Abstract
In this paper we present a rapid and simple one-pot method to obtain gold nanoparticles functionalized with folic acid using a photochemistry method. The bioconjugate folic acid-gold nanoparticle was generated in one step using a photo-reduction method, mixing hydrogen tetrachloroaurate with folic acid in different ratios and varying the illumination time of a mercury lamp (λ = 255 nm). Scanning electron microscopy showed a particle size of around 40–50nm and dynamic light scattering exhibited that the zeta potential varies from −41 to −50mV with different illumination times. Storage in the dark at 4°C prolongs the stability of folic acid-gold nanoparticle suspensions to up to 26 days. Ultraviolet visible and Fourier transform infrared spectroscopy showed a surface plasmon band of around 534nm and fluorescence spectroscopy exhibited a quenching effect on gold nanoparticles in the fluorescence emission of folic acid and thus confirmed the conjugation of folic acid to the surface of gold nanoparticles. In this study we demonstrate the use of a photochemistry method to obtain folic acid-gold nanoparticles in a simple and rapid way without the use of surfactants and long reaction times. The photochemical synthesis of FA-AuNPs opens new perspectives for creating novel functional nanomaterials for biomedical applications.
1. Introduction
In recent years the number of applications of gold nanoparticles in biology and medicine has increased due to its excellent physical and chemical properties, such as strong absorption, ease of synthesis, simplicity of conjugation chemistry and excellent biocompatibility [1–5]
Among the conventional methods for preparation of gold nanoparticles (AuNPs) [6–8], the most commonly used is the citrate reduction of HAuCl4 [9]. One of the main requirements for the application of AuNPs in medicine is the possibility to be functionalized with biological molecules [10, 11]. Different types of biomolecules, such as proteins [12], peptides and DNA [13], have been immobilized on the surface of AuNPs for targeting and detecting cancer cells. AuNPs have been activated to target various cancer cells using folic acid (FA). FA is a ligand that is useful for targeting cell membranes and enhancing AuNP endocytosis by the folate receptor. FA receptors can be direct targets for drug delivery [14–18], which explains the diversity of strategies used for folate conjugation.
Numerous attempts using poly(a-midoamine) (PAMAM) dendrimers and poly(ethylene glycol) (PEG) to develop biodegradable hydrophilic folate-nanoparticle systems for cancer targeting have been reported [19]. Another strategy to anchor FA makes use of glutathione to form GSH-capped AuNPs with carboxylic groups as a linker thus allowing the anchoring of FA [20]. Li et al. reported for the first time a facile one-step synthesis to obtain FA-AuNPs by heating an aqueous solution of HAuCl4/FA in which FA acts as both a reducing and stabilizing agent [21]. In an approach Zhang et al. developed a rapid method to fabricate in one step FA-AuNPs under microwave irradiation [14]. Although the obtained AuNPs were stable and displayed uniform size in both methods, the synthesis is time consuming and strict control of the temperature is necessary.
In this work, we synthesized in one step an FA-AuNP bioconjugate using a simple and rapid one-pot photochemical method. SEM was used to characterize the size distribution and DLS was used to determine zeta potential and the stability of the FA-AuNP bioconjugate. UV-Vis, FTIR and fluorescence spectroscopy was employed to confirm the presence of the plasmon band and to show evidence of the immobilization of FA on AuNPs. The synthesis of these nanoconjugate systems opens new possibilities for their use in the treatment of infectious diseases and cancer cells.
2. Experiment
2.1 Photochemical synthesis
FA was dissolved in water. FA powder (0.029g) was mixed with H2O (25mL) and 50μL of NaOH (1M) due to the poor solubility of FA. The solution was magnetically stirred until a yellow-to-clear transition was observed. 611μL of FA solution (2.7mM) were adding slowly to 9089μL of H2O double distilled under magnetic stirring for seven minutes in a photochemical reactor (Ace-Glass model T-121m) with a UV mercury lamp λ=255nm 5.5W (UVP, Corporate Headquarters) (Figure 1). Then 300μL of HAuCl4 (1mM) were adding slowly to the photochemical reactor containing the FA solution. The light yellow colour of the HAuCl4 solution became dark and a precipitate was observed. This precipitate could be due to the poor solubility of FA at low pH produced by the HAuCl4 solution. For this reason, a further amount of NaOH (100μL 1M) was adding to obtain a clear orange solution.

Photochemical reactor for the synthesis of the bioconjugate FA-AuNP
The reaction was magnetically stirred for 15 minutes, with constant illumination using a UV lamp at room temperature. Samples were collected after 10, 45 and 120 minutes from the photochemical reactor to be analysed and characterized. A cooler system was adapted to the photochemical reactor to avoid the reaction overheating. The solution FA-GNP was then dialyzed three times with distilled water (MCWO 8000) to ensure the complete removal of unconjugated FA.
2.2.1 Characterization of the bioconjugate
UV-Vis absorption spectra were taken in a quartz cell with a 1.0cm path length using an Agilent-HP 8453 spectrophotometer. FTIR spectra were taken in a NICOLE AVATAR 360 FTIR spectrophotometer. The fluorescence emission from FA-AuNP solutions were measured using a standard luminescence spectrophotometer Perkin Elmer LS-55. A Zetasizer Nano particle analyser series (Malvern, model ZEN3500) was used to observe the dispersion state measuring the zeta potential of the FA-AuNP bioconjugate. Finally the SEM images were obtained using a Quanta 650 FEG scanning electron microscope (Oregon, USA).
3. Results and discussion
The absorption spectra of different solutions of FA-AuNPs are shown in Figure 2. The solutions were obtained with different molar ratios of FA/HAuCl4 (0.4:1, 0.5:1, 0.56:1, 0.63:1) and different irradiation times (10, 45 and 120 minutes). At FA concentrations below 0.17mM (0.4:1) the plasmon band did not display a well-defined absorption peak and some precipitate appeared, which might be due to insufficient FA to protect and reduce the auric salt. A similar result was observed by Li et al. [21]. On the other hand, when the molar ratio FA/HAuCl4 was increased to 0.63:1 the absorption peak associated with the plasmon band was also observed with an undefined shape. When a molar ratio of 0.56:1 was used, the absorption spectra showed a more well-defined plasmon band and a uniform size distribution was observed by SEM (around 50nm), see Figure 3.

Absorption spectra of the bioconjugate FA-GNP obtained with different molar ratios FA/HAuCl4 (a) 0.4:1, (b) 0.5:1, (c) 0.56:1, (d) 0.63:1 and irradiation times

SEM micrograph (a) and size distribution of FA-AuNP (b) at 10 min of irradiation time
A plasmon band of around 534nm is shown in Figure 2c. The absence of a band indicates no formation of nanoparticles when a ratio of 0.63:1 was used, shown in Figure 2d
The effect of UV light irradiation time in the photochemical reactor was also studied. Figures 2a and 2b show an increase of the absorption band around 530–543nm with the increase of illumination time. On the other hand, Figure 2c (0.56:1) shows no dependence of the irradiation time. Furthermore, the intensity of the plasmon resonance band remains unchanged. This result indicates that at ten minutes of irradiation, UV light is enough to form the bioconjugate FA-AuNP with a well-defined plasmon band. A different approach, which makes use of heating and microwave irradiation to prepare FA-GNP, requires longer times (between two and eight hours) [14].
Zeta potential is a physical property that can be related to the stability of colloidal dispersions and indicate the degree of repulsion between adjacent, similarly charged particles in the dispersion [22]. Figure 4 portrays the zeta potential and particle size for different days of the FA-AuNP at a concentration of 0.17mM. Ten minutes of irradiation was enough to form a stable colloidal nanoparticle (-41 mV) due to the negative charge on the surface of AuNPs. The bioconjugate remained stable for 26 days with sizes ranging from 40.4 to 46.4nm and the values of zeta potential evidenced the stability of the FA-AuNP. Thus FA has served as a reducing and stabilizing agent to form AuNPs with good stability. This result is an indication that the FA-AuNP conjugate is formed in stable dispersion and can be useful for biomedical applications that involve physiological conditions.

Size and zeta potential of FA-GNP stored in the dark at 4°C.
The comparison of FTIR spectra of free and conjugated FA is shown in Figure 5. The bands between 3600 and 3400cm−1 in Figure 5a are due to the hydroxyl (OH) stretching bands of glutamic acid moiety and the NH- group of the pteridine ring of FA. These groups of vibrations are not present in Figure 5b (FA-AuNP), probably due to the fact that the –NH
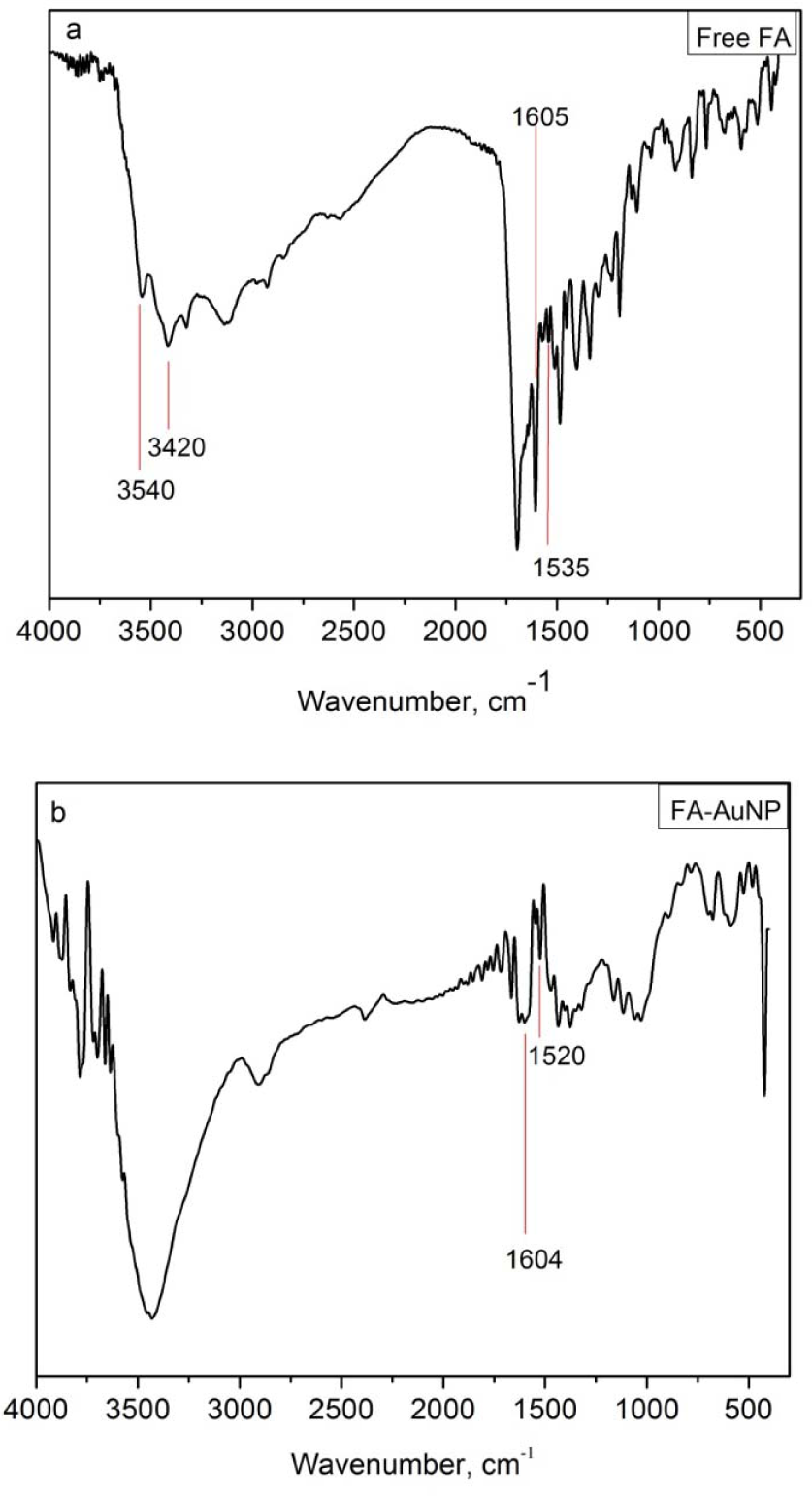
FTIR of free (a) FA and (b) FA-AuNP
The IR absorption peaks at 1605 (bending mode of –NH vibration) and 1635cm−1 (stretching vibration of C=O) of free FA are present in the spectrum of FA-AuNP, indicating the binding of FA to the AuNPs. The interval of vibrations between 1464 and 1535cm−1 due to the benzene ring of FA are present in both spectrums.
AuNPs have been considered as an effective quencher of a variety of molecules, the decrease of their fluorescence is observed through a variety of transfer electron mechanisms [22, 23]. The conjugation of FA to AuNPs was further studied by fluorometric measurement. A solution of free FA and FA-AuNP was determined by fluorescence emission using 364nm as the excitation wavelength. From Figure 6 a low intensity emission from FA-AuNP can be observed, compared with the emission from FA. The fluorescence emission of FA was quenched by AuNPs, as is shown in Figure 6. In a similar work a protein bounded to AuNPs with a size of 40nm showed a decrease in the intensity emission of the bioconjugate [24, 25] and the quenching effect was explained by a decrease in the electric field presented by the AuNP at a close distance to the protein (4nm). Analogously, in our system the short distance between FA and our 40–50nm sized AuNPs probably could induce the quenching of FA.

Fluorescence emission spectra of free FA (solid line) and FA-GNP (dashed line)
Furthermore, quenching of FA could be attributed to the dipole energy around the GNP, which reduces the ratio of the radiative to non-radiative decay and the quantum yield of FA, thus producing the quenching effect [26].
Compared with the previous typical synthetic process of FA-AuNPs [4, 8, 14, 27], the photochemical reduction of HAuCl4 with FA is more convenient since this approach proposes the shortest time required for the generation of bioconjugate FA-AuNP. The amino (-NH2) and hydroxyl (-OH) groups of FA probably help photo reducing the gold ion into gold nanoparticles, thus acting as both the reducing and stabilizing agent. Finally, FA adsorbed on the surface of the AuNPs provided stability due to the presence of negative charges around the bioconjugate.
4. Conclusions
We have developed a new and rapid one-pot method to obtain AuNPs functionalized with FA using photochemical reduction. The irradiation of HAuCl4 and FA solution leads to obtaining FA-AuNPs with a well-defined plasmon band and a size distribution ranging from 40–60nm. This study reports a shorter time reaction method for the generation of FA-AuNPs without additional reagents such as surfactants and polymers. Thus, these FA-AuNPs bioconjugates may be used as suitable and efficient materials for biomedical applications such as infectious disease treatments and cancer diagnosis.
Footnotes
5. Acknowledgments
The Colombian Administrative Department of Science, Technology and Innovation, COLCIENCIAS, (Project 110245921468) is gratefully acknowledged for financial support.
