Abstract
This study aimed to identify risk factors for sarcopenia and develop a nomogram to predict its probability among community-dwelling older adults. This cross-sectional secondary data analysis used the 2023 Korea National Health and Nutrition Examination Survey, which employed a stratified multistage clustered probability sampling method. Of the 6929 respondents, 1836 community-dwelling adults aged 65 years and older were included. Complex-sample logistic regression was used to identify significant risk factors, and a nomogram was developed using the resulting regression coefficients. Predictive accuracy was evaluated using the area under the receiver operating characteristic curve (AUC). Sarcopenia was significantly associated with the following risk factors: age, kidney disease, obesity status, chewing discomfort, and sedentary time. Compared to adults aged 65 to 69 years, those aged 75 to 79 had 2.80 times higher odds, and those aged 80 years and older had 5.01 times higher odds. Participants with kidney disease had 3.29 times higher odds than those without. Compared to obese individuals, the odds of sarcopenia were 17.04, 6.80, and 2.72 times higher for underweight, normal weight, and overweight individuals, respectively. Chewing discomfort increased the odds by 1.65 times, and each additional hour of sedentary time increased the risk by 7%. The nomogram included obesity, sedentary time, age, kidney disease, and chewing discomfort, with an AUC of 0.809. Obesity, sedentary behavior, age, kidney disease, and chewing discomfort were key factors affecting the risk of sarcopenia in older adults. The developed nomogram provides a practical tool for early identification of high-risk community-dwelling older adults. Future research should incorporate longitudinal data and objective indicators to enhance predictive models and validate the tools in practice.
This study used nationally representative 2023 Korea National Health and Nutrition Examination Survey data and complex-sample logistic regression to identify risk factors for sarcopenia and construct a predictive nomogram among community-dwelling older adults.
Sarcopenia risk was strongly associated with both non-modifiable factors (older age, chronic kidney disease) and modifiable factors (non-obese BMI categories, prolonged sedentary time, chewing discomfort), with underweight status showing the highest odds of sarcopenia.
The nomogram, incorporating obesity status, sedentary time, age, kidney disease, and chewing discomfort, demonstrated good discrimination (AUC = 0.809) and provides a practical tool for early identification and preventive management of sarcopenia in community and primary care settings.This study used nationally representative 2023 Korea National Health and Nutrition Examination Survey data and complex-sample logistic regression to identify risk factors for sarcopenia and construct a predictive nomogram among community-dwelling older adults.
Introduction
The global population is aging rapidly. South Korea will become a super-aged society by 2025, reflecting one of the fastest demographic transitions worldwide. 1 This shift increases the need for strategies that maintain health, independence, and functional capacity among older adults. 2 Promoting health in later life aligns with the World Health Organization’s (WHO) concept of healthy aging, which emphasizes maintaining functional ability and participation rather than merely preventing disease. 3 Sarcopenia is a degenerative condition marked by progressive loss of skeletal muscle mass and strength.4,5 Its prevalence among older adults varies widely, ranging from 5.5% to 25.7% in Asian populations 4 and 4% to 45% in studies conducted in rapidly aging societies, 5 depending on diagnostic criteria. Despite increasing awareness, sarcopenia remains underrecognized.6-9
The WHO has recognized sarcopenia as a disease affecting older adults. 3 It impairs mobility and independence and contributes to frailty, falls, fractures, disability, depression, and higher mortality.9-14 Meta-analyses confirm a significant association between sarcopenia and all-cause mortality,15,16 underscoring the importance of early detection and prevention. Effective management of sarcopenia not only prevents adverse health outcomes but also supports physical autonomy and social participation, which are central to healthy aging. 4
Sarcopenia develops through interactions between physiological aging and behavioral/lifestyle factors.17-20 Non-modifiable factors include age and chronic conditions such as chronic kidney disease, whereas modifiable factors include low physical activity, inadequate nutrition, and chewing difficulty.18-22 Muscle mass typically begins to decline after age 60, with up to 30% to 50% lost by age 70.19,21 Addressing modifiable factors through early screening and preventive interventions is essential to delay functional decline and reduce the societal burden of aging.
Despite extensive research,5,19-22 few studies have developed predictive models integrating multiple risk factors for sarcopenia among community-dwelling older adults. In particular, studies focusing on community-dwelling older adults remain limited. 2 Most existing studies are limited to small-scale or single-population samples, leaving uncertainty regarding their generalizability. Community-dwelling older adults often rely on self-management and possess varying levels of health literacy,²² which complicates the early detection of sarcopenia. 23 Generating population-based evidence on key risk factors can enhance global understanding of sarcopenia prevention strategies and inform cross-cultural comparisons in aging research.
Therefore, this study aimed to identify key risk factors and develop a nomogram predicting the probability of sarcopenia among community-dwelling adults aged 65 years and older, using a large-scale population health survey. A nomogram is a visual predictive tool that integrates multiple variables to estimate disease probability and can be easily applied in clinical and community healthcare.24,25 It is expected to support early identification of high-risk older adults and guide personalized preventive interventions. These findings may inform nursing practice, clinical decision-making, and public health policy aimed at promoting healthy aging in diverse populations.
Methods
Research Design
This cross-sectional secondary data analysis used the 2023 Korea National Health And Nutrition Examination Survey (KNHANES) to identify risk factors for sarcopenia among community-dwelling older adults aged 65 years and older and develop a nomogram for predicting the probability of sarcopenia.
Samples Used for Analysis
The KNHANES has been conducted since 1998 under the supervision of the Korea Disease Control and Prevention Agency (KDCA) to assess health status, nutritional intake, and prevalence of major chronic diseases among the Korean population. Data from the 2023 survey cycle were used for this study. Subjects were recruited using a stratified multistage clustered probability sampling method, taking into account both region and household type. To ensure statistically representative sampling, the first stage was to select the primary sampling units, and the second stage was to select the final sample households using systematic sampling. Of the 6929 participants in the 2023 KNHANES, only those aged 65 years and older were selected for the present study, resulting in an analytic sample of 1836 community-dwelling older adults. Participants within this subgroup who had missing data on skeletal muscle mass, handgrip strength, or any covariates were excluded using listwise deletion. All analyses applied complex-sample weights to maintain national representativeness.
Measures
Sarcopenia
According to the criteria established by the Asian Working Group for Sarcopenia (2019), 4 sarcopenia is defined as the coexistence of reduced muscle mass and decreased handgrip strength. Muscle mass was assessed using bioelectrical impedance analysis, with appendicular skeletal muscle mass (excluding bone mineral content) divided by height squared (ASM/height 2 ). Sarcopenia was diagnosed when the ASM/height 2 was less than 7.0 kg/m² for men and less than 5.7 kg/m² for women. Handgrip strength was measured using a dynamometer, with diagnostic thresholds of <28 kg for men and <18 kg for women.
Socio-Demographic Characteristics
Sociodemographic variables included age, sex, income quartile, educational level, living arrangement (living alone or not), and type of health insurance. These variables were categorized for analysis. Age was classified into 4 groups: 65 to 69, 70 to 74, 75 to 79, and 80 years old.
Health-Related Lifestyle Factors
Health-related lifestyle variables included perceived health status; diagnoses of hypertension, diabetes, or dyslipidemia; presence of kidney disease; current smoking status; history of alcohol consumption; number of walking days per week; engagement in aerobic physical activity; obesity status; reported chewing difficulties; sedentary time; and health literacy. All variables were categorized for analysis. Perceived health status was classified as good, fair, or poor. Obesity status was categorized as underweight, normal-weight, pre-obese, or obese.
Data Analysis
Data were analyzed using SPSS/WIN version 26.0 (IBM Corp., Armonk, NY, USA). A complex sample design file was constructed by defining the stratification variables and primary sampling units (PSUs) based on the 2023 KNHANES sampling structure and applying integrated sample weights to ensure national representativeness. Accordingly, all statistical analyses were performed using complex-sample procedures. Participants’ sociodemographic and health-related lifestyle characteristics were analyzed using unweighted frequencies, weighted percentages, weighted means, and standard errors. Differences in sarcopenia prevalence according to these variables were examined using the Rao–Scott chi-square test and complex-sample general linear model t-tests. To identify the key risk factors for sarcopenia among community-dwelling older adults, complex sample binary logistic regression analysis was performed. Variables with a significance level of P < .05 in the univariate analyses were included in the multivariate model. A nomogram was subsequently developed based on the β-coefficients obtained from the final multivariable logistic regression model. Model performance was evaluated using the receiver operating characteristic (ROC) curve and the area under the curve (AUC) to assess discrimination, and the Hosmer–Lemeshow goodness-of-fit test to verify calibration.
Ethical Considerations
This study was a secondary data analysis and was exempted from review by the Institutional Review Board (IRB) of the researchers’ affiliated institution (JBNU 2024-11-017). The raw data used in this study were obtained after receiving approval for use from the KNHANES website. Anonymized data with unique identification numbers were provided in accordance with data disclosure regulations.
Results
General Characteristics of the Participants
Most of the participants in this study (34.7%) were aged 65 to 69 years old. Women accounted for 56.1% of the sample. Regarding socioeconomic status, 26.8% were in the highest income quartile, and the majority (45.6%) had an education level of elementary school or below. A total of 72.6% were not living alone, and 92.0% were covered by the National Health Insurance Service.
In terms of health-related characteristics, 46.9% of participants perceived their health status as average. The prevalence rates of hypertension, diabetes, dyslipidemia, and kidney disease were 56.6%, 22.7%, 45.4%, and 3.9%, respectively. Current smokers accounted for 9.9% of the sample, and 75.0% reported having consumed alcohol at some point in their lives. Approximately 33.0% of the participants engaged in aerobic physical activity. The average sedentary time per day was 8.81 h, and participants walked an average of 4.22 days per week. In total, 35.5% of the sample was classified as obese. Additionally, 29.6% reported experiencing chewing discomfort, and the average health literacy score was 26.47 out of 40 (Table 1).
Demographic Characteristics of the Subjects (N = 1836).

M = mean; SE = standard error.
Unweighted count.
Weighted %.
Differences in Sarcopenia by Participant Characteristics
Significant differences in sarcopenia prevalence were observed according to general characteristics, such as age (P < .001), educational level (P = .024), living arrangement (P = .038), and type of health insurance (P = .005). In terms of health-related factors, statistically significant differences were found in diabetes (P = .028), kidney disease (P = .008), sedentary time (P < .001), obesity status (P < .001), chewing discomfort (P = .008), and health literacy scores (P = .003). Specifically, the non-sarcopenia group had the highest proportion of individuals aged 65 to 69 years, whereas the sarcopenia group had the highest proportion of individuals aged 80 years and older. Compared to the non-sarcopenia group, individuals with sarcopenia were significantly more likely to have an elementary school education or less, to live alone, and to be covered by medical aid. Additionally, the sarcopenia group showed higher prevalence rates of diabetes and kidney disease and greater chewing discomfort. They also had a longer average daily sedentary time. By contrast, the non-sarcopenia group had higher obesity rates and better health literacy scores (Table 2).
Differences in Factors According to Sarcopenia (N = 1836).
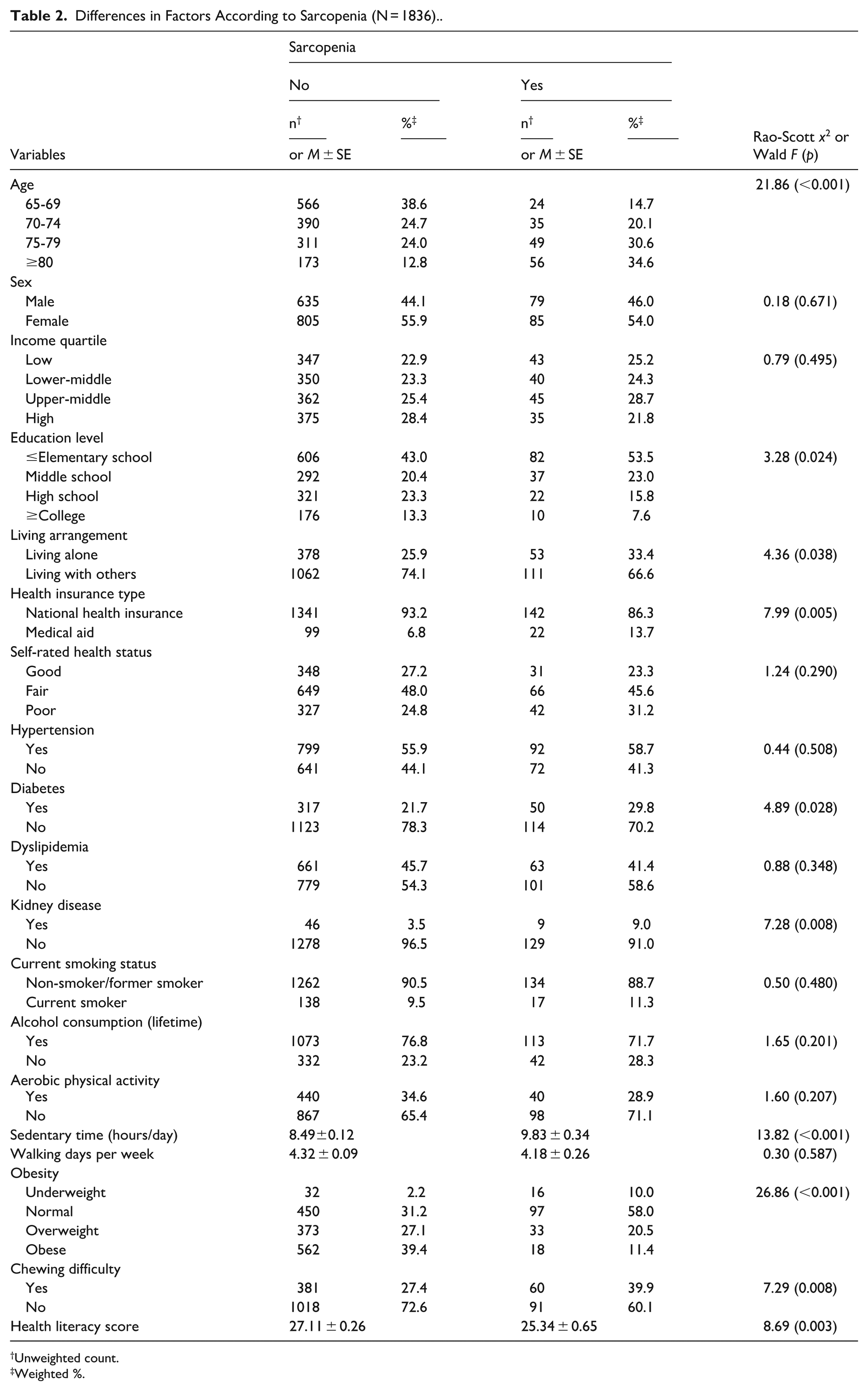
Unweighted count.
Weighted %.
Factors Affecting Sarcopenia in Older Adults
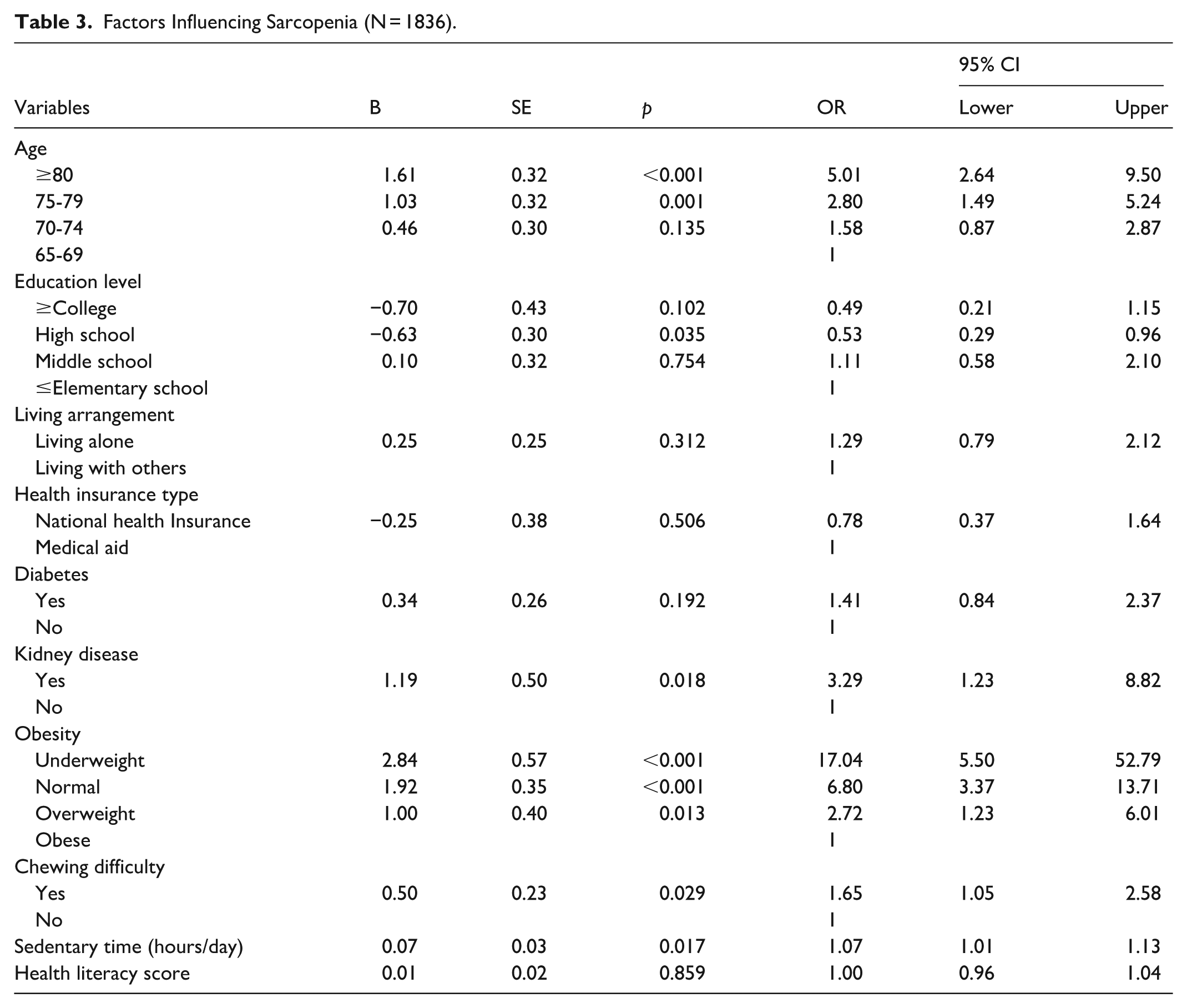
Factors influencing sarcopenia were identified using complex sample logistic regression analysis. Compared to individuals aged 65 to 69 years, those aged 75 to 79 years had 2.80 times higher odds of having sarcopenia (95% CI = 1.49-5.24), and those aged 80 years and older had 5.01 times higher odds (95% CI = 2.64-9.50). Although the overall model effect for educational level was not statistically significant (P = .082), post-hoc comparisons showed that individuals with a high school diploma had 0.53 times lower odds of sarcopenia compared to those with elementary school education or less (95% CI = 0.29-0.96).
Participants diagnosed with kidney disease had 3.29 times higher odds of sarcopenia than those without a diagnosis (95% CI = 1.23–8.82). Regarding obesity status, compared to the obese group, the odds of sarcopenia were 2.72 times higher in the overweight group (95% CI = 1.23–6.01), 6.80 times higher in the normal weight group (95% CI = 3.37–13.71), and 17.04 times higher in the underweight group (95% CI = 5.50-52.79). Additionally, those who reported chewing discomfort had 1.65 times higher odds of sarcopenia than those without discomfort (95% CI = 1.05-2.58). Each 1-h increase in daily sedentary time was associated with a 1.07 times higher likelihood of sarcopenia (95% CI = 1.01-1.13; Table 3).
Factors Influencing Sarcopenia (N = 1836).
Sarcopenia Nomogram
A nomogram was developed using variables significantly associated with sarcopenia to quantify the influence of each factor as a point score. The total score, calculated by summing the individual points, was used to estimate the probability of sarcopenia using a probability scale. Among the included variables, obesity status had the widest score range (0-100), indicating the highest discriminatory power. This was followed by sedentary time (0-65 points), age (0-63 points), kidney disease (0-30 points), and chewing discomfort (0-25 points; Figure 1). To assess how well the predicted values based on the nomogram aligned with actual sarcopenia cases, a receiver operating characteristic (ROC) curve was plotted, and the area under the curve (AUC) was calculated. The resulting AUC was 0.809, indicating good predictive accuracy (Figure 2). In addition, the Hosmer–Lemeshow goodness-of-fit test showed no significant difference between observed and predicted probabilities (χ² = 13.36, P = .100), confirming adequate model calibration.

Nomogram predicting the probability of sarcopenia.

ROC curve showing the predictive performance of the nomogram for sarcopenia.
Discussion
This study utilized data from the 2023 KNHANES to identify risk factors for sarcopenia in community-dwelling older adults and develop a predictive nomogram based on significant risk factors. The results revealed that age, kidney disease, obesity, chewing difficulty, and sedentary time were significantly as sociated with sarcopenia. These findings align with those of previous studies,26,27 suggesting that sarcopenia does not result from a single physiological cause, but rather from a complex interplay of chronic diseases, functional decline, and lifestyle factors.
The risk factors identified in this study can be broadly categorized as non-modifiable factors (such as age and chronic kidney disease) and modifiable factors (such as obesity status, sedentary behavior, and chewing difficulty). This distinction highlights the importance of preventive interventions targeting lifestyle modification, while emphasizing the need for continuous management in individuals with chronic diseases or advanced age.
Among the non-modifiable factors, aging showed a strong association with sarcopenia risk. The present study found that individuals aged 75 to 79 years had a 2.80 times higher risk, and those aged 80 years and older had a 5.01 times higher risk than those aged 65 to 69 years. These findings suggest that sarcopenia may accumulate over time and that functional deterioration becomes more pronounced beyond a certain age threshold. Aging has been identified as a major risk factor for reduced muscle quality and functional impairment. 28 In addition, predictive modeling has shown that age serves as a key variable explaining the onset of sarcopenia, reinforcing the current findings. 29 Aging is accompanied by various physiological changes, including reduced skeletal muscle fibers, increased intramuscular fat infiltration, hormonal alterations, and diminished regenerative capacity of muscle tissue.28,30 These mechanisms collectively contribute to the pathophysiology of sarcopenia. Therefore, among community-dwelling older adults, early screening and health education for sarcopenia should be implemented alongside targeted nursing interventions that focus on functional assessment and fall prevention, particularly for those aged 75 and older. Structured exercise programs and nutritional strategies aimed at preserving muscle mass should be systematically reinforced to support healthy aging in this population.
In addition, individuals diagnosed with chronic kidney disease (CKD) exhibited a significantly higher risk of developing sarcopenia. This finding aligns with previous research demonstrating notable reductions in muscle mass and strength among dialysis patients with kidney disease, highlighting the role of CKD in directly affecting muscle metabolism and protein balance.31-33 As kidney function deteriorates, increased permeability of the glomerular filtration barrier can lead to proteinuria, resulting in the continuous loss of essential proteins from the body. This loss impairs muscle protein synthesis and induces a negative nitrogen balance, ultimately contributing to skeletal muscle depletion and reduced muscle strength.32,33 Given these mechanisms, older adults with CKD face compounded risks, such as restricted protein intake, decreased appetite, and reduced physical activity. Therefore, a multidisciplinary approach, including registered dietitians, is recommended in community-based nursing practice. Regular monitoring of body composition and nutritional status and personalized dietary and exercise interventions that consider protein balance are essential to mitigate the elevated risk of sarcopenia in this vulnerable population.
Among the modifiable factors, obesity status showed the strongest association with sarcopenia risk. The prevalence of sarcopenia was lowest among obese individuals, whereas the risk increased progressively in the overweight, normal-weight, and underweight groups. These findings are consistent with previous studies suggesting the existence of the “obesity paradox,” whereby being overweight or mildly obese may offer a protective effect on muscle function.29,34,35 However, because BMI alone does not adequately reflect muscle quality or composition, combining BMI with indices such as skeletal muscle mass or fat-free mass can improve the predictive accuracy of sarcopenia risk models. 36 From a clinical nursing perspective, older adults with low BMI should be recognized as a high-risk group for sarcopenia. Individualized nutrition and resistance exercise interventions should be implemented to preserve muscle mass and promote healthy weight gain. Furthermore, community-based education and early screening initiatives are essential to enhance awareness and facilitate early identification of sarcopenia risk, particularly among underweight older adults. 37
In addition to obesity, self-reported chewing difficulty was also significantly associated with sarcopenia, emphasizing the critical link between oral health, nutrition, and muscle maintenance. Masticatory dysfunction can negatively influence dietary quality and energy intake, ultimately accelerating muscle loss in older adults (Kugimiya et al). 38 Therefore, community and clinical nurses should monitor oral health and nutritional status to identify individuals at risk and provide timely interventions, including referrals to dental and nutritional care services.
Prolonged sedentary behavior was another significant modifiable factor associated with increased sarcopenia risk. Extended sitting time reduces muscle activity and energy expenditure, contributing to impaired mobility and metabolic dysregulation.39-43 Nurses can play a key role in promoting behavior modification strategies, such as encouraging frequent posture changes, light-intensity activities, and regular walking. Incorporating digital health tools to monitor activity levels or sedentary time may further improve adherence to physical activity recommendations and enhance the effectiveness of sarcopenia prevention programs. 22
Beyond clinical practice, the developed nomogram may be integrated into digital health tools or mobile applications that facilitate self-assessment among older adults. Such integration could enable continuous monitoring of sarcopenia risk and support personalized preventive interventions in both community and home-care settings. The incorporation of this predictive tool into electronic health record systems could further assist healthcare professionals in identifying high-risk individuals and tailoring interventions accordingly.
A key strength of this study lies in its use of nationally representative data from the 2023 KNHANES, which enhances the reliability and generalizability of the findings. By integrating both modifiable and non-modifiable risk factors, the study provides a practical framework for early identification and prevention of sarcopenia among community-dwelling older adults. Furthermore, the use of a nomogram offers a visually intuitive and clinically applicable approach that can be easily adopted in public health and nursing practice.
Nevertheless, several limitations should be acknowledged. The cross-sectional nature of the data precludes causal inference, and several variables, including sedentary time, chewing difficulty, and health behaviors, were based on self-reported data, which may introduce recall bias. Additionally, although internal validation demonstrated good predictive accuracy (AUC = 0.809), external validation using independent datasets was not performed, which may limit the generalizability of the model to different populations.
Future research should employ longitudinal study designs and incorporate objective indicators such as physiological biomarkers, wearable activity data, and detailed dietary intake measures to enhance predictive precision. Further validation and refinement of the developed nomogram in various settings—including hospitals, community centers, and long-term care facilities—will be essential to establish its clinical utility. Ultimately, integrating such predictive models into digital and nursing care platforms could play a pivotal role in promoting healthy aging and preventing sarcopenia among community-dwelling older adults.
Conclusions
This study used data from the 2023 Korea National Health and Nutrition Examination Survey (KNHANES) to analyze risk factors associated with sarcopenia among community-dwelling older adults living in South Korea and provide the foundation for constructing a predictive nomogram. The analysis identified obesity, sedentary time, age, kidney disease, and masticatory discomfort as significant risk factors for sarcopenia. Therefore, to facilitate early prediction and management of sarcopenia in this population, public health nurses should assess individuals’ overall health status, including oral health, and monitor for potential nutritional imbalances related to aging. Furthermore, it is essential to establish effective collaborative systems with local community resources to support appropriate physical activity interventions. Based on the findings of this study, the following recommendations are proposed: first, future research should adopt longitudinal designs and incorporate objective indicators such as physiological biomarkers, physical activity tracking, and dietary intake records to enhance the predictive accuracy of models. Second, empirical studies are needed to assess the validity and effectiveness of the nomogram developed in this study by applying it to real-world settings, such as public health institutions.
Footnotes
Author Contributions
Conceptualization, SKP, HJK, YML, and HYK; methodology, SKP, HJK, YML, and HYK; formal analysis, SKP & HJK; investigation, and data curation, SKP & HJK; writing—original draft, SKP, HJK, YML, and HYK; writing—review and editing, SKP, HJK, YML, and HYK; visualization, HJK. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National University Development Project at Jeonbuk National University in 2024.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional Review Board Statement
This study was performed after receiving approval from the Institutional Review Board (IRB) of the Jeonbuk National University to which the researcher belongs (JBNU 2024-11-017).
