Abstract
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are increasingly used for managing type 2 diabetes and obesity. As their clinical applications expand, interest in their safety and adverse effects has grown. This study provides a comprehensive bibliometric analysis of global research trends, collaboration patterns, and thematic evolution on GLP-1RA-related adverse effects from 2006 to 2025. This study is a bibliometric analysis. Data were extracted from the Scopus database and analyzed using Bibliometrix (R package) and VOS viewer. Indicators assessed included publication and citation metrics, institutional productivity, and keyword co-occurrence and mapping. Correlations were evaluated using Pearson and Spearman tests, and the Durbin–Watson statistic was applied to assess the independence of residuals. A total of 1075 articles published in 389 journals were identified, authored by 6068 researchers across 85 countries. The annual growth rate was 32.06%, with no single-author papers and 34.23% international co-authorship, indicating strong global collaboration. The United States (30.5%), the United Kingdom (10.6%), and Denmark (7.8%) led in publication output and total citations. Institutional analysis identified Novo Nordisk A/S (60 papers, 11 207 citations) and Eli Lilly & Co. (45 papers, 9948 citations) as the most influential contributors. Diabetes, Obesity and Metabolism published the most articles (n = 106), followed by Diabetes Care and The Lancet Diabetes & Endocrinology. Significant correlations were found between article count and h-index (r = .677, P < .001) and total citations (r = .779, P < .001). Keyword analysis revealed 2 main thematic clusters-one pharmacological (drug safety, efficacy, liraglutide, semaglutide) and 1 clinical (human, male, female, adult)—with an emerging focus on population-specific safety since 2021. Research on GLP-1RA-related adverse effects has expanded rapidly, shaped by strong international collaboration and industry-academic partnerships. Future efforts should prioritize balanced global participation, real-world safety data, and mechanistic insights to inform clinical practice and pharmacovigilance.
Keywords
Introduction
Historical Overview
Glucagon-like peptide-1 (GLP-1) is a gastrointestinal hormone consisting of 30 to 31 amino acids, produced from the digestive tract and initially characterized in 1984. It increases the amount of insulin released while lowering the amount of glucagon in the body. 1
GLP-1 is essential for standard glucose tolerance. It works through GLP-1 receptors, which are part of the G protein-coupled glucagon receptor family. These receptors are found in pancreatic β-cells, as well as in the stomach, small intestine, heart, mucosa, and other tissues.
Exenatide was the first GLP-1 receptor agonist (GLP-1RA) to get FDA approval. It was approved in 2005 for type 2 diabetes mellitus. Post-marketing research showed that many users lost significant weight. Nonetheless, definitive evidence about its effectiveness for weight loss in patients with simple obesity is still lacking.2,3
In 2014, Liraglutide was approved as an additional medication for long-term weight management in obese patients aged ≥12 years, predicated on BMI thresholds (≥30 kg/m2 in adults >60 kg). It was also approved for treating diabetes.3,4 Semaglutide was introduced in 2021 for a similar use in weight loss. 5
Tirzepatide, a dual GIP/GLP-1 receptor agonist, was FDA-approved in 2022 for the treatment of type 2 diabetes .In 2023, the FDA approved tirzepatide for obesity management, representing a significant expansion of incretin-based therapies for weight management. 6
Between 2018 and 2023, prescriptions for GLP-1RAs went up by 300%. This shows that they work, are safe, and are easy to use. But their high price raises many worries about how easily they can be obtained and how they might affect healthcare equity. 7
Lixisenatide, which was approved in 2016 for adults with type 2 diabetes as a once-daily addition to diet and exercise, helped people lose weight and was generally well tolerated. Nonetheless, it has not received approval for obesity treatment. 8
In 2017, Semaglutide was approved for type 2 diabetes; in 2020, it was approved to lower the risk of major cardiovascular events in people with diabetes and cardiovascular disease; and in 2025, it was approved to lower the risk of kidney disease progression and cardiovascular death in people with diabetes and chronic kidney disease. 9
In 2021, it was also approved for long-term weight control in individuals who are overweight or obese and have at least one weight-related health problem, such as high blood pressure, type 2 diabetes, or dyslipidemia.10 -13
The FDA authorized Dulaglutide, another long-acting GLP-1RA, for type 2 diabetes in 2014, but it is not recommended for obesity. 14 Up to date, only Liraglutide, Semaglutide, and Tirzepatide have been approved by the FDA for obesity and overweight. 6
Research repeatedly demonstrates that GLP-1RAs are generally safe and efficient for facilitating moderate weight loss.15 -18 Their off-label usage for weight loss has become common, which often makes it hard for people with type 2 diabetes to get them when they need them most. 19 Nonetheless, apprehensions regarding their safety have emerged in multiple instances.20 -22
Even with these benefits, GLP-1RAs can cause a wide range of side effects, the most prevalent of which are problems with the digestive system. There are also rare but substantial hazards to the pancreas, thyroid, and heart.23 -25 It is essential to understand these safety issues to maximize the benefits of therapy, encourage patients to stick with it, and reduce the risk of problems.
Adverse Effects of GLP-1 Receptor Agonists
Gastrointestinal (GI) disturbances are the most frequently reported adverse effects of GLP-1 receptor agonists (GLP-1RAs), including nausea, vomiting, diarrhea, constipation, and reflux. These effects are typically dose-dependent and constitute a major reason for treatment discontinuation.23,26 -28 GI symptoms generally appear early in therapy, affecting approximately 50% to 60% of patients, but tend to diminish over time.29,30 The slowing of gastric emptying associated with GLP-1RAs raises concerns about intestinal obstruction and perioperative aspiration, and regulatory agencies have issued warnings regarding serious GI complications and potential risks such as pancreatitis and thyroid safety concerns.31,32
Hypoglycemia was initially considered uncommon with GLP-1RAs because their glucose-lowering effects are glucose-dependent; however, evidence indicates that hypoglycemia can occur even in normoglycemic patients, particularly when GLP-1RAs are combined with other antidiabetic agents (eg, in combination with insulin or Sulfonylureas). 33 A systematic review of 34 trials involving 14 464 participants found that most GLP-1RAs (except Albiglutide) were associated with increased hypoglycemia rates (10.4%-19.4%) compared with placebo. 34 Nevertheless, the risk remains lower than that associated with sulfonylureas or insulin therapy.35,36 Although randomized trials report treatment adherence rates of 65% to 85% for agents such as Liraglutide, Semaglutide, and Tirzepatide, real-world adherence is substantially lower due to adverse effects, complex dosing, and economic or patient–provider factors.37 -41
Rare but serious adverse events have been reported. Acute pancreatitis and elevated pancreatic enzymes are infrequent, and meta-analyses generally show no significant increase in pancreatitis or pancreatic cancer risk.40,42 -44 Similarly, concerns regarding medullary thyroid cancer (MTC) stem primarily from preclinical findings; no confirmed human cases have been documented, though boxed warnings remain as a precaution. 25 Long-term gastrointestinal complications such as gastroparesis or ileus have been reported in pharmacovigilance databases, but randomized trials rarely capture these events and real-world evidence remains limited. 45 Psychiatric events, including rare reports of suicidal ideation, have prompted ongoing regulatory monitoring, although causal relationships have not been established.31,46
GLP-1RAs are also associated with an increased risk of gallbladder and biliary disease, particularly in high-dose weight-loss trials, and semaglutide has been linked to worsening diabetic retinopathy in patients with pre-existing disease, possibly due to rapid HbA1c reductions.21,47 -50 Despite these potential risks, the overall safety profile of GLP-1RAs remains favorable relative to older antidiabetic therapies, and their cardiovascular and metabolic benefits continue to drive clinical adoption. Ongoing pharmacovigilance and long-term studies are necessary to clarify rare adverse signals and optimize patient safety.
The Aim of This Review
This study addresses a significant need to evaluate research productivity, impact, and theme progression in the field of GLP-1RA safety, given the growing demand for bibliometric evaluations, especially by policymakers, funding organizations, and regulatory bodies. A thorough understanding of international research on GLP-1RA-related side effects is crucial for assessing the existing database and guiding future studies, especially given the increasing clinical application of these medicines in diabetes, obesity, and metabolic diseases. Bibliometric analysis provides a comprehensive methodology for assessing research trends, delineating collaborations among authors and organizations, and recognizing subject clusters within the scientific literature. Consequently, the current study analyzes the attributes of publications regarding GLP-1RA adverse effects indexed in Scopus, illustrates worldwide research hotspots and developmental trajectories, and investigates trends in authorship, international collaboration, and thematic progression. The project aims to enhance the database on the safety of GLP-1RA and to bolster national and global initiatives focused on rational usage, pharmacovigilance, and patient safety through an integrative strategy.
The goal of this review is to use bibliometric indicators and thematic science-mapping techniques to map and measure the global research landscape on the adverse effects of GLP-1 RAs. This will help, 51 find publication trends, geographic and institutional contributions, and highly cited works;, 52 describe the main safety topics and adverse-event clusters (like gastrointestinal events, pancreatitis, gallbladder disease, and rare signals) and how they have changed; and highlight evidence gaps and research priorities to inform clinicians, regulators, and researchers about areas needing further primary studies or pharmacovigilance. This study combines quantitative bibliometric metrics and network visualization (co-authorship, co-citation, and co-word/topic mapping) with qualitative thematic analysis to provide an objective, reproducible map of the field and to contextualize clinical safety findings reported in systematic reviews and trials.
Methods
Sources of Data
Data were collected to explore global research trends on GLP-1RA-related adverse effects using the Scopus database (https://www.scopus.com/). The Scopus database is a platform that provides a unique, advanced search of the world’s most trusted citation index for scientific and scholarly research. It is used to provide researchers with a comprehensive dataset used subsequently for bibliometric analysis across many disciplines.
On October 11th, 2025, we searched the Scopus Database for articles on GLP-1RA-related adverse effects in peer-reviewed journals indexed in the Scopus Database from 2006 to 2025 (Data for 2025 are partial). A flowchart of the article screening process is presented in Figure 1.
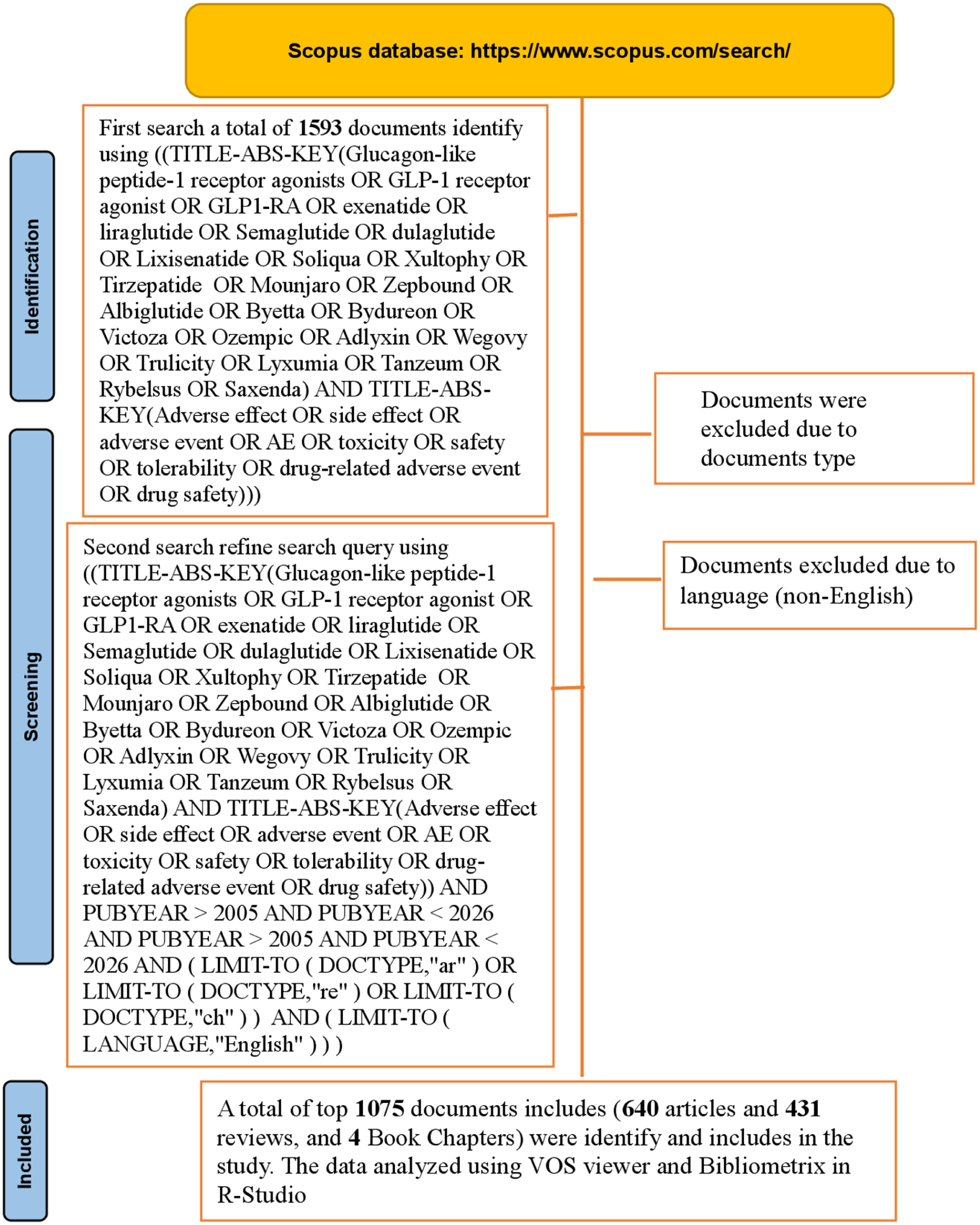
Flow chart of the search approach use in data collection process from Scopus database.
A Boolean search strategy was applied using the subject term formula (TITLE-ABS-KEY) in Scopus to retrieve studies related to GLP-1RAs’ adverse effects. The following query string was used: ([TITLE-ABS-KEY[Glucagon-like peptide-1 receptor agonists OR GLP-1 receptor agonist OR GLP1-RA OR exenatide OR liraglutide OR Semaglutide OR dulaglutide OR Lixisenatide OR Soliqua OR Xultophy OR Tirzepatide OR Mounjaro OR Zepbound OR Albiglutide OR Byetta OR Bydureon OR Victoza OR Ozempic OR Adlyxin OR Wegovy OR Trulicity OR Lyxumia OR Tanzeum OR Rybelsus OR Saxenda] AND TITLE-ABS-KEY[Adverse effect OR side effect OR adverse event OR AE OR toxicity OR safety OR tolerability OR drug-related adverse event OR drug safety]] AND PUBYEAR >2005 AND PUBYEAR <2026 AND PUBYEAR >2005 AND PUBYEAR <2026 AND [LIMIT-TO [ DOCTYPE, “ar” ] OR LIMIT-TO [ DOCTYPE, “re” ] OR LIMIT-TO [DOCTYPE, “ch” ]] AND [LIMIT-TO [ LANGUAGE, “English” ]]).
The time frame from 2006 to 2025 was selected to ensure comprehensive coverage of research on GLP-1 receptor agonists (GLP-1 RAs) and their adverse effects. Although the first GLP-1 RA, exenatide, was approved for clinical use in 2005, the earliest studies reporting safety and tolerability outcomes, including gastrointestinal adverse effects, began to appear in 2006. Therefore, this period captures the complete publication history relevant to post-marketing safety observations and emerging adverse effect profiles up to the most recent year available in the Scopus database. Although the inclusion of publications from 2025 may influence publication trends and citation impact due to their recent nature, these records were retained to ensure that the analysis reflects the most current research activity in the field, particularly given the growing global scientific focus on GLP-1 receptor agonists.
Inclusion and Exclusion Criteria
The articles, reviews, and book chapters on GLP-1RA-related adverse effects from the Scopus database were included in our analysis. Scopus was used as the sole data source because of its broad multidisciplinary coverage and extensive indexing of biomedical literature, including the majority of records available in PubMed. This allowed comprehensive retrieval of peer-reviewed studies relevant to GLP-1 receptor agonist safety within a single dataset while reducing duplication across databases. Access to additional databases such as Web of Science was not possible due to institutional access and data extraction constraints. This limitation is acknowledged, and future bibliometric analyses incorporating multiple databases may improve coverage and generalizability. The exclusion criteria included all types of reported documents, such as letters, notes, conference papers, short surveys, editorials, errata, early access, proceedings papers, and undefined publications. These publications were excluded from Scopus; moreover, other non-English records were removed due to language.
Bibliometric Analysis
This bibliometric study was conducted and reported in accordance with the Biblio (Bibliometric Analysis Reporting Guidelines) framework, 53 in line with the EQUATOR Network recommendations for transparent reporting of research. The completed Biblio checklist has been provided as a Supplemental file.
Descriptive statistical analyses of key data, including publication trends, sources of local impact, author productivity, leading institutions and countries, and overall publication performance, were conducted using the Bibliometrix R-package (R for Windows) and RStudio (version 2025.05.1-513) with its online analysis platform. VOS viewer was used to visualize co-authorship networks, keyword co-occurrence, and citation and co-citation maps.54,55 The search records of global documents associated with GLP-1RA-related adverse effects -indexed in Scopus included 1075 documents after filtering by document type (review OR article) and language (English). The search was performed, and the results from the Scopus database were collected and exported in RIS, BibTeX, and CSV formats. Most publications were in Medicine (54%), followed by Biochemistry, Genetics and Molecular Biology (17%) and Pharmacology, Toxicology and Pharmaceutics (9%). Nursing accounted for 72 publications (4%), with smaller contributions from other fields.
Thematic map visualization and evaluation were conducted via Bibliometrix to carry out conceptual network analysis, factorial analysis, social network analysis, and collaboration network analysis. In contrast, country collaboration maps were analyzed via R Studio version 4.4.3 software, Bibliometrix, the R package, and an online analysis platform. GraphPad Prism 9 (version 9.2.0, GraphPad Software LLC, United States) and OriginPro 2019 v9.6 were used for inferential statistical analysis and to correlate study variables. A P value less than or equal to .05 was considered statistically significant. To assess the assumption of independence among residuals, the Durbin-Watson (DW) statistic was calculated for each correlation model.
Results
Trends in Global Publication and Main Characteristics
Figure 2 shows the descriptive summary statistical analysis of the 1075 identified global publications on GLP-1RA-related adverse effects, including year, sources, annual growth, document contents, authors, and document types during the period of 2006 to 2025. The included articles and reviews were published in 389 sources by 6068 authors, with zero single-author documents and 34.23% international co-authorships. The annual growth rate was 32.06%.

General information and the trend of publication.
Performance Analysis of GLP-1RA-Related Adverse Effects -Associated Publications
As illustrated in Figure 3, the evolution of publications on GLP-1RA-related adverse effects between 2006 and 2025 shows an initial steady rise in scientific output up to 2023, after which production rises sharply to reach a peak in 2025 (200 articles, until October). Citation performance drops sharply from a peak in 2006 to a bottom in 2007, and remains near this bottom until 2025. These results indicate that years with greater publication counts did not necessarily correspond to higher citation rates per year. A weak positive correlation was found between the number of articles and the mean total citations per article (r = .245, P = .007; ρ = .211, P = .021). The number of articles was also weakly correlated with the mean total citations per year (r = .228, P = .012; ρ = 0.197, P = .031).

Annual growth of globally cited articles by year on GLP-1RA-related adverse effects indexed in Scopus (A); annual trend of the global production by year in GLP-1RA-related adverse effects described by the average mean of total citations per article and total citations per year for articles indexed in Scopus (B).
Authorship Analysis
A total of 6068 authors contributed to global research on GLP-1RA-related adverse effects. Among the most influential contributors, Schwartzbard, A., achieved the highest h-index (155), followed by Holst, J. (h-index = 143), as well as Nauck, M. (h-index = 142; Table 1). Collaboration measures showed consistent positive associations with impact indicators 3. A strong positive correlation was observed between the number of authors and the total number of citations (r = .814, P < .001; ρ = .792, P < .001), h-index (r = .703, P < .001; ρ = 0.688, P < .001), and g-index (r = .711, P < .001; ρ = 0.692, P < .001). Table 2 shows the top 10 most cited documents on GLP-1RA-related adverse effects -associated publications from the Scopus database.
Top 10 Authors’ Local Impact on GLP-1RA-Related Adverse Effects-Associated Publications from the Scopus Database (2006-2025) Based on the h_index.
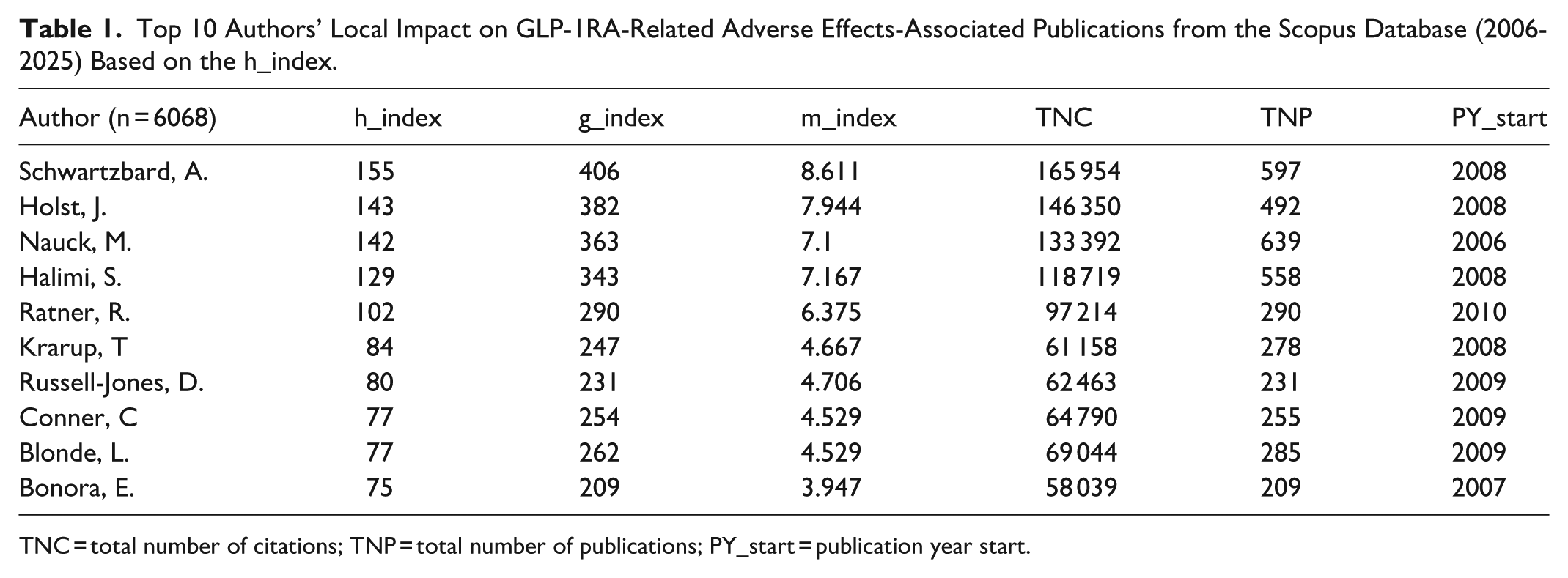
TNC = total number of citations; TNP = total number of publications; PY_start = publication year start.
Top 10 Most Cited Documents on GLP-1RA-Related Adverse Effects -Associated Publications from the Scopus Database (2006-2025).

Country Production Analysis
An analysis of global GLP-1RA-related adverse effects publications revealed contributions from 85 countries, resulting in 34.23% international co-authorships (Table 3 and Figure 2). A bibliometric assessment of global research productivity showed that the United States (USA) leads with 470 publications, accounting for the largest share of output in the dataset. The United Kingdom follows with 164publications, reflecting its rapidly growing contribution to the field. China (135), Denmark (121), and Germany (114) also demonstrate strong research engagement, rounding out the top 5 contributors.
Corresponding Author Countries for GLP-1RA Adverse Effect Publications (2006-2025, Scopus Database).

AAC = average article citations; TNP = total number of publications; SCP = single country publications; MCP = multiple country publications.
Top 10 Journals Published Articles on GLP-1RA-Related Adverse Effects
A total of 389 journals published the 1075 identified articles on GLP-1RA-related adverse effects (Table 4). Diabetes, obesity, and metabolism ranked first, with 106 documents and 4742total citations, followed by Diabetes Care Journal (33 papers) and 6240 citations, and the lancet diabetes and Endocrinology (23 articles, 7549 citations). The number of articles demonstrated a strong correlation with the h-index (r = .677, P < .001; ρ = .662, P < .001). The correlation between the number of articles and the m-index was weak and not statistically significant (r = .169, P = .064; ρ = .142, P = .103), while a strong correlation was observed between the number of articles and total citations (r = .779, P < .001; ρ = .765, P < .001). Across analyses, the Durbin–Watson statistics ranged from 1.69 to 1.91, suggesting no substantial autocorrelation among residuals. Lag-1 autocorrelations of the predictor and response variables ranged between 0.15 and 0.28, indicating mild non-independence but within acceptable limits.
Top 10 Journals on GLP-1RA-Related Adverse Effects -Associated Publications from the Scopus Database By the Number of Local Citations h_index (2006-2025).

TNC = total number of citations; TNP = total number of publications; PY_start = publication year start.
Analysis of the Top 10 Relevant Affiliations or Institution Influence (2006-2025)
A total of 2227 institutions were reported. Many scientific institutions have provided evidence that research institutions have played a crucial role in driving research productivity on GLP-1RA-related adverse effects, as indicated by publications in the Scopus database over the past 2 decades. With 12 as a minimum number of publications for the institution,23 met the criteria. “Novo Nordisk A/S, Bagsvard, Denmark,” had a greater influence on these institutions, with 60 documents with 11 207citations and 57 total link strengths, followed by “Eli Lilly and Company, Indianapolis, United States,” with 45 papers and 9948 citations, and “Københavns Universitet, Copenhagen, Denmark,” with 38 documents and 2637 citations (Table 5).
Analysis of the Top 10 Most Influential Institutions Contributing to Articles on GLP-1RA-Related Adverse Effects (2006-2025).

TLS = total length strength.
Keyword Analysis
According to the co-occurrence analysis shown in Table 6 and Figure 4 via VOS viewer, the total number of identified author keywords was 1490, and the number of keywords plus was 6172. When analyzed the author keyword with minimum occurrence of 25; 25 met the criteria; “type 2 diabetes” appears 225 times with a total link strength (TLS) of 312, followed by “obesity” with 121 occurrences and 211 TLS, then “type 2 diabetes mellitus " with 100 occurrences and 129 TLS, “liraglutide” occurs 78 times with 186 link strength, “semaglutide” arriving 73 times with 165 TLS, the keyword “glp-1” appears 62 times with 103 TLS. Safety occurs 60 times with 106 TLS (Figure 4A).
Co-occurrence of Keywords Through VOS Viewer.


Visualization of keywords (A), keyword plus (B), and overlay visualization of the co-occurrence analysis of keywords plus over the years (C).
In the keyword plus analysis, with minimum occurrences of 355, 25 met the criteria, “human,” with 1038 occurrences, have 12 494 TLSs. The words indicate that “non-insulin dependent diabetes mellitus” occurred in total in 839, with 10 823 TLSs. The words “drug safety” occurred in 820, with TLS 10509. The word “humans” occurred 816 times, with 10 499 TLS (Figure 4B).
Overlay visualization of the co-occurrence analysis of Keywords Plus over the years in Figure 4C shows that new and emerging topics starting in 2021 concern the words “glucagon-like peptide 1 receptor,” “male,” and “female,”” middle-aged”, and “adult.” Figure 5 shows the word cloud of publications on GLP-1RA-related adverse effects (2006-2025).

Word cloud of publications on GLP-1RA-related adverse effects (2006-2025).
Thematic Map Analysis
Thematic mapping of the author keyword, as shown in Figure 6A, showed 4 main themes that represent different stages of conceptual development in the research field. In the Motor Theme, which constitutes 1 cluster, the main topics were “type 2 diabetes mellitus,” “GLP-1 receptor agonists,” and "cardiovascular diseases.”

Thematic map of authors’ keywords (A) and keywords plus (B).
The Niche Theme, with 3 clusters, included specialized but cohesive topics like “type 2,” “sodium-glucose cotransporter 2 inhibitors,” “post-marketing surveillance,” “real-world evidence,” and “Japan”. The 3 clusters in the basic theme included subjects such as “type 2 diabetes,” “obesity,” and "liraglutide”. Lastly, the 1 cluster in the emerging theme featured “endocrinology,” which has low centrality and density, indicating either gaining or losing attention, with fewer studies being conducted on it.
The thematic mapping of the keyword plus identified 2 distinct clusters.
In the Motor Theme, which represents topics that are both well-developed and central to the research field, the dominant cluster comprised “human,” “non-insulin-dependent diabetes mellitus,” and “drug safety.”
The cluster in the emerging theme included general descriptors such as “article,” “female,” and “male.” These represent peripheral or methodological keywords that are either declining in research prominence or serve as background indexing terms rather than conceptual research foci.
No clusters were identified in the niche theme or basic theme, suggesting a concentrated but thematically narrow research landscape within this subset.
Keyword Co-occurrence Network
The first cluster, shown in red, shows the pharmacological and clinical focus of the field. It is made up of terms like drug safety, drug efficacy, antidiabetic agents, glucagon-like peptide-1 receptor agonists, and type 2 diabetes mellitus. The second cluster, shown in blue, includes methodological and demographic descriptors like male, female, adult, aged, and randomized controlled trial. This indicates that human-based clinical studies are the most common type of research.
The connecting nodes of human and non-insulin-dependent diabetes mellitus serve as bridges between these clusters, reflecting how pharmacological investigations are closely linked to patient-level clinical outcomes. This structure underscores a research field that is both clinically grounded and pharmacologically focused, emphasizing real-world safety and efficacy evaluations of GLP-1 receptor agonists across diverse populations (Figure 7).
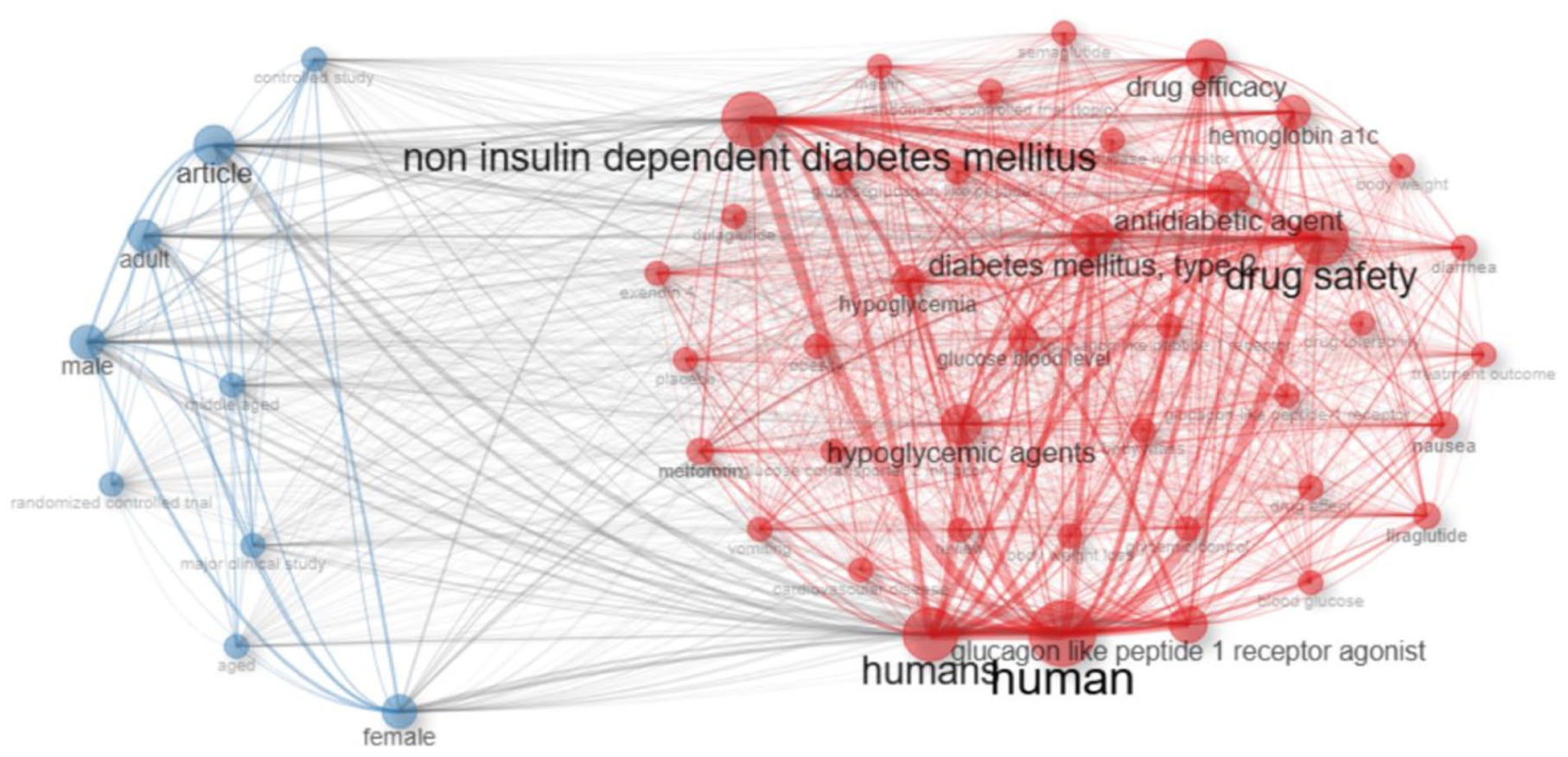
Keyword co-occurrence network of GLP-1 receptor agonist adverse effects research.
Thematic Evolution
The thematic evolution analysis (Figure 8) shows how research themes have changed over time in 5 different periods: 2006 to 2016, 2017 to 2020, 2021 to 2022, 2023 to 2024, and 2025. The thematic evolution map (Figure X) shows how research themes on type 2 diabetes mellitus (T2DM) and its pharmacological management have changed from 2006 to 2025.
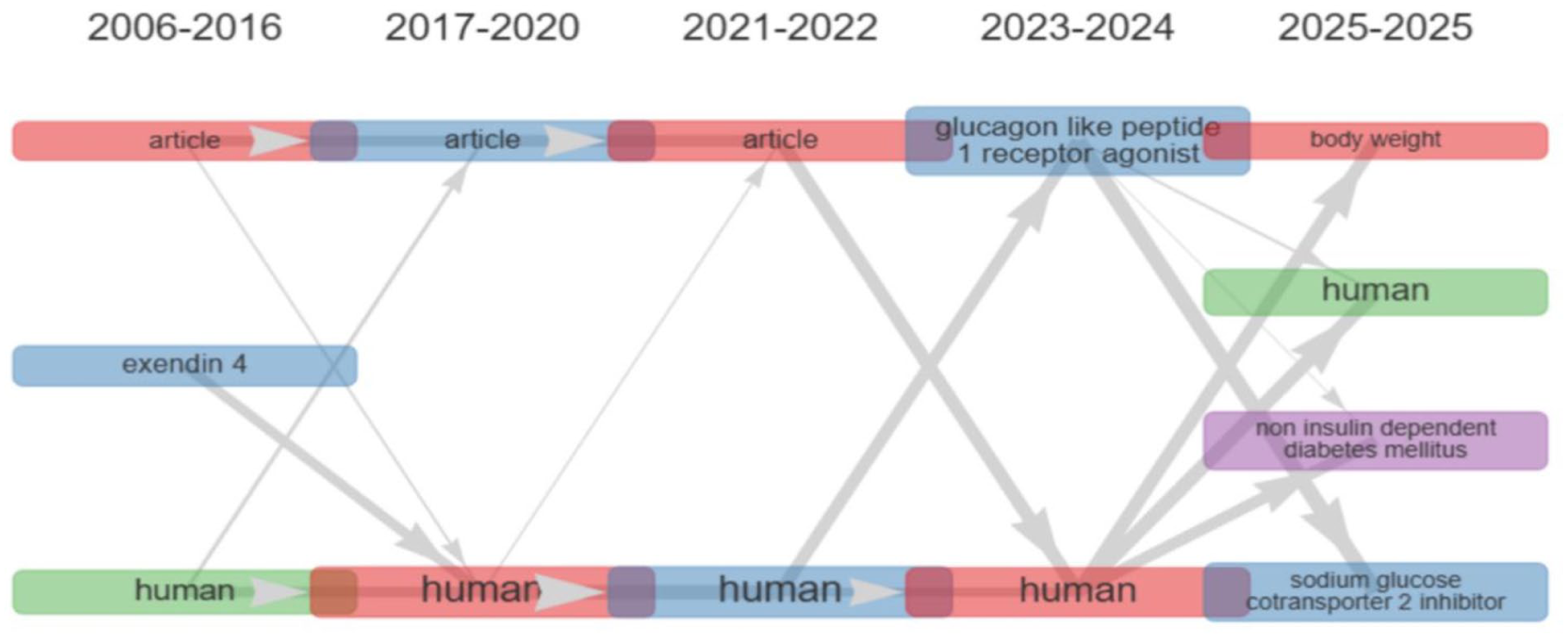
Thematic evolution.
From 2006 to 2016, the main topics were “exendin 4” and “human.” These show that research was focused on the early study of glucagon-like peptide-1 (GLP-1) analogs, especially exenatide, which led to incretin-based medicines.
The keywords “article” and “human” remained the same from 2017 to 2020, indicating an increase in clinical publications and experimental validation. This period also saw more clinical trials and meta-analyses examining the safety and effectiveness of GLP-1 receptor agonists and sodium-glucose cotransporter-2 (SGLT2) inhibitors.
The word “article” remained consistent in 2021 to 2022, indicating that clinical evidence and synthesis efforts were coming together. The word “human” remained unchanged, indicating that the focus on patient-centered outcomes was still strong.
By 2023 to 2024, the concept of “glucagon-like peptide-1 receptor agonist” became a central idea, indicating that significant research was being conducted on incretin-based therapies and cardiovascular outcome trials. The strong connection between the previous “human” and “article” themes suggests that research is moving from experimental studies to translational and clinical applications.
The map for 2025 highlights new themes such as “body weight,” “non-insulin-dependent diabetes mellitus,” and “sodium-glucose cotransporter-2 inhibitor.” These show that research is moving toward integrated metabolic management, cardiorenal outcomes, and obesity-related aspects of T2DM. The word “human” remains present in all periods, indicating that clinical research is still fundamental.
Discussion
Various quantitative systematic reviews and meta-analyses have examined the safety of GLP-1 receptor agonists.6,56 However, to the best of our knowledge, no qualitative or bibliometric analysis has been conducted to explore research trends, thematic evolution, or collaboration networks in this field. The present study represents the first bibliometric analysis to visualize the global research landscape, identify emerging topics, and highlight influential authors, institutions, and collaborative clusters related to GLP-1 receptor agonists’ adverse effects.
This study helps lay the groundwork for new or emerging aspects of the research on GLP-1 receptor agonists’ adverse effects.
This bibliometric analysis provides a comprehensive overview of global research trends on GLP-1 receptor agonists’ adverse effects from 2006 to 2025. The findings demonstrate a highly collaborative research environment, with no single-author publications and widespread international co-authorship. This reflects the multidisciplinary and cross-institutional efforts required to advance understanding of diabetes therapeutics. Strong global research networks and shared scientific priorities underpin the field, particularly in the study of emerging therapeutic classes such as GLP-1 receptor agonists and related agents. The rapid expansion of research in this domain is likely driven by increasing clinical interest, accumulating evidence of cardiometabolic benefits, and the growing role of these therapies in diabetes and obesity management. 57
The results highlight that research productivity and collaboration strongly influence scientific impact. Journals publishing more articles tend to achieve higher citation visibility, suggesting that productivity can enhance research dissemination and perceived quality. Broader authorship networks also correlate with greater citation impact and sustained scholarly influence, reinforcing the lesson that multidisciplinary collaboration contributes to knowledge generation and visibility. 58 However, citation performance is not determined by output volume alone; study quality, topical relevance, and audience reach play critical roles in shaping impact. 59
While higher publication activity is associated with stronger citation metrics such as the h-index and g-index, long-term citation consistency depends on sustained research quality rather than quantity alone. 60 The cumulative relationship between publication volume and total citations indicates that journals maintaining consistent output over time tend to build enduring scientific influence. 61 These findings emphasize that impactful research ecosystems balance productivity with methodological rigor and collaborative engagement. The key lesson is that scientific progress is maximized when high-quality research is disseminated through collaborative networks, ensuring both innovation and broad knowledge transfer.
Autocorrelation diagnostics (Durbin-Watson = 1.69-1.91) indicated the absence of substantial autocorrelation among residuals, suggesting that the regression models adequately satisfied the assumption of independent errors. The corresponding lag-1 autocorrelations of the predictor and response variables (ranging between 0.15 and 0.28) reflected only mild non-independence, remaining well within acceptable analytical limits. 62 These findings affirm the robustness of the correlation estimates and imply that temporal or sequential dependencies among bibliometric indicators, such as publication volume and citation metrics, did not meaningfully bias the observed relationships. This stability supports the reliability of the derived associations in capturing genuine structural patterns within the data rather than artifacts of serial dependence.63,64
The analysis at the country level showed that the United States had the most research output and citation impact, with 30.5% of all publications and the highest citation count (TNC = 42 359). The United Kingdom, China, and Denmark followed, with Denmark having the most international collaboration (MCP% = 47.3%). European countries like Germany, Italy, and France showed strong patterns of cooperative research, while China and Japan had lower rates of collaboration, which shows that research is more focused on their own countries. Even though they had fewer publications, Denmark and Canada had a high citation impact compared to their output, which shows the quality and international visibility of their work. Overall, the data show that research is spread out around the world, but led by the West, with strong global partnerships and growing contributions from Asia. 65
The overall distribution of research output underscores the apparent dominance of high-income countries in this domain, aligning with patterns commonly observed across other areas of clinical pharmacology. 66 This imbalance highlights persistent disparities in research capacity and resource allocation between high-income and low- and middle-income countries (LMICs). Strengthening international collaborative frameworks, fostering equitable funding mechanisms, and promoting capacity-building initiatives within LMICs could help bridge these gaps and contribute to a more globally representative understanding of GLP-1 receptor agonists and their adverse effects. 67
The journal-level analysis revealed that a relatively small number of journals accounted for a substantial proportion of publications and citations related to GLP-1 receptor agonist (GLP-1RA)-associated adverse effects. Diabetes, Obesity, and Metabolism emerged as the leading outlet by publication count. At the same time, Diabetes Care and The Lancet Diabetes & Endocrinology achieved higher citation impact, reflecting their broad readership and reputational influence within the field of endocrinology. The strong correlation observed between the number of articles and both the h-index and total citations indicates that journals with higher productivity tend to maintain a more substantial and sustained impact, likely reflecting selective editorial standards and high research visibility. Conversely, the weak and nonsignificant association between publication count and the m-index suggests that journal productivity alone does not predict consistent long-term citation performance, emphasizing that sustained impact is driven more by research quality and topical relevance than by output volume. The acceptable Durbin–Watson and lag-1 autocorrelation values further confirmed the statistical robustness of these associations, indicating no significant violation of model independence assumptions. 68
Our analysis of the top 10 contributing institutions revealed that industry-affiliated organizations such as Novo Nordisk A/S, Bagsværd, Denmark and Eli Lilly, United States had substantial influence, with 60 and 45 publications, respectively, and high citation counts (11 207 and 9948). While these contributions have clearly advanced research on GLP-1RA-related adverse effects, the strong presence of industry-affiliated institutions may also introduce potential publication or citation bias, potentially shaping research trends and visibility in this field.
Among academic institutions, Københavns Universitet (University of Copenhagen) stood out as a key contributor, reflecting Denmark’s central position in GLP-1RA research. The concentration of output among a few highly productive institutions suggests a field driven by close collaboration between academia and the pharmaceutical industry. This alignment between research output and commercial innovation highlights the translational orientation of the GLP-1RA research domain, where pharmacovigilance and therapeutic optimization remain central priorities.
The author keyword co-occurrence analysis revealed the dominant research themes and evolving focus areas within the literature on GLP-1RA-related adverse effects. The prevalence of terms such as “type 2 diabetes,” “obesity,” and “type 2 diabetes mellitus” underscores the central therapeutic and metabolic contexts in which GLP-1RAs are primarily studied. The frequent occurrence of drug-specific terms such as “liraglutide,” “semaglutide,” and “GLP-1” reflects the clinical emphasis on evaluating individual agents within this pharmacological class, particularly concerning safety and efficacy outcomes. The strong total link strengths (TLS) associated with these keywords suggest a high level of thematic interconnectedness among publications, consistent with integrated research trends in metabolic and cardiovascular endocrinology.
The Keyword Plus analysis highlighted broader and more general biomedical terms such as “human,” “non-insulin dependent diabetes mellitus,” and “drug safety,” indicating that the field is grounded in clinical investigation rather than preclinical experimentation. The high TLS values for these terms reinforce the translational orientation of GLP-1RA research, emphasizing patient-level outcomes and real-world pharmacovigilance. Overlay visualization revealed the emergence of newer descriptors since 2021, such as “glucagon-like peptide 1 receptor,” “male,” “female,” “middle-aged,” and “adult,” suggesting an increasing focus on demographic-specific analyses and mechanistic investigations into receptor biology and gender-based safety profiles. These trends align with a broader movement toward precision medicine and individualized therapeutic strategies in diabetes care. 69
Thematic map analysis highlighted various patterns of conceptual progression within the study domain concerning T2DM and associated pharmacotherapies.
In the author keywords map, Motor Themes like “type 2 diabetes mellitus,” “GLP-1 receptor agonists,” and “cardiovascular diseases” show the main areas of current research, which is in line with the fast progress in clinical and pharmacological therapies that use incretins and lower cardiometabolic risk.70,71 The presence of both “GLP-1 analogs” and “clinical trials” simultaneously highlights the importance of translational and outcomes-based research, aligning with the larger literature that provides evidence from large cardiovascular outcome trials (CVOTs). 35
The Niche Themes (“sodium–glucose cotransporter 2 inhibitors,” “post-marketing surveillance,” “real-world evidence,” and “Japan”) are very specific but still fit together well. These are likely studies that examine the real-world safety and effectiveness of newer antihyperglycemic agents in particular areas, with a focus on pharmacovigilance (98). Even though these themes have a lot of internal density, their limited external centrality suggests that these studies, while detailed, have not yet been fully integrated into the global research discourse.
The Basic Themes, such as “type 2 diabetes,” “obesity,” and “liraglutide,” are the main ideas that make up the field. Their high centrality but low density indicates that they are often cited, though not limited to, one area of research, which is common for general mechanistic or treatment-oriented topics that connect different fields. 72 On the other hand, the Emerging Themes, such as “endocrinology” and “diabetes & endocrinology,” indicate that some subfields are growing or fading, which may suggest that research is shifting from broad endocrinological studies to more pharmacocentric and outcome-driven approaches.
The keyword plus thematic map, on the other hand, shows a more limited research structure, with the Motor Theme cluster (“human,” “non-insulin-dependent diabetes mellitus,” and “drug safety”) being the most important. This suggests that human studies are more focused on clinical safety evaluations, which is in line with the growing regulatory focus on pharmacovigilance and adverse event monitoring for antidiabetic medications.73 -75 The fact that “drug safety” is a central term indicates a greater focus on integrating real-world data into risk-benefit assessments (103). On the other hand, the Emerging Themes (“article,” “female,” “male”) exhibit demographic or indexing-related tags with low conceptual connectivity, indicating they don’t significantly contribute to topic development.
These 2 maps show a change in the way T2DM research is done. It used to be broad and multidimensional, looking at both mechanistic and therapeutic perspectives. Now, it is more focused on patient safety and treatment optimization. This change is in line with what has been seen in global bibliometric analyses of diabetes research, which have shown that pharmacotherapy and post-marketing outcomes have become more specialized since 2015.76 -78
The keyword co-occurrence network illustrates a well-structured and advanced research domain focused on both the pharmacological and clinical aspects of GLP-1 receptor agonist (GLP-1RA) studies. The prominence of keywords associated with “drug safety,” “efficacy,” and “type 2 diabetes mellitus” signifies that the essence of GLP-1RA research continues to concentrate on enhancing therapeutic outcomes while monitoring adverse effects. This clustering pattern reflects the dual clinical objectives of improving glycemic control and reducing treatment-related risks such as hypoglycemia, gastrointestinal disturbances, and weight fluctuation.79,80
Thematic evolution analysis shows a clear and consistent path in T2DM research, moving from molecular pharmacology to clinical validation and finally to holistic metabolic therapy.
During the initial phase (2006-2016), research predominantly involved experimental studies on incretin hormones, especially exendin-4 and other GLP-1 analogs, which established the mechanistic foundation for incretin therapies. 81 This foundational phase facilitated subsequent translational research aimed at clinical applicability.
The intermediate phase (2017-2022) demonstrates the consolidation of clinical evidence, bolstered by extensive trials and real-world studies evaluating the cardiovascular and renal outcomes of GLP-1 receptor agonists and SGLT2 inhibitors. 82 The recurring theme of “human” during this period highlights the field’s ongoing shift toward patient-centered outcomes and safety profiling.
The most recent period (2023-2025) shows a change in the focus of research. The emergence of “body weight” and “non-insulin-dependent diabetes mellitus” themes indicate the incorporation of obesity and metabolic health into T2DM management. This change is in line with current treatment guidelines that stress weight loss and cardiovascular protection as key therapeutic goals. 83
The sustained linkage between “glucagon-like peptide 1 receptor agonist” and “sodium–glucose cotransporter 2 inhibitor” demonstrates the synergistic clinical importance of these 2 drug classes in modern diabetes care. 84
The strengths of this study include, to the best of our knowledge, being the only qualitative and thematic analysis of the literature on GLP-1RA adverse effects. It highlights the extent of scientific research and scholarly contributions over the past 2 decades.
This study has several limitations that warrant consideration. First, the analysis relied exclusively on the Scopus database to retrieve relevant publications. Although Scopus is a comprehensive and widely recognized source for bibliometric studies, restricting the search to a single database may have led to the omission of pertinent studies indexed in other databases such as Web of Science, PubMed, MEDLINE, or Google Scholar. This limitation could have influenced the overall completeness of the dataset and potentially affected the accuracy of the observed research trends related to GLP-1RA adverse effects. Future investigations are encouraged to integrate data from multiple databases to achieve a more exhaustive and balanced bibliometric assessment in this domain.
Second, the restriction to English-language publications may have introduced linguistic bias, omitting pertinent studies published in other languages and consequently underrepresenting specific regional or cultural research contributions. Subsequent bibliometric studies could address these limitations by performing cross-database and multilingual analyses to achieve a more thorough and globally representative comprehension of GLP-1RA adverse effects research trends.
Conclusion and Future Research Directions
This bibliometric analysis provides the first qualitative overview of global research trends on glucagon-like peptide-1 receptor agonist (GLP-1RA) adverse effects between 2006 and 2025. The findings reveal a rapidly expanding and highly collaborative research landscape, with strong international co-authorship and multidisciplinary contributions. However, research activity remains concentrated in high-income countries and major industry–academic networks, underscoring persistent disparities in global research capacity. Addressing these gaps requires enhanced funding mechanisms and capacity-building initiatives in low- and middle-income countries (LMICs) to broaden scientific participation and improve the generalizability of safety evidence. Future bibliometric studies should integrate multiple databases and multilingual search strategies to overcome single-database and language limitations, thereby achieving a more comprehensive assessment of global research output and regional contributions.
Thematic analyses demonstrate an evolution from mechanistic investigations toward translational and real-world evaluations of GLP-1RA safety, with emerging attention to demographic-specific safety profiles and long-term adverse effects. Journals and institutions with sustained productivity exhibit greater citation visibility and scientific influence, highlighting the role of research ecosystems in shaping knowledge dissemination. Nevertheless, important evidence gaps remain, particularly regarding population-specific safety outcomes and rare adverse signals. Future research should employ longitudinal analyses and topic modeling to track thematic evolution and detect emerging safety trends. Comparative studies across incretin-based therapies and pharmacovigilance datasets would further clarify risk–benefit profiles and inform clinical decision-making. Strengthening open-access publishing and data-sharing frameworks will enhance transparency, reproducibility, and equitable access to scientific knowledge, supporting evidence-based policy and therapeutic innovation in GLP-1RA research.
Supplemental Material
sj-docx-1-inq-10.1177_00469580261441498 – Supplemental material for Mapping Global Research on Adverse Effects of GLP-1 Receptor Agonists (2006-2025): A Scopus-Based Bibliometric and Thematic Analysis
Supplemental material, sj-docx-1-inq-10.1177_00469580261441498 for Mapping Global Research on Adverse Effects of GLP-1 Receptor Agonists (2006-2025): A Scopus-Based Bibliometric and Thematic Analysis by Riad Mohammed Abdelrahman, Taha Hussein Musa, Ismail Adam Arbab, Eltieb Omer Ahmed, Sahar Ibrahim Gasmallah, Mohammed Jalal, Wafaa Ramadan Ahmed and Khalid Hamid Fadul in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
The authors would like to express their sincere appreciation to their colleagues at the Faculty of Pharmacy, Libyan International Medical University, for their valuable academic support, insightful discussions, and constructive feedback throughout the development of this manuscript.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available on the websites mentioned in the References section.
The Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the authors used ChatGPT and Grammarly to improve the readability, grammar, and clarity of the text, specifically in the Introduction and Discussion sections. No part of the data analysis, interpretation of results, or generation of original scientific content was performed by any AI tool. Following the use of these tools, the authors thoroughly reviewed, verified, and edited all AI-assisted content to ensure its accuracy and integrity. The authors take full responsibility for the final content and confirm that this manuscript represents their original intellectual contribution.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
