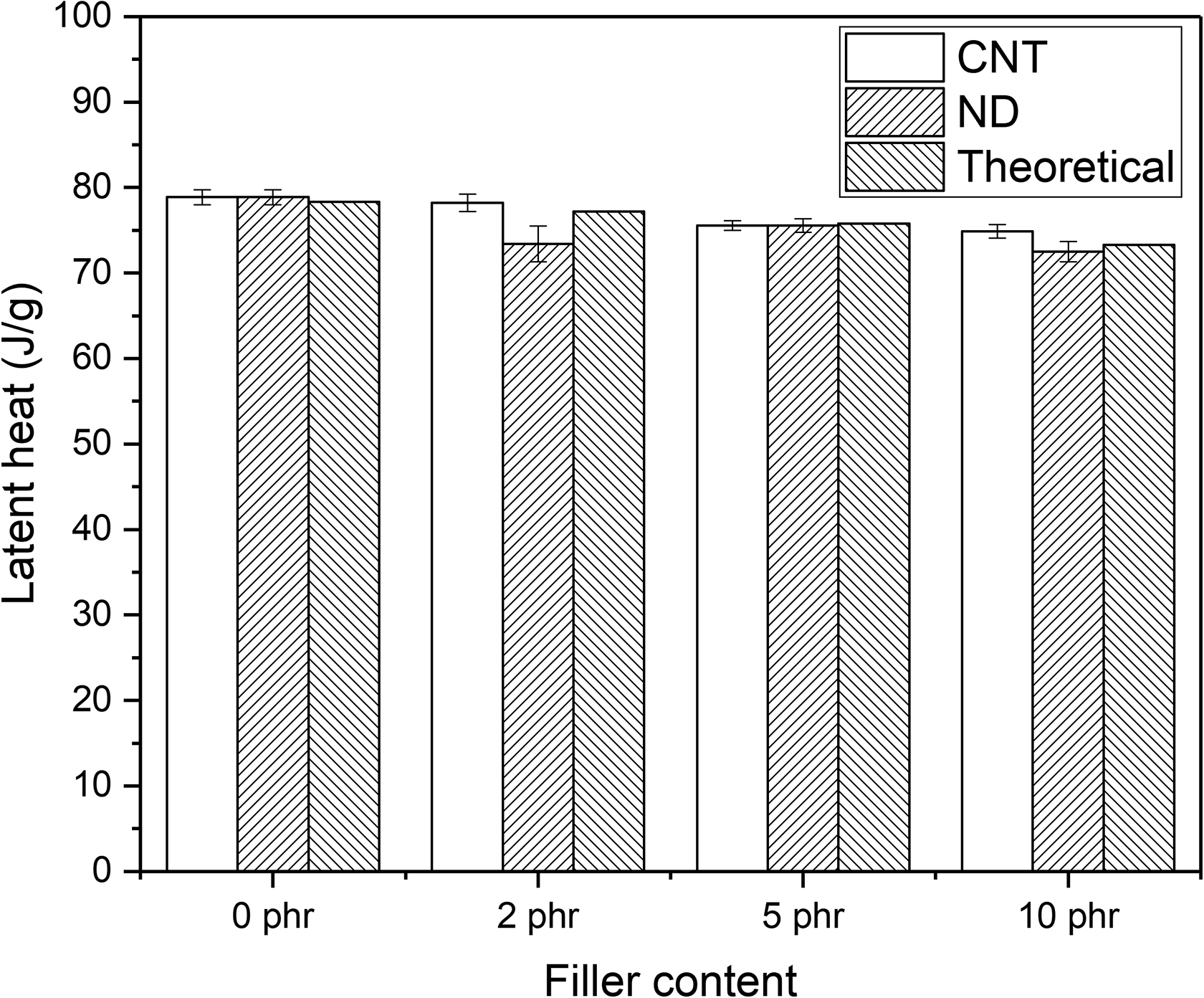
Latent heat of PCM-NR composites filled with different amount of fillers. PCM: phase change material; NR: natural rubber.
Overall, CNTs work better than NDs. The composites containing CNTs have higher latent heats than the composites containing NDs. This can again be due to the higher volume fraction of CNTs as the larger volume fraction of fillers can bind paraffin better as well as due to better dispersion of paraffin after addition of CNTs as shown in Figure 4.
To analyze a reason for the low heat of fusion values of the PCM-NR composites filled with NDs, TGA measurements were performed. The assumption was that a part of paraffin was lost during the vulcanization process. The results are shown in Figure 7. According to the TGA, the paraffin content is 10–11% lower than the theoretical amount in the composites containing CNTs. For NDs, this value varies between 11% and 13% being the lowest for the composite containing 10 phr NDs. The unfilled composite contains only 86% paraffin out of the theoretical value. This supports the results presented earlier. It confirms that nanofillers and especially CNTs prevent the migration of the paraffin. The fact that the paraffin loss was high in the composite containing 10 phr NDs can be explained by poor dispersion of fillers. The fillers have remained as large agglomerates which prevents interaction with paraffin.
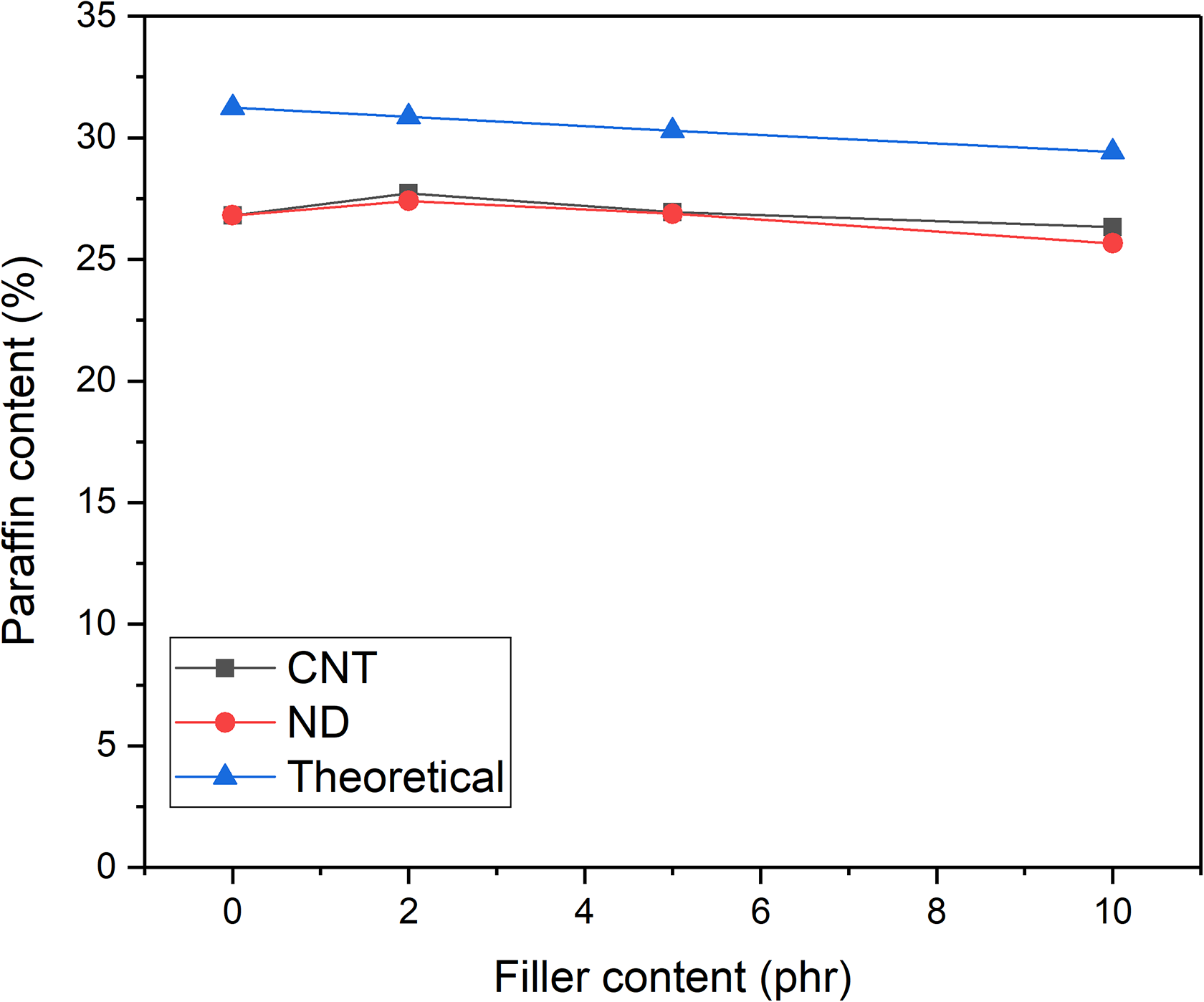
The measured and theoretical CNT and ND contents in PCM-NR composites. CNT: carbon nanotube; ND: nanodiamond; PCM: phase change material; NR: natural rubber.
Conclusions
The effect of CNTs and NDs on the thermal conductivity and heat storage capacity of paraffin-NR composite was studied. Over 40% improvement in thermal conductivity was achieved with 10 phr CNTs. Although, the addition of CNTs decreased the total amount of the latent heat, it was observed that the amount of latent heat is higher than it should theoretically be due to the paraffin-CNT interaction. It was also observed that CNTs prevent the loss of paraffin during the processing.
Furthermore, NDs improve thermal conductivity and prevent the loss of paraffin but the effect is much lower than with CNTs. This is due to the lower volume fraction and aspect ratio. NDs would require higher filler concentration to form a filler network and therefore to get significant improvements in thermal conductivity.
Footnotes
Acknowledgements
Paavo V. Suominen Foundation is acknowledged for funding this study. Clara Lessa Belone is thanked for her assistance in DSC, TGA, and LFA measurements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Paavo V. Suominen Foundation.