Abstract
There is a paucity of evidence-based information regarding healthcare professionals’ awareness and views toward counterfeit medicines in developing countries. Therefore; this is aimed to assess health care providers’ knowledge, attitude, and practice toward counterfeit medicines in Mizan-Tepi University Teaching Hospital, South West Ethiopia. A cross-sectional study was conducted among health care providers working in Mizan-Tepi University Teaching Hospital from December 2020 to January 2021. A total of 171 health care providers participated in the study. Data were collected through self-administered structured questionnaires developed by reviewing similar surveys with some modifications. The data analyzed using a Statistical Package for Social Sciences (SPSS, version 21). Variables with P value <.05 were considered as statistically significant. Large proportions of the study participants (84.2%) have information on counterfeit drugs and 15.8% of them described counterfeit medicine as product with toxic impurities. 50.3% of them were able to distinguish a counterfeit drug from the genuine drug. 8.2% of the participants demonstrated that counterfeit medicine can be identified by physical observation of labeling, color appearance and packaging. 61.4% of the study respondents revealed that strong legal action can prevent circulation of counterfeit medicine. Professional distribution was significantly associated with the attitude of health care providers toward counterfeit medicines (P < .05). Accordingly, public health officers and midwifery health care providers were more likely to have poor attitude toward counterfeit medicines (AOR = 6.09, CI (1.798-20.69) and (AOR = 3.98(1.54-10.25)) respectively as compared to nurses. This study demonstrated the importance of awareness creation to all health care providers. Drug regulatory bodies and concerning bodies must play an active role in designing appropriate program and policy to enhance health professionals’ knowledge and attitude toward CFM. There is also a need of empowering the practitioners in identifying counterfeit drugs by simple observations.
Keywords
Substandard and counterfeit drugs are known to invade the health care system due to a number of problems including increase in morbidity and mortality as well as loss of public confidence in existing health care structures.
It assesses health care providers’ knowledge, attitude and practice toward counterfeit drugs in Mizan-Tepi University Teaching Hospital, South West Ethiopia. Professional distribution was significantly associated with the attitude of health care providers toward counterfeit medicines.
This study will provide enough information on knowledge, attitude and practice regarding counterfeit which are often undermine and given less concern in the area of research. There is a need for information conserving awareness and practice of counterfeit drugs especially on health care providers. This study provides useful information for scientific community on perception and practice of counterfeit drugs in Ethiopia.
Introduction
Substandard and counterfeited drugs are 2 types of poor-quality medicines. Authorized drugs that do not meet quality standards are known as substandard drugs. 1 Whereas counterfeit medicines (CFM) are medicines which are deliberately and fraudulently mislabeled with respect to their identity and/or source, and may include products with correct or wrong ingredients, without active ingredients, with insufficient or inadequate quantities of ingredient(s) or with fake packaging. 2 Counterfeiting can apply to both branded and generic products.2,3 It covers a wide range of medications, from lifesaving to lifestyle items.4,5 Antibiotics and anti-malarial medications are the most frequently reported drugs found to be of poor quality.6-8
Moreover, poor quality medicines are the global problems with the highest prevalence in low and middle income countries.1,6 Despite the lack of precise data on the magnitude of counterfeit medicines, World Health Organization (WHO) has estimated that approximately 10% of the global pharmaceuticals market consists of counterfeit drugs, but this estimate increases to 25% in developing countries, and may exceed 50% in certain countries. 9 Furthermore, it is well recognized that substandard and counterfeit medicines infiltrate the health-care system as a result of a number of issues, including an increase in morbidity and mortality, as well as a loss of public trust in existing health-care systems. 10 Additionally, it raises health-care expenses for patients as well as the health-care system.1,11 As a result, developing countries should pay close attention to the issue of substandard and counterfeit medicines available for public use. 12
The probable reason for the presence of poor quality drugs in developing countries like Ethiopia might be due to the lack of well-developed regulatory framework to assess and take action to solve the problems of poor quality medicines. 13 To detect and combat counterfeit drugs, Ethiopia has regulations and methods in place, as well as several sources of information. 14 However, there is information on the circulation of CFM in this country, and the majority of illegally imported drugs were anti-infective medicines including anti-malarial and antibiotics. Furthermore, the quality, safety, and efficacy of these medications are unknown, and they may be counterfeit, substandard, or degraded. 15
Government and health care workers jointly play a substantial role in protecting public health from the problems of counterfeit drugs. 16 More interestingly, studies suggested that designing and implementing educational programs for health professionals including pharmacists are important to resolve the distribution of counterfeit medicine.17,18 However, a review of knowledge and attitudes regarding counterfeit medication among diverse health care providers from various countries revealed that only a tiny percentage of participants knew the precise definition of counterfeit medicine.17,19,20 They also stated that they thought CFM was a concern for the profession.17,21
Awareness and opinion of health care providers play a pivotal role in mitigating the circulation of counterfeit drugs in the country.22,23 However, there is a paucity of evidence-based information regarding healthcare professionals’ awareness and views toward counterfeit medicines in developing countries. 24 Taking this fact into consideration, this study is aimed to assess health care providers’ knowledge, attitude and practice toward counterfeit medicines in Mizan-Tepi University Teaching Hospital, South West Ethiopia.
Method
Study Design and Study Setting
A cross-sectional study was conducted among health care providers working in Mizan-Tepi University Teaching Hospital from December 2020 to January 2021. This hospital is located in Mizan-Aman town, Southwest Ethiopia. It provides different inpatient and outpatient services, and known to have a range of specialties including pediatrics, internal medicine, surgery, gynecology, and ambulatory care services. Besides, the hospital has 4 different pharmacies such as outpatient pharmacy, antiretroviral therapy pharmacy, emergency pharmacy, and inpatient pharmacy to render pharmaceutical services for the patients. Furthermore, at the time of data collection, the hospital had 363 health care providers.
Sample Size and Eligibility Criteria
The study included all health care providers working at Mizan-Tepi University Teaching Hospital, including pharmacists, public health officials, physicians, midwives, and nurses. A total of 171 health care providers took part in the study.
Inclusion Criteria
Health care provider who was on duty during the data collection period was included of the study.
Exclusion Criteria
Health care provider that was not willing to participate in the study was excluded from the study.
Data Collection Tools and Procedures
Data were collected through self-administered structured questionnaires developed by reviewing similar surveys with some modifications.17,18,24-27 The questionnaire addressed the basic demographic characteristics of the study participants’, health professionals’ knowledge, attitude and practice toward CFM. To ensure the validity of the study of the instruments and respondent’s understanding of the questions, questionnaire was pretested before actual data collection with 18 health professionals. Minor modifications were made based on the feedback obtained from the pilot testing. The pilot study data were excluded from the study results. To check internal consistency, the Cronbach’s alpha coefficient of the data collection tool was calculated, and it was .81, which is within the acceptable range. To help with the data collection, 2 trained pharmacy professionals were hired. Supervisors double-checked the accuracy and consistency of the data collected.
Data Analysis
The collected data were coded, entered, and analyzed using a Statistical Package for Social Sciences (SPSS, version 21). Descriptive statistics such as frequency, percentage, and mean were used to summarize demographic characteristics, knowledge, attitude, and practice of the study participants’ toward CFM. The health care providers’ attitude was assessed by using 6 items rated on a 5-point Likert scale as agree, neutral, disagree, and strongly disagree. A response of “Strongly agree” was given a score of 5, “Agree a score of four,” “Neutral” a score of 3, “Disagree” a score of 2, and “Strongly disagree” a score of 1 for all positive questions, and reverse scored for negatively worded questions during the analysis. The overall level of attitude was categorized by using mean score. Participants who score above or equal to the mean were classified as having good attitude and scores below the mean were classified as having a poor attitude. Furthermore, the logistic regression model was fitted to see the association between attitude items and demographic characteristics. From the output of binary logistic regression, variables with P-value < .25 were entered into multivariate logistic regression, and variables with P value <.05 were considered as statistically significant.
Ethics Approval and Consent to Participate
Ethical clearance was obtained from School of Pharmacy, College of Medicine and Health Sciences, Mizan-Tepi University and was forwarded to the hospital officers (Ref. no CP0054/13). Furthermore, written informed consent to participate in the study was secured from the study participants before data collection. To ensure the confidentiality of participants, name and address of the study participant was not recorded in the data collection format.
Results
Demographic Characteristics
Among 171 health care providers participated in the study, majority (57.3%) of them was males. Around half (48.0%) of the study participants were married. Meanwhile, similar figure was observed for marital status, in which 48.0% of the participants were single. Furthermore, 55% of the participated professionals were nurse followed by midwives and pharmacists which representing of 15.8% and 11.1%, respectively. Out of all participants, 47.4% were Orthodox Christian and 11.7% were Muslims. About 37% of the health care providers were Amhara by ethnicity, followed by Oromo and Bench with a proportion of 29.2% and 26.3%, respectively (Table 1).
Demographic Profile of the Participated Health Care Providers’ in Mizan-Tepi University Teaching Hospital From December 2020 to January 2021.
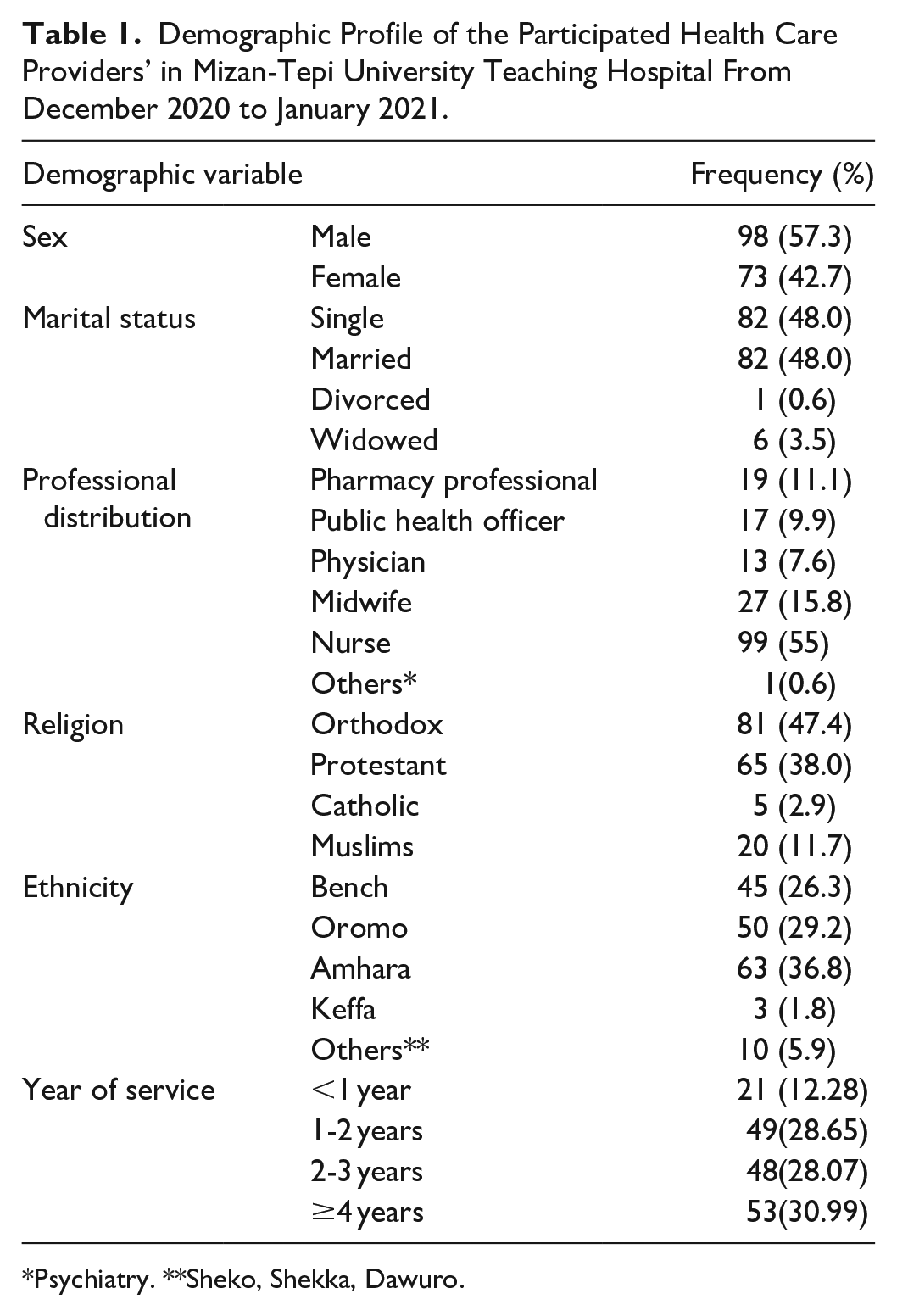
Psychiatry. **Sheko, Shekka, Dawuro.
Health Care Providers’ knowledge Toward Counterfeit Medicines
As shown in Table 2, a substantial percentage of study participants (84.2%) are aware of counterfeit medicines, with 65.5% defining counterfeit medicines as a medicine with correct packaging but incorrect active ingredient identity or quantity. Similarly, 15.8% of them characterized CFM as a product that contains toxic impurities. 2.9% of the study participants, on the other hand, were unable to describe CFM. When asked about their major source of information on counterfeit medicines, roughly 45% of study participants said solely the media, followed by both media and education with a combined figure of 40.9%. Consumption of counterfeit drugs is linked to at least one problem, according to nearly all of the participants. As a result, 53.8% of them demonstrated that CFM has a safety issue, and 5.8% of the involved health professionals were aware of CFM’s economic issue. Only one participant, on the other hand, stated that counterfeit medicines are not a threat (Table 2).
Health Care Provider’s knowledge Toward Counterfeit Medicines in Mizan-Tepi University Teaching Hospital From December 2020 to January 2021.

Practices of Health Care Providers Toward Counterfeit Drugs
Out of 171 the study participants included in this study, around half (50.3%) of them were able to distinguish a counterfeit drug from the genuine drug. In connection, 13.5% of the study respondents said that CFM can be identified by its effect, and 8.2% of the participants demonstrated that CFM can be identified physical observation of labeling, color appearance and packaging. However, large proportions (48.5%) of the study participants were not familiar with the method used for identification of CFM. 61.4% of study participants believe that substantial legal action may be taken to prevent the consumption of counterfeit medicines. Similarly, 16.4% of health care practitioners believe drug regulatory agencies should be better equipped to prevent the sale of counterfeit medicines. On the other hand, only 24% of the participants were shown how to combat counterfeit medicines. Further, more than 85% of the participated health care providers were ready to report when they encounter counterfeit medicines during their practice (Table 3).
Practices of Health Care Providers Toward Counterfeit Medicines in Mizan-Tepi University Teaching Hospital From December 2020 to January 2021.

Health Care Providers’ Attitude Toward Counterfeit Drugs
As it can be observed from Table 4, among the study respondents enrolled; about 72 (42.1%) strongly agree that dispensing and prescribing counterfeit medicine is unethical. With respect to legality of dispensing and prescribing counterfeit medicines, 50.9% of the study participants agreed that dispensing and prescribing unregistered or counterfeit medicine is illegal. On the other hand, 28.7% of the study participants disagreed in similarity between efficacy of counterfeit and genuine medicines. 31.6% study participants disagreed that CFM is more cost effective than its genuine counterpart. Furthermore, 31% of health care providers disagreed that CFM intake is as safe as their genuine medicines, whereas 5.3% claimed that counterfeit medicine consumption is as safe as genuine original drug consumption. In this regard, 34.5% of study participants felt that unregistered medications are likely to be ineffective.
Health Care Providers’ Attitude Toward Counterfeit Medicines in Mizan-Tepi University Teaching Hospital From December 2020 to January 2021.

The mean attitude of the study participants was 21.62 and 75 (43.9%) of the study participants have a good attitude whereas 96 (56.1%) have a bad attitude.
Factors Associated With Attitude of Health Care Providers’ Toward Counterfeit Medicines
Out of 171 health care providers, the majorities (56.1%) of them were found to have a poor attitude toward counterfeit medicines. On the other hand, about 44% of the study respondents had a poor attitude toward counterfeit medicines. Multivariate logistic regression analysis performed revealed that professional distribution was significantly associated with the attitude of health care providers toward counterfeit medicines (P < .05). Accordingly, public health officers and midwifery health care providers were more likely to have poor attitude toward counterfeit medicines (AOR = 6.09, CI (1.798-20.69)) and (AOR = 3.98 (1.54-10.25)) respectively as compared to nurses (Table 5).
Factors Associated With Attitude of Health Care Provider’s Toward Counterfeit Medicines in Mizan-Tepi University Teaching Hospital From December 2020 to January 2021.

Discussion
A large proportion of the study participants have information on counterfeit medicines, and majority of them described counterfeit medicines as a medicine with correct packaging but with incorrect identity or quantity of active ingredient. Similar findings are reported in study from Jordan done by Abu Taleb and Al Madadha 17 in which 76% are aware of the CFM problem, and current laws and regulations. The finding from this study indicated that the majority of the study participants were aware about counterfeit medicines and their effects. This result was similar with the study conducted in Iran, which indicated that 21.5% (158/734) of pharmacists were aware of CFM. 18
Contrary to a study conducted in Sudan on community pharmacists’ awareness and attitudes toward counterfeit medicines, which reported that a high percentage of participants define counterfeit medicines as unknown source and poor quality, 25 in our study, the majority of study participants described counterfeit medicines as a medicine with correct packaging but incorrect identity or quantity of active ingredient. This disparity could be due to the fact that the studies were conducted on different study subjects; in our case all health care providers who may not have enough information about counterfeit medicines, whereas the study in Sudan was conducted on pharmacists who had adequate information on counterfeit medicines.
The Food and Drug Administration (FDA) defines counterfeit medicines as “fake medicines that may be contaminated or contain the wrong or no active ingredients,” 28 but only 15.8% and 2.9% of study participants described counterfeit medicines as “toxic impurities” and “medicine with fake packaging and active ingredient.” Similar findings were also reported from Sudan, where only 27% of study participants identified counterfeit medicines as potentially contaminated, containing the erroneous or no active ingredients. 25
In our study, about 45% of the study participants mentioned only media as their main source of information on counterfeit medicines, followed by both media and education with a figure of 40.9%. This is deviated from the study conducted in Sudan 25 and Lebanon 29 in which CFMs education is not included in the curriculum of most faculties. Study from California recommended that academic institute should have a key role in educating health care professionals on counterfeit medicines. 20 In addition, the current study discovered a problem associated with counterfeit medicines/unregistered drugs on the market, and a high percentage of study participants indicated that CFM has a safety problem, with a low percentage of participating health professionals aware of CFM’s economic problem.
Physical examination of a drug’s label and packaging design, which might reveal hints regarding counterfeit status, is the first stage in combatting counterfeit medicine. Any drug product’s label must carry the following information as a bare minimum: the brand name of the product, the drug’s generic name, indications, size/weight, warnings/cautions, usage instructions, manufacturing details, country of origin, batch number, and a clear barcode. The absence of any of these features is a clear indication that the product is counterfeit. Providing education about these drug labeling standards would be a critical step in combating counterfeit products and limiting their use. 30
In our investigation, about half of the participants were able to tell the difference between a counterfeit and a genuine medicine. Large sections of the study participants were unfamiliar with the approach utilized to detect CFM when it came to recognizing CFM. CFM may be identified by its effect, according to a quarter of the survey participants, and 8.2% of the individuals demonstrated that CFM can be identified by physical inspection of labeling, color appearance, and packaging. These findings contradict a study conducted in Sudan, which found that participants distinguish counterfeit drugs from genuine ones based on packaging and supplier characteristics, 25 and that 21.5% of participants in Iranian pharmacists were given the correct answer for the selective method for counterfeit drugs. 18
Furthermore, our findings contradicted a WHO advice that counterfeit medicines can be discovered by looking at the box, checking the manufacturer, expiration date, and the appearance of the medicine. 2 This could be owing to the fact that none of the study participants had ever encountered counterfeit medications in their carrier. In this survey, the majority of study participants stated that strong legal action can curb the spread of counterfeit drugs. Similar findings have been reported from Sudan 25 and Lebanon, 29 where harsh action is required against pharmacists who deal with counterfeit drugs, ranging from penalties to the closure of their businesses.
In terms of health-care providers’ attitudes, roughly half of those questioned strongly agreed that dispensing and administering counterfeit medication is unethical. When it came to the legality of dispensing and prescribing unregistered or counterfeit pharmaceuticals, 50.9% of participants believed that doing so is against the law. Only 43.9% of participants have a positive attitude, indicating that health care professionals’ attitudes are negative. These findings contradict a study conducted in Iran, which found that pharmacists had poor awareness and practice of counterfeit pharmaceuticals despite having a positive attitude toward the subject. 18 This deviation is due to the difference in study subjects and sample size used in our study.
In this study, multivariate logistic regression analysis performed revealed that professional distribution was significantly associated with the attitude of health care providers toward counterfeit medicines (P < .05). Accordingly, public health officers and midwifery health care providers were more likely to have poor attitude toward counterfeit drugs (AOR = 6.09, CI (1.798-20.69) and (AOR = 3.98 (1.54-10.25)) respectively as compared to nurses. This could be due to the fact that nurses have direct contacts with drugs like that of pharmacists and they have an ability to differentiate the drugs using physical inspection. This leads to the improvement of their attitude toward differentiating counterfeit medicines. Correspondingly, different results are reported from the study conducted in Iran in which increasing age resulted in attitude improvement (P = .013) and gender also had a marginal relationship (at .05 level) with attitude, so that women attitudes were better than that of men (P < .05). 18 This difference could be due to the fact that the study from Iran is conducted on pharmacists only where as our study conducted on all health care providers. These findings show that strengthening communication between health care practitioners and regulatory authorities, as well as reporting suspicions and preventing counterfeit pharmaceuticals from entering the pharmaceutical distribution chain, are more important than ever. In addition, there is a significant need for training on the counterfeiting for health-care professionals.
Limitations of the Study
As our study is conducted on a single facility, small sample size of the study might oversimplify generalizability of our findings. Meanwhile, a cross sectional study design has a weakness for ascertaining the way participants perceive, but it simply describes what and doesn’t describe cause and effect relationship.
Conclusions
A large proportion of the health care providers had information on counterfeit drugs, and a substantial number of the study respondents were able to distinguish between genuine and counterfeit medicines. The study findings also revealed that a large proportion of the study participants had poor attitude for counterfeit medicines. Professional distribution was significantly associated with the attitude of health care providers toward counterfeit medicines. This study demonstrated the importance of awareness creation to all health care providers. Drug regulatory bodies and concerning bodies must play an active role in designing appropriate program and policy to enhance health professionals’ knowledge and attitude toward CFM. There is also a need of empowering the practitioners in identifying counterfeit drugs by simple observations as a major step toward discouraging the marketing of counterfeit medicines.
Footnotes
Acknowledgements
We would like to extend our gratitude to Mizan Tepi University teaching hospital for allowing us to conduct this study. We are also extended our gratitude to participants who involved in data collection.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Data Availability Statement
All relevant data are within the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Ethical clearance was obtained from School of Pharmacy, College of Medicine and Health Sciences, Mizan-Tepi University and was forwarded to the hospital officers (Ref. no CP0054/13). Furthermore, written informed consent to participate in the study was secured from the study participants before data collection. To ensure the confidentiality of participants, name and address of the patient was not recorded in the data collection format.
