Abstract
Three-dimensional (3D) pile loop knit fabrics made up of polyethylene terephthalate (PET) yarns are encouraging scaffolds for engineering cardiac patches aiming at preserving the native heart following acute myocardial infarction (AMI) in survivors. However, the poor conductivity of PET limits the cell housing ability of the scaffold. In this study, the depositing of gold nanoparticles (AuNPs), with a thickness of 45 nm, on the back surface of the knitted scaffold was shown to change the dielectric properties of the scaffold, and increased its conductivity by approximately twofold at 1 MHz without compromising its mechanical properties. The values of the Young’s modulus of AuNP–scaffold hybrids both in the warp (240.7 kPa) and weft directions (62.24 kPa) at 20% strain were revealed not to be far from those of the pristine scaffold (warp 177.78 kPa; weft 59.21 kPa). Transmission electron microscopy images obtained after subjecting the AuNP–scaffold hybrid to collagenase degradation assay demonstrated that the diameters of the degraded AuNPs were in the range of 3.16–92.98 nm. Furthermore, C2C12 cells on AuNP–scaffold hybrids exhibited higher cell proliferation compared with the pristine scaffold, which was attributed to conductive AuNPs providing connections between groups of cells and good alignment of connexin-43 proteins leading to a better electrical signal propagation across the scaffold. Here, it has been shown that the integration of AuNPs onto the 3D knitted scaffold can improve the biological performance of an engineered cardiac patch, contributing to its therapeutic potential for myocardial tissue engineering.
Acute myocardial infarction (AMI), a coronary artery disease that has been declared to account for approximately one-sixth of global deaths, has been announced as one of the leading fatal diseases worldwide.1,2 Current cardiac therapies (e.g., reperfusion within an hour and a half after AMI, angiotensin-converting enzyme inhibitors, and ventricular assist devices) fail to preserve the native heart following AMI in survivors. 3 Furthermore, cell therapy has been researched since its birth in the 1990s through preclinical and clinical studies.4–6 Although various approaches, e.g., formation of new cardiomyocytes (CMs), new blood vessels, paracrine mechanisms (secretion of growth factors, chemokines, exosomes, cytokines by transplanted cells), an acute response by the immune system, have been proposed to explain cell delivery induced enhancement in myocardial healing, the exact mechanism clarifying the therapeutic role of safely transplanted cells into the heart is still unknown.7–9 In addition, housing the injected cells at the infarcted site is a challenge in cell therapy, as only a small fraction of injected cells, less than 1%, survive within the first month following transplantation.10–12 In this respect, patch therapy in cardiac tissue engineering consisting of biocompatible scaffold, cells, and bioactive molecules (e.g., growth factors) has been researched both in vitro and in vivo to increase transplanted cell survival and promote cardiac recovery.13,14
An ideal biocompatible scaffold should serve as a temporary housing for delivered cells, preventing them from being washed out at the infarct site until they produce their own extracellular matrix (ECM). 15 Researchers continue to design scaffolds with a porous structure as well as with an appropriate anisotropic mechanical strength (Young’s modulus of approximately 50 kPa end-diastole at a strain of 15–22% and 10–20 kPa end-systole at a strain of <10%) and electrical conductivity (5 × 10-5 S/cm and 1.6 × 10-3 S/cm in the direction of transverse and longitudinal, respectively) resembling and integrating well into native myocardium tissue.14,16
Electrically conductive biomaterials, either in particle form including gold nanoparticles (AuNPs), carbon nanotubes, graphene, or in polymer form containing polypyrrole (PPy) or polyaniline (PANi), are incorporated into the scaffold design to provide the continuation of electrical coupling of CMs in the myocardium.14,17 Since AMI induced nonconductive tissue that could block the electrical signaling of the infarct site with the rest of the myocardium, developing scaffolds using electroconductive biomaterials would help restore electrical signal propagation across the whole myocardium and contraction of CMs synchronously by minimizing cardiac rhythm disorders after cardiac patch implantation.14,17
AuNPs (e.g., gold nanospheres, gold nanorods, and gold nanowires), approved by the United States Food and Drug Administration (FDA) in the biomedical field, have been studied for decades in medicine including tissue engineering and regenerative medicine, biosensing (e.g., optical biosensor), bioimaging (e.g., contrast agent in computed tomography imaging), and drug and gene delivery.18–20 The optical properties (e.g., surface plasmon resonance [SPR] influenced by the size and shape of AuNPs), surface chemical characteristics (e.g., ability to be conjugated with various molecules such as drugs, peptides, and nucleic acid), and biocompatibility characteristics (e.g., in vivo low toxicity driven by especially size, shape, surface charge, surface coating, and concentration of AuNPs) of AuNPs make them potentially appropriate candidates for biomedical applications.21–23
Dvir et al. 24 developed alginate scaffolds in which gold nanowires were embedded and seeded with neonatal rat cardiac cells. They demonstrated that the integration of gold nanowires into the macroporous scaffold improved the electrical conduction across the pore walls of the alginate scaffold, increasing the expression of heart-specific electrical coupling (connexin-43 [Cx-43]) and contractile proteins (cardiac troponin I, α-sarcomeric actinin), and enhanced the contractile behavior of the cardiac patch. 24 Fleischer et al. 25 cultured neonatal rat cardiac cells within coiled electrospun poly(ε-caprolactone) (PCL) scaffolds covered with and without AuNPs. They exhibited that the AuNP-based engineered cardiac patch led to a larger cell area, higher CM aspect ratio, and higher engineered tissue contractions per minute than the cardiac patch without AuNPs. 25 You et al. 26 cultured neonatal rat CMs on a hydrogel scaffold composed of thiol-2-hydroxyethyl methacrylate (thiol-HEMA) mixed with HEMA at from 0% to 40% thiol-HEMA weight/HEMA volume synthesized with spherical AuNPs. They showed that the expression of Cx-43 increased on scaffolds with 40% thiol-HEMA weight to HEMA volume compared with 0% in the case of both the presence and absence of electrical stimulation, where the gold content increased in parallel with the increase in the thiol-HEMA weight to HEMA volume. 26 Qiu et al. 27 developed a L-dopamine (Dopa)-based methacrylate grafted gelatin (GelMa)/poly(ethylene glycol) diacrylate (PEGDA)/AuNPs cardiac patch, where the AuNPs were biosynthesized from bacteria (Staphylococcus aureus), and had spherical shapes and a size range of 5–20 nm. The AuNP-integrated cardiac patch improved heart function and reduced infarct area in a rat heart model experimentally exposed to myocardial infarction (MI). 27 Furthermore, there have been studies regarding the toxicity of AuNPs, which are conflicting, mainly due to the variability in experimental conditions, including size, shape, concentration, surface modification, surface charge of AuNP, cell lines used, administration routes, and animal employed.28,29 Chen et al. 30 investigated the toxicity effects of naked AuNPs with a diameter range of 3–100 nm for 4 weeks using BALB/C mice via intraperitoneal injection at a given dose. While particles with a diameter of 3, 5, 50, and 100 nm did not cause a serious detrimental effect on mice, lethality was induced by 8, 12, 17, and 37 nm particles. 30 Modica et al. 31 evaluated the oral toxicity of gold nanocrystals with a hydrodynamic radius range of 8–28 nm for 60 days by an administered repeated dose to Wistar rats, and no deaths or adverse harmful effects on rats were observed. Balfourier et al. 32 demonstrated that citrate-coated AuNPs with a size 4, 15, and 22 nm were degraded in vitro by primary human fibroblast cells with the key role of lysosome after up to 6 months observation, in contrast to the belief of the intracellular inertness of gold. They argued that this would help elucidate the metabolism of toxicity of AuNPs. 32
Recently, it was shown that anisotropic porous pile loop knit scaffolds were not cytotoxic to C2C12 myoblast cells. 33 This study aimed to improve the performance of the knitted scaffold further by integrating the AuNPs to assist the propagation of the electrical signal throughout the heart after implantation of the cardiac patch. First, the ability of AuNPs to deposit on a polyethylene terephthalate (PET) knitted scaffold and alter the structural properties, including the electrical properties of a scaffold, were investigated. Next, the effect of the AuNPs on cell proliferation and organization was examined. Overall, the integration of AuNPs onto the surface of the scaffolds were thought to be favorable for obtaining a functional cardiac patch.
Methods
Materials
The half-pile loop fabrics comprised 100 deniers (≈11 tex), nonintermingled, textured PET yarns, where one yarn contained 48 filaments in cross-section. The mean values of the physical and mechanical properties of the knit fabric are given in Table 1. 34 It is referred to as full-pile loop when ground and pile loops are knitted together at every round on the machine at full length of the fabric, and half-pile loop when pile loops join the formation of knitting structure one in two rounds. 35
Physical and mechanical properties of the knit structure.
The knit structure had a pore network consisting mainly of 100–500 µm macropores that were well interconnected with micropores (<90 µm).
To assess the effect of the AuNPs on the mechanical and wetting performances of the engineered AuNP–scaffold hybrids, the uniaxial tensile (ASTM D 5035-11 (2019), 36 Standard Test Method for the Breaking Force and Elongation of Textile Fabrics, utilizing a crosshead speed of 10 mm/min, and an initial gauge length of 25 mm), and contact angle (the sessile drop method with 4 µl distilled water on a Theta Lite Optical Goniometer; KSV Instruments Ltd, Finland) tests were performed using the same procedures as detailed in a previous study. 35
The equipment used for the analysis are a thermal evaporator, atomic force microscopy (AFM), broadband dielectric spectroscopy (BDS), scanning electron microscopy (SEM), and transmission electron microscopy (TEM), where the test procedures are given in the following sections.
AuNP–scaffold hybrid preparation
The 38 cm2 knitted circular samples were mounted in a Nanovak NVTH-350 thermal evaporator. Gentle rotation of the substrate plate aided uniform deposition. Gold films were prepared by evaporation of Au pellets (purity, 99.99%) from a tungsten boat at 1–3 × 10-6 torr at a deposition rate of 0.5 A/s. The thickness readings were obtained by adjusting the deposition time using a quartz crystal microbalance (QCM) through a thin-film deposition monitor (SQM-160, Inficon). The schematic representation of the AuNP–scaffold hybrid preparation is shown in Figure 1.

Schematic representation of AuNP–scaffold hybrid preparation.
Topography measurement by AFM
Topography measurement was performed using a VEECO Mulltimode 8 AFM Bruker, RTESP-300 scanning probe, where a resonance frequency of 300 kHz was used.
Conductivity measurement by BDS
Conductivity (σ', S/cm) was measured by an impedance dielectric spectroscopy analyzer Novocontrol GmbH Concept 50 Broadband Dielectric Spectrometer (Germany) with a frequency range of 0.1 Hz to 3 MHz connected to the standard sample cell BDCS 140 at temperature 22°C.
SEM
Samples were imaged using a ZEISS GeminiSEM 500 scanning electron microscope without additional gold coating. Elemental mapping was accomplished using EDAX Octane Electra energy-dispersive x-ray spectroscopy (EDX) and imaged via INCA software, without additional gold coating.
Biological assessment
Cell culture experiment and MTT assay
C2C12 mouse myoblast cells (ATCC® CRL-1772™) were seeded in a 75 cm2 flask with culture media contained Dulbecco’s Modified Eagle’s Medium (DMEM, Sigma, USA; with 4500 mg/l glucose) supplemented with 10% fetal bovine serum (FBS, Biological Industries, USA), 1% L-glutamine (Biological Industries, USA) and 1% Penicillin–Streptomycin–Amphotericin B solution (Sartorius, Germany) and incubated at 37°C, humidified in 5% CO2 in an incubator. When cells reached the required number, they were detached from the flask surface by using Trypsin-EDTA, and then cells were centrifuged at 300 g for 5 minutes.
Two groups, AuNP–scaffold hybrid and pristine scaffold, were prepared. Knitted samples from each group as 28 mm2 circular slices were placed in non-treated polystyrene 96-well plates (Corning, USA). The scaffold samples were prewetted with 1% Penicillin–Streptomycin–Amphotericin B (Sartorius, Germany) solution in DPBS for 2 hours. Then, all the media was aspirated and washed with phosphate-buffered saline (PBS, Biological Industries, USA) three times. Then 4 × 104 C2C12 cells with culture media were seeded onto scaffold samples in each group. Cells were incubated with samples for 3 and 5 days.
In MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) assay, eight samples were prepared for each group, where four samples were used for days 3 and 5 of the MTT assay. A 0.5 mg/ml final concentration of MTT was added to the wells, and 3 h later, all MTT solution was aspirated carefully. Then, dimethyl sulfoxide (DMSO) was added to solve the formazan crystals. The absorbance values were read at 560 nm using an enzyme-linked immunosorbent assay (ELISA) reader (Promega, Glomax, USA).
Furthermore, AuNP–scaffold hybrid samples were subjected to 7-day cell-free in vitro culture conditions in a cell culture incubator to ensure the binding of AuNPs onto the scaffold. Then, samples were washed and dried in air, and EDX-elemental mapping was carried out.
Cell immunostaining
Scaffolds were fixed by using 2.5% glutaraldehyde for 2 hours at room temperature, then washed with PBS and permeabilized with 1% triton X for 5 minutes. Then they were washed again. The cells were stained with Connexin 43 (1:250 in PBS; Elabscience, USA) as a primary antibody, incubated at +4°C overnight. The scaffolds were washed again and incubated with secondary antibody (1:1000; cat. no. 21207, Lifetech, USA) and DAPI (0.1 µg/ml, cat. no. D9542, Sigma, USA) for 1 hour at room temperature. Then scaffolds were washed again and imaged under a confocal microscope (Zeiss, LSM 900).
Collagenase degradation assay
A procedure from the literature was followed. 37 Here, scaffold samples were incubated in DMEM with 4500 mg/l glucose with collagenase type II (95 U/ml; Worthington, Lakewood, NJ) and pancreatin (0.6 mg/ml; Sigma-Aldrich) in a humidified, 37°C, 5% CO2 incubator for 1 week.
TEM
TEM experiments were conducted in a FEI TALOS F200S TEM, operating at 200 kV. Images were captured using TEM imaging and analysis (TIA) software.
Statistical analysis
The one-way analysis of variance (ANOVA) was used for statistical analysis by utilizing the trial version of JMP 18 statistical software. p-values less than 0.05 were considered significant.
Results and discussion
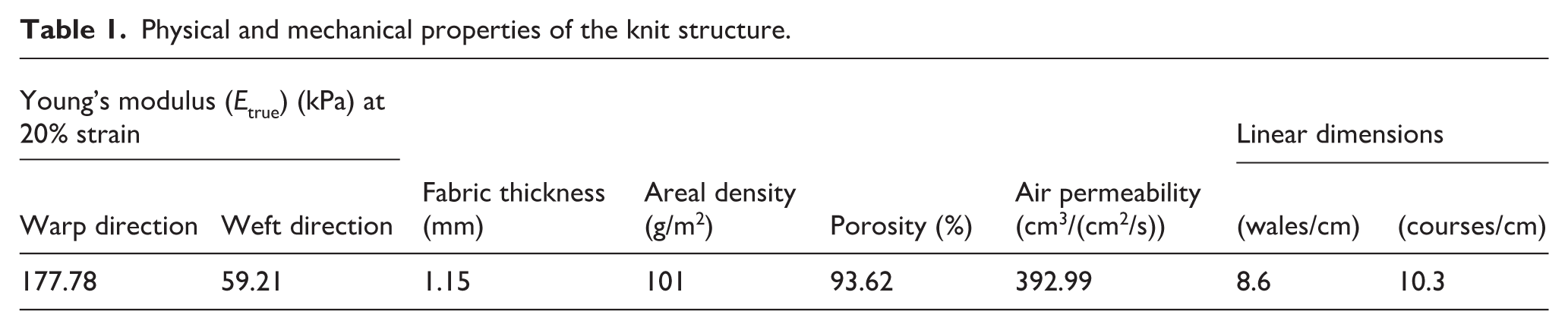
AuNPs, with a thickness of 45 nm, were evaporated onto the back surface of the fabric using a thermal evaporator, changing the color of the knitted scaffold as shown in Figures 2(a) and (b). Although the porous knitted structure allows the depositing of the nanoparticles in between the yarns, controlling the deposition speed assisted to uniformly coat the surfaces of the fabric filaments with nanoparticles layer by layer by providing a good adhesion. Here, the AuNPs were deposited homogeneously on the filaments without affecting the morphology of the fabric, which was visualized by SEM without additional Au coating (Figures 2c and d). Figures 2(e) and (f) show the topography of a pristine and AuNP-supplemented filament by AFM, respectively. While the pristine filament had a smooth surface, the AuNPs were relatively visible on the surface of the modified filament. Furthermore, elemental composition analysis performed using EDX confirmed that the AuNPs observed on the filaments were certainly Au (weight % of Au: 63.19; Figure 2g). In addition, Figure 2(h) shows Au-related peaks (weight % of Au: 58.27) obtained by EDX after the 7-day treatment with subsequent thorough washing. This indicates that Au content was kept on the filaments during the 7-day cell-free in vitro culture period.
(a) Macroscopic picture of a pristine scaffold. (b) Macroscopic picture of an AuNP–scaffold hybrid. (c), (d) SEM images of the AuNP–scaffold hybrids. (e) Topography of a pristine filament by AFM. (f) Topography of an AuNP–scaffold hybrid by AFM. (g) EDX spectrum of AuNP–scaffold hybrid. (h) EDX spectrum of AuNP–scaffold hybrid after 7 days of treatment with subsequent thorough washing.
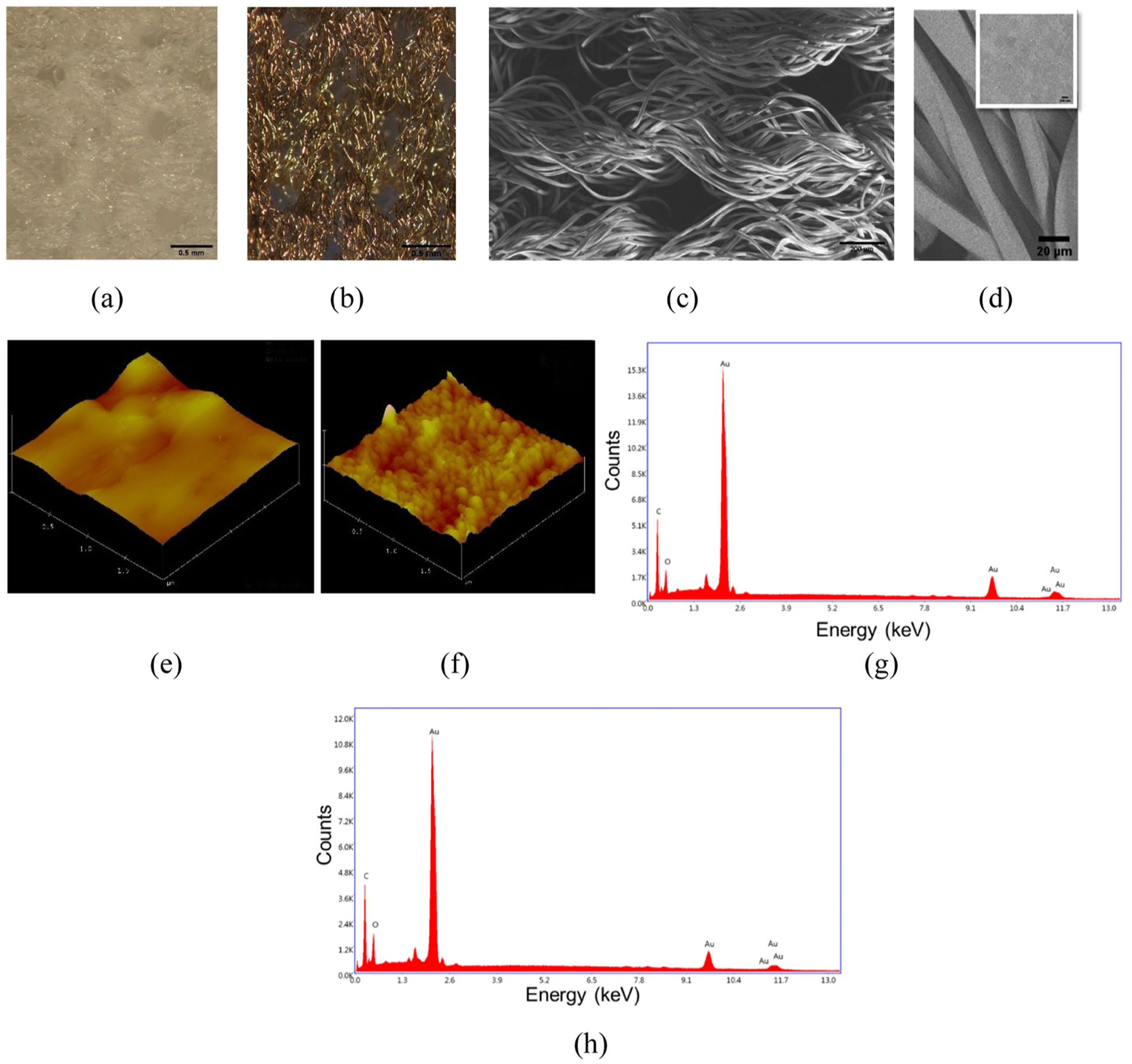
The values of the Young’s modulus of the AuNP–scaffold hybrids at 20% strain considering the true stress–strain curves were observed to be similar to the pristine scaffold both in the warp (240.7 kPa) and weft (62.24 kPa) directions (Figure 3). Thus, this would result in no considerable shift in the stiffness of the scaffold and consequently in cell behavior including cell proliferation, migration, and differentiation. Furthermore, the contact angles of the pristine scaffold and the AuNP–scaffold hybrid were measured as 146° and 137°, respectively. Here, it was revealed that adding AuNPs to the knitted scaffold did not change the hydrophobic character of the scaffold.
Stress–strain curves of the pristine and AuNP–scaffold hybrid (adapted from Haroglu 34 ).
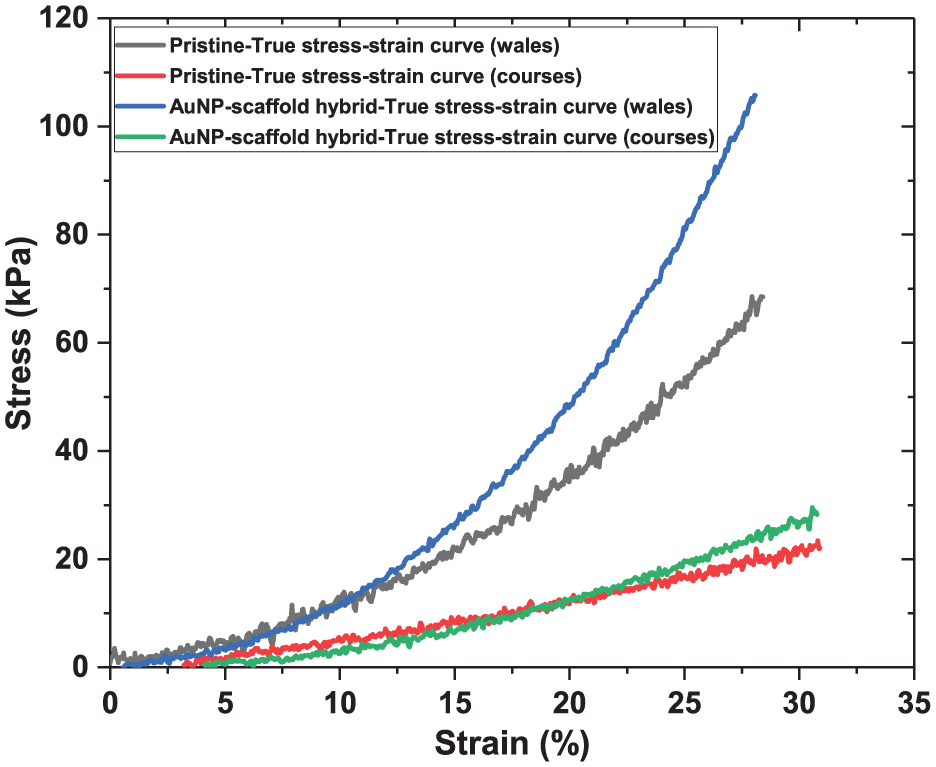
The alternating current (AC) conductivity (σ') measurements of the prepared AuNP–scaffold hybrid and the pristine scaffold were done over a frequency range from 100 to 106 Hz using BDS (Figure 4).
Frequency sweep for conductivity (σ′) of AuNP–scaffold hybrid (red line) and pristine scaffold (black line).
In Figure 4, the conductivity increases with frequency, which is in agreement with the literature.38,39 AuNP–scaffold hybrid has higher conductivity in the whole measured range due to the presence of conductive AuNPs. The maximum conductivity was observed at 1 MHz frequency, where the conductivity values of the AuNP–scaffold hybrid and pristine scaffold were 1.883 × 10−9 and 9.5 × 10−10 S/cm, respectively. Thus, the presence of the AuNP coating on the filaments changed the dielectric properties of the scaffold and increased its conductivity approximately twofold (1.883 × 10−9/9.5 × 10−10 ≈ 2). At lower frequencies, the difference in conductivities was more pronounced with σ' values of 1.08 × 10-13 and 5.65 × 10-15 at 10 Hz for AuNP–scaffold hybrid and pristine scaffold, respectively. Here, PET is a dielectric material that exhibits high electrical resistance. The deposited AuNPs are not expected to form a continuous film due to the irregular and rough nature of fabric surfaces.
Since AuNPs can be enzymatically disassembled in in vivo conditions, where collagenase type II is reported to be highly active after ventricular remodeling following the initial few days of MI, a collagenase degradation assay was applied.40,41 Figures 5(a) and (b) show the TEM images of the degraded AuNPs and the histogram of the particle size of the AuNPs, respectively. The diameters of the almost spherical AuNPs were observed to be in the range of 3.16–92.98 nm. The samples used for the assay were washed, and then EDX analysis was performed (Figure 5c), where AuNPs were still able to be observed (weight % of Au: 53.46) on the filaments. Here, when Figure 5(c) is compared with Figure 2(g) (weight % of Au: 63.19) and Figure 2(h) (weight % of Au: 58.27), the reduction rate in gold content might be affected by the coating thickness. Nevertheless, the issue of size-related toxicity of AuNPs is still under discussion and research in this area is continuing.
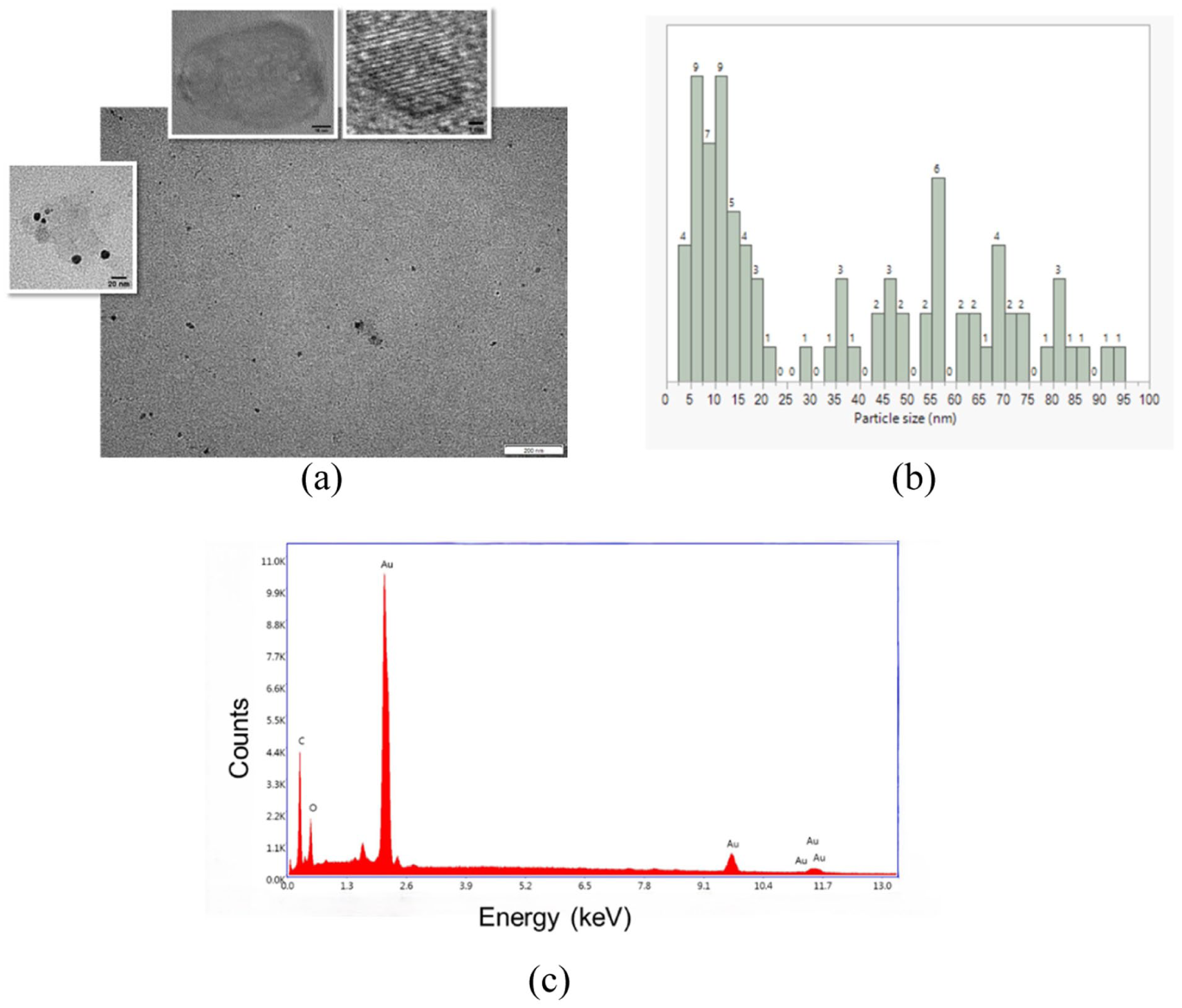
(a) TEM images of AuNPs after degradation assay. (b) Histogram of AuNPs. The numbers on the rectangles display the number of AuNPs (counts). (c) EDX spectrum of AuNP–scaffold hybrid after collagenase degradation assay.
The MTT assay evaluated the cytocompatibility of the AuNP–scaffold hybrids with C2C12 myoblasts, and the results are shown in Figures 6 and 7. The absorbance values at 560 nm of the MTT assay conducted on day 3 and day 5, both between the pristine scaffold and AuNP–scaffold hybrid and within themselves, were observed to be statistically different (p < 0.05). In Figure 7, the green diamond and the red box represent the 95% confidence interval and the outlier box plot, respectively. Furthermore, the absence of intersections of the circles in the right column of the graphs demonstrates that the related means are significantly different.
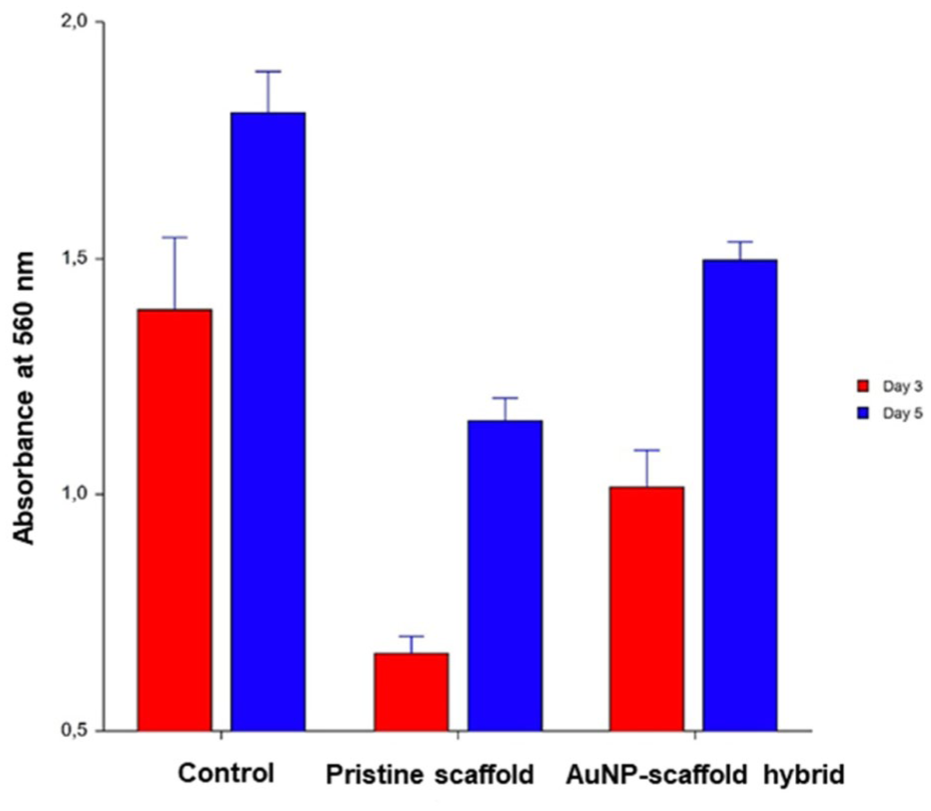
Absorbance values of MTT assay for day 3 (red column) and day 5 (blue column) in control, pristine scaffold, and AuNP–scaffold hybrid samples.
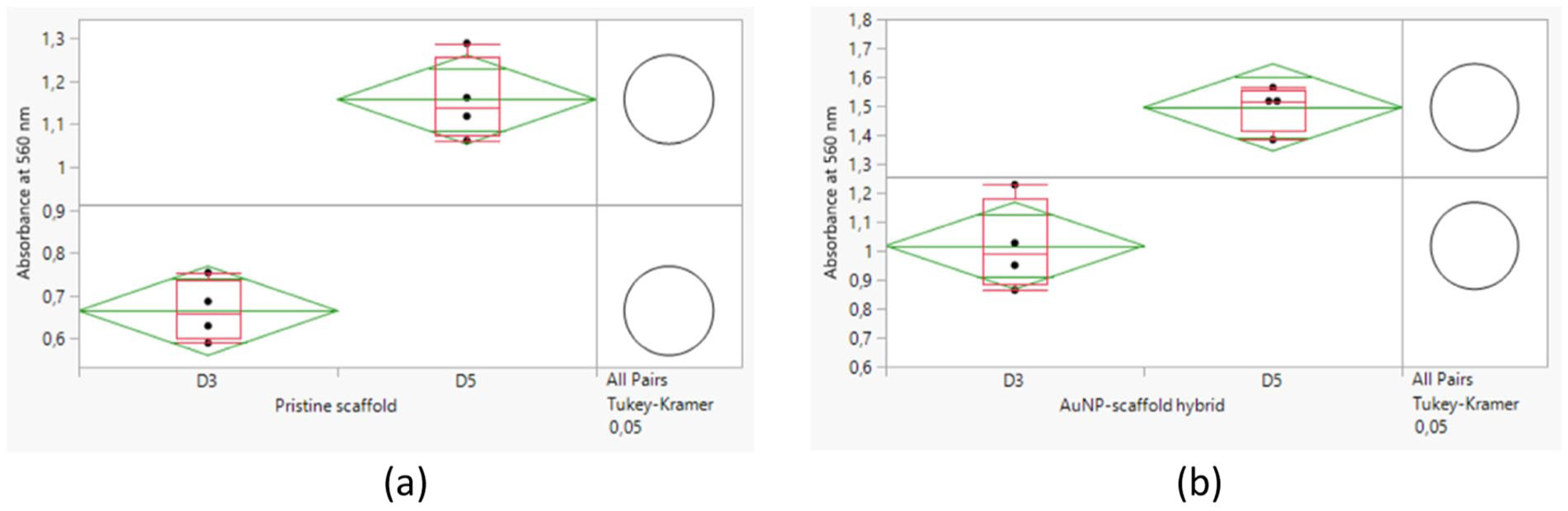
Results of cell proliferation assay for scaffolds: (a) MTT assay for pristine scaffold and (b) MTT assay for AuNP–scaffold hybrid. D3, day 3; D5, day 5.
The AuNP–scaffold hybrid had no cytotoxic effect on C2C12 cells. Unlike the recent study ECM protein (e.g., fibronectin) was not used to coat the hydrophobic scaffolds, both the pristine and AuNP–scaffold hybrid, to promote cell adhesion. Here, multiple large pores in polygonal geometries (pore diameters 200–500 µm) of the scaffold may have contributed to the improved microenvironment for cell attachment. Furthermore, depositing AuNPs onto the filament surface was observed to lead to higher cell proliferation compared with the pristine scaffold and enhance scaffold functionality. These effects can be attributed to the fact that AuNPs can construct conductive connections across the scaffold by making bridges between adjacent groups of cells and increasing the expression of Cx-43, gap junction protein.26,42
On day 5, knitted scaffolds were stained for Cx-43, an electrical coupling protein, to assess the AuNP–scaffold hybrid versus pristine structure. Figure 8 shows that Cx-43 proteins were distributed to a great extent in order between adjacent C2C12 cells grown in the AuNP–scaffold hybrids (white arrows in Figure 8) when compared with those in pristine scaffolds. The organized alignment of Cx-43 proteins may lead to better electrical signal propagation in an anisotropic structure, which may improve the performance of engineered cardiac patches. 43
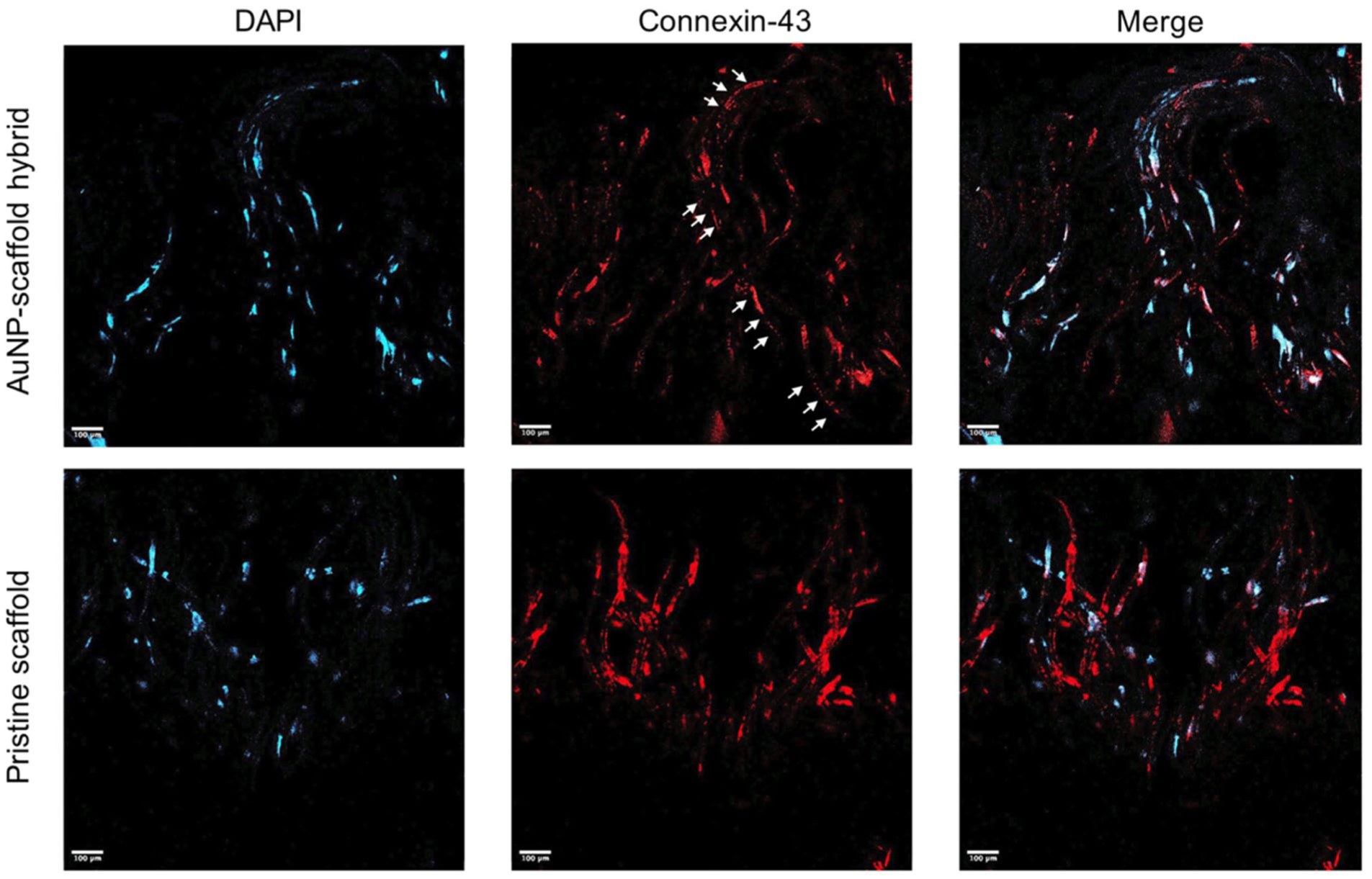
Confocal images of C2C12 cells on pristine scaffold and AuNP–scaffold hybrid stained with DAPI, nuclei (blue), and Cx-43 (red) on day 5.
Conclusion
In this study, AuNP–scaffold hybrids were designed to improve the conductivity of a cardiac patch. A uniform distribution of AuNPs on the back surface of the knitted fabrics was achieved, which was demonstrated by SEM images. In addition, AFM images showed that the surface roughness increased due to the AuNPs, while the pristine filament had a smooth surface. Furthermore, the preservation of the gold content on the surface of the filaments was confirmed by EDX analysis after a 7-day cell-free in vitro culture period.
The electrical property was analyzed by BDS. The conductivity of the knitted scaffold increased approximately twofold at a frequency of 1 MHz, where the conductivity values of the AuNP–scaffold hybrid and pristine scaffold were 1.883 × 10-9 and 9.5 × 10-10 S/cm, respectively (1.883 × 10−9/9.5 × 10−10 ≈ 2). The values of the Young’s modulus of AuNP–scaffold hybrids both in the warp (240.7 kPa) and weft directions (62.24 kPa) at 20% strain were revealed not to be far from those of the pristine scaffold (warp 177.78 kPa; weft 59.21 kPa). This would not cause any significant change in the stiffness of the scaffold and, hence, in cell behavior including cell proliferation, migration, and differentiation. Furthermore, integrating AuNPs into the knitted scaffold did not change the wettability–hydrophobicity of the scaffold.
TEM images obtained after subjecting the AuNP–scaffold hybrid to collagenase degradation assay showed that the diameters of the degraded AuNPs were in the range of 3.16–92.98 nm, where the size-dependent toxicity of AuNPs is still being investigated. Biological experiments revealed that all of the scaffolds displayed good cell adhesion. Furthermore, AuNP–scaffold hybrids showed higher cell proliferation when compared with the pristine scaffold, which was attributed to the deposited AuNPs building connections between cell groups and good alignment of Cx-43 proteins, leading to better electrical signal propagation throughout the scaffold.
The biocompatibility and increased conductivity of three-dimensional AuNP–scaffold hybrids make them well-applicable candidates for cardiac tissue engineering in the future.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the University of Erciyes Research Council (Project code: FHD-12936) for providing funds.
