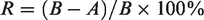Abstract
Plant-based antimicrobial finishing of textiles is comparable in efficacy, can replace synthetic antimicrobial agents, and is environmentally safe and effective. However, developing durable antimicrobial finishes on cotton-based textiles is a challenge. This research reports the development and characterization of nano-emulsions obtained from Karanja [Milletia pinnata] and Gokhru [Pedalium murex Linn] plants. The nano-emulsions were produced using Milletia pinnata, coconut oil, and curry leaves (nano-emulsion 1) and Pedalium murex, coconut oil, and curry leaves (nano-emulsion 2). The nano-emulsions were characterized for their thermal stability, particle size, pH, and percentage add-on. Two different oils, with surfactant (polysorbate) ratios [1:1 and 1:2], were finished on organic cotton fabrics using a batch process. Scanning electron microscopy images were evaluated to determine the surface morphology of the finished fabrics, and gas chromatography-mass spectrometry images of nano-emulsions were studied to determine the specific chemical constituents of nano-emulsions 1 and 2. The finished fabrics were evaluated for their antimicrobial resistance using various gram-positive bacteria [Staphylococcus aureus, Staphylococcus epidermidis], which are found on human skin and cause nosocomial infections, gram-negative bacteria [Escherichia coli and Klebsiella pneumoniae], which cause urinary tract infections, and fungi [Aspergillus niger]. The antimicrobial resistance was in the range of 98.62–99.87%, even after 10 washes, indicating that the finishes were effective and durable. The finished and unfinished fabrics were also evaluated for their performance properties, tensile strength, and moisture vapor transmission rate, and the results indicated good durability and comfort characteristics. Our findings highlight the potential of plant-based antimicrobial agents for durable finishing of cotton textiles with antimicrobial properties, thus preventing the spread of infections.
Keywords
The recent global pandemic has raised awareness of hygiene and increased the need to control the transmission of infection through textiles. Textile industry professionals and academics have focused on developing various antimicrobial and antiviral compounds for textiles. Textiles intended for hygiene and medical applications are finished with a range of antimicrobial agents, such as triclosan, metallic salts (TiO2 and ZnO), chitosan, phenolic compounds, N-halamines, and quaternary ammonium compounds (QACs), mainly for the fiber types of cotton, polyester, nylon, wool, acrylic, and polypropylene. 1 Textiles made of natural fibers, such as cotton, wool, and silk, provide excellent conditions for the growth of bacteria and fungi compared to synthetic filaments. 2 Bacteria such as Staphylococcus epidermidis and Corynebacterium cause body and clothing odor, whereas Candida albicans and Epidermophyton floccosum cause skin irritation and infections. 3 Staphylococcus aureus is a gram-positive bacterium that causes community and hospital-acquired infections. 4 Klebsiella pneumoniae and Escherichia coli are gram-negative bacteria and belong to the Enterobacteriaceae family, which causes community and hospital infections and has high antimicrobial resistance (AMR). 5 Klebsiella pneumoniae causes pneumonia, sepsis, and inflammation of the urinary tract. 6 E. coli also cause diseases in the intestinal tract, renal system, and central nervous system. 5 AMR is a global challenge, and it is estimated that 1.2 million people die from drug-resistant infections. 7 AMR occurs when bacteria, fungi, and viruses no longer respond to medicines, making them harder to treat, spreading infection and illness, and causing death. 8
Textiles are finished with antimicrobial agents using several surface modification techniques – continuous and exhaust methods, nanotechnology, micro-encapsulation, layer-by-layer and vapor deposition, and the sol–gel method. 9 Antimicrobial agents that generally kill bacteria are called biocidal, 10 and they inhibit the growth of microorganisms, which also prevents odor formation, also referred to as biostatic. In the biostatic mechanism, although it inhibits microorganisms, it may leave the surface with live bacteria, causing transmission of infection. Most antimicrobial agents used in textiles are biocidal. 11 The mechanism of antimicrobial activity is based on controlled release and barrier formation mechanisms. In the controlled release mechanism, the antimicrobial agent is incorporated into the textile fibers or chemically bonded, and a gradual release of antimicrobial agent kills the microbes.2,12–14 However, the release of the antimicrobial substance decreases in concentration over time (possibly due to repeated laundering), and its effectiveness is lowered. In the barrier formation, the antimicrobial agent bonds onto the textile surface through chemical bonding and they do not leach or release the antimicrobial agent but rather serve as a barrier to the microbes and kill when in contact with the textile surface.13,15
The use of synthetic chemicals (silver-based and mineral-based) as antimicrobial agents may affect health and the environment. For instance, QACs such as benzalkonium chloride are widely used in hand sanitizers and facial cleansers. When released into waterways, these chemicals may not biodegrade easily, and their increased use of QACs may lead to bacteria developing resistance and may spread infections. 16 Many textiles finished with antimicrobials that have intimate contact with the skin affect the skin-derived microbes, causing intolerance of the human body and allergies, and affecting human skin flora. 17 Triclosan, widely used as an antimicrobial chemical in textiles, is found to be toxic and affects human health (endocrine-disrupting chemical, skin irritations, cancer development). 18 Therefore, it is necessary to identify antimicrobial agents that have the most negligible impact on human health and the environment. The use of plant-based antimicrobial compounds is environmentally friendly, safe, and non-toxic19–21 compared with synthetic chemicals and inhibits bacterial pathogens. Several herbal extracts possess bioactive compounds, such as the salvation plant, Salvia officinalis L., which possesses bioactive compounds with antimicrobial, antioxidant, anti-inflammatory, and antiviral effects. 22 Bioactive substances in plants include basil (eugenol as the active substance), neem (limonoids such as azadirachtin), and turmeric (curcumin). 23 Active bio components in plants enable an antimicrobial effect on textiles, such as terpenoids, which possess resistance to S. aureus, P. aeruginosa, and V. cholera.24,25 Flavonoids possess resistance to bacteria including K. pneumoniae, S. centerica, P. aeruginosa, S. aureus, E. coli,26,27 while quinones inhibit S. aureus, B. subtilis, and P. aeruginosa.28,29 Plant/herbal extracts from leaves, stems, fruits, roots, seeds, and bark offer antimicrobial properties. 30 Previous studies reported antibacterial and antifungal efficacy using neem (Azadirachta indica), 31 tulsi leaves (Ocimum sanctum), 32 aloe vera, 33 and eucalyptus leaves. In this study, two herbal nano-emulsions were prepared using a combinations of herbal oils, namely Milletia pinnata L., curry leaves, and coconut oil, and another combination using Pedalium murex, curry leaves, and coconut oil.
Milletia pinnata, commonly called Karanja, belongs to the Fabaceae family, 34 which originated in India and is found in parts of Southeast Asia and Australia. 35 Karanja is also known as Pongamia glabra, Derris indica, and Pongamia pinnata L. 34 Different parts of the Karanja tree (leaves, flowers, fruits, bark, and seeds) are used; they possess medicinal properties and are used for nutritional, medical, and pharmacological applications. 36 The leaves of Karanja contain bioactive molecules, such as phenolic acids and flavonoids, which exhibit antimicrobial activity. 37 The aqueous methanol extracts obtained from the bark of Karanja exhibited higher AMR, with the zone of inhibition ranging from 16.7 to 26.2 mm, compared with leaves with a range of 10.2–16.2 mm. Authors posit that the antimicrobial activity of aqueous methanol extract of Pongamia pinnata is high in phenolic acids and flavonoids. 37 Flavonoids are known to retard the growth of microorganisms through inhibiting nucleic acid synthesis. 38 Pongamia pinnata leaf extracts of methanol, chloroform, and ethyl acetate have shown antibacterial effects against food-borne pathogens B. subtilis, S. aureus, L. monocytogenes, P. aeruginosa, and S. typhimurium. Ethyl acetate and methanol extracts exhibited a strong antibacterial effect against all four gram-positive bacteria, namely B. subtilis, S. aureus, L. monocytogenes, and L. monocytogenes, with their respective diameter zones of inhibition of 13–18 mm. 39 Authors suggested that the antibacterial activity of Pongamia pinnata leaf extracts was due to bioactive phytochemicals (flavonoids, alkaloids, flavones, and furanodiketone). 39
Pedalium murex, commonly called Gokhru, belongs to the Sesame family and grows in South India, Sri Lanka, Mexico, and Pakistan. 40 Different parts of the plant – leaves, roots, and seeds – have medicinal value and treat various diseases, such as the urinary tract and reproductive system. 40 Moreover, it is widely used in Ayurvedic medicine to treat various symptoms, including coughs, asthma, skin diseases, and piles. 40 Earlier studies have shown that aqueous leaf extracts of Pedalium murex showed resistance to fungal growth (Aspergillus niger) with a diameter zone of inhibition of 15 mm. 41 In addition, several studies have reported the phytochemical constituents of Pedalium murex, such as fatty acids, flavonoids, triterpenoids, steroids, tannins, saponins, ursolic acid, and vanillin,42–47 which inhibits microbial growth AMR.
Murraya koenigii (Linn), commonly called curry leaves, is widely used in cuisine due to its aromatic flavor. 48 Root extracts of Murraya koenigii in organic solvents (methanol, chloroform, and hexane) showed good inhibition to microbial growth. 48 Authors reported that the plant possesses carbazole alkaloids, which are cytotoxic and antioxidative, and have anti-inflammatory properties,49–51 and the leaves are rich in mono-terpenoids and sesquiterpenoids, which exhibit antifungal properties. 52 Curry leaf extracts inhibited the growth of E. coli and S. aureus, which showed zones of inhibition of 3.2 and 5.9 mm, respectively, when using a 10 mg/mL concentration of curry leaves. 53
Coconut oil is an edible oil extracted from the dried kernels of coconuts 54 with 92% saturated fatty acids. There are different types of coconut oil obtained from different parts of the coconut. 55 Coconut oil showed antibacterial activity against Pseudomonas aeruginosa, Escherichia coli, Proteus vulgaris, and Bacillus subtilis. 56 More recently, virgin coconut oil (VCO) was made from fresh kernels without heating, mainly by fermenting with microbes (Lactobacillus fermentum and Lactobacillus plantarum). VCO contains high lauric acid, 54%, whereas coconut oil has only 2.81%. 57 Moreover, VCO has lactic acid bacteria (LAB; Lactobacillus plantarum and Lactobacillus paracasei), inhibiting bacterial growth of Pseudomonas aeruginosa, Klebsiella pneumoniae, Staphylococcus aureus, S. epidermidis, and Escherichia coli. 57 It is also interesting to note that coconut oil, when formulated as a cream, is found to exhibit antibacterial and antifungal properties, as the emulsifying agent in the cream aids in the penetration of the active compound monolaurin. 58
It can be noted from the above review that plant-based constituents derived from Milletia pinnata, Pedalium murex, Murraya koenigii, and coconut oil have the potential to be used as antimicrobial agents. However, none of these herbal extracts have been used in combination and reported for their usage in organic cotton fabrics, particularly in the preparation of nano-emulsion, determining the method of finishing on the fabrics, and evaluating their percentage add-on. In addition, the evaluation of finished fabric performance parameters, including wash durability, antibacterial resistance, antifungal efficacy, tensile strength, and comfort properties, has not been reported before, and it adds to the literature. Therefore, for the first time, this research reports the potential of nano-emulsions derived from these herbal combinations, which produce an eco-friendly antimicrobial finishing of organic fabrics suitable for hygiene applications. Therefore, this research aims to determine the antimicrobial efficacy and performance of GOTS (global organic test standards) certified organic cotton fabrics finished with nano-emulsions produced using mixtures of Milletia pinnata L., curry leaf, and coconut oil, and Pedalium murex, curry leaf, and coconut oil.
Methodology
Materials
Based on the extensive analysis of literature and trials of plant-based herbs and their relative constituents, Pedalium murex, Milletia pinnata, curry leaves (Murraya koenigii), and pure, odorless coconut oil were obtained from Matunga, Mumbai. Polysorbate 80 (glycol), a non-ionic surfactant, was selected for this research and was obtained from Loba Chemicals, Mumbai, and locally sourced organic cotton fabrics, a 20 gsm (grams per square meter) plain-woven fabric and 60 gsm twill-woven fabric, were used for this research and were supplied by Test Fab India (Vapi, Gujarat).
Herbal oil extraction
Fresh leaves of Pedalium murex (Gokhru) and Milletia pinnata (Karanja) were washed thoroughly with distilled water and dried in an oven at 105°C for 1 h to remove any dirt and impurities. Dried herbs, namely 10 g of Pedalium murex, 5 g of curry leaves, and 100 ml of coconut oil, were heated together using steam supplied from a steam generator. The steam distillation process releases the aromatic components of the essential oils.
Solvent extraction
Pure ethanol (99%) was used for solvent extraction; dried herbs were placed in the thimble of a Soxhlet extractor, and ethanol was added. The solvent extract collected was filtered and, as the solvent was evaporated, oil was collected in a pot. The oil yield was calculated using the following formula: [amount of extracted oil (g)/the amount of dry herbs and oil (g)] × 100%. The above process was repeated for Milletia pinnata. Both extracted oils were stored in glass bottles for further analysis.
Preparation of nano-emulsion and characterization
Nano-emulsion was prepared using the herbal oil mixture, which was mixed with the surfactant in the ratios 1:1 (100 ml of distilled water, 1 ml of oil mixture, and 1 ml of polysorbate) and 1:2 and, using a high-speed homogenizer Tool-Tech that mixes at 1000–5000 rpm, the mixture was homogenized for 1 h until the nano-emulsion was stable. The particle sizes of nano-emulsion 1 (Milletia pinnata, curry leaves, and coconut oil) and nano-emulsion 2 (Pedalium murex, curry leaves, coconut oil) were assessed using a particle size analyzer (Shimadzu SALD-7500 nano, Kyoto, Japan). The pH of the nano-emulsions was also evaluated using a standard pH meter (EquipTronic) at 37°C. The thermal stability of the nano-emulsions was determined using a Metal-Lab MSI-17B (Metal-lab Scientific Industries, Mumbai, India) at varying temperatures, observing the oil separation from the constituents. The whiteness index of the finished fabrics was evaluated using a SpectraScan 5100+ computer color matching system (Rayscan, Australia), which measures the light reflected from the surface of the fabric at each wavelength to calculate the index.
Gokhru/Karanja extracts was subjected to gas chromatography-mass spectrometry (GC-MS) analysis with an Agilent system, 7890B GC and 5977A MSD detectors with the following parameters: fused silica capillary column 30 m × 0.320 μm × 0.25 μm thickness. Helium was used as a carrier gas with a 2 ml/min flow rate, average velocity of 52.54 cm cm/s, hold-up time of 0.970 min, and a split ratio of 1:10. The injector temperature was 250°C. The oven temperature was programmed at 160°C with an increase of 5°C/min until 270°C and then 15°C/min to 320°C and a hold time of 5 min at the final temperature of 320°C. Mass spectra were obtained in the range width of m/z 35–600, interface temperature of 290°C, solvent cut time of 3 min, and normal scan speed. In addition, the mass spectra of the separated components were compared with the NIST database (NIST, Version 11), and ChemStation GC/MSD software was used.
Finishing of fabrics with nano-emulsions
Both fabrics were finished with nano-emulsions using the exhaust method (batch process) that uses a Rota dryer machine (Figure 1). The fabrics pass through a winch and are immersed in a bath containing the nano-emulsions. The fabric samples were maintained in exhaustion in a material-to-liquor ratio of 1:50 at 60°C for 1 h. During the exhaust process, the material-to-liquor ratio was trialed at varying temperatures, 60–80°C for 1 h, and it was observed that a more uniform application of nano-emulsion was observed at 60°C for 1 h. However, any increase in the temperature caused the nano-emulsions to break down, and the finishing of the fabric was not uniform. This is to allow the penetration of nano-emulsion into the fabric structure. The finished fabric passes through the rollers to squeeze out any excess nano-emulsions, and is washed at room temperature.
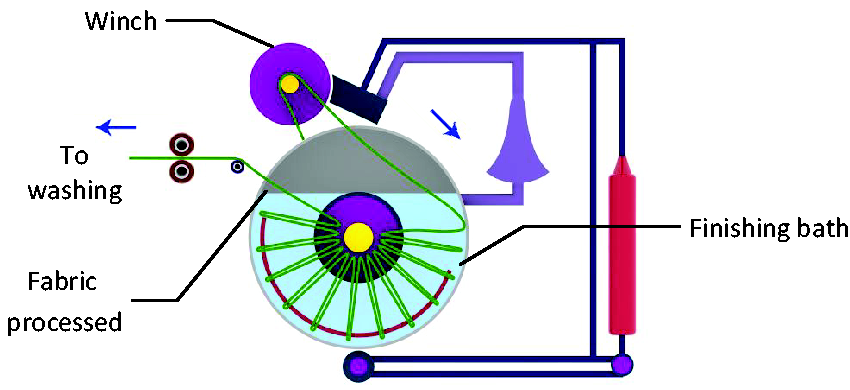
Graphical illustration of the exhaust method.
Fabric characterization
The surface morphologies of the organic cotton fabrics (20 and 60 gsm) were analyzed using scanning electron microscopy (SEM; Carl Zeiss Supra 40 VP, Oberkochen, Germany); a variable pressure of 30 Pa was used. In addition, energy-dispersive X-ray spectroscopic analysis (EDX) was carried out using an Apollo 40 SDD (Tilburg, Netherlands). EDX enables the identification of the elemental composition of fabric finished with nano-emulsions.
Antibacterial and antifungal resistance tests
Antibacterial tests were evaluated using the quantitative method
59
(AATCC 100-2019) and the qualitative method (AATCC 147:2016)
60
with Staphylococcus aureus strain no. ATCC 6538 (gram-positive bacteria), Escherichia coli strain no. ATCC 10799 (gram-negative bacteria), Staphylococcus epidermidis strain no. ATCC 12228 (gram-positive bacteria), and Klebsiella pneumoniae strain no. ATCC 4352 (gram-negative bacteria). For quantitative tests, 1.0 ml of the test inoculum (S. aureus, E. coli, S. epidermidis, K. pneumoniae) was loaded on fabric swatches of 4.8 ± 0.1 cm diameter. The strains were cultured in nutrient agar and sterilized using an autoclave. After the test samples were allowed to contact for 24 h, the percentage reduction of bacteria was determined using the following formula:
Fabric performance tests
The finished fabrics were evaluated for their durability to determine their performance post finishing with both nano-emulsions. Tensile strength was determined in the warp and weft directions of woven fabrics, and its breaking extension (%) and elongation (mm) were reported (ASTM D 5035-11 2019). 63 The moisture vapor transmission rates (MVTRs) of the finished fabrics were analyzed at a temperature of 32°C, relative humidity of 50%, and air velocity of 0.02–0.3 m/s (ASTM 96/96 M: 2005). 64
Results and discussion
Particle size, pH, thermal stability, percentage add-on, and whiteness index
As highlighted in the Preparation of nano-emulsion and characterization section, nano-emulsions 1 and 2 were prepared following several trials, and in this section, the findings of nano-emulsions 1 and 2 in terms of particle size analysis, pH, thermal stability on nano-emulsions, percentage add-on to finished organic fabrics, and whiteness index of the finished fabrics are reported.
The particle size of nano-emulsions
It can be noted that for both nano-emulsions, the particle size decreased from ‘immediately after preparation' to weeks one and two, and this reduction was in the range of 66–70% of the initial particle size in week two. It can be noted for the 1:1 ratio, the particle size for nano-emulsion 1 was in the 106–36 nm region, and for nano-emulsion 2, it was marginally similar in the range of 115–33 nm (Figure 2). The above trend was similar for ratio 1:2 for both nano-emulsions. However, there was a marginal decrease in particle size when the oil-to-surfactant ratio increased from 1:1 to 1:2. Like in previous research, 65 active antioxidants in herbs reduce the particle size. The addition of the surfactant also reduces the particle size. The surfactant plays a crucial role in the formation of a stable nano-emulsion. It helps in reducing the interfacial surface tension between oil and water by absorbing the liquid–liquid interface (it is the junction between two immiscible liquids). The surfactant has a hydrophilic head and hydrophobic tail. During nano-emulsion formation, the increase in the surfactant results in the decrease of surface tension between oil and water, and it accumulates around the droplets and prevents droplet aggregation throughout the preparation process and storage. Therefore, as the level of surfactant increases, it reduces the surface tension between oil and water and helps form a stable nano-emulsion with the reduction in the particle size.
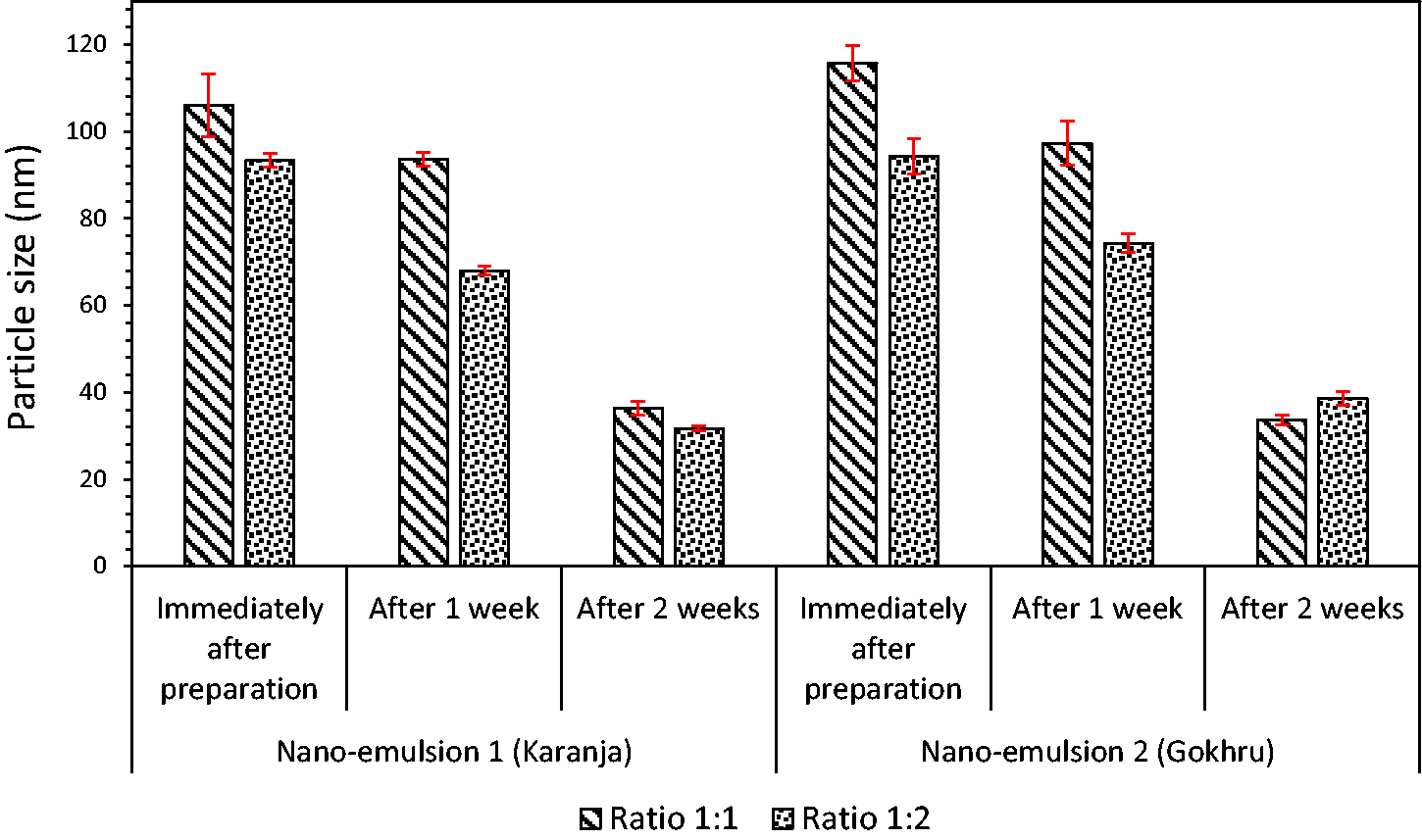
Particle size analysis.
As the particle size decreases, the ease of penetration into the fabric structure is much better and offers stability and prevents the aerial oxidation of oils that results in malodor and taste.
pH optimization
The optimization of the pH of both nano-emulsions was carried out for both ratios by adding 0.1% hydrochloric acid and 0.1% sodium hydroxide. The pH for nano-emulsion 1 was 6.07 for both 1:1 and 1:2 ratios, and for nano-emulsion 2, the pH was in the range of 6.2–6.3 for ratios 1:1 and 1:2. Overall, it can be noted that the pH of the nano-emulsions was in the range of 6–6.3, which indicates they are acidic and have a similar pH to that of groundwater.
Thermal stability
The thermal stabilities of nano-emulsions 1 and 2 were studied by placing the beakers containing nano-emulsions 1 and 2 separately in a water bath at a temperature of 95°C. The thermal stabilities of nano-emulsion 1 for ratios 1:1 and 1:2 were 56°C and 57.6°C, respectively, while for nano-emulsion 2, they were 57°C and 58.3°C, respectively. Beyond this temperature range, the nano-emulsions become turbid and break down, and cannot be used again. It should be noted that the nano-emulsions are thermodynamically unstable, and the two immiscible liquids (oil and water) used in this study will break at higher temperatures. In the exhaust process, a uniform application of nano-emulsion was observed at 60°C for 1 h. However, the thermal stability experiments revealed that nano-emulsions 1 and 2 were stable in the region of 56–58.3°C. In the thermal stability experiment, the nano-emulsions were heated directly in a glass beaker, causing them to break down when the temperature was above 58°C; however, in the exhaustion process, the heat was applied with the fabric/nano-emulsion together, and it retained its stability up to 60°C, but any increase in temperature caused the nano-emulsion to break down.
Percentage add-on
In the case of nano-emulsion 1, the percentage add-on was marginally higher for 20 gsm fabric than 60 gsm fabric; this could be related to the fabric structure of the fabrics, the closeness of the weave pattern (fabric count), and the cover factor of the fabric (the area covered by a set of threads) (Figure 3). The SEM images in Figure 4(b) show that yarns are tightly spaced, as indicated in the fabric counts of 60 gsm (117 ends per inch × 38 picks per inch) and 20 gsm with a more open structure (75 ends per inch × 27 picks per inch) (Figure 4(a)). The cover factor (K) for 20 gsm was low 21 (warp) + 64 (weft), whilst for 60 gsm, it was marginally high at 63 + 21. The interaction between the fabric surface and nano-emulsions is simple mechanical adsorption without crosslinking. This occurs between the interspaces between fibers/yarns in the fabric structure. This study used two different types of weaves with varying fabric weights to demonstrate percentage add-on for lightweight fabrics. The above trend was similar for nano-emulsion 2. However, when the oil-to-surfactant ratio was increased to 1:2, the percentage add-on was higher for both nano-emulsions 1 and 2. As the surfactant ratio increases, the particle size reduces, which enables the nano-emulsion to disperse within the fabric more easily and uniformly. Overall, it can be inferred that the 20 gsm fabric with the 1:2 ratio had a higher percentage add-on of nano-emulsion than the 60 gsm fabric.
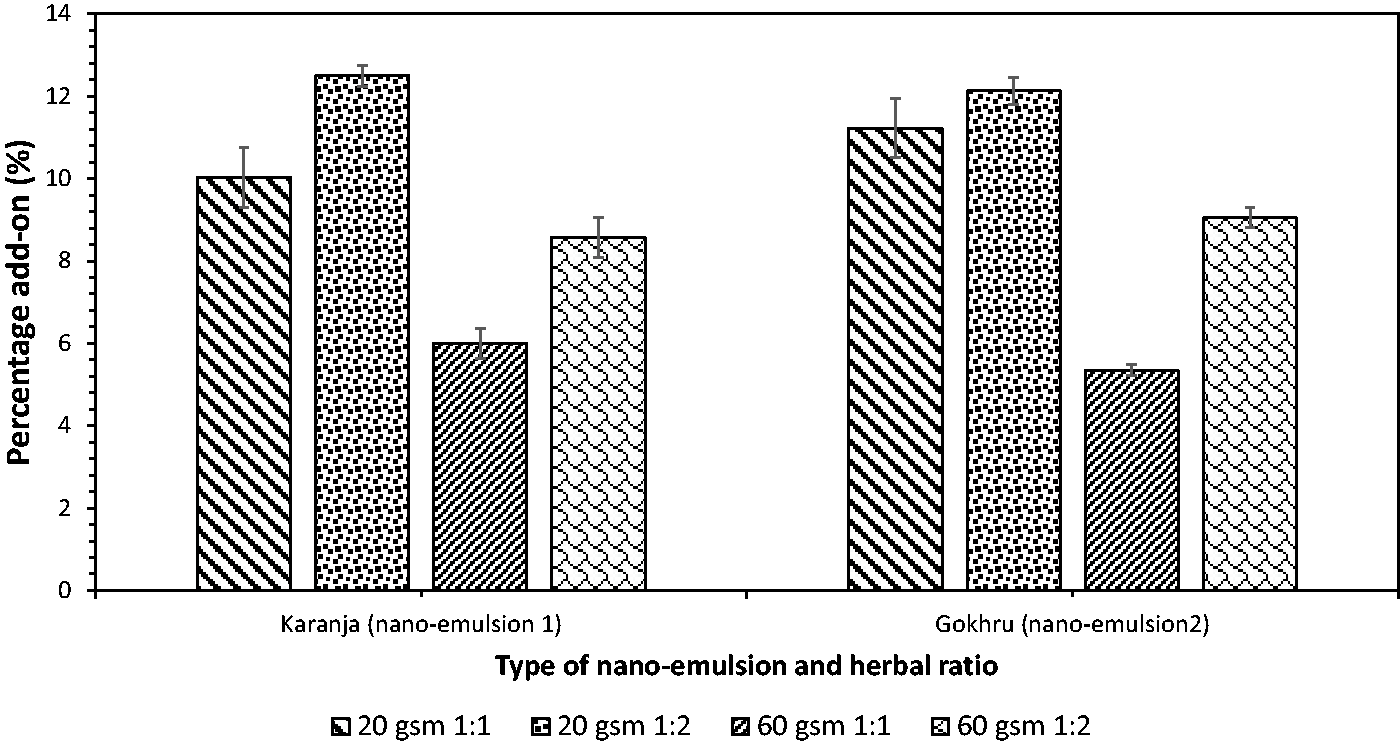
Percentage add-on of nano-emulsions on organic fabrics.

Scanning electron microscopy images of organic fabrics: (a) 20 gsm plain weave and (b) 60 gsm twill weave before finishing.
Whiteness index
The whiteness index measurements of finished fabrics with nano-emulsions 1 and 2 revealed marginal variations from unfinished fabrics. The percentage change in the whiteness index for the 20 gsm fabric finished with nano-emulsion 1 being marginally higher for the 1:2 ratio compared to the 1:1 ratio. In the case of the 60 gsm fabric, the percentage change in the whiteness index was relatively lower than that of the 20 gsm fabric, with a slightly higher change in the index for the 1:2 ratio than for the 1:1 ratio. As seen earlier in the percentage add-on, the penetration of nano-emulsion was more pronounced for 20 gsm fabric than for 60 gsm fabric due to its open structure and low cover factor, especially for the 1:2 ratio. There was a similar trend for nano-emulsion 2. Overall, it can be noted that the change in whiteness index was well below 10% for both nano-emulsions 1 and 2 and is not noticeable to the naked eye, which is indicated by the dotted blue line in Figure 5.
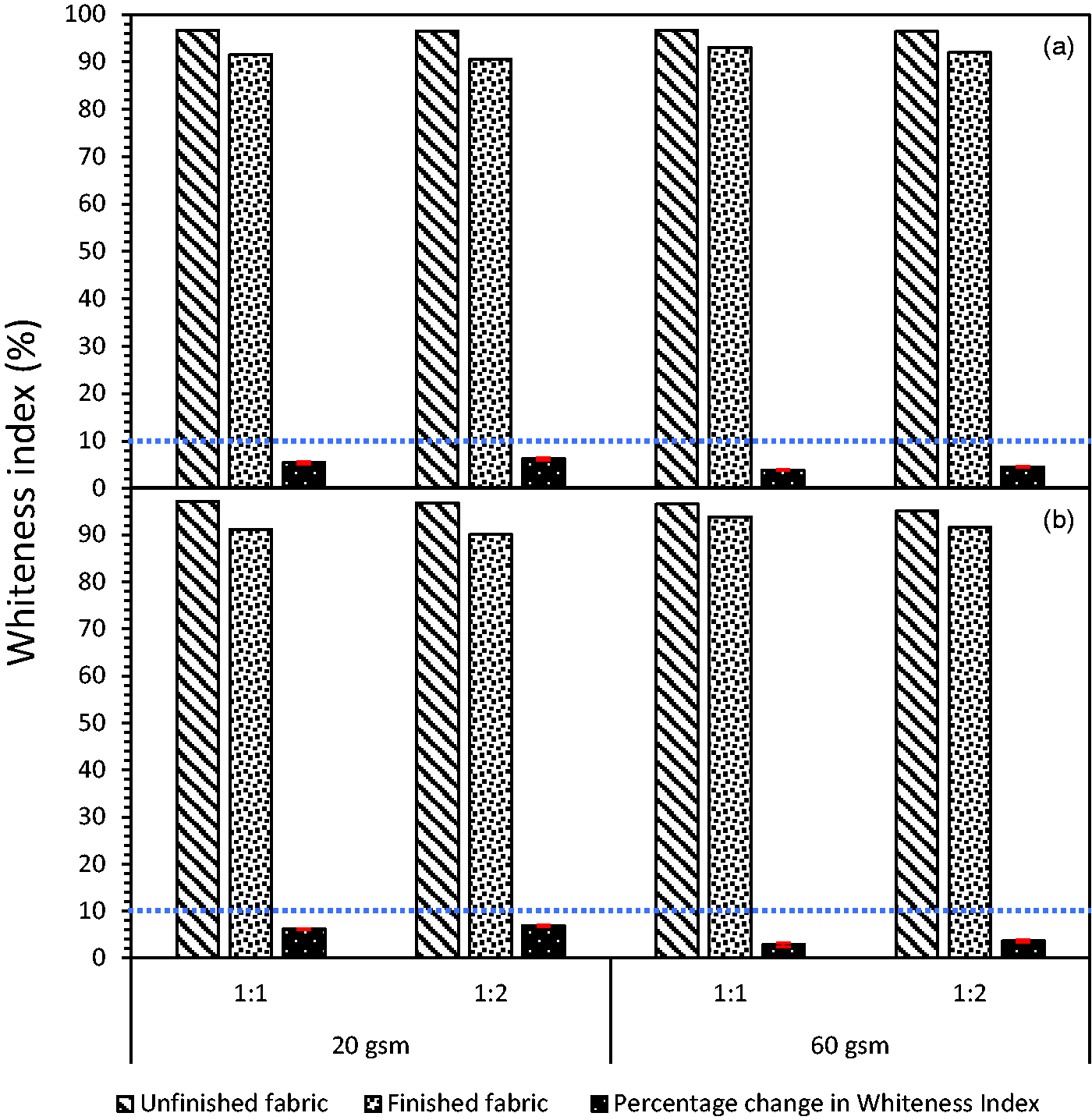
Whiteness index: (a) nano-emulsion 1 (Karanja); (b) nano-emulsion 2 (Gokhru) for 20 and 60 gsm fabric; the blue dotted line indicates the 10% index that is not noticeable to the naked eye (color online only).
Antibacterial tests
The quantitative antibacterial tests (AATCC 100) revealed that both nano-emulsions 1 and 2 showed excellent resistance (the percentage reduction in microorganisms is approximately 99%) to gram-positive and gram-negative bacteria. In the case of the 20 gsm fabric finished with nano-emulsion 1, there is a marginal difference in the percentage reduction of microorganisms for ratios 1:1 and 1:2 for unwashed samples (Table 1). These were in the range of 99.30–99.85%. For the 60 gsm fabric, the percentage reduction was in the range of 99.29–99.89%. In the case of nano-emulsion 2, the percentage reduction of microorganisms was marginally higher than for nano-emulsion 1 in the range of 99.73–99.84% for both 20 and 60 gsm fabrics.
Reduction of microorganisms (AATCC 100) – exhaust method*
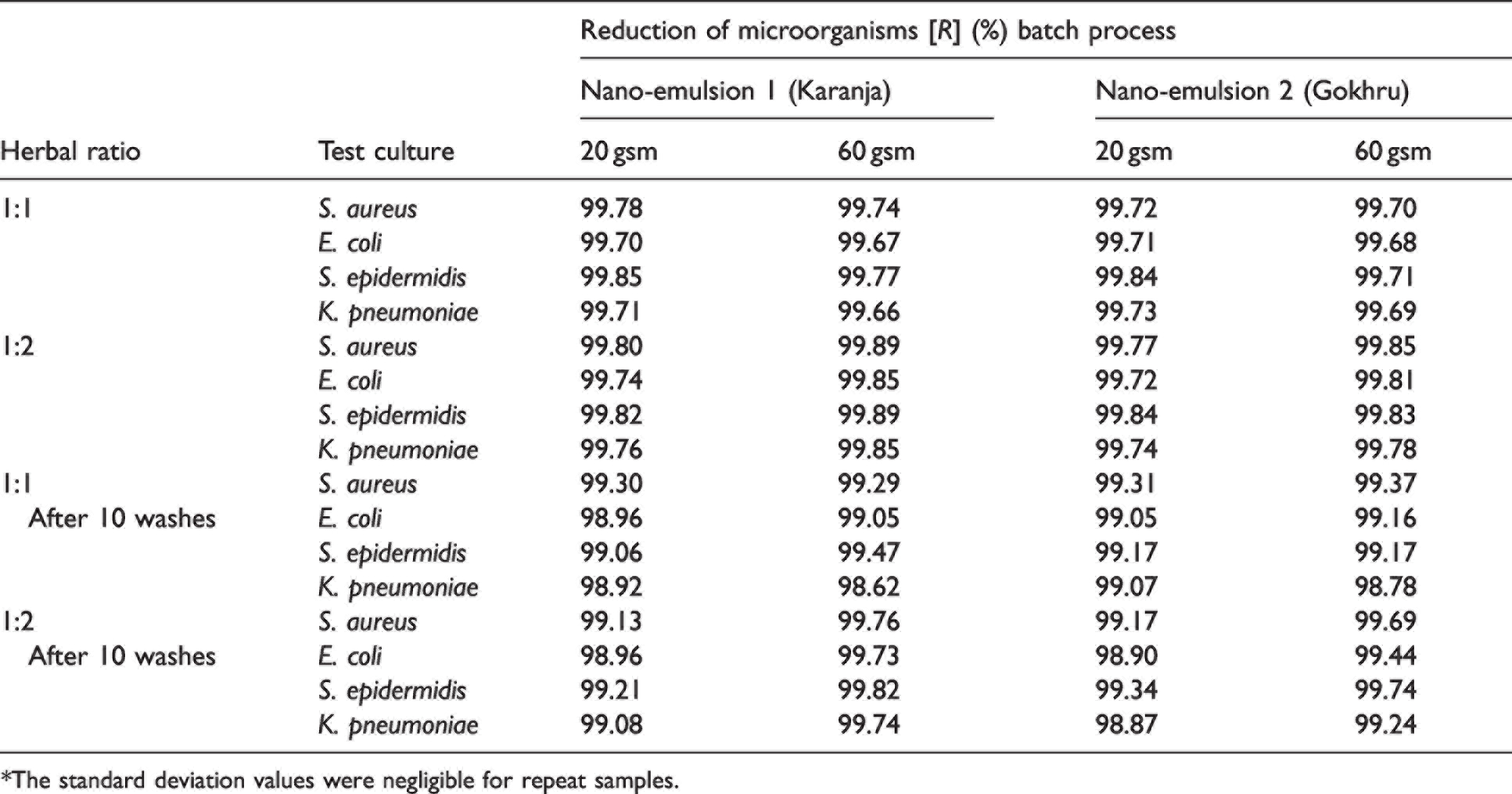
*The standard deviation values were negligible for repeat samples.
For the 20 gsm fabric, the decrease in the percentage reduction of microorganisms for the 1:1 ratio after 10 washes was in the range of 0.5–0.79%, which is marginal, demonstrating good efficacy in resisting the growth of microorganisms (gram-positive and gram-positive bacteria). In the case of the 1:2 ratio, the drop or reduction in the percentage of microorganisms after 10 washes was in the range of 0.61–0.78%, which also showed the antimicrobial efficacy after washing. For the 60 gsm fabric, the decrease in the percentage reduction of microorganisms for the 1:1 ratio decrease was in the range of 0.30–0.45% for gram-positive bacteria, whilst for gram-negative bacteria, the decrease was in the range of 0.62–1.04%. A marginal decrease in the percentage reduction of microorganisms (1.04%), especially for gram-negative bacteria, is noted after 10 washes. Generally, the biocidal effect drops slightly. After 10 washes, the antimicrobial agent on the fabric surface washed off from the fabric surface, causing a decrease in antimicrobial reduction. In the case of the 1:2 ratio, for gram-positive bacteria, the percentage of reduction was in the range of 0.13–0.42%, and for gram-negative bacteria was in the range of 0.12–0.11%. In the case of nano-emulsion 2, for the 20 gsm fabric, for the 1:1 ratio the decrease in the percentage reduction of microorganisms after 10 washes for gram-positive bacteria was in the range of 0.41–0.67%, and for gram-negative bacteria it was 0.66%. For the 1:2 ratio, the decrease for gram-positive bacteria was 0.55–0.82%, and 0.5–0.87% for gram-negative bacteria. For the 60 gsm fabric, with the 1:1 ratio, the decrease in percentage after 10 washes was 0.33–0.54% for gram-positive bacteria and 0.52–0.91% for gram-negative bacteria; however, in the case of the 1:2 ratio, the decrease was much lower. Overall, it can be inferred that a percentage reduction after 10 washes of less than 1% indicates good wash durability for 20 and 60 gsm fabrics. The Appendix illustrates the antimicrobial efficacy of nano-emulsions 1 and 2, before and after 10 washes. In the case of the parallel streak method, there was no growth of bacteria below the specimen for nano-emulsions 1 and 2, particularly for S. aureus and E. coli.
Antifungal tests
The antifungal tests were conducted using Aspergillus niger, and as a representative sample, the 1:1 ratio 60 gsm fabric was used for both the nano-emulsions. There was a 4.3 mm zone of inhibition for nano-emulsion 1 and no fungal growth. However, in the case of nano-emulsion 2, there was no zone of inhibition, and minor traces of fungal growth (rating 1) were noticed.
SEM and EDX
SEM of the finished fabrics with nano-emulsion 1 (Figure 6) reveals that at 1000× magnification, the round ribbon-shaped cotton fibers were stretched, flattened, and observed to have striations. This can be noticed especially when comparing the Figure 6(a) with Figures 6(c) and (e), and similarly when comparing Figure 6(g) with Figures 6(i) and (k) for the 60 gsm fabric. This visible flattening is more visible for 20 gsm than 60 gsm fabrics, especially for the 1:2 ratio. This stretching of finished fabrics is due to the mechanical adsorption of the fabrics with the nano-emulsions and passing between the nip rollers. The above morphological changes were observed on the fabric/yarn structure for 20 and 60 gsm fabrics finished with nano-emulsion 2 (Figure 7).

Scanning electron microscopy for 20 and 60 gsm fabrics finished with nano-emulsion 1.

Scanning electron microscopy for 20 and 60 gsm fabrics finished with nano-emulsion 2.
Figure 8(a) illustrates the SEM image where a portion of the fabric is examined using EDX to determine the elemental composition of fabric finished with nano-emulsion 1, 1:1 ratio, and for 20 gsm fabric. Three random portions were identified for analysis for each fabric finished with nano-emulsion. Some minor traces of sodium, aluminum, silica, and calcium were observed. This observation was similar for nano-emulsion 2 for the 20 gsm fabric, Figure 8(b), and for the 60 gsm fabric for both nano-emulsions 1 and 2 (Figures 8(c) and (d)). There were also some minor traces of magnesium in the fabric finished with nano-emulsion 2. Generally, most green leaves (spinach) have good magnesium content and are widely consumed. It is worth mentioning that minor traces of silicon and aluminum were also noted. This could have occurred during the finishing process, as the fabric could have picked up when passing between the silicon rubber nip rollers and wet pick aluminum rollers. Based on the analysis, it can be confirmed that there were no harmful elements in the finished fabrics, and they are safe for use.
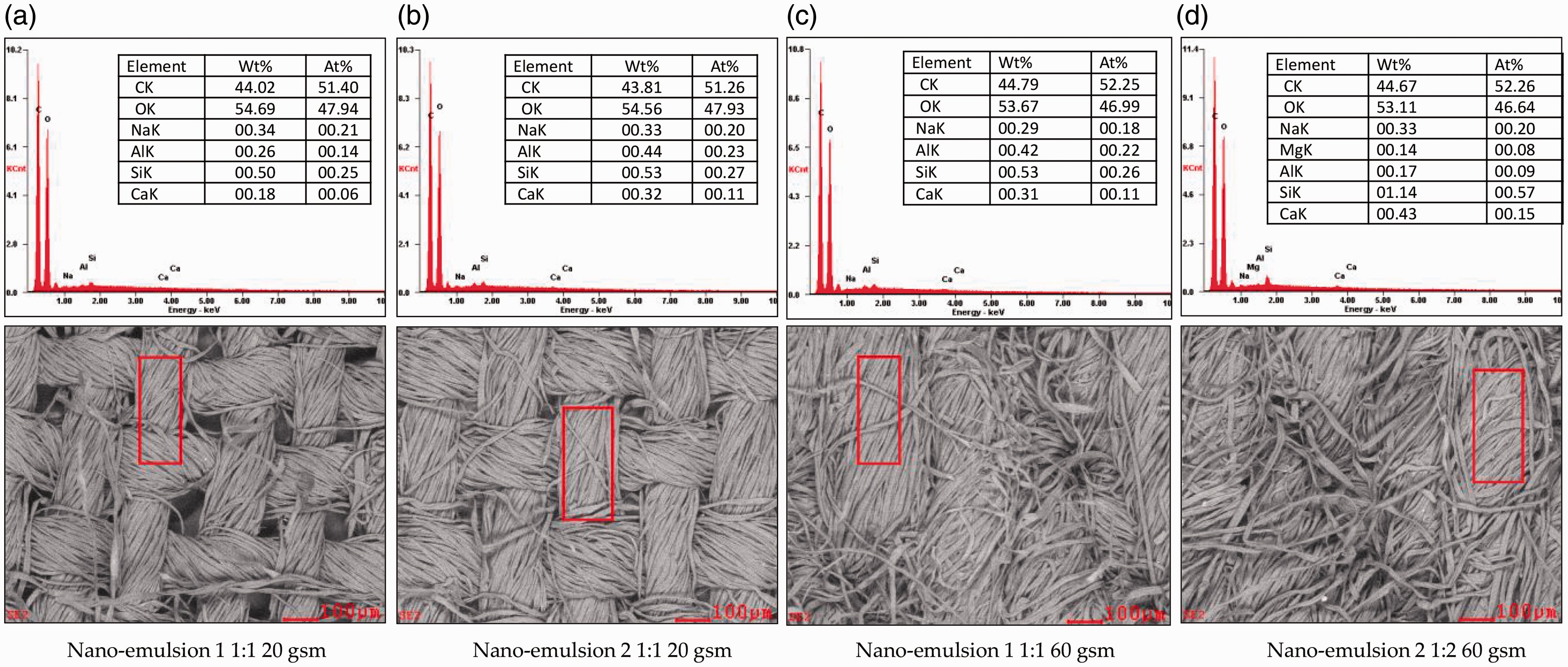
Energy-dispersive X-ray spectroscopic analysis for 20/60 gsm fabrics finished with nano-emulsions 1 and 2.
Tensile strength
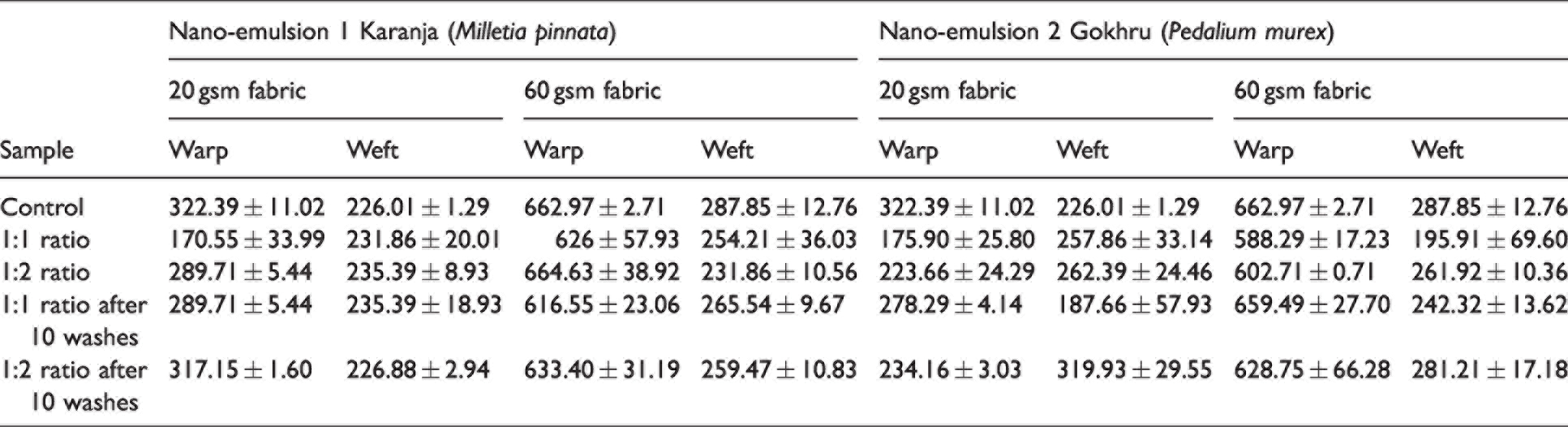
In the case of nano-emulsion 1, the tensile strength of the 20 gsm fabrics in the warp direction was reduced for the 1:1 ratio significantly, and for the 1:2 ratio, there was a moderate drop in the tensile strength compared to the unfinished fabric (Table 2). In the case of the 60 gsm fabric (warp direction), there was a gradual reduction in strength for both ratios 1:1 and 1:2. For the 20 gsm fabric in the weft direction, there was a marginal increase in the tensile strength. However, for the 60 gsm fabric, there was a drop in the strength for both ratios. In the case of nano-emulsion 2 and for both 20 and 60 gsm fabrics, the tensile strength was reduced for both ratios for the warp direction. Compared to unfinished fabrics, the tensile strength was reduced post-washing (10 times) and for both ratios in warp and weft directions. Similar to nano-emulsion 1, for the 20 gsm fabric in the weft direction, there was a marginal increase in the tensile strength. It should be noted that the finishing of fabrics with nano-emulsions was due to the simple mechanical adsorption of emulsion on the fabric structure, whereby the nano-emulsions remain between the voids of the fibers in the fabric. Moreover, the pH of the emulsions was slightly acidic (6.0–6.3), and as cotton fibers are sensitive to acids, a reduction in tensile strength is anticipated.
Tensile strength (N)
Moisture vapor transmission rate analysis
The MVTR is defined as the ability of the fabric to allow moisture to penetrate through the fabric over a particular period (ASTM 96/96 M: 2005). The MVTR of finished and unfinished fabrics were evaluated to determine if the finishing of fabrics with nano-emulsions were affected. For both nano-emulsions 1 and 2, 20 and 60 gsm fabrics, for both ratios 1:1 and 1:2, the MVTR was reduced compared to unfinished fabrics (Table 3). This reduction in MVTR also reveals that nano-emulsions were adsorbed by the cotton fabrics, affecting the interspaces between the fibers within the yarn/fabric structure and reducing water vapor transmission through the fabric.
Moisture vapor transmission rate (g/m2/24 h)

GC-MS analysis
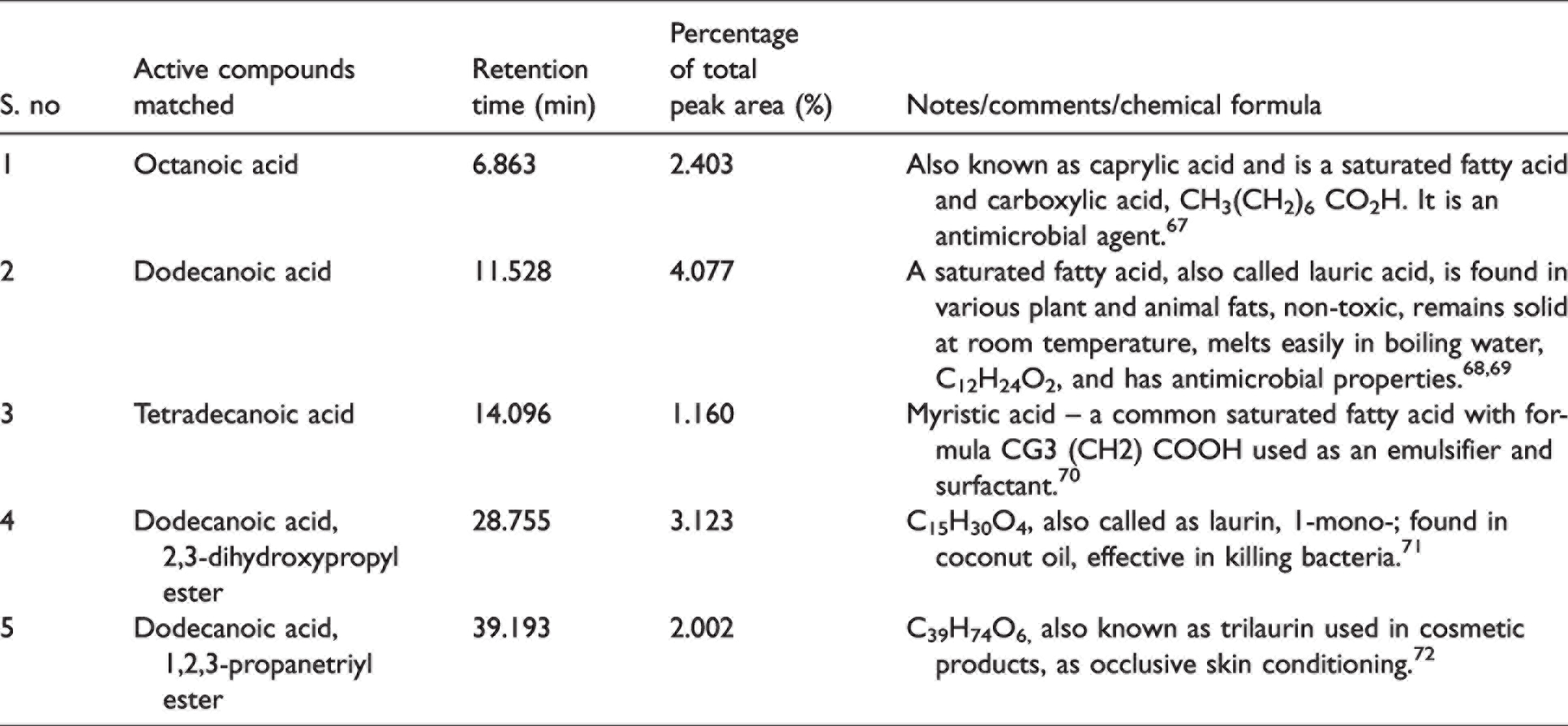
GC-MS analysis of Karanja extract revealed (Table 4) the presence of saturated fatty acids – caprylic acid, lauric acid, myristic acid, and 1-mono-laurin. These acids possess antimicrobial properties, which could inhibit the growth of bacteria. In addition, the presence of trilaurin was observed, which is widely used in skin conditioning and cosmetic products, and it reveals that the finishing of nano-emulsion 1 is safe on the skin with its antimicrobial properties. Figure 9 illustrates the GC-MS with peaks (constituents matched against the NIST database), retention time (minutes), and abundance of the constituent in percentage for Karanja extract. Some of the peaks shown in Figure 9 highlight that active compounds matched, and these were octanoic acid (6.91%); dodecanoic acid (11.53%); carbamic acid (28.70%); dodecanoic acid (31.26%); carbamic acid and dodecanoic acid (33.78%). Carbamic acid can be used as a herbicide and insecticide. When used correctly, it can help society prevent insect vector diseases. 66
Gas chromatography-mass spectrometry analysis of Karanja (Milletia pinnata)
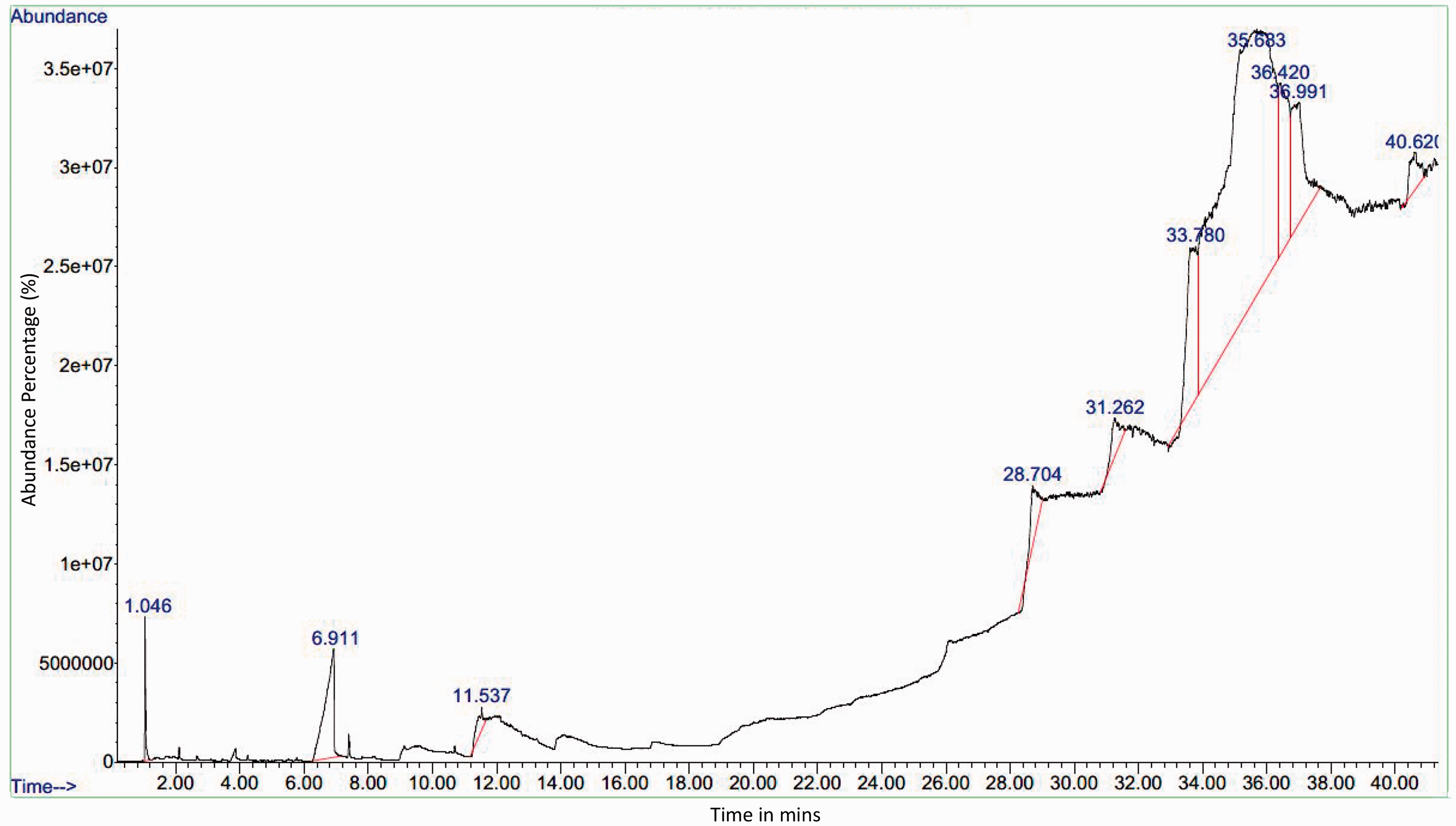
Gas chromatography-mass spectrometry of the ethanolic extract of Karanja leaves.
GC-MS analysis of Gokhru extract (Table 5) revealed saturated fatty acids dodecanoic acid, tridecanoic acid, octanoic acid (caprylic acid), and undecylic acid, which serve as fungicides that can inhibit the growth of fungus. Most of the saturated fatty acids have bactericidal properties. In addition, compounds such as 2-decenal (a nematicide – can be used as pesticide) and 2-undecanone (insect repellent), possess insect repelling properties. Figure 10 illustrates the GC-MS with peaks (constituents matched against the NIST database) and retention times for Gokhru extract. Some of the peaks shown in Figure 10 highlight that active compounds matched, and these were octanoic aid (7.73%); dodecanoic acid (13.11%, 33.3% and 35.62%); and tetradecanoic acid (14.8%). These are fatty acids and dodecanoic acid was observed in higher abundance, which has antimicrobial properties.
Gas chromatography-mass spectrometry analysis of Gokhru (Pedalium murex)
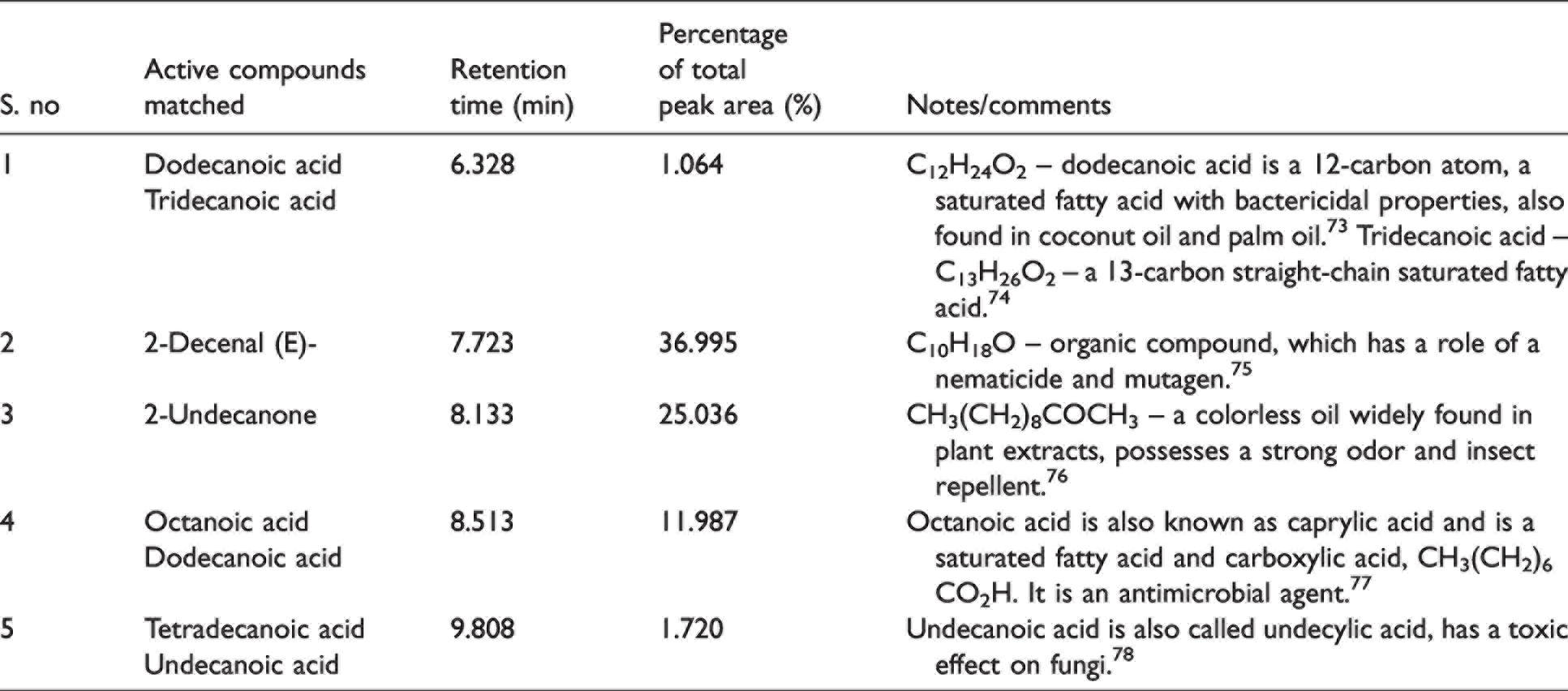
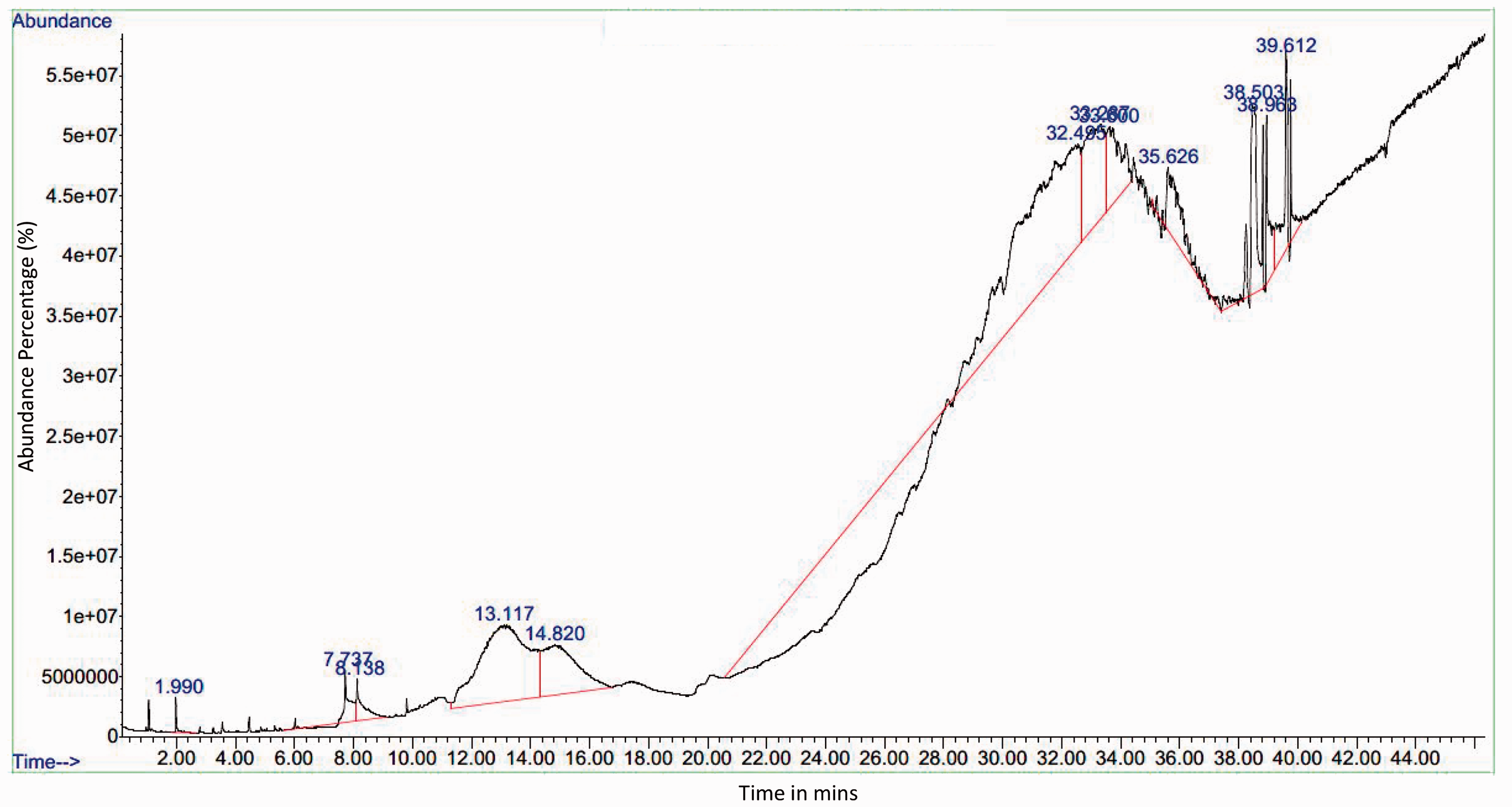
Gas chromatography-mass spectrometry of the ethanolic extract of Gokhru leaves.
Conclusions
Finishing cotton textiles for imparting antimicrobial properties for hygiene applications is one of the top priorities for many textile industries and technical textile professionals. During the current pandemic, public awareness of improving personal hygiene and sanitation was at a peak level. In addition, the increased consumer awareness of the environmental impact of disposable products on the community and the need for durable finishes with environmentally friendly finishing are also of utmost importance. 79 Antimicrobial finishing with synthetic chemicals could affect health 17 and the environment, 16 and there is a pressing rationale to identify plant-based antimicrobial compounds that can resist a broad spectrum of bacteria and fungi. Plant-based extracts possess bioactive compounds with antibacterial, antifungal, antioxidant, and antiviral properties. In this research, a combination of herbal extracts was identified based on a comprehensive literature survey. Two different nano-emulsions were developed and characterized for particle size, pH, thermal stability, percentage add-on, and whiteness index. Nano-emulsion 1 (Milletia pinnata, curry leaves, and coconut oil) and nano-emulsion 2 (Pedalium murex, curry leaves and coconut oil) were mixed with a surfactant to develop two ratios, 1:1 and 1:2. GOTS certified organic cotton fabrics were finished using an exhaust method (batch process).
Results indicated that the particle size decreased from immediately after preparation to weeks 1 and 2. The particle size decreased, enabling it to penetrate the fabric structure easily. The overall change in the whiteness index for both nano-emulsions 1 and 2 and both fabrics was lower than 10%, which is not noticeable to the naked eye. Antibacterial tests for both nano-emulsions 1 and 2 showed excellent resistance to a range of gram-positive and gram-negative bacteria. The percentage reduction of bacteria was 99.30–99.89% for both nano-emulsions 1 and 2 and for both fabrics. The antibacterial efficacy was not affected after 10 washes, showing a <1% reduction in the percentage reduction of microorganisms for both nano-emulsions. This also indicated that the finishing of organic cotton fabrics with herbal nano-emulsions was durable. The 60 gsm finished fabrics with nano-emulsion 1 (1:1 ratio) showed resistance to fungal growth with a clear zone of inhibition. The tensile strength of the finished fabrics (both 20 and 60 gsm fabrics) marginally dropped compared to that of unfinished fabrics, and this drop in the strength values is anticipated with any finishing. SEM analysis revealed minor stretching and flattening of cotton fibers in the case of finished fabrics with no major surface morphological changes. EDX of finished fabrics also indicated there were minor traces of sodium and calcium. The MVTR for both the fabrics and nano-emulsions was reduced after finishing, indicating that interspaces between the fibers in the fabric structure were adsorbed by nano-emulsions.
GC-MS analysis of Karanja and Gokhru extract revealed the presence of saturated fatty acids and bioactive compounds, which have bactericidal and fungicidal properties, highlighting why these finishes on cotton fabrics are antimicrobial. This study adds to the extant literature that demonstrates the potential of the novel combination of herbal nano-emulsions that have durable bacterial resistance to a range of gram-positive and gram-negative bacteria. AMR is a major challenge globally, and if plant-based antimicrobial finishes can inhibit the growth of gram-positive bacteria (S aureus and S epidermidis), gram-negative bacteria (E coli and K pneumoniae) and fungi (A niger), which are responsible for the spread of infections through textiles in the community, then this research demonstrates a significant potential of the findings in preventing the transmission of infections in an eco-friendly manner. Furthermore, these finishes using plant-based antimicrobial compounds are easy to finish or replenish, safe next to the skin, and have a negligible impact on the environment compared with synthetic chemicals. Further work on different combinations of herbal constituents and ratios to enhance the antibacterial efficacy of various other blended textiles could open new opportunities for durable and environmentally friendly antimicrobial textiles.
Footnotes
Acknowledgments
The authors would like to acknowledge the kind contribution from the following technical/support colleagues: Ms Hayley Andrews, MMU, UK, for providing SEM and EDX images and analysis; all staff of the Department of Fibres and Textile Processing Technology, ICT, Mumbai.; Dr Mrinal Choudhary (Director), WRA (Wool Research organisation), and Dr Madhura Nerurkar, Calantha Biotech, Mumbai India; Dr Ketan Merchant SAS Labs, Mumbai, India, and Mr V Dhanapal, India, for providing support toward development of three-dimensional images.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by QR funding from the Global Challenges Research Fund (GCRF) from the Manchester School of Art Research Centre [MSARC], Faculty of Arts and Humanities (Project ID 328682), Manchester Metropolitan University, UK.