Abstract
Globally, more than 20 million newborns are born with low birth weight (LBW) every year. Most of the LBW occurs in low- and middle-income countries. It is the most critical risk of neonate mortality. Therefore, this study aims to identify determinants of low birth weight among women who gave birth in public health facilities in the North Shewa zone. Institutional-based unmatched case–control study was conducted from February to June 2020 to select 180 cases and 380 controls. Interviewer-administered questionnaire was used to collect data. Data were entered through EPI Info and exported to Statistical Package for Social Science (SPSS) for analysis. Text, percentage and tables were used to present data. Bivariate and multivariate logistic regression analyses were performed to see the association and adjusted odds ratios with 95% confidence interval (CI), and P-value < .05 was considered to declare statistical significance. Lack of nutritional counseling (adjusted odds ratio [AOR] = 2.14; 95% CI = [1.13, 4.04]), unable to take iron-folate supplement (AOR = 2.3.78; 95% CI = [2.1, 6.85]), insufficient additional meal in take (AOR = 6.93; 95% CI = [3.92, 12.26]), restriction of foods (AOR=2.29; 95% CI =[1.81, 4.09]), maternal mid upper arm circumference (MUAC) < 23 cm (AOR=2.85; 95% CI = [ 1.68, 4.85]), maternal height ≤155 cm (AOR=3.58; 95% CI = [1.92, 6.7]), anemia (AOR = 2.34; 95% CI = [1.21, 4.53]), pregnancy-related complications (AOR=3.39; 95% CI = [2.02, 5.68]), and alcohol drinking during pregnancy (AOR = 2.25; 95% CI = [1.24, 4.08]) were significantly associated with LBW. Nutritional counseling, iron-folate supplementation, additional meal intake, restriction of some foods in pregnancy, MUAC of the mother, maternal stature, maternal anemia status, pregnancy-related complications, and a history of alcohol drinking during pregnancy were identified as determinants of low birth weight. The intervention-targeted nutritional counseling, early detection and treatment of anemia, and behavioral change communication to pregnant women are mandatory.
What do we already know about this topic?
The prevalence of low birth weight was already assessed, and factors like socio-demographic, disease-related and pregnancy-related were identified.
How does your research contribute to the field?
This research finding was contributed to the current research field because our study findings are unique in obtaining determinants of low birth weight like food taboo and substance use (alcohol drinking) which are new findings in Ethiopia.
What are your research’s implications toward theory, practice, or policy?
We anticipated that our findings provide basis for low birth weight determinants that are significant for intervention of pregnant mothers.
Background
The World Health Organization (WHO) defined LBW as weight at birth less than 2500 g. 1 Birth weight is the main indicator of the child’s susceptibility to childhood diseases. Most of the time, the cause of LBW is premature birth, intrauterine growth restriction, or the combination of both. 2 In developing countries, the cause of LBW is premature birth, whereas intrauterine growth restriction in developed nations. 3
Low birth weight is the most critical risk for neonate mortality, 4 which contributes 60-80% of neonatal morbidity and mortality. 5 LBW newborns are almost 20 times at high risk to die than newborns with normal birth weight. 6
Long-term complications of LBW include hypertension, diabetes mellitus, later-age renal diseases, eye problems, deafness, neurologic complications like cerebral palsy, developmental delay, seizure, and psychological disturbances.7-9 Another impact of LBW is being at higher risk of infection, poor mental development, stunting, and developing non-communicable diseases. 5
More than 20 million newborns are born with LBW globally every year. 10 Most of the LBW occurs in low- and middle-income countries. 11 The prevalence of LBW is ranging from 9 to 35.1% in developing countries. 12 The prevalence of LBW in Africa was 22% 13 and 13% in Ethiopia. 14 Some of the factors that are positively associated with LBW in developing countries include advanced maternal age (35-49 years), mothers who have informal education, female newborns, delayed conception, inadequate antenatal care, primiparity, living in rural areas, and low socioeconomic status. 12 Other different studies from Ethiopia reported that pregnancy-induced hypertension, low maternal educational status, low maternal height, incomplete antenatal care, household food insecurity, lack of iron supplementation, anemia, unable to take additional food, preterm birth, medical problems during pregnancy, MUAC of mother <23 cm, lack of nutritional counseling, and previous history of abortion are factors that are associated with LBW.15-19
The WHO planned to achieve a reduction of LBW by 30% in 2025, but the prevalence of LBW is not decreasing to achieve the WHO target. 10 Even though the Ethiopian Federal Ministry of Health also developed a national nutrition strategy to improve the micronutrient deficiency among pregnant women through increasing the system to give comprehensive and routine nutritional assessment intervention, the magnitude of LBW was not decreased yet. Additionally, the Ministry of Health of Ethiopia provided routine iron–folate supplementation and deworming during pregnancy to mitigate adverse birth outcomes like LBW, but the prevalence of LBW was not reduced. 20 Even if this strategy strives to improve LBW, it is still a public health problem among newborns in Ethiopia. Despite food taboo is very common in Ethiopia, its relationship with the occurrence of adverse birth outcome like low birth weight was not studied. Therefore, this study aimed to identify low birth weight determinants among women who gave birth in public hospitals in the North Shewa zone, central, Ethiopia.
Methods
Study Design, Period, and Setting
Unmatched case-control was conducted from February to June 2020 in five public hospitals and 14 health centers in the North Shewa zone. Fiche is the capital city of the zone and 114 km away from Addis Ababa. The North Shewa zone has a total population of 1.6 million according to 2007 census. From the total population, 49,667 were pregnant women and expected to give birth according to the North Shewa Zone health office report. The Zone has five hospitals and 50 health centers that provide antenatal care, delivery, and postnatal services to the North Shewa Zone communities.
Source and Study Population
All women who gave birth in public hospitals in the North Shewa zone during data collection were source population for both cases and controls, whereas those included in the study were the study population for both cases and controls. Women who were deaf, unable to speak or communicate, severely ill (comatose women and women who had postpartum hemorrhage), and those who delivered deformed newborns were excluded from the study. Cases were mothers who gave live birth weighed less than 2500 g, whereas mothers who gave live birth weighed greater than 2500 g were controls. 1
Sample Size Calculation and Sampling Procedure
The sample size was calculated by using double population proportion formula through EPI INFO version 7 statistical software package with the assumption of confidence level 95% (Zα/2 = 1.96), power 80% (Zβ = .84), case to control ratio 1:2, where P1 is the proportion of cases exposed, and P2 is the proportion of controls exposed. By taking unable to take iron and folic acid supplementation during pregnancy as identified, the study was conducted in Dessie town, Ethiopia in 2017 (proportion of cases that did not take iron-folate supplementation is31.2%, and the proportion of controls that did not take iron-folate supplementation is 10.8%, and AOR = 2.84). 18
This gave 95 cases and 190 controls with a total of 285. After multiplying this by design effect 2, the final sample was 190 cases and 380 controls with a total of 570 study participants. The study was conducted at five hospitals and randomly selected 14 health centers. The number of study participants was allocated proportional to each health facility based on estimations obtained from the average of previous quarter delivery services by referring to delivery registration books at each hospital. Therefore, the sample of each hospital was calculated by multiplying the average number of pregnant women who delivered in each hospital per 5 months with the total sample size, dividing by a total number of pregnant women attending antenatal care units for 5 months of all hospitals, which was obtained from delivery registration of the previous months. Multi-stage sampling was used to select health facilities. Cases were selected by taking all cases until the required sample size obtained, and controls were selected using systematic random sampling.
Data Collection Tools, Procedure, and Management
The data were collected by an interviewer-administered questionnaire, which was developed from different similar works of the literature.12,9,16 The questionnaire was prepared in English and translated to a local language Afan Oromo for a better understanding of both data collectors and respondents. It was translated back to the English language by language experts to check for consistency. The questionnaire contained socio-demographic characteristics (age, marital status, educational status, place of residence, religion, ethnicity, occupation, and monthly income), maternal nutritional factors (nutritional counseling, iron–folic acid (IFA) supplementation), restriction of food or food taboo (restriction of food made from butter, fruits, and vegetables during whole pregnancy due to fear of fetus overdevelopment and attachment to the fetus during pregnancy), food frequency (meal), additional food (adding frequency of food and amount of food intake during pregnancy), fasting (unable to take any food and fluid for 9 h on Wednesday and Friday), and substance use-related factors (cigarette smoking, alcohol drinking, and chat chewing during pregnancy) were obtained by face-to-face interview. MUAC of the mother, height of mother, obstetrics and gynecologic factors (previous history of abortion, number of abortion, previous history of adverse birth outcomes [LBW, premature birth, and stillbirth]), gravidity, parity, birth interval, pregnancy status, antenatal care (ANC) visit, number of ANC visits, medical illness for recent pregnancy (hypertension, diabetes, urinary tract infection, and sexually transmitted diseases) and pregnancy-related complications (gestational hypertension, premature rupture of membrane, antepartum hemorrhage, gestational age, and anemia status) were obtained from patients’ ‘records.
Ten BSc mid-wives were recruited for data collection, and five BSc public health officers were superviced data collection. Three days of training was given for data collectors and supervisors on the objective of the study and how to maintain the confidentiality of the respondents. Additionally, data collectors and supervisors were trained about COVID-19 prevention technique and offered the prevention materials (mask, alcohol or sanitizer, and soap) to implement during data collection. Pretest was conducted on 5% of the sample at Chencho Hospital which is outside the study area. Investigators, data collectors, and supervisors discussed the questionnaire and made necessary correction where necessary after pretest before actual data collection. Close supervision was done during data collection.
The interview was conducted 1 h after delivery and after the newborn’s weight was measured. The weight of newborn was measured 1 h after delivery using a balanced digital Seca scale (Germany). The scales were always calibrated using the materials with the standard weight and the reading on each scale by taking to zero levels before weighing each newborn. Mother’s height was also measured using a height board while the mother was in the standing position which was taken from height measured before delivery. The height of each mother was taken to the nearest .1 cm. Mother was asked to stand without shoes in front of the height board with head erect and arms hanging naturally at the sides. The MUAC of the mother was measured using a flexible, non-stretchable standard tape meter to the nearest .1 cm to determine the nutrition of the mother. The outcome (low birth weight) is identified according to the WHO definition, weight at birth less than 2500 g. 1 The reliability of the questionnaire was checked with Cronbach’s alpha with the value of .876.
Data Processing and Analysis
After data collection, data were checked for completeness and coded, cleaned, and entered using EPI INFO version 7 and were exported to SPSS version 23 for data cleaning and analysis. After cleaning data for inconsistencies and missing values, texts, tables, and proportions were used to present data. Logistic regression was carried out to identify independent predictors of low birth weight. Bivariate analysis was carried out to determine a significant association between each predictor variable and low birth weight at a P-value < .25. Bivariate and multivariate logistic regression analyses were carried out to determine an association between low birth weight and independent variables.
Multivariate logistic regressions were carried out to identify determinants of low birth weight among women who gave birth in public hospitals. The goodness-of-fit model (Hosmer and Lemeshow) was used to select the best multivariate model. Multicollinearity was checked by using standard error. Finally, AOR with 95% CI and P-value < .05 were considered to declare statistical significance.
Ethical Consideration
The Salale University Ethical Review Committee approved the ethical clearance with Reference number SLUERC/019/2020. A formal letter of cooperation was written to each hospital. Written consent was obtained from study participants who were above 18 years old, and written assent was obtained from their parents or guardians where participants were women under 18 years of age. The privacy and confidentiality of study participants were also protected strictly. Data collectors were informed about coding the questionnaire and not to write the name of the study participants. Only data collectors saw the records of the study participants.
Result
Socio-Demographic Characteristics of the Study Participants
Socio-Demographic Characteristics of Women Who Gave Birth at Public Health Facilities in the North Shewa Zone from February to June 2020 (n = 185 Cases and n = 370 Controls).
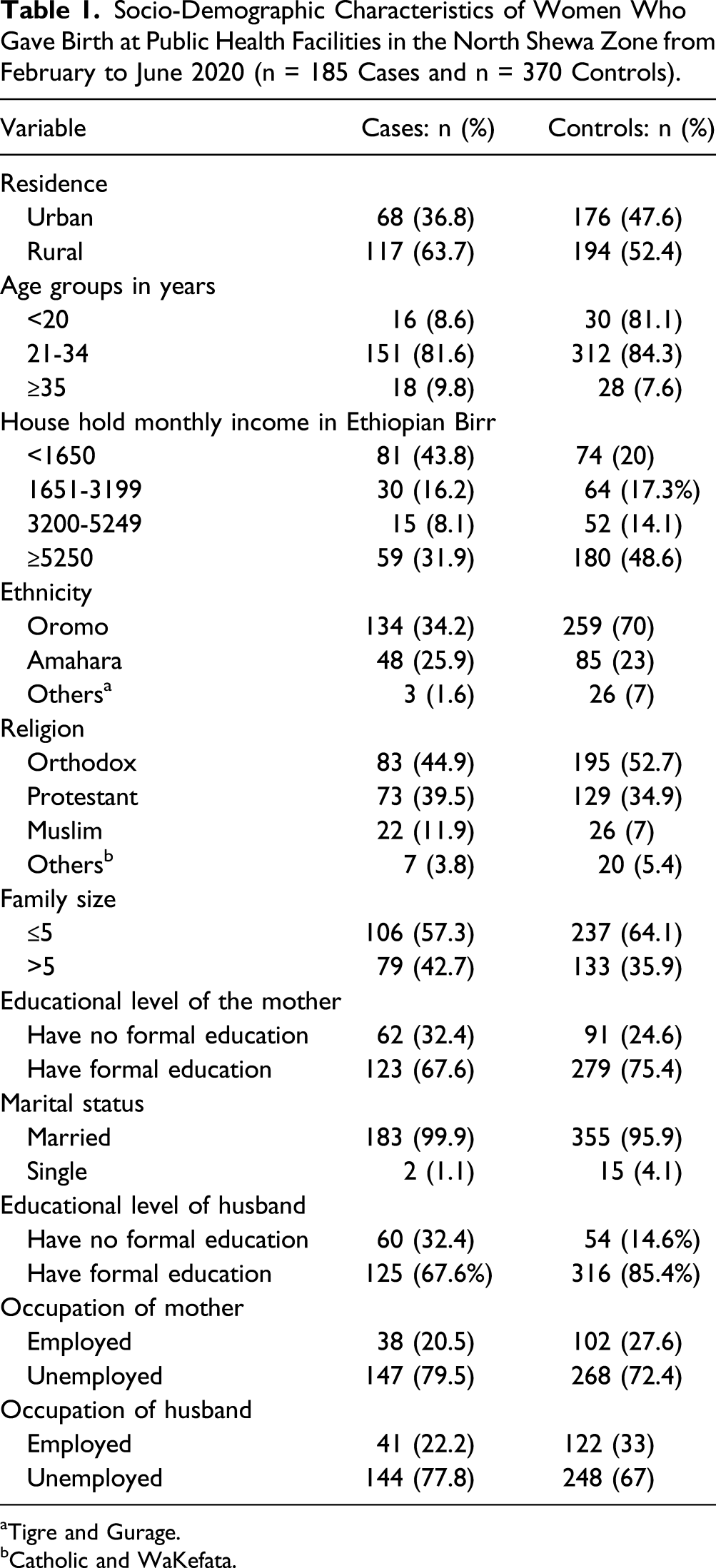
aTigre and Gurage.
bCatholic and WaKefata.
Nutritional-Related Characteristics of Study Participants
Nutritional-Related Characteristics of Women Who Gave Birth at Public Health Facilities in North Shewa Zone, from February to June 2020 (n = 185 Cases, and n = 370 Controls).

Food Frequency of the Respondents
Food Frequency of Women Who Gave Birth at North Shewa Public Health Facilities From February to June 2020 (n = 185 Cases, and n = 370 Controls).

Obstetrics and Gynecologic Characteristics of the Study Participants
Obstetric and Gynecologic Characteristics of Women Who Gave Birth at Public Health Facilities in North Shewa Zone, from February to June 2020.

Substance Use-Related Characteristics of the Study Participants
Substance Use-Related Characteristics of Women Who Gave Birth at Public Health Facilities in North Shewa Zone, from February to June 2020 (n = 185 Cases, and n = 370 Controls).

Determinants of Low Birth Weight
Factors Associated With Low Birth Weight Among Women Who Gave Birth at Public Health Facilities in the North Shewa Zone from February to June 2020 (n = 185 Cases, and 370 Controls).

Note. 1= reference; COR = crude odds ratio; AOR = adjusted odds ratio.
aStatistically significant at P-value < .05.
Discussion
Unable to get nutritional counseling during pregnancy was a significant determinant of low birth weight in this study. This finding is similar to a case-control study conducted in Dassie town, 18 where nutritional counseling was an independent predictor of LBW. This might be because nutritional counseling enables and improves women’s food intake, improves their nutritional status, and finally decreases the risk of giving LBW babies. The present study revealed that mothers who did not take iron-folate supplementation during their recent pregnancy had higher odds of giving LBW babies than mothers who received iron-folate supplementation. It is in line with a study conducted in Dassie town, 18 Kambata, 16 Amhara, Ethiopia, 17 Rural Ethiopia, 21 Gahana, 22 Auckland, New Zealand, 23 and Bangladesh 24 that showed unable to take iron-folate during pregnancy associated positively with LBW. This might be due to intake of iron helps women not to develop anemia and risk of delivering LBW babies since the required amount of iron cannot be obtained from nutrients alone during pregnancy. 25
Intake of additional meals at recent pregnancy had a significant association with LBW in our study. It is consistent with the study conducted in Kambata, 16 Dassie town, 18 and Ghana 26 where the chance of giving LBW newborns is higher among mothers who did not eat additional food compared to their counterparts. Taking additional meals is important for women themselves and their fetuses in pregnancy. 27 The only way the fetus obtains nutrients from the mother is through the placenta. When the mother did not get additional meals during pregnancy, the nutrients transferred to the fetus through the placenta are reduced, which causes restriction of fetal growth that leads to LBW. 28 Unable to take additional meals also attributes to anemia, premature birth, and pregnancy-related complications that, in turn, lead to LBW. 29
Women who had MUAC <23 cm have higher odds of delivering LBW neonates than those who had MUAC ≥23 cm. It is similar to studies conducted in Dassie town, Ethiopia, 18 Amhara, Ethiopia, 17 Guinea-Bissau, 30 and Bangladesh 12 where maternal under-nutrition is positively associated with LBW. The reason behind this fact is that low nutritional status of the mother leads to different diseases that may cause LBW. Moreover, maternal under-nutrition may also cause fetal retardation because of the very low transfer of nutrients from the mother to her fetus, which leads to LBW. 31
Food taboo was positively associated with occurrence of LBW. This is because these restricted foods are vital food groups whose deficiencies cause malnutrition like anemia, which leads to LBW. Furthermore, when mothers do not take enough foods, nutrients transferred to the fetus through the placenta can be decreased, leading to fetus growth restriction and LBW. In contrast, a study conducted in Addis Ababa, Ethiopia, found the opposite. 9 The reason for the disparity between the two findings is due to the socio-cultural characteristics of the two populations; the current study was conducted in rural and town settings, whereas the previous study was conducted in the country’s capital city, where the community may have extensive knowledge. The pregnancy-related complication was another predictor of LBW. It is in agreement with a case-control study conducted in Addis Ababa, 9 a case-control study conducted at public hospitals in the Amhara region, 17 at referral hospitals in North Ethiopia, 32 where pregnancy-related complication was positively associated with LBW. The reason behind this fact is that pregnancy-related complications can cause a reduction in nutrients and oxygen to the fetus, which in turn leads to LBW. 17
Low maternal stature was an independent predictor of LBW. This finding is in line with studies conducted in Addis Ababa, 9 Morocco, 33 Cameroon, 34 Nigeria, 35 Eastern Nepal, 36 and India,37,38 where maternal stature is significantly associated with the occurrence of LBW. This might be because mothers with short stature may also have narrow pelvis with limited intrauterine space that restricts the fetus’s intrauterine growth, leading to LBW. 39
Anemia of mother was statistically significantly associated with LWB. This finding is in agreement with a case-control study conducted at Kambata, 16 Dassie town, 18 Gahana, 22 Nigeria, 35 and Nepal 40 that found a statistically significant association between anemia and LBW. This is because anemia is caused due to the reduction of red blood cells (hemoglobin), and it can cause the amount of maternal blood production, which leads to the reduction of nutrients received by the fetus. This reduced amount of fetal nutrient causes restriction of fetal growth and leads to LBW.
Drinking alcohol was positively associated with occurrence of LBW. It is similar to another study where consuming one drink at least once per day was associated with LBW. 41 This is because due to unknown factor that leads to an increased risk of fetal retardation. Finally, the limitation of this study is that it may be susceptible to recall and social desirability bias.
Conclusion
Nutritional counseling, iron-folate supplementation, additional meal intake, restriction of some foods in pregnancy, mid-upper arm circumference of the mother, maternal stature, maternal anemia status, pregnancy-related complications, and a history of alcohol drinking in pregnancy were identified determinants of low birth weight. Intervention on LBW prevention should consider nutritional counseling, additional meal intake, iron-folate supplementation, early detection, and treatment of anemia, avoiding alcohol drinking in pregnancy. Behavioral change communication targeting pregnant women to reverse food taboos by health professionals.
Footnotes
Acknowledgments
We would like to express our great thanks to Salale University’s Research and community service coordinators for facilitating this proposal work. Second, we would like to acknowledge health facilities administrative staff for their cooperation and support to get the baseline data used to contract the sampling procedure. Last but not least, we would like to acknowledge my friends, who supported me all the time from the start to the end of this proposal work, all health institutions from which data were obtained, and research participants.
Authors Contributions
BSD and KJ were involved in idea creation, proposal development, supervising, data collection, data analysis, interpreting results, editing, supervision, guiding during the whole research proposal development, research result writing, and manuscript preparation. BSD and KJ made important contributions to the work’s idea and design, as well as the gathering, analysis, and interpretation of data. They also contributed to the work’s writing or critical revision for key intellectual substance. BSD and KJ both gave their approval for the final version to be published. They committed to take responsibility for all elements of the work, including ensuring that any issues about the truth or integrity of any portion of it are thoroughly examined and addressed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Salale University provided funding for this study.
Ethical Approval
The Salale University Ethical Review Committee approved the ethical clearance with Reference number SLUERC/019/2020. A formal letter of cooperation was written to each hospital.
Informed Consent
Written consent was obtained from every study participants who were above 18 years old, and written assent was obtained from their parents or guardians where participants were women under 18 years of age. The privacy and confidentiality of study participants were also protected strictly. Data collectors were informed about coding the questionnaire and not to write the name of the study participants. Only Data collectors saw the records of the study participants.
Data Availability
The data for this study cannot be made publically available at present. It will be made available from the corresponding author on a reasonable request.
