Abstract
The availability of direct-to-consumer, at-home medical tests has grown over the last decade, but it is unknown how frequently older adults purchase at-home tests, how they perceive such tests, and how interested they are in using at-home tests in the future. We conducted a cross-sectional, nationally representative survey of non-institutionalized US adults aged 50 to 80 about their previous use of, perceptions of, and future intentions to use at-home medical tests. We found that nearly half of older adults (48.1%) have purchased an at-home medical test (95% CI 45.2%-51.0%), including 32.0% (95% CI 29.3%-34.8%) who purchased a COVID-19 test, 16.6% (95% CI 14.7%-18.7%) who purchased an at-home DNA or genetic test, 5.6% (95% CI 4.5%-7.0%) who purchased a screening test for cancer, and 4.4% (95% CI 3.4%-5.6%) who purchased a test for an infection other than COVID-19. Compared with White, non-Hispanic adults, Black, non-Hispanic adults were less likely to have purchased an at-home test (35.5% vs 49.6%, P < .01). Those with a college degree and those with an annual household income greater than $100K were more likely than others to have purchased at-home tests (55.5% vs 42.0%, P < .01; 60.6% vs 39.0%, P < .001, respectively). Most older adults had positive perceptions about at-home tests and expressed interest in using at-home tests in the future. At-home medical testing is now common among older adults. Clinicians should be familiar with different tests that patients can purchase and be prepared to discuss the potential advantages and disadvantages of at-home testing.
The availability of direct-to-consumer, at-home medical tests has grown over the last decade and increased during the COVID-19 pandemic.
This study presents the first national estimates for purchase of, perceptions of, and interest in future use of at-home medical tests among older US adults.
Clinicians should be prepared to discuss the potential advantages and disadvantages of at-home tests with their patients; and when these tests are reliable and serve a potential public health benefit, policymakers, insurers, and healthcare systems should consider the racial, ethnic, and socioeconomic differences highlighted in this research.
Introduction
Early restrictions due to the COVID-19 pandemic limited access to routine primary care services and yet, also necessitated a way to provide patients with access to rapid, reliable medical tests that have historically been offered via primary care. 1 In 2021 the Food and Drug Administration (FDA) granted emergency use authorization (EUA) to several companies to sell and market at-home COVID-19 antigen tests. 2 In January 2022, the Biden administration began making COVID-19 test kits freely available to every US household, marking a massive, nationwide initiative to increase direct-to-consumer, at-home testing. 3
Throughout the US, individuals can now purchase a variety of screening, diagnostic, and other informational medical tests directly from pharmacies, supermarkets, and online companies without the assistance of a clinician. Using saliva, respiratory secretions, urine, or blood, individuals can test for a variety of conditions and biomarkers of disease such as urinary tract infections, sexually transmitted infections, hormone levels, certain cancers, and genetic polymorphisms. 4 Some of these tests can be conducted completely at home (eg, COVID-19 antigen tests) and others require a self-collected specimen to be mailed to outside companies and laboratories for testing (eg, direct-to-consumer genetic tests). Although different, common to these various types of at-home tests is their potential to quickly provide individuals with information that can impact the health and well-being of themselves and others.5 -8
However, numerous concerns have been raised about the use of at-home medical tests. For example, the validity, reliability, and utility of at-home tests is often uncertain.9 -12 Some individuals may have difficulty understanding the implications of their results.13 -15 Because at-home tests depart from the traditional model of medical testing within an established patient-clinician relationship, when at-home tests are used clinicians can be excluded from health-related evaluations and decisions. This can result in a range of unintended consequences16,17 including a potential cascade of increased costs to patients and healthcare systems. 6 Older adults, who on average have a greater number of medical conditions than other populations, 18 may be particularly vulnerable to such negative consequences.
Despite these concerns, little is known about how many patients have purchased at-home tests and their perceptions of these tests. Existing studies have generally focused on a single type of at-home test, 19 have not specifically addressed use among older adults, 20 or have focused on a different demographic of high-risk patients. 21 We conducted a nationally representative survey to better understand how many older US adults have purchased at-home tests, their perceptions of these tests, how often they shared their results with their primary care clinicians, and their intentions to use at-home tests in the future.
Methods
Study Design
This study was conducted through the University of Michigan National Poll on Healthy Aging (NPHA). The NPHA is a recurring, cross-sectional survey of US adults aged 50 to 80 years on health and health care issues affecting older adults. 22 Samples are drawn from the NORC AmeriSpeak Panel®, a probability-based panel designed to be representative of the non-institutionalized, US population aged 50 to 80. 23 Panel participants can complete surveys online or by phone. Data were collected in January 2022. Overall, 2163 respondents completed the survey (1971 by web and 192 by phone). Median survey duration was 12 min. The survey completion rate was 75.2%. This study was determined to be exempt by the University of Michigan Institutional Review Board.
Survey Instrument
Participants were asked “Have you ever bought an at-home test for COVID-19?” and “Have you ever bought any other types of at-home medical tests?” Options for other types of at-home tests included: (1) test for an infection other than COVID-19 (such as HIV, urinary tract infection); (2) screening test for cancer (such as colon cancer, prostate cancer); (3) DNA or genetic test (such as 23andMe, Ancestry.com); (4) hormone test (such as testosterone, menopause); (5) food sensitivity test, (6) allergy test; or (7) some other test. Participants were asked about purchase of at-home tests rather than use of at-home tests in order to draw a distinction from any tests ordered by a doctor, those covered by insurance and the free COVID-19 test kits made available by government agencies and community organizations. Home monitoring tests such as ambulatory blood pressures and glucose monitoring were excluded. Participants who had purchased an at-home test were asked if they shared their results with their primary care provider. All participants were asked how interested they were in using each type of at-home test in the future. To assess participants’ overall perceptions of at-home tests, they were asked to indicate their level of agreement that at-home tests “are more convenient than tests through my health care providers”; “can be trusted to give reliable results”; “are regulated by the government”; “are a good value”; and “should be discussed with my doctor.” The full survey instrument is available as a Supplemental Material.
Sociodemographic and Health Outcome Measures
Data on sociodemographic characteristics were collected by NORC from AmeriSpeak® panel participants and assigned to categories for age group, gender, race and ethnicity, marital status, education, and annual household income. Data on self-reported physical health status and mental health status were also collected.
Statistical Analysis
Multivariable logistic regression models were estimated to measure associations between respondents’ age, gender, race/ethnicity, income, education, marital status, and health status with the following dependent variables: any previous purchase of an at-home medical test, any previous purchase of specific types of at-home tests, overall perceptions of at-home tests, and interest in future use of different types of at-home tests. Given the relatively low prevalence of purchase of allergy, food sensitivity, and hormone tests, purchase of these tests was combined into a single variable for the models estimating previous purchase of at-home tests and future interest in at-home tests. Previous purchase of any at-home test was added as an independent variable in the models to predict overall perceptions of at-home medical tests and interest in future use. The primary outcomes are reported as marginal estimates of the models. Frequencies of sharing results of at-home tests with primary care providers are reported as unadjusted bivariate associations due to relatively small sample sizes for previous purchase of some types of at-home tests. Responses for level of interest in future use of different types of at-home tests were dichotomized as being interested or not interested in each test type. Responses to statements about at-home test perceptions were dichotomized into agree or disagree. All analyses used sample weights to yield nationally representative estimates. Missing or “no answer” responses were excluded. All analyses were performed using STATA 17.
Results
Purchase of At-Home Medical Tests
Estimates of population characteristics are shown in the Supplemental Material. Nearly half of adults aged 50 to 80 (48.1%) reported having purchased any type of at-home medical test (95% CI 45.2%-51.0%, Table 1). Almost one-third (32.0%, 95% CI 29.3%-34.8%) had purchased a COVID-19 test, 16.6% (95% CI 14.7%-18.7%) had purchased an at-home DNA or genetic test, and 5.6% (95% CI 4.5%-7.0%) had purchased a screening test for cancer. Few older adults (4.4%, 95% CI 3.4%-5.6%) had purchased a test for an infection other than COVID-19, and 7.8% (95% CI 6.3%-9.5%) had purchased some other type of at-home test (eg, a hormone, allergy, or food sensitivity test).
Adjusted Prevalence of Purchase of Different Types of at Home Medical Tests.

Note. Prevalences reported as marginal estimates with 95% confidence intervals. Models also adjusted for self-reported mental health, physical health, and marital status.
Unadjusted, overall weighted estimate with 95% CI.
P < .05. **P < .01. ***P < .001.
In multivariable analyses, Black, non-Hispanic adults were less likely to have purchased any type of at-home test compared to White, non-Hispanic adults (35.5% vs 49.6%, P = .006). Black, non-Hispanic adults were also less likely to have purchased a test for an infection other than COVID-19 (0.5% vs 4.9%, P < .001) and a DNA/genetic test (9.6% vs 17.3%, P = .026). Older adults with a college degree (vs high school or less) and those with a household income greater than $100 000 (vs less than $30 000) were more likely to have purchased any type of at-home test (55.5% vs 42.0%, P = .001; 60.6% vs 39.0%, P < .001, respectively). Similar differences in purchase of an at-home tests by education and income were observed for COVID-19 tests (36.4% vs 28.5%, P = .034; 44.9% vs 24.5% P < .001, respectively) and DNA/genetic tests (23.6% vs 9.6%, P < .001; 21.9% vs 12.1% P = .011, respectively). Women were more likely than men to have purchased an at-home test for an infection other than COVID-19 (5.6% vs 2.7%, P = .016).
Perceptions of At-Home Medical Tests
Most older adults agreed that at-home medical tests are more convenient than tests through their health care provider (75.1%, 95% CI 72.5%-77.6%), can be trusted to give reliable results (59.9%, 95% CI 57.1%-62.8%), are regulated by the government (54.8%, 95% CI 51.9%-57.7%), and are a good value (66.0%, 95% CI 63.2%-68.7%). In multivariable analyses, Black, non-Hispanic and Hispanic adults were less likely than White, non-Hispanic adults to agree that at-home tests are more convenient (65.2% vs 78.4%, P = .002, 66.9% vs 78.4% P = .002, respectively), but Black, non-Hispanic adults were more likely than White, non-Hispanic adults to agree that at-home tests are a good value (76.7% vs 65.0%, P = .012). Those with some college or at least a bachelor’s degree were more likely than those with high school education or less to agree that at-home tests are more convenient (77.4% and 82.4% vs 68%, P = .002 and P < .001, respectively). Those with annual household incomes of at least $100 000 were more likely to agree that at-home tests are more convenient (80.7% vs 71.9%, P = .043) and are a good value (71.2% vs 61.2%, P = .04). Compared with those who had not previously purchased an at-home test, those who had previously purchased an at-home test had significantly higher prevalence of agreement with each of the perception statements (Table 2).
Adjusted Prevalence of Perceptions About At-Home Medical Tests.
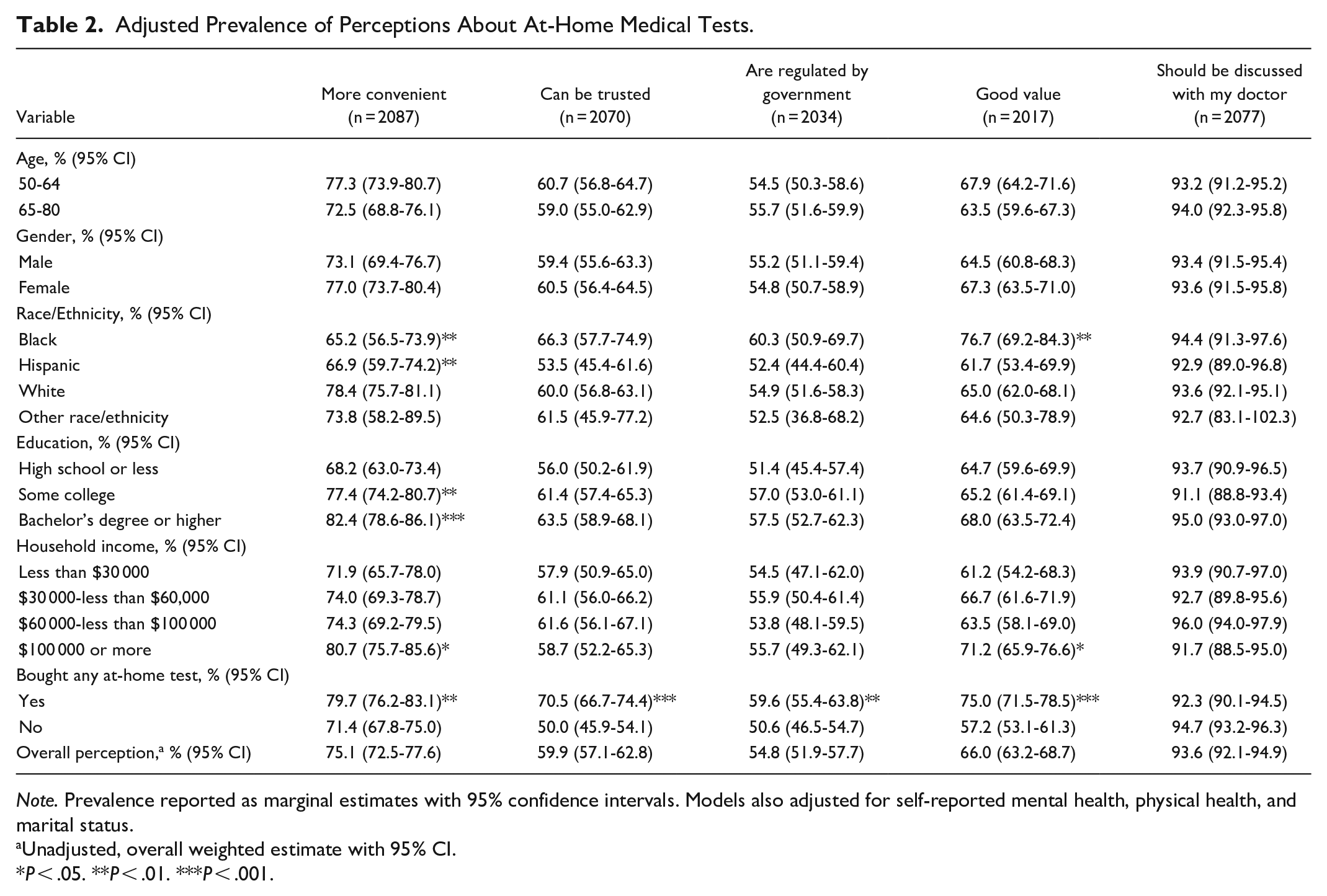
Note. Prevalence reported as marginal estimates with 95% confidence intervals. Models also adjusted for self-reported mental health, physical health, and marital status.
Unadjusted, overall weighted estimate with 95% CI.
P < .05. **P < .01. ***P < .001.
A large majority of older adults agreed that the results of at-home tests should be discussed with one’s doctor (93.6%, 95% CI 92.1%-94.9%). Among those who had purchased each respective type of at-home test, 90.5% (95% CI 82.4%-95.0%) shared the result of an at-home cancer screening test with their primary care provider (PCP), 63.1% (95% CI 46.7%-76.9%) shared the result of some other type of at-home test (eg, hormone, food sensitivity, or allergy tests) with their PCP, 55.4% (95% CI 41.4%-68.5%) shared the result of an at-home test for an infection other than COVID-19 with their PCP, and 9.0% (95% CI 5.6%-13.9%) shared the result of an at-home DNA/genetic test with their PCP (Figure 1).

Percentage of those who shared the results of an at-home test with their primary care provider (blue) versus those who purchased an at-home test but did not share the result (orange) by test type.
Interest in Future Use
Most older adults (82.4%, CI 80.1%-84.4%) were interested in using some type of at-home medical test in the future. Women and those with a bachelor’s degrees or higher were more likely to report interest in using any type of at-home test in the future (87.4% vs 76.1%, P < .001; 88.5% vs 77.8%, P = .011, respectively). Compared with those with annual household incomes less than $30,000, all other income groups were more likely to be interested in future use of at-home testing (Table 3).
Adjusted Prevalence of Interest in Future Use of At-Home Medical Tests.

Note. Prevalence reported as marginal estimates with 95% confidence intervals. Models also adjusted for mental health and marital status.
Unadjusted, overall weighted estimate with 95% CI.
P < .05. **P < .01. ***P < .001.
Adults aged 65 to 80 were less likely than those aged 50 to 64 to report interest in future use of at-home tests other than COVID-19 tests (Table 3). Women were more likely than men to express interest in future use of COVID-19 tests (74.6% vs 63.1%, P < .001), tests for infections other than COVID-19 (50.0% vs 34.6%, P < .001), DNA/genetic tests (44.2% vs 34.6%, P = .001), and other types of at-home tests such as hormone, allergy, or food sensitivity tests (50.1% vs 38.7, P < .001). Compared with White, non-Hispanic adults, Black, non-Hispanic adults reported greater future interest in use of COVID-19 tests (79.3% vs 66.9%, P = .005) and DNA/genetic tests (49.8% vs 38.6%, P = .033). Hispanic adults, compared to White non-Hispanic adults, reported greater interest in future use of at-home tests for cancer screening (63.6% vs 53.9%, P = .029) and other at-home tests such as hormone, allergy, or food sensitivity tests (53.8% vs 43.0%, P = .015).
Those with bachelor’s degrees or higher were more likely than those with lower levels of education to report interest in future use of every type of at-home test (Table 3). Individuals with household incomes of at least $100 000 were more likely to report interest in future use of COVID-19 tests (76.0% vs 62.4, P = .003) and other at-home tests such as hormone, allergy, or food sensitivity tests (48.0% vs 38.4%, P = .049). Those reporting fair or poor physical health were more likely than those reporting better physical health to be interested in purchasing other types of at-home tests such as hormone, allergy, or food sensitivity tests (51.8% vs 42.7%, P = .013). Those who had previously bought an at-home test were more likely to express interest in purchasing any type of at-home in the future (Table 3).
Discussion
In this nationally representative survey, we found that about half of US adults aged 50 to 80 have purchased some type of direct-to-consumer, at-home medical test. About one third have purchased a COVID-19 test, and just over 16% have purchased at-home genetic tests. There were important socioeconomic and racial/ethnic differences in prior purchase and perceptions of at-home tests, though most older adults across subgroups had positive perceptions. We also found that a considerable percentage of older US adults are interested in future use of at-home tests, although this too varied among different demographic groups. To our knowledge, this is the first study to generate national estimates for purchase of, perceptions of, and interest in future use of at-home tests among older US adults.
We found significant disparities in prior purchase of at-home tests by education and income. In a survey on use of at-home COVID-19 tests from fall 2021 to spring 2022, the US Centers for Disease Control and Prevention similarly found increased test use among those with higher education and income. 24 Differences in income and education level have also been found regarding direct-to-consumer genetic testing awareness in the US 25 and at-home testing in the Netherlands. 26 Given the convergence of these prior results with the current study, we hypothesize differences in income may be attributable to the often high out-of-pocket costs of at-home tests. 6 Furthermore, the differences observed by education level may be due to a greater sense of self-efficacy to use and interpret at-home test results without the assistance of a clinician among respondents with a higher education level. 27
Income and education-level disparities were also observed regarding future interest in at-home tests, and to some extent regarding perceptions. These differences may be due to greater financial ability and confidence to purchase, perform, and interpret at-home tests among those with higher levels of education and income. Among these subgroups, it is also possible that at-home testing could be viewed as more favorable than the time it takes to schedule, attend, and await results from a doctor’s appointment.
We also found significant racial/ethnic differences in at-home test purchase. This is in contrast to a 2019 study within the Kaiser Permanente system that found no difference in prior use of direct-to-consumer genetic testing between Hispanic, Black, non-Hispanic, and White, non-Hispanic participants. 28 However, their overall estimates of such testing were much lower than in our study (5% compared with 16%), and other studies have shown lower use of certain at-home tests among black individuals compared with White individuals. 24 At the same time, Black, non-Hispanic older adults reported significantly greater future interest in COVID-19 and DNA genetic testing compared with White, non-Hispanic older adults. Some have hypothesized that at-home testing could assist in decreasing health disparities,6,29 but these findings suggest that more research is needed before this is realized.
Most older adults stated that at-home tests are more convenient than those from their doctors, which is consistent with prior hypotheses that a strength of at-home tests is their accessibility.5,6 Particularly in a time when primary care services are more difficult to access due to greater patient volumes and a shrinking primary care workforce, 30 at-home tests could at times provide greater, more timely access to health information. Assuming valid and reliable at-home tests, the growing availability of telemedicine may be an especially apt place for more at-home testing as this would enable clinicians to continue to be a part of the decision-making process.
Most adults thought the results of at-home tests could be trusted and that these tests were regulated by the government. The validity, reliability, and overall utility of at-home tests has been debated among experts, and much of this debate has focused on the regulation of such tests.9 -11 The FDA has approved a number of diagnostic at-home tests for “over-the-counter” use, 4 but many tests on the market remain unregulated or under-regulated.6,31 Our results suggest, however, that patients generally believe at-home tests are regulated by government but a substantial minority did not, which may reflect public confusion in how at-home testing is regulated. Indeed, the nuances between some regulatory policies and exemptions created confusion about the meaning of EUAs during the COVID-19 pandemic among both patients and clinicians. 32 Additionally, there are a number of exclusions for FDA oversight of at-home tests that makers of direct-to-consumer, at-home tests could use to advertise them without formal approval. 33 Greater clarity surrounding the regulation of at-home tests from the FDA and retailers alike could assist individuals in better understanding the veracity of at-home test results. Broader public messaging or more explicit marketing standards about non-FDA approved tests could assist in this.
Limitations
Purchase of at-home tests was self-reported and is vulnerable to recall and social desirability biases. Our results may not be generalizable to adults beyond 50 to 80 years or those older adults residing in long term care or other institutions. Our survey was administered in January 2022 during the White House’s announcement to make COVID-19 tests freely available, and this may have affected perceptions and interest in future use as well as overestimated the purchase of COVID-19 tests specifically.
Conclusion
Half of older adults have used some form of at-home medical test, and such use is likely to grow given positive perceptions and interest in future use of at-home tests. Clinicians should be aware of the different types of at-home tests now available, be prepared to discuss with patients the risks and benefits of using at-home tests, and consider asking about prior at-home testing. When at-home tests have the potential to mitigate personal and public health risk, policymakers should explore strategies to promote equity in access to evidence-based at-home tests that have potential to reduce health care disparities.
Supplemental Material
sj-docx-1-inq-10.1177_00469580241284168 – Supplemental material for Use of At-Home Medical Tests Among Older US Adults: A Nationally Representative Survey
Supplemental material, sj-docx-1-inq-10.1177_00469580241284168 for Use of At-Home Medical Tests Among Older US Adults: A Nationally Representative Survey by Joshua B. Rager, Matthias Kirch, Dianne C. Singer, Erica Solway, Preeti N. Malani, J. Scott Roberts and Jeffrey T. Kullgren in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
None
Data Availability Statement
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Kullgren reports grant funding from the US National Institutes of Health, the US Department of Veterans Affairs, the Healthwell Foundation, and the State of Michigan Department of Military and Veterans Affairs; receiving consulting fees from the Washington State Office of the Attorney General; receiving honoraria from the American Diabetes Association, the Luxembourg National Research Fund, the Donaghue Foundation, the National Science Foundation, the University of California-Los Angeles, and the University of Pennsylvania. Dr. Kullgren is also supported by the Department of Veterans Affairs, Veterans Health Administration, Health Services Research and Development Service and the views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for study is provided AARP and Michigan Medicine. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethical Statement
This study was determined to be exempt by the University of Michigan Institutional Review Board.
Informed Consent
This study was determined to be exempt by the University of Michigan IRB, thus informed consent for this individual study was not necessary. All panelists are consented by NORC prior to participating on the AmeriSpeak panel.
Trial Registration Number/Date
N/A
Grant Number
N/A
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
