Abstract
Objective
Childhood trauma can lead to major depressive disorder (MDD) in adulthood. Based on the hypothesis that childhood trauma disrupts white matter integrity and predisposes individuals to MDD, this study investigated the relationship between childhood trauma, white matter integrity disruption, and the presence of MDD.
Methods
This study included 30 patients with MDD in a depressive episode and 30 control subjects aged 18-65. All subjects underwent magnetic resonance diffusion tensor imaging (MR-DTI) and completed the Hamilton Depression Rating Scale (HAM-D) and the Childhood Trauma Questionnaire (CTQ).
Results
Controlling for age and education, no difference was found between the patient and control groups for the mean fractional anisotropy (FA) values of the forceps major, forceps minor, and the right and left corticospinal tracts. However, the apparent diffusion coefficient (ADC) value of the forceps minor (a white matter fiber tract in the brain, located in the anterior part of the corpus callosum that connects the two frontal lobes) was significantly higher in the patient group than in the control group. There was also a positive correlation between the ADC value of the left corticospinal tract and physical neglect in the patient group.
Conclusion
The significantly higher ADC values observed in the forceps minor group compared to the control group suggests impaired white matter integrity in depressed patients. The positive correlation between the ADC value of the left corticospinal tract and physical neglect in the patient group supports the possibility that childhood trauma affects the integrity of brain white matter, thereby predisposing individuals to psychopathology. Prospective studies involving larger samples are needed to replicate the initial findings from this case-control study.
Introduction
Childhood adversities, such as abuse or neglect, are well-established risk factors for depression. 1 Evidence suggests that alterations in neural circuits resulting from early traumatic experiences may mediate the development of depression in adulthood, a view supported by neuroimaging studies.2,3
Diffusion tensor imaging (DTI) is a widely used method for investigating white matter structure in psychiatric populations, providing greater detail of microstructural features than conventional structural magnetic resonance (MR) imaging. 4 The principle of diffusion imaging is based on measuring the movement of water molecules within tissues. 5 DTI is particularly useful for identifying connections between brain regions. Among DTI-derived metrics, fractional anisotropy (FA) is the most commonly used. FA quantifies the anisotropic diffusion of water molecules on a scale between 0 and 1: values approaching 1 reflect diffusion along neural fibers restricted by intact myelin sheaths, while values closer to 0 indicate random Brownian motion. Reduced FA values suggest axonal loss and/or demyelination, whereas values near 1 indicate high anisotropy and preserved axonal integrity. 6
Another parameter that quantifies diffusion is the ADC, also referred to as mean diffusivity. ADC reflects the rate at which water molecules diffuse within cerebral tissues and is expressed per unit area per unit time. Higher ADC values indicate less restricted diffusion and therefore a less organized white matter structure. Conversely, lower ADC values suggest the presence of organized pathways and fibers that impede water diffusion or thick myelin layers that restrict water mobility along axonal tracts.6,7
Numerous DTI studies in patients with depression have demonstrated FA abnormalities in specific brain regions, suggesting structural and functional disruptions of white matter.8-11 The corticospinal tracts are likely to contribute to functions relevant to MDD, including somatosensory, emotional, and cognitive processes. 12 Psychomotor disturbances commonly observed in depression, such as impaired concentration, decision-making difficulties, and increased sensitivity to negative stimuli, further point to potential pathology in motor pathways. 13 Supporting this, two DTI studies evaluating the bilateral and right corticospinal tracts in depressed patients reported significantly reduced FA values.14,15
The forceps minor connects the prefrontal cortex (PFC) and anterior cingulate cortex (ACC), forming part of the fronto-limbic circuit. Because it links the ACC with other brain structures, disruption of its integrity may alter ACC activity.16,17 It can therefore be hypothesized that the forceps minor is affected in patients with MDD, given its proposed regulatory role in emotional experience. 18 Consistent with this, previous studies have reported significantly lower FA values in the forceps minor of depressed patients compared with healthy controls.16,18,19
The forceps major is a fiber bundle that connects the occipital lobes and crosses the midline through the splenium of the corpus callosum. Dysfunction in this pathway may disrupt the integration of somatosensory and visual information between the hemispheres and impair the recognition of social emotions. 20 Several studies have reported decreased FA values in the forceps major of patients with MDD.19,21
Studies have demonstrated reduced integrity in multiple white matter pathways, including the corpus callosum, uncinate fasciculus, cingulum, fornix, superior longitudinal fasciculus, inferior longitudinal fasciculus, and inferior fronto-occipital fasciculus, in individuals with a history of childhood trauma. 22 A longitudinal study showed that adolescents exposed to maltreatment had impaired integrity of the cingulum bundle extending to the hippocampus compared with healthy controls, and this alteration predicted the development of depression at a 5-year follow-up. 23
This study aimed to examine the relationship between childhood trauma, white matter integrity, and the development of MDD. This objective was based on the assumption that childhood trauma disrupts white matter pathways, thereby increasing vulnerability to MDD.
Methods
Sampling and Data Collection
This case-control study was conducted in the inpatient and outpatient psychiatry clinics of a university hospital, which is a tertiary care center, and included 30 MDD patients (20 females, 10 males) and 30 healthy controls (20 females, 10 males) aged 18-65, recruited between December 2019 and February 2021. All patients met DSM-5 criteria for MDD and were experiencing a depressive episode at the time of participation. Exclusion criteria were comorbid psychiatric disorders other than MDD, neurological or systemic diseases affecting neurophysiology, recent ECT or neuromodulatory treatment, pregnancy or breastfeeding, intellectual disability, alcohol/substance use disorder (except smoking), illiteracy, or contraindications to MRI. Sociodemographic and clinical data were collected using a structured form. Diagnostic evaluations were conducted through face-to-face clinical interviews based on DSM-5 criteria and were confirmed independently by two clinicians. Psychometric scales were administered by a psychiatrist. Blood samples were obtained, and diffusion tensor magnetic resonance imaging (DTI) was performed to assess white matter integrity.
Scales
Hamilton Depression Rating Scale (HAM-D)
This scale was developed by Hamilton in 1960 24 . Akdemir et al. 25 established the validity and reliability of the Turkish version.
Childhood Trauma Questionnaire (CTQ)
The CTQ is a 28-item, 5-point Likert scale developed by Bernstein et al. 26 and adapted to Turkish by Şar et al.. 27 It assesses five domains of childhood trauma: physical abuse, sexual abuse, emotional abuse, physical neglect, and emotional neglect. It also includes three minimization items. Higher total scores indicate more severe trauma. The scale has shown high internal consistency (Cronbach’s α = 0.79-0.94). 26
Acquisition of MR Imaging Data
MRI scans were acquired on a 1.5 T Signa system (General Electric Healthcare Technologies, Milwaukee, WI, USA) using a 16-channel head coil. Participants were scanned in the supine position without sedation. A high-resolution T1-weighted anatomical sequence was followed by DTI with 20 gradient directions (b = 0 and 1000 s/mm2).
DTI Data Processing and Analysis
All MRI scans were reviewed and analyzed by an experienced radiologist who was blinded to participants’ group allocation (patient vs control). DTI data were processed with AW Server Version 3.2 Ext. 1.0 (General Electric Healthcare Technologies, Milwaukee, USA). Tractography of the right and left corticospinal tracts, forceps major, and forceps minor was reconstructed by a radiologist using the ROI-based method described by Wakana et al (2007). Mean FA and ADC values of each tract were calculated. 28
Bilateral Corticospinal Tract
Bilateral corticospinal tracts were identified between the primary motor cortex and the medulla oblongata. Three ROIs were placed along the tract following the method of Wakana et al (2007): the cerebral peduncle at the level of the superior cerebellar peduncle decussation, the precentral gyrus at the level of the central sulcus, and the medullary pyramid. Mean FA and ADC values were then calculated for the right and left corticospinal tracts.
Forceps Major
The splenium of the corpus callosum was selected as the first ROI. For the second ROI, a parasagittal plane was selected at the level of the cingulum, and the parieto-occipital sulcus was identified. A coronal plane was taken at the most posterior edge of this sulcus, such that it included only the right occipital lobe. For the third ROI, the left occipital lobe was defined in a coronal section. The mean FA and ADC values of the reconstructed pathway were then measured.
Forceps Minor
The first ROI was placed in the genu region of the corpus callosum. The anterior edge of the genu and the anterior midpoint of the frontal lobe were selected in the sagittal plane. Both frontal lobes were completely surrounded in the section corresponding to this point in the coronal plane, creating the second and third ROIs. The mean FA and ADC values of the obtained tract were then measured.
Statistical Analysis
Normality of continuous variables was assessed using the Shapiro–Wilk test, skewness values, and visual inspection of Q–Q plots and boxplots. Categorical variables are presented as n (%). Continuous variables are presented as mean ± standard deviation (SD), as all main study variables showed approximately normal distributions. Group differences in HAM-D and CTQ scores were examined using independent samples t-tests. Group differences in FA and ADC values were examined using multivariate analysis of covariance (MANCOVA), with age and duration of education included as covariates. Pearson correlation coefficients were calculated to assess correlations between variables. The statistical significance level was accepted as P < .05.
Results
Clinical Characteristics of the Patients (n = 30)
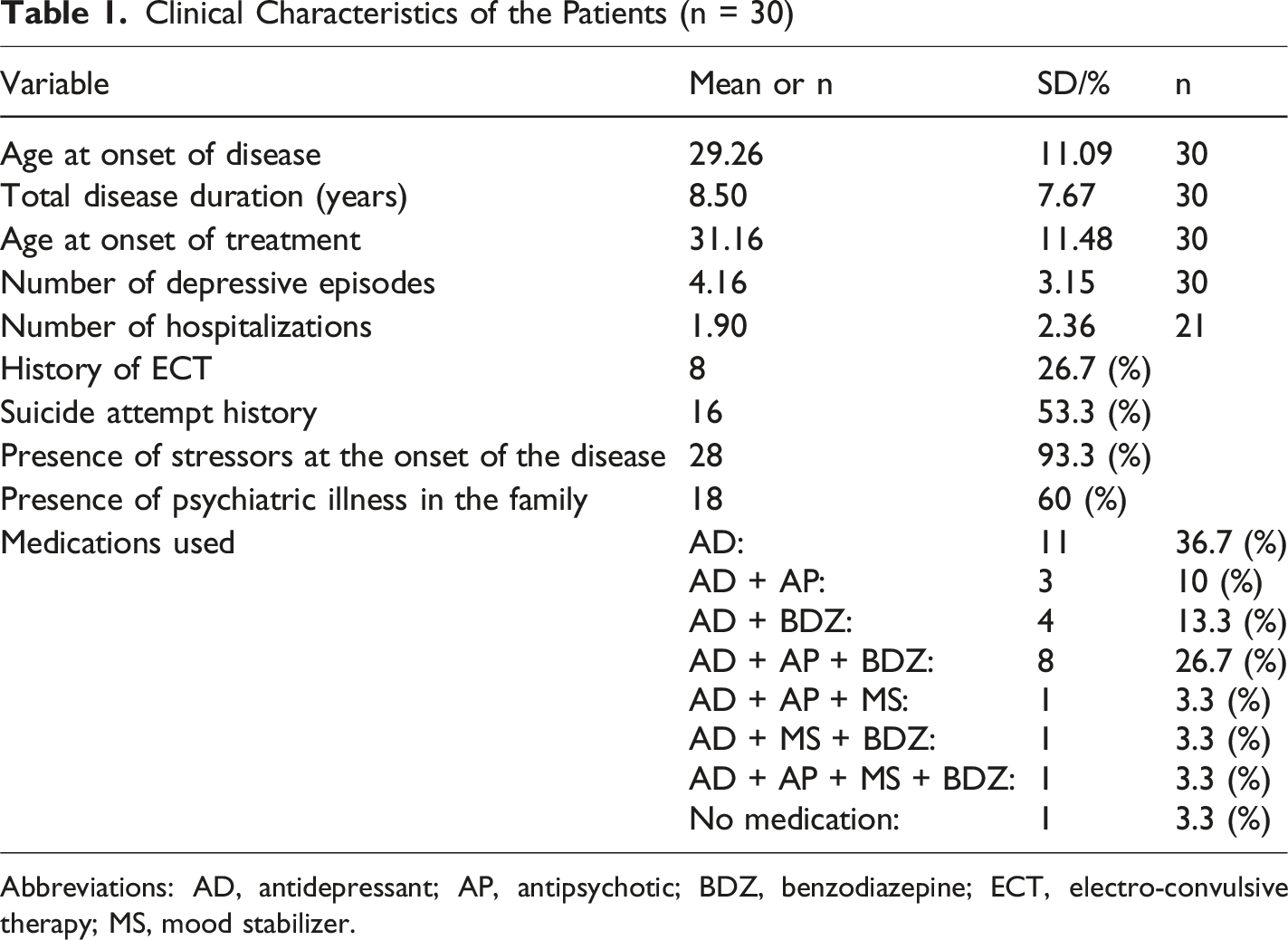
Abbreviations: AD, antidepressant; AP, antipsychotic; BDZ, benzodiazepine; ECT, electro-convulsive therapy; MS, mood stabilizer.
Comparison of Psychometric Test Scores of Patients and Controls
Comparison of HAM-D and CTQ Scores of Patients and Controls
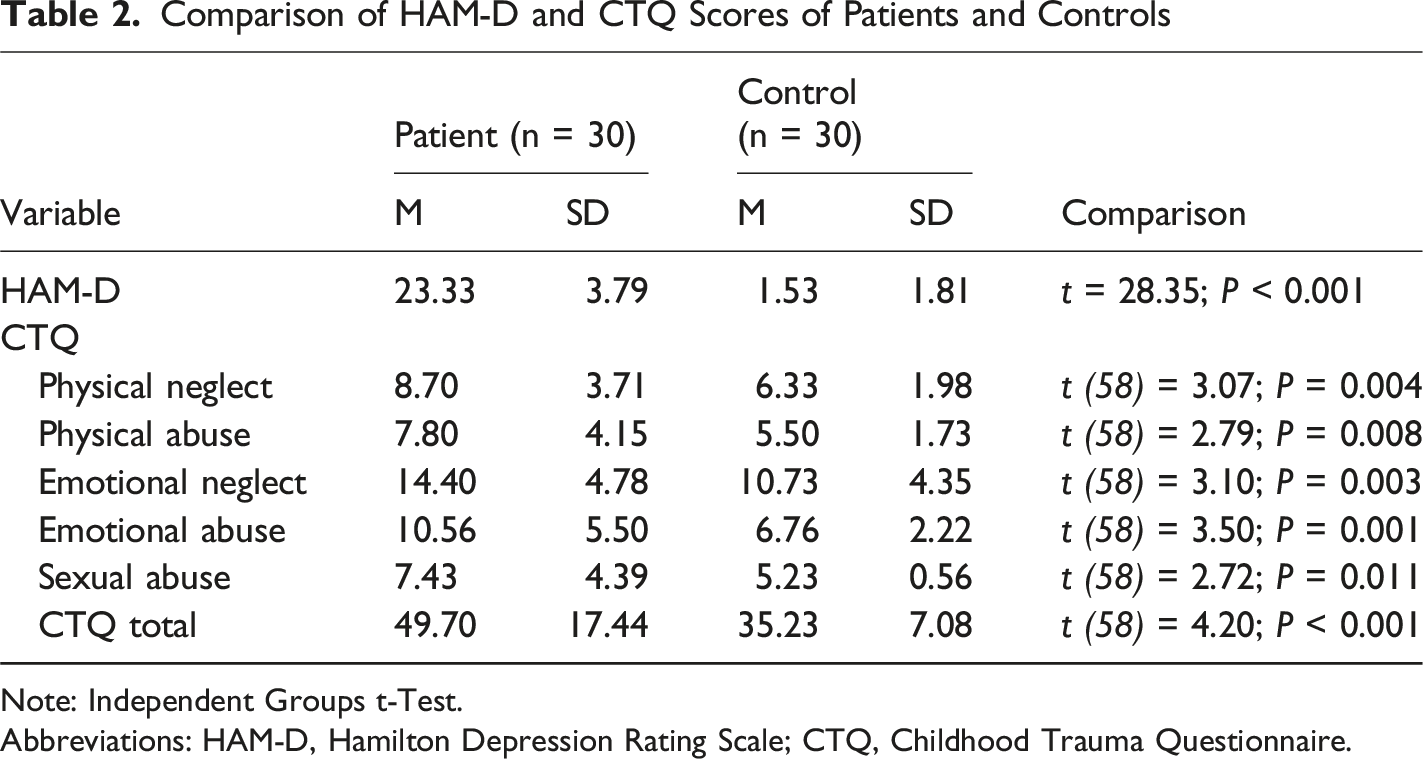
Note: Independent Groups t-Test.
Abbreviations: HAM-D, Hamilton Depression Rating Scale; CTQ, Childhood Trauma Questionnaire.
Comparison of Mean FA and ADC Values of Patients and Controls
Comparison of Mean (±SD) FA and ADC Values Between Patients (n = 30) and Controls (n = 30)
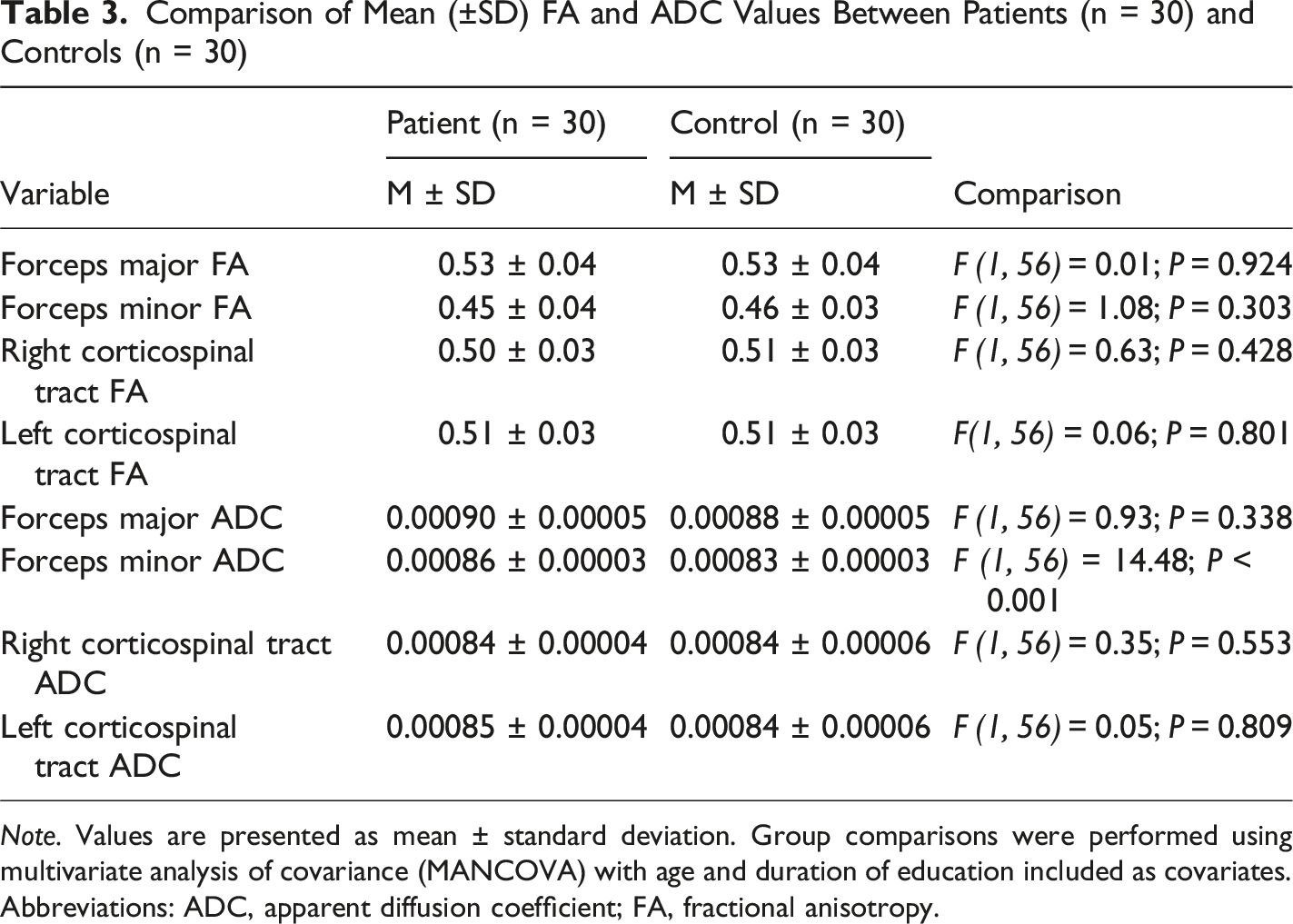
Note. Values are presented as mean ± standard deviation. Group comparisons were performed using multivariate analysis of covariance (MANCOVA) with age and duration of education included as covariates.
Abbreviations: ADC, apparent diffusion coefficient; FA, fractional anisotropy.
Correlation Studies in the Patient and Control Groups
A statistically significant positive correlation was found between the ADC value of the left corticospinal tract and physical neglect in the patient group (r = 0.48, P = .008). In the control group, a negative correlation was found between the forceps minor FA value and physical abuse, emotional abuse, and CTQ total score (r = −0.54, P = .002; r = −0.46, P = .011; r = −0.38, P = .037, respectively). There was also a negative correlation between the right corticospinal tract ADC value and the emotional abuse score (r = −0.39, P = .032), as well as a statistically significant negative correlation between the left corticospinal tract FA value and the emotional abuse score and the CTQ total score (r = −0.48, P = .008; r = −0.39, P = 0.035).
Discussion
The present study examined the relationship between childhood trauma and white matter integrity in patients with MDD. No significant differences were observed in FA values of the bilateral corticospinal tracts between patients and healthy controls. Previous DTI studies investigating white matter abnormalities in depression have used heterogeneous methodologies and yielded inconsistent results. Considering that depression is often accompanied by impairments in concentration, decision-making, negative cognitive biases, and psychomotor symptoms, motor pathways were specifically evaluated in this sample. 13
In a DTI study of untreated first-episode depression, FA values of the bilateral corticospinal tracts were significantly reduced. 14 In contrast, most patients in our sample were receiving multiple psychotropic medications, which may have contributed to relatively preserved FA values and the absence of significant group differences. Supporting this assumption, FA has been shown to increase following antidepressant treatment. 29 Similarly, another study of medicated patients with depression reported reduced FA in the right corticospinal tract compared with controls, 15 while other reports have found no significant differences between groups.21,30,31 Conversely, Sacchet et al (2014a, 2014b) observed increased bilateral corticospinal tract FA in MDD.12,32 The findings of the present study may have been influenced by the heterogeneity of clinical characteristics in our patient group. Since motor pathways such as the corticospinal tract are closely related to psychomotor functioning, it is reasonable to suggest that different outcomes might have been observed if subgroups of depressed patients with psychomotor retardation or agitation had been specifically included.
Although no group differences were found in forceps major and minor FA values, forceps minor ADC values were significantly higher in patients than in controls. Given that the PFC and ACC are central to motivation and reward processing, and that the forceps minor connects these regions, disruption of this pathway may contribute to impaired emotional experience and anhedonia.16-18 The elevated ADC values observed in our patients, therefore, suggest impaired white matter integrity and weakened PFC–ACC connectivity.
Lyden et al (2014) similarly reported lower FA and higher mean diffusivity in the forceps minor of patients with MDD, which improved following ECT treatment, supporting the idea that reduced FA and increased diffusivity in fronto-limbic pathways may serve as biological markers of depression. 16 However, previous studies have yielded inconsistent results. Medication use may have contributed to the absence of FA differences in our sample, as psychotropic treatment could normalize values. Indeed, while reduced FA without mean diffusivity changes has been reported in drug-naïve first-episode patients,18,19 Shen et al (2017) found no difference in forceps minor FA between groups. 21 Collectively, these findings indicate that FA and ADC may capture distinct aspects of white matter pathology and may hold different predictive value.
Childhood trauma, particularly sexual abuse and bullying, is a well-established risk factor for depressive disorders. 33 Exposure to interparental violence has also been linked to lower educational attainment and higher rates of depressive symptoms in adulthood, suggesting that adverse experiences disrupt emotional, psychological, and social development. 34 Neuroimaging studies support these associations: maltreated adolescents show white matter abnormalities that increase vulnerability to depression, 23 and DTI investigations have reported widespread FA reductions in individuals with a history of trauma.35,36 Notably, previous neuroimaging studies examining the effects of childhood trauma have predominantly focused on adolescent samples, often before the onset of a formal diagnosis of major depressive disorder, whereas the present study was conducted in an adult sample with a confirmed diagnosis of MDD and well-matched patient and control groups in terms of age and gender. These differences in sample characteristics and study design may partly account for discrepancies across findings and underscore the distinct contribution of the present study. In contrast, this study did not reveal significant correlations between childhood trauma and FA values in patients with MDD. Instead, we found a positive association between physical neglect and the ADC value of the left corticospinal tract. This finding supports the hypothesis that childhood trauma may contribute to the development of psychopathology in MDD by disrupting white matter integrity.
A study on MDD patients reported unexpected findings, showing a positive correlation between childhood trauma and white matter integrity, such as increased FA or decreased mean diffusivity. 37 Other studies comparing depressed patients with healthy controls found the opposite pattern, with reduced FA and increased diffusivity, while some reported that trauma-exposed depressed patients had higher FA and lower diffusivity than non-exposed individuals. These inconsistencies have led to the hypothesis of a compensatory mechanism in response to trauma.20,38 In the present study, healthy controls showed negative correlations between childhood trauma scores and FA values in the forceps minor and corticospinal tracts. By contrast, such associations were absent in the patient group. This pattern may further support the compensation hypothesis, suggesting that while trauma is associated with disrupted white matter integrity in healthy individuals, patients with MDD may exhibit compensatory changes, such as increased myelination, that mask these associations.
Strengths and Limitations of the Study
A key strength of this study is the careful exclusion of psychiatric comorbidities when forming the patient and control groups. The groups were also matched for gender, thereby minimizing potential hormonal influences, and their comparable mean ages contributed to homogeneity, given the known effects of age on white matter integrity. Additional strengths include the use of tractography-based analysis.
A major limitation, however, is that most patients were receiving multiple psychotropic medications. Moreover, heterogeneity in clinical features, such as illness duration and number of episodes, may have reduced the reliability of the findings. Finally, the absence of a standardized method for image analysis and variability across approaches could have contributed to inconsistencies in DTI results.
In addition, the relatively small sample size, the lack of a longitudinal study design, and the restriction of the sample to a single academic medical center should be considered important limitations, as these factors may limit the generalizability of the findings.
Moreover, internal consistency indices for the HAM-D and CTQ (eg, Cronbach’s alpha/McDonald’s omega) could not be recalculated in the present sample because item-level responses were not available and only total scale scores could be accessed.
Finally, an a priori statistical power analysis was not performed; therefore, the relatively modest sample size may have limited statistical power, particularly for detecting small effect sizes.
Clinical Implications
The findings of this study suggest that early-life adverse experiences, particularly childhood trauma, may be associated with underlying neurobiological alterations in patients with MDD. These results underscore the importance of systematically assessing childhood trauma in patients presenting with depressive symptoms.
From a clinical standpoint, a trauma-informed approach may be especially relevant for primary care physicians and mental health professionals, who often serve as the first point of contact. Although advanced neuroimaging techniques are not part of routine clinical practice, increased awareness of the potential long-term impact of childhood trauma may support more comprehensive evaluation, individualized treatment planning, and appropriate referral for psychosocial interventions when indicated.
Conclusion
This case-control diffusion tensor imaging (DTI) study suggests that major depressive disorder may be associated with subtle microstructural alterations in white matter pathways involved in fronto-limbic regulation, particularly within anterior callosal connections.
Taken together, the findings indicate that diffusion-derived indices may provide complementary information about white matter organization and that early-life trauma, especially physical neglect, may be linked to neurobiological vulnerability relevant to depressive illness. From a clinical perspective, these preliminary results highlight the importance of routinely assessing childhood trauma in patients with depression and incorporating trauma-informed approaches into evaluation and treatment planning.
Larger, longitudinal, multi-center studies, ideally including medication-free samples, are needed to replicate these observations, clarify temporal relationships, and determine whether white matter metrics have prognostic value or treatment-related utility.
Footnotes
Acknowledgments
We would like to thank the Proofreading & Editing Office of the Dean for Research at Erciyes University for the copyediting and proofreading service for this manuscript.
Ethical Considerations
This research was approved by the Erciyes University Faculty of Medicine Ethics Committee (Decision Number: 2019/798, 13/11/2019). Written consent was obtained from all participants in the patient and control groups after they were informed about the aims of the study and how it would be conducted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Erciyes University Scientific Research Projects Commission (Project Number: TTU-2020-9891).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
