Abstract
Optimal medication management is important during hospitalization and at discharge because post-discharge adverse drug events (ADEs) are common, often preventable, and contribute to patient harms, healthcare utilization, and costs. Conduct a cost analysis of a comprehensive pharmacist-led transitions-of-care medication management intervention for older adults during and after hospital discharge. Twelve intervention components addressed medication reconciliation, medication review, and medication adherence. Trained, experienced pharmacists delivered the intervention to older adults with chronic comorbidities at 2 large U.S. academic centers. To quantify and categorize time spent on the intervention, we conducted a time-and-motion analysis of study pharmacists over 36 sequential workdays (14 519 min) involving 117 patients. For 40 patients’ hospitalizations, we observed all intervention activities. We used the median minutes spent and pharmacist wages nationally to calculate cost per hospitalization (2020 U.S. dollars) from the hospital perspective, relative to usual care. Pharmacists spent a median of 66.9 min per hospitalization (interquartile range 46.1-90.1), equating to $101 ($86 to $116 in sensitivity analyses). In unadjusted analyses, study site was associated with time spent (medians 111 and 51.8 min) while patient primary language, discharge disposition, number of outpatient medications, and patient age were not. In this cost analysis, comprehensive medication management around discharge cost about $101 per hospitalization, with variation across sites. This cost is at least an order of magnitude less than published costs associated with ADEs, hospital readmissions, or other interventions designed to reduce readmissions. Work is ongoing to assess the current intervention’s effectiveness.
Keywords
Optimal medication management is important during hospitalization and at discharge because post-discharge adverse drug events are common, often preventable, and contribute to avoidable patient harms, healthcare utilization, and costs.
In this cost analysis, a pharmacy-led intervention that addressed medication reconciliation, medication review, and medication adherence among hospitalized older adults involved a median of 67 min of pharmacist time, equating to $101 per hospitalization.
The cost of comprehensive medication management around hospital discharge is at least an order of magnitude less than published costs associated with an ADE, a hospital readmission, or prior quality improvement interventions designed to reduce readmissions.
Introduction
Optimal medication management is important during hospitalization and at the discharge transition because post-discharge adverse drug events (ADEs, injuries due to medication therapy) are common, often preventable, and contribute to patient harms, hospital readmissions, and avoidable costs. 1 Post-discharge ADEs represent 70% of all adverse events occurring within 30 days after hospitalization.2 -4 Older adults, particularly those with multimorbidity and complex medication regimens, are at particularly high risk for post-discharge ADEs, with rates of 17% to 19%.5,6 About one quarter of post-discharge ADEs are preventable and another third could be ameliorated, that is, decreased in duration or severity, through improvements in care.4,7 Hospital readmissions are attributable to post-discharge ADEs 23% to 38% of the time.6,8,9
In this context, hospitals have been employing pharmacist-led medication management strategies to prepare for discharge and prevent post-discharge ADEs.10,11 The strategies predominantly focus on 3 domains. The first domain is medication reconciliation, which involves establishing a best possible medication history (ie, what medications a patient was taking and should have been taking prior to admission), and identifying and correcting discrepancies between this history and ordered medications on admission and discharge. This is now commonly performed upon admission and discharge, as recommended by The Joint Commission. 12 Both admission and discharge medication reconciliation serve as foundations for medication-management strategies in the other 2 domains. The second domain, medication review, involves reviewing a patient’s medical conditions and medication history to consider what medications should and should not be prescribed. Medication review is especially useful to detect polypharmacy, the phenomenon where patients, especially older patients with multimorbidity, are prescribed multiple medications that are unnecessary, duplicative, and/or risky. Medication review also detects under-prescribing, which is common among older adults as well. 13 The third domain involves identifying and addressing barriers to medication adherence (eg, cost, side effects, patient/caregiver understanding). Systematic reviews show that interventions involving these strategies can reduce ADEs, including after discharge.14 -16 In addition to being effective, these pharmacist-led practices are becoming more commonplace, yet little is known about the time and costs involved in implementation, particularly the time and costs of implementing multiple strategies together.
Our team developed a comprehensive set of evidence-based, pharmacist-led best practices in peri- and post-discharge medication management, in prior work funded by the American Society for Health-System Pharmacists.14 -16 We then worked with pharmacy leaders at 2 large academic hospitals to develop a protocol for implementation, focusing on older adults with chronic comorbidities. The present cost analysis sought to quantify the time and costs involved in implementing the intervention at these sites as part of a federally funded, 2-site, randomized controlled trial. To achieve this objective, we employed a micro-costing approach, 17 which involved conducting a time-and-motion study of pharmacists and using these data to estimate the total per-patient cost of implementation, relative to usual care at the study sites.
Methods
We report this cost analysis in accordance with the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) Statement. 18 This study occurred in the context of a pragmatic randomized controlled trial examining the effectiveness of these practices at reducing 30-day post-discharge hospital utilization 19 (PHARMacist Discharge Care (PHARM-DC) trial, clinicaltrials.gov (NCT04071951). 20
Setting
The study occurred at 2 large urban medical centers in the United States: an academic hospital in the Northeast (Brigham and Women’s Hospital [BWH]) and a dual academic and community hospital in the West (Cedars-Sinai Medical Center [CSMC]). At both sites, pharmacist leaders committed institutional resources to support implementation of the medication management practices, and they were willing to randomly allocate use of the practices to test their benefit. Both hospitals have very high patient volumes, including patients meeting study eligibility criteria (below). Additionally, both sites have robust pharmacy programs, including residency programs for pharmacists, as well as training programs for physicians and nurses, although only one (CSMC) had an established transition of care (TOC) pharmacy program. Both sites employ Epic electronic health record (EHR) systems with clinical decision support and other advanced features. Across CSMC and BWH, respectively, 43% and 41% of patients were covered by Medicare, 42% and 47% had private insurance, and 13% and 12% had Medicaid. 19
Population
For the current time-and-motion analysis, we employed additional exclusion criteria: (1) patients for whom the medication management practices planned for them were not completed (eg, rapid discharges, unplanned readmissions), (2) patients for whom we were unable to observe key parts of the planned practices (eg, practices performed concurrently or outside of observer visibility), and (3) patients for whom the pharmacists were supervising trainees during delivery of the intervention (ie, to avoid contamination of time and cost estimates by training activities). Because the intervention was tailored for individual patients, the nature and scope of medication management practices varied across patients in the time-and-motion analysis.
For the analysis of costs, we excluded patients when the time-and-motion analysis did not capture all of the intervention activities delivered to the patient.
Intervention
Prior publications provide details about the intervention. 19 To summarize, the intervention involved 12 specific best practices predominantly falling into 3 domains, as noted in the Introduction above: (1) medication reconciliation, (2) medication review, and (3) medication adherence. Pharmacists employed specific component practices during the hospitalization, on the day of hospital discharge, and within 1 to 3 days after discharge. See Table 1.
Intervention Design: Pharmacist-led Medication Management Strategies for Preventing Post-discharge Adverse Drug Events Among High-risk Patients (N = 40 Observed Patients Over 36 Pharmacist Workdays).

No patients received 100% of all components because the intervention was tailored to individual participants’ needs and because patients may be discharged before all components planned for them have been completed.
Although 0 of the 40 patients in the time and motion analysis received this component, we report it in the table to show the complete list of components that pharmacists were able to use as part of the intervention.
Comparator: Baseline Medication Management Practices (Usual Care)
Before and during the study, pharmacy staff at CSMC obtained a “best possible medication history” for nearly all patients as part of admission medication reconciliation, while pharmacy staff at BWH did so only for higher-risk patients based on availability. This involved determining which medications each patient was prescribed prior to admission and which medications each patient was actually taking by interviewing patients and/or caregivers/family members and, as necessary, calling the patient’s community pharmacy and/or consulting the Surescripts pharmacy fill history. Additionally, the CSMC pharmacists used a Medication Adherence and Literacy scoring tool.23,24 Pharmacists then updated the EHR and communicated this information to treating physicians to inform or adjust the ordering of inpatient medications. High-risk and other prioritized patients at CSMC received post-discharge phone calls and/or bedside delivery of medications prior to discharge; the latter was available at BWH based on patient need and clinician request.
Measures
Data Collection
We trained observers with clinical backgrounds (research nurses, medical students) to document time use by study pharmacists on a minute-by-minute basis in real time. We developed detailed instructions that explained the time-and-motion procedures as well as categories of activities. The observers used an electronic data collection tool to record minutes spent per hospitalization on each of the 7 categories of activities listed above (TimeCaT 3.9; Lopetegui et al). 29 For pharmacist activities related to patients who had already been discharged, the observer also recorded the number of days since discharge (eg, post-discharge day 4). The observers followed study pharmacists during entire workdays because study pharmacists’ workdays included a mixture of activities, including unrelated activities. For a period of time at each site, we had dual observers assigned to each pharmacist, to assure reliability. During data collection, the research team met with the observers regularly to answer questions and advise on classification of activities.
Observations included 36 sequential workdays (Mondays to Fridays) of study pharmacists between both study sites (247 hours = 14 846 min total). We chose this sample size because we expected that it would allow for collection of at least 30 observations. We expected that this would allow the central limit theorem to hold in estimating the mean time necessary for one intervention for one patient, which would allow us to estimate costs.
We conducted observations at CSMC from July 7 to 29, 2021 (site total of 7916 min), with a mean shift for the observer of 7 h 20 min. At BWH, we conducted observations from November 15 to December 10, 2021 (site total of 6930 min), with a mean shift of 6 h 7 min. Observations occurred during year 3 of the trial to ensure that study procedures were well-established and pharmacists were experienced with the intervention. In a minority of cases at CSMC, it was difficult to separate non-intervention admission medication reconciliation activities from the intervention, especially when these were performed together.
Analyses
For wage data, we used national data sources to increase the generalizability of results. In the base case, we used the national median hourly wage of a hospital pharmacist in 2020 ($61.88), as per the Bureau of Labor Statistics. 30 No discounting was performed because the analysis was focused on short-term costs per hospitalization.
In sensitivity analyses, we used the 25th and 75th percentiles from the Bureau of Labor Statistics ($54.18 and $71.01, respectively) 30 as well as the hourly wage for a PharmD from ZipRecruiter.com ($53 per hour). 31 We used a benefit rate of 46.6%, the average for private sector workers at this wage range. 32
We used Spearman’s rank-order correlation coefficient (rs) to assess the strength and direction of the correlation between minutes of pharmacist time per hospitalization and patient age in years. Kendall’s tau-b correlation coefficient (τb) was used to assess the strength and direction of the correlation between minutes of pharmacist time per hospitalization and the number of outpatient medications at discharge. Kendall’s tau-b is more conservative and favored over Spearman’s correlation coefficient when there are many tied ranks in the data. 33 Correlation coefficient calculations were limited to patients with complete observation data.
Ethics Review: This study used a single Institutional Review Board from Cedars-Sinai to oversee work at both sites, including waiving informed consent for patients. Study pharmacists who implemented the intervention received a written information sheet and gave verbal consent to participate in the time-and-motion observations.
Conflict of Interest: The investigators have no conflicts of interest with the work.
Results
Across the 2 sites, the observers documented 8 study pharmacists delivering care to 117 unique patients. For 40 patients’ hospitalizations (CSMC: 29, BWH: 11), the time-and-motion observations captured all intervention activities delivered to the patient for a given hospitalization; all subsequent results focus on this subpopulation. For the other 74 patients, some intervention activities occurred on days when time-and-motion observations were not performed.
Table Listing Characteristics of Patients Observed in the Time-and-Motion Analysis.
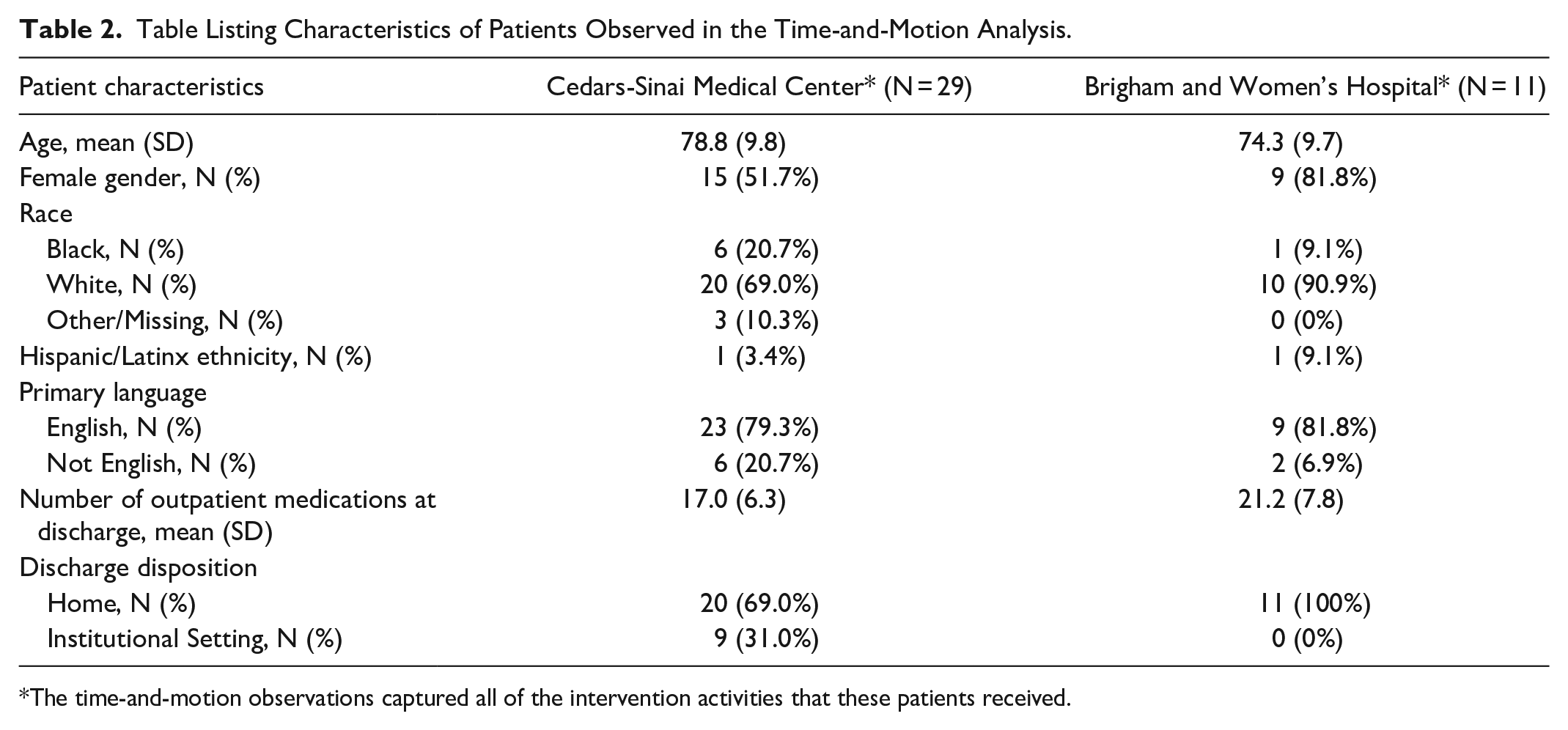
The time-and-motion observations captured all of the intervention activities that these patients received.

Categories of pharmacist activities involved in implementing comprehensive pharmacist-led transitions-of-care medication management intervention for older adults around hospital discharge, by study site (total N = 40 hospitalizations).*
Based on the median minutes spent by study pharmacists per hospitalization and using the 25th and 75th percentiles from the Bureau of Labor Statistics and the wage estimate from ZipRecruiter.com, alternative per-hospitalization cost estimates were $88, $116, and $86, respectively.
There were no statistically significant correlations between minutes of pharmacist time per hospitalization and the number of outpatient medications at discharge (τb = 0.205, P = .067), nor between minutes of pharmacist time per hospitalization and patient age (rs = −0.112, degrees of freedom = 38, P = .493). See Figure 2.
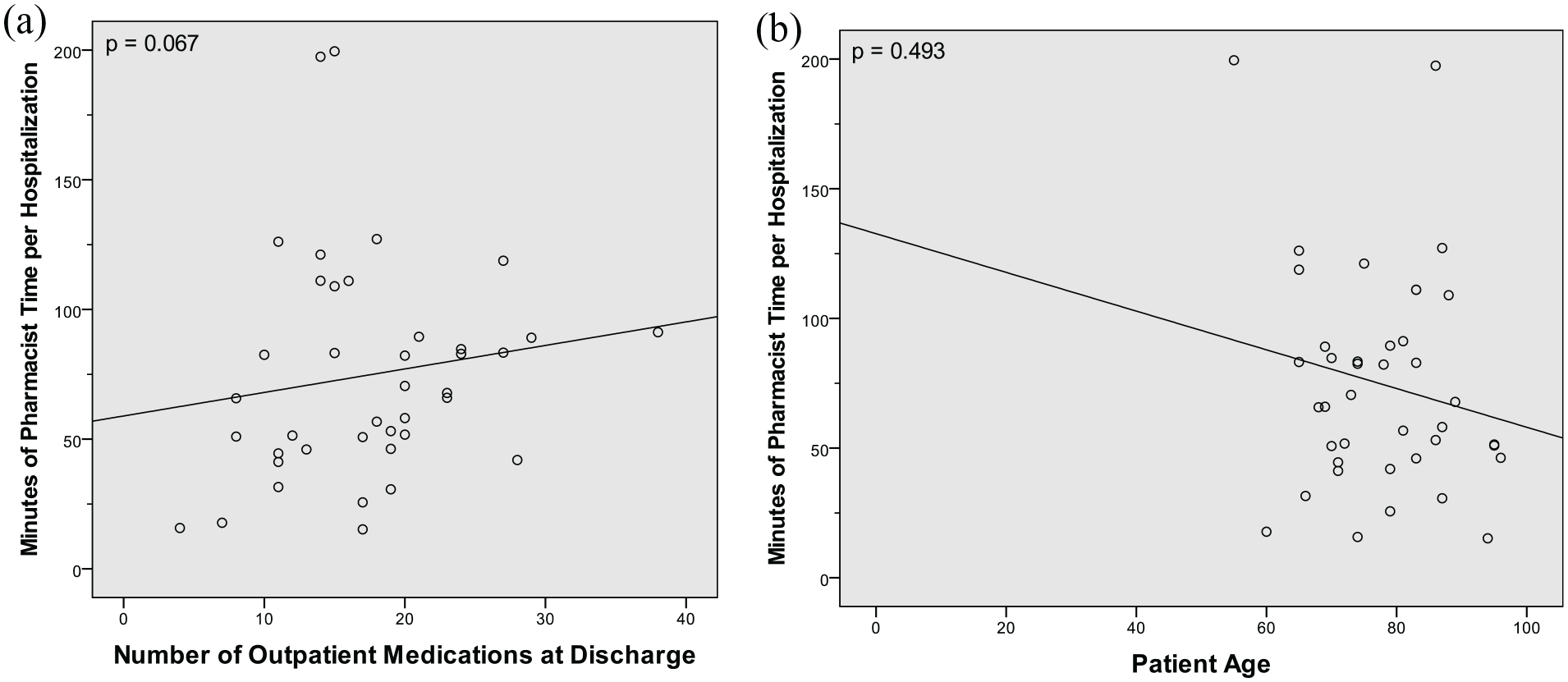
Minutes pharmacists spent on intervention per individual hospitalization (N = 40*), stratified by (a) number of outpatient medications at discharge and (b) patient age.
We did not observe significant (unadjusted) differences in minutes of pharmacist time per hospitalization by patient language or discharge disposition. The median times to deliver the PHARM-DC intervention among primary English speakers and non-primary English speakers were 66.9 min (IQR = 47.4-90.7) and 66.8 min (IQR = 26.9-105.7), respectively; the Mann-Whitney U test was not significant (U = 111.0, P = .565). The median times to deliver the PHARM-DC intervention among patients discharged to home versus an institution were 82.7 min (IQR = 51.3-111.0) and 48.5 min (IQR = 39.1-60.5), respectively; the Mann-Whitney U test was not significant (U = 82.0, P = .063), despite the 41% lower median time investment for patients discharged to an institution.
Discussion
This study examined the time and costs associated with a transitions-of-care intervention through which trained, experienced pharmacists comprehensively managed medications in high-risk older adults around the time of hospital discharge. We found that the pharmacists spent a median of 67 min per hospitalization, corresponding to $101 per hospitalization ($86-$116 in sensitivity analyses). The mean minutes and costs per hospitalization varied greatly: from 15 to 200 min ($23-$302) across individual patients and from 58 to 118 min ($98-$199) between the 2 hospitals. It appears likely that the intervention components employed for a patient may have influenced variation in the minutes spent, since patients who were discharged to institutional settings generally received a narrower set of components, as compared with patients discharged home, although the difference in time spent was not statistically significant. Neither the number of outpatient medications before admission nor patient age was associated with higher resource expenditures.
Prior trials have tested comprehensive medication management interventions for hospitalized patients, with the goal of reducing post-discharge ADEs and/or hospital readmissions. Results have been mixed. A 2021 trial by Gurwitz et al randomized 361 inpatients (age ≥50, discharged on ≥1 high-risk medication) to a multifaceted, moderately comprehensive intervention or control condition. There was no difference in clinically important medication errors (primary outcome). 34 In contrast, the 2018 OPTIMIST trial randomized 1467 adults taking ≥5 medications to 3 arms: usual care, medication review alone, or a more comprehensive multifaceted intervention. Thirty-day readmissions declined (primary outcome, hazard ratio 0.62; 95% CI 0.46-0.84); the number needed to treat was 12. 35 However, a 2021 trial randomized 1702 patients age ≥ 85 years and/or taking >10 medications at hospital admission to 2 arms: medication reconciliation (alone) versus usual care; they found no difference in 30-day readmissions. 36 The Medication Reviews Bridging Healthcare trial cluster randomized 2637 patients (age ≥ 65) to 3 arms: medication review, medication review plus post-discharge follow-up, or usual care. Return hospital visits within 12 months (primary outcome) did not differ among the arms. These trials build on a large number of earlier studies examining medication management strategies and largely demonstrating improvements in medication appropriateness.14 -16,37
Despite the potential clinical benefits, only a few prior time-and-motion studies have examined the time invested in medication management at discharge. A 2013 study reported that discharge medication reconciliation involved a mean of 19 min of pharmacist or physician time per hospitalization for patients on Internal Medicine services and 29 min for patients on Geriatrics services, in a Canadian population that was taking medication prior to admission and discharged home. 38 In a 2021 French study of hospitalized adults age 65 and above, medication reconciliation at discharge involved 30 min of pharmacist or physician time per patient. 39 In Australia, a 2022 study found that pharmacists spent a mean of 26 min per “discharge medication handover” (a similar concept to reconciliation) on inpatient medical and surgical services. 40 These time estimates are remarkably similar, despite differences in geography and implementation of medication reconciliation. Our comprehensive medication management intervention involved more time, 67 min per patient hospitalization, likely because we selected patients at the greatest risk for ADEs and because our intervention included not only medication reconciliation but also in-depth medication review and efforts to assess and enhance medication adherence. Our intervention also occurred at several time-points in a patient’s journey: shortly after admission (ie, at the time of randomization), at discharge, and after discharge.
Pharmacy leaders may be concerned that comprehensive medication management around hospital discharge would require substantial time and costs, given that the work is labor intensive and performed by highly-skilled, highly-paid practitioners. Such concerns might partly explain why comprehensive medication management practices are not universally implemented, despite the incentives to prevent hospital readmission created by the Medicare Hospital Readmissions Reduction Program. However, our current results rebut these concerns: $101—or even $302—per hospitalization is low relative to the costs associated with other relevant quality improvement interventions, of ADEs, and of readmissions themselves. The mean cost for a multifaceted quality improvement intervention designed to prevent readmission (+/− medication management), is $554, per a recent meta-analysis. 41 The cost of an ADE ranges from $153 to $15 712 in older populations, according to a 2022 systematic review, with most primary studies reporting values between $1200 and $2500. 42 Hospital readmissions cost >$15 000 each. 43 The current intervention might, therefore, be net cost-neutral if it prevents ≥1 ADE per 12 to 25 hospitalizations or ≥1 readmission per 149 hospitalizations. We are in the midst of conducting a randomized controlled trial designed to assess whether the current intervention avoids >1 in 40 readmissions. 19 Thus, this intervention would be cost saving with a decline in readmissions that the current RCT is powered to detect.
The cost of comprehensive medication management around discharge could be lower than $101 in other populations and hospital settings, for several reasons. As noted above, the current intervention was particularly comprehensive, with multiple labor-intensive components provided to most patients. It targeted the highest risk patients, who were taking, on average, 16 to 20 medications each at the time of hospital admission. It was implemented, furthermore, at 2 quaternary academic referral centers with particularly complex patient populations. Additionally, we obtained substantially lower cost estimates at one study site, where implementation reflected more real-world conditions. That hospital had established comprehensive medication management as an operational priority over the long term, with a cadre of dozens of dedicated transitions of care pharmacists, potentially enabling study pharmacists to learn how to implement the intervention more efficiently. Estimates at that hospital may have even over-estimated the amount of time required for the intervention since it was not always possible to separate out admission medication reconciliation activities that were not part of the intervention. At the other hospital, the intervention was implemented primarily as a research activity, so the pharmacists had less experience and were more focused on being comprehensive in intervention delivery.
Future research could examine the effectiveness and costs of the interventions using lower-cost clinicians, such as pharmacy technicians. In contrast to the median wage of $61.88 per hour for highly trained pharmacists with doctoral (PharmD) degrees, pharmacy technicians earn $16.87 per hour and have a high-school education. 44 Although not all pharmacy technicians have the training and experience to perform higher level tasks like comprehensive medication review, we previously showed that pharmacy technicians can be very effective in performing the complex task of obtaining a complete medication history at hospital admission. 45 We expect that other activities, like identifying and addressing medication adherence barriers, may also be aided by the efforts of pharmacy technicians, with oversight and completion of tasks by pharmacists.
This analysis bears several limitations. First, although the number of minutes of observation in this time-and-motion analysis was relatively high (>14 000), the results analyzed are from only 40 patients cared for during a brief segment of the four-year study. One hospital had only 11 patients with complete data. A larger sample could yield a wider range of estimates but also greater precision as to the true median/mean for time spent and costs. Second, pharmacists may not perform at their maximum speed due to pressure from being directly observed, although this concern may diminish as they become more comfortable with being observed over time. Third, our estimates of minutes per hospitalization and costs may tend toward overestimates because the true cost reflects the difference between intervention and control groups. We did not conduct time-and-motion analyses in the control group patients, who routinely receive admission medication histories and who receive the other medication management practices when there is a strong indication.
Conclusion
This study estimated that a comprehensive intervention designed to optimize medication management in the highest-risk patients around the time of hospital discharge required investments of 67 min of skilled pharmacist time per hospitalization, at a cost of $101. These costs varied between study sites, but not with primary language, discharge disposition, number of outpatient medications, or patient age. Nonetheless, the cost of the intervention was low relative to the costs associated with other quality improvement interventions designed to prevent hospital readmission, of an ADE, and of a hospital readmission. Next steps in this work involve assessing its effectiveness.
Footnotes
Authors’ Note
PHARM-DC Group (collaboration group) includes Michael Corrado, Lilly Thao, Kelsey O’Neil, and Nicole Conti at Brigham and Women’s Hospital; Michelle S. Keller, James Guan, Yervant Malkhasian, Emily Phung, Audrienne Salandanan, Shirley Wu at Cedars-Sinai Medical Center; and Korey A. Kennelty, Logan T. Murry at the University of Iowa College of Pharmacy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Aging (R01 AG058911). In-kind resources were contributed by Cedars-Sinai Medical Center and Brigham and Women’s Hospital. Prior phases in the research were supported by the American Society of Health System Pharmacists.
Ethics and Consent
The Institutional Review Board at Cedars-Sinai Medical Center approved human subjects considerations including informed consent procedures.
