Abstract
Polypropylene as one of the world's top commodity polymers is also widely used in the textile industry. However, its non-polar nature and partially crystalline structure significantly complicate the process of industrial coloring of polypropylene. Currently, textiles made of polypropylene or with a significant proportion of polypropylene are dyed under quite harsh conditions, including the use of high pressures and temperatures, which makes this process energy intensive. This research presents a three-step synthesis of coloring agents, capable of adhering onto synthetic polypropylene yarns without harsh energy-consuming conditions. This is possible by encapsulation of organic pigments using trimethoxyphenylsilane, introduction of surface double bonds via modification of the silica shell with trimethoxysilylpropylmethacrylate and final attachment of highly adhesive anchor peptides using thiol-ene chemistry. We demonstrate the applicability of this approach by dyeing polypropylene yarns in a simple process under ambient conditions after giving a step-by-step guide for the synthesis of these new dyeing agents. Finally, the successful dyeing of the yarns is visualized, and its practicability is discussed.
Polypropylene is an industrial multipurpose polymer used in a wide variety of technical applications. Its most important advantages are low cost, light weight and excellent chemical stability, being the reason why its main use is found in the packaging industry.1–3 However, it suffers greatly from the lack of dyeability with commonly used methods because of its non-polar nature and partially crystalline structure. As dyeability is a very important property for the usage in textile applications, a considerable amount of research has been conducted in order to find possible solutions to this problem.4,5 The blending of polypropylene with different homopolymers has been investigated, searching for an improvement of dye adsorption, by reducing the amount of crystalline areas. Polymers used included polystyrene, polyester, 6 polyamide, 7 poly(ethylene-co-vinyl acetate), 8 and poly(vinyl alcohol) 9 and were also extended to copolymers. 10 The same goal was achieved using additives such as clay derivates.11–13 Further concepts included chemical treatment using sodium hypochlorite, which led to the chlorination of the polypropylene fabric and therefore increasing the wetting of fibers with dye solutions, 14 and dyeing using supercritical carbon dioxide as a solvent. 15
In this research we will present a new dyeing approach, which does not need a pre-treatment of the polypropylene fabric. In contrast to common dyeing techniques for textiles using high pressure and/or temperature, the proposed method can be conducted at ambient temperature and pressure leading to a considerable decrease of production costs. 16 This new method is a result of combining basic chemistry concepts and newly attained biotechnological achievements, namely the discovery and application of anchor peptides. Originally, anchor peptides were intensively investigated as antimicrobial coatings, 17 but since then their application as surface modification agents for different materials and objects, including polymersomes 18 and polypropylene, 19 has shown their potential also for the textile industry. 20 The antimicrobial effect of anchor peptides is based on interactions with cell membranes and was analyzed by Brogden et al. who postulated three different mechanisms. 17 Depending on the type of cell membrane, peptides are able to adsorb and to penetrate, leading to the creation of channels, to dissolution into fragments or to the complete destruction of the cell. The ability to adsorb and to penetrate the lipid bilayer in the cell membrane is based on the combination of certain amino acids in the peptide sequence. The peptide can adjust its structure to a surface to be adhered by varying its folding, creating hydrophilic, hydrophobic and even charged areas for an attractive interaction. 21 The anchoring effect of the peptide named LCI was used in this research to stain polypropylene fibers.
To combine dyeing capabilities with the binding effect of anchor peptides, organic pigments in powdered form were chosen as the coloring agent. In general, these colloids are characterized by their good chemical resistance and high opacity and are non-soluble in common solvents. 22 In particular in this research, the C.I. Pigment Violet 19 was used, which belongs to the group of quinacridones. It possesses a high thermal stability and is non-soluble in almost all solvents due to its strong hydrogen bonding and π–π stacking between the aromatic ring structures. Its strong red color is a result of the crystal lattice interaction inside the pigment, which achieves a maximum yield for individual pigment particles in an optimal size range. Therefore, it is very important to keep the size distribution of these colloids during their further modification, and no changes in the chemical structure of these organic pigments need to be done.23,24 For that reason, an encapsulation and shell growth approach using silanes was utilized to introduce the chemical coupling system required for the binding of the anchor peptides to the pigments surface.25,26 This encapsulation approach with silanes was already investigated using different dyes embedded in colloids27–30 and also conducted with pigments.31,32 In contrast to these examples, the presented method aimed at decreasing the utilized chemicals and purifications steps. For this purpose, the surface-active trimethoxyphenylsilane (TMPS) was used, which can adhere in an aqueous dispersion of Violet 19 to the pigment surface, due to its low solubility in water and π–π interaction between the aromatic rings. Afterwards, the encapsulation of individual pigment particles in silica–silane shells is initialized by the addition of ammonia as a catalyst. A further advantage of this approach is the flexibility concerning the organic pigments used. An exchange of the dye used is straightforward and will not need additional adaptations of the concept. From this point, the silica shell modification using another functional silane is possible.
To couple the anchor peptide onto the pigment shell, a suitable coupling system had to be found. It was possible to include a single thiol functionality into the peptide structure, by adding a cysteine.18,19 A very important reaction for thiol groups, which is also inert to oxygen, is the thiol-ene click reaction.33–36 It is a fast, irreversible reaction and no harsh conditions are needed, which could interfere with the peptide stability. Another important factor is the feasibility of this reaction in water. 37 The reaction can be conducted following three different mechanisms: 37 nucleophilic addition using a basic catalyst, electrophilic addition using an acidic catalyst and the radical addition using a radical initiator. As the last mechanism is the most insensitive to variations of the chemical structure, which is an important property for the reaction with peptides, the radical addition was chosen. An ultraviolet (UV) sensitive free-radical initiator, namely 4,4-Azobis(4-cyanovaleric acid) (ACVA), was used to start the coupling between the modified pigments and the anchor peptide LCI. The corresponding double bond needed for this reaction was introduced on the surface of the encapsulated pigment using trimethoxysilylpropylmethacrylate (TMSPMA) in a second shell growth reaction.
This research presents detailed information on the encapsulation of organic pigments, including solvent influence on shell growth, followed by the surface modification using methacrylate silanes which were investigated by zeta potential measurements and infrared (IR) spectroscopy. The addition of the peptide onto the pigment surface using thiol-ene reaction was followed by fluorescence microscopy. Furthermore, the adsorption ability of the anchor peptide LCI on a polypropylene model system was investigated using atomic force microscopy (AFM). Finally, polypropylene fibers were stained in a one-step approach at ambient conditions and compared with the fibers colored with the pristine organic pigment.
Materials and methods
Chemicals
The organic pigment Violet 19 was obtained from Clariant. Ethanol (reinst.) was purchased from Th. Geyer. Ammonia solution, Triton X-100 and ACVA were purchased from Sigma-Aldrich. Tetraethylorthosilicate (TEOS), Trimethoxyphenylsilane (TMPS), Trimethoxysilylpropylmethacrylate (TMSPMA) and Octyltriethoxysilane (OTES) were purchased from ABCR. Silica wafer (Orientation 100, p-type boron) was purchased from CrysTec. The anchor peptide LCI was synthesized and provided by Joanna Weber from evoxx technologies GmbH. The fluorescent labeled anchor peptide LCI-eGFP was synthesized and provided by Kerstin Rübsam from the group of Uli Schwaneberg at DWI-Leibniz Institute for Interactive Materials.
Encapsulation
One hundred and twenty-five milligrams of pigment Violet 19 (0.40 mmol) were dispersed in 500 mL water using a sonication bath (Elmasonic P60H: 37 kHz, 21W/L) for 180 min in a 1 L round-bottom flask. After this, the dispersion was continuously stirred at 400 rpm; 170 µL of TMPS (0.91 mmol) were quickly injected into the dispersion using a syringe. After 60 min, to ensure a sufficient accumulation of the hydrophobic silane on the pigment particles, 3 mL of ammonia solution (30 wt%) were added to start the encapsulation process. Additional amounts of 150 µL TMPS (0.80 mmol) were added after 24 h and 48 h, respectively. After 72 h the dispersion was washed three times in water and ethanol respectively, using centrifugation (10 min, 12,000 rpm) and sonication.
Shell growth
The encapsulated pigment particles were dispersed in 200 mL ethanol using a sonication bath (Elmasonic P60H: 37 kHz, 21W/L) in a round-bottom flask. The dispersion was stirred at 400 rpm while 500 µL of TMSPMA (2.1 mmol) were injected to induce shell growth. After 24 h, another additional 500 µL of TMSPMA (2.1 mmol) were added to the dispersion. After 72 h, the dispersion was washed three times in ethanol using centrifugation (10 min, 12,000 rpm) and sonication. The pigment particles were collected using centrifugation and thoroughly dried (30 ℃, 0.2 mbar).
LCI modification
One hundred and twenty-five milligrams of the modified pigment were dispersed in 100 mL water using a sonication bath (Elmasonic P60H: 37 kHz, 21W/L) for 15 min in a 250 mL round-bottom flask. The dispersion was stirred continuously at 400 rpm and flushed with nitrogen; 14 mg of ACVA (50 µmol) were dissolved in 4 mL water and added to the dispersion. After 30 min, the nitrogen flow was switched off; 1 mg pure LCI or fluorescent LCI-eGFP conjugate was dissolved in 1 mL water and injected into the dispersion. The reaction medium was irradiated with a UV lamp (LF-206-LS, UVitec: 365 nm, 6 W) for 24 h. The protein–pigment conjugate was washed three times in water using centrifugation (10 min, 12,000 rpm) and stirred with a vortex mixer (VV3-S40, VWR).
Synthesis of silica nanoparticles
One hundred and fourteen milliliters of ethanol, 5.7 mL ammonia solution and 3.8 mL TEOS (17 mmol) were stirred at 250 rpm for 24 h in a round-bottom flask. The silica nanoparticles were washed three times in ethanol using centrifugation (10 min, 12,000 rpm) and sonication. The centrifuged particles were dried for 12 h at 80 ℃ before dispersing them in ethanol.
Modification of silica nanoparticles
The silica particles were dispersed in 200 mL ethanol using a sonication bath (Elmasonic P60H: 37 kHz, 21W/L) in a round-bottom flask. The dispersion was stirred at 400 rpm while 500 µL of TMSPMA (2.1 mmol) were injected to cause the shell growth. After 24 h the dispersion was washed three times in ethanol using centrifugation (10 min, 12,000 rpm) and sonication. The particles were collected using centrifugation and thoroughly dried (30 ℃, 0.2 mbar).
Modification of silica wafer
Polished silica wafers were cut into 1 × 1 cm2 pieces, cleaned using a snow jet cleaning device (tectra GmbH) and activated using air plasma treatment (PlasmaFlecto 10: 60 s, 300 W, 0.2 mbar). The pieces were placed into a desiccator with a solvent reservoir containing 500 µL of OTES. The pressure was lowered to 2 mbar for 30 min, when the desiccator was closed. After 72 h the desiccator was opened, and the silica pieces were washed with ethanol using sonication and dried for 12 h at 80 ℃.
Adhesion of LCI
An aqueous LCI solution (75 µg/mL) was prepared. A modified silica wafer was rinsed with water and submerged into the LCI solution for 150 min at ambient conditions. The wafer was removed and rinsed with water to remove excess non-bound LCI. After drying at ambient conditions, the LCI monolayer was scratched with a cannula and characterized using AFM. After measurement, the same wafer was placed into a Triton X-100 solution (2 mmol/mL) for 5 min and rinsed with the same solution followed by pure water. The wafer was dried at ambient conditions and investigated with AFM.
Dyeing of synthetic yarn
The textiles, consisting of polypropylene and polyester, were washed for 15 min at 20 ℃ with Mucasol inside a Zeltex Polycolor laboratory dyeing apparatus. After that, the textiles were washed in water and dried at 25 ℃. The textiles were placed in the dyeing apparatus filled with 100 mL of pigment dispersion for 150 min at 20 ℃. The pristine pigment was tested at a concentration of 0.85 wt% and the protein–pigment conjugate with a concentration of 1 wt%. After dyeing, the textiles were rinsed with water and dried at ambient conditions. An additional washing step was conducted using a standard color detergent at 60 ℃ for 30 min and dried at ambient conditions. The textiles were examined using two different resistance tests: a washing fastness test according to DIN EN ISO 105-C06 and a rubbing fastness test according to DIN EN ISO 105-X12 / DIN 54021.
Characterization
Scanning electron microscopy (SEM) images were taken with a JSM6330F from JEOL at an acceleration voltage of 5 kV. Before measuring, the samples were sputtered with platinum (4 nm thickness). Fluorescence microscopy pictures were taken with a DMi8 from Leica. For image processing LAS X software (Version 2.0.0) from Leica was used. AFM images were taken with a Bruker Dimension Icon using Tapping Mode with OTESPA tips (k = 42 N/m, f0 = 300 kHz). Nanoscope (Version 9.1) and Nanoscope Analysis (Version 1.5) software were used for measurements and for the image processing, respectively. The contact angle was measured using Drop Shape Analyzer OCA 15 from Dataphysics. IR measurements were conducted using an RFS 100/S from Bruker Optik GmbH. Dynamic light scattering (DLS) and zeta potential results were obtained with a Zetasizer Nano ZS from Malvern Instruments. Thermogravimetric analysis (TGA) was done using a TA Instruments Thermogravimetric Analyzer Q500 at a heating rate of 10 K/min under a nitrogen atmosphere.
Results and discussion
The developed approach for the staining of polypropylene textiles with Violet 19 pigment consists of three subsequent steps, illustrated in Figure 1. Each step will be described in detail in the following sections, explaining the synthetic and analytical challenges and their solution.
Schematic showing the synthesis route for highly adhesive coloring agents: encapsulation of organic pigment Violet 19 with trimethoxyphenylsilane, shell growth using trimethoxysilylpropylmethacrylate and modification step with the anchor peptide LCI.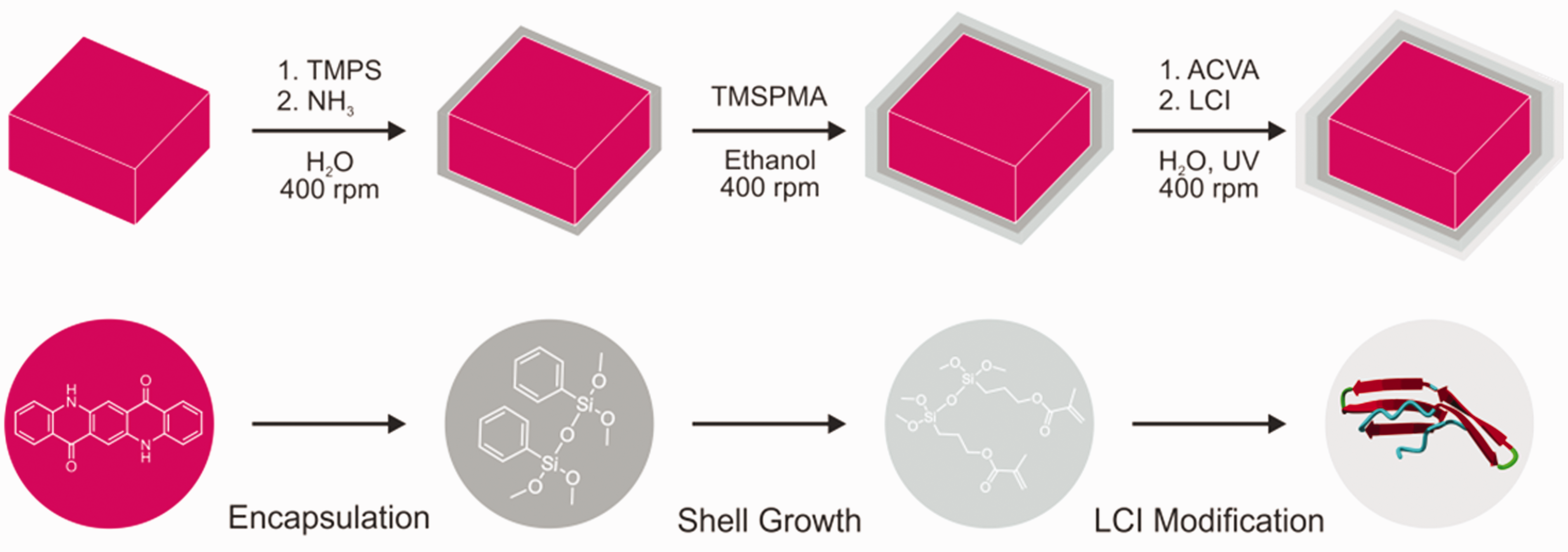
Encapsulation
Typically, the encapsulation of colloids in the silica/silane shells is carried out using TEOS and auxiliary surfactants supporting the shell formation, for example CTAB. Although this approach leads to the successful encapsulation, the resulted colloids demonstrate usually an incorporation of the applied surfactant into the silica shell changing their charge and other physicochemical properties. 38 To avoid this, the silane used should possess, at least partially, a certain surface activity. As a suitable candidate for this concept, the silane TMPS was selected. Due to its aromatic ring and its low solubility in short-chain alcohols as well as in water, a successful encapsulation was conceivable. Prior to the encapsulation step, a suitable dispersion method had to be found, to achieve a narrow size distribution of the pigment particles in solution (Compare in the electronic supplementary information section (ESI) Figure S1 and S2). An extended treatment of the dispersion using an ultrasonic bath was able to achieve the necessary quality.
SEM images in Figure 2 show two types of encapsulated pigment particles catalyzed by ammonia using TMPS in methanol and purified water, respectively. The findings in methanol show an irregular and non-continuous coverage of particles of the organic pigment. In contrast to this, the results obtained in purified water show a uniform shell with smooth surface around each pigment particle. Apparently, the solubility of TMPS in methanol is still too high to induce a sufficient phase separation between the silane and the surrounding solvent for a complete encapsulation, which takes place in water.
Scanning electron microscopy images showing the first encapsulation step of the pigment Violet 19 using trimethoxyphenylsilane conducted in different solvents: (a) methanol and (b) purified water.
In order to achieve a complete coverage of the pigment with a silica shell, but to keep its thickness as low as possible to prevent a strong alteration of the coloring abilities of the dye, kinetic experiments were conducted. An important parameter for the successful encapsulation using this two-phase approach was the low concentration of pigments in the aqueous dispersion (<0.025 wt%). Higher concentrations resulted in a significant increase of agglomerates (size > 1 µm). Also, a high initial concentration of TMPS leads to the same effect combined with an unacceptable thick silica shell. After investigation using DLS and zeta potential measurements (compare ESI), the most effective and successful approach was to combine a relatively long reaction time and a stepwise addition of low amounts of TMPS.
Shell growth
The second step was the introduction of functional groups for the coupling of anchor peptides onto the colloid surface. In this case, the double bond of TMSPMA was used for a silane shell growth step. For this, the washed encapsulated pigment from the first synthesis step was redispersed in ethanol and subsequently stirred during the addition of the silane. Similar to the first encapsulation step, a longer reaction time accompanied by stepwise addition of TMSPMA proved to be successful, which again was monitored by DLS measurements (compare ESI). During this step, a successful functionalization could not be verified using zeta potential measurements, as no change was anticipated and detectable (compare ESI). For this reason, additional spectroscopic measurements needed to be conducted. IR measurements using the encapsulated pigment are not possible, due to signal overlap (compare ESI Figure S3). Therefore, a model system using blank silica nanoparticles was treated under identical conditions, to test the shell growth step. Afterwards, IR measurements before and after the functionalization step were conducted and the results are displayed in Figure 3.
Infrared measurement of blank silica particles and silica particles functionalized with trimethoxysilylpropylmethacrylate. Inset shows the magnification of the region between 1500 and 1750 cm−1.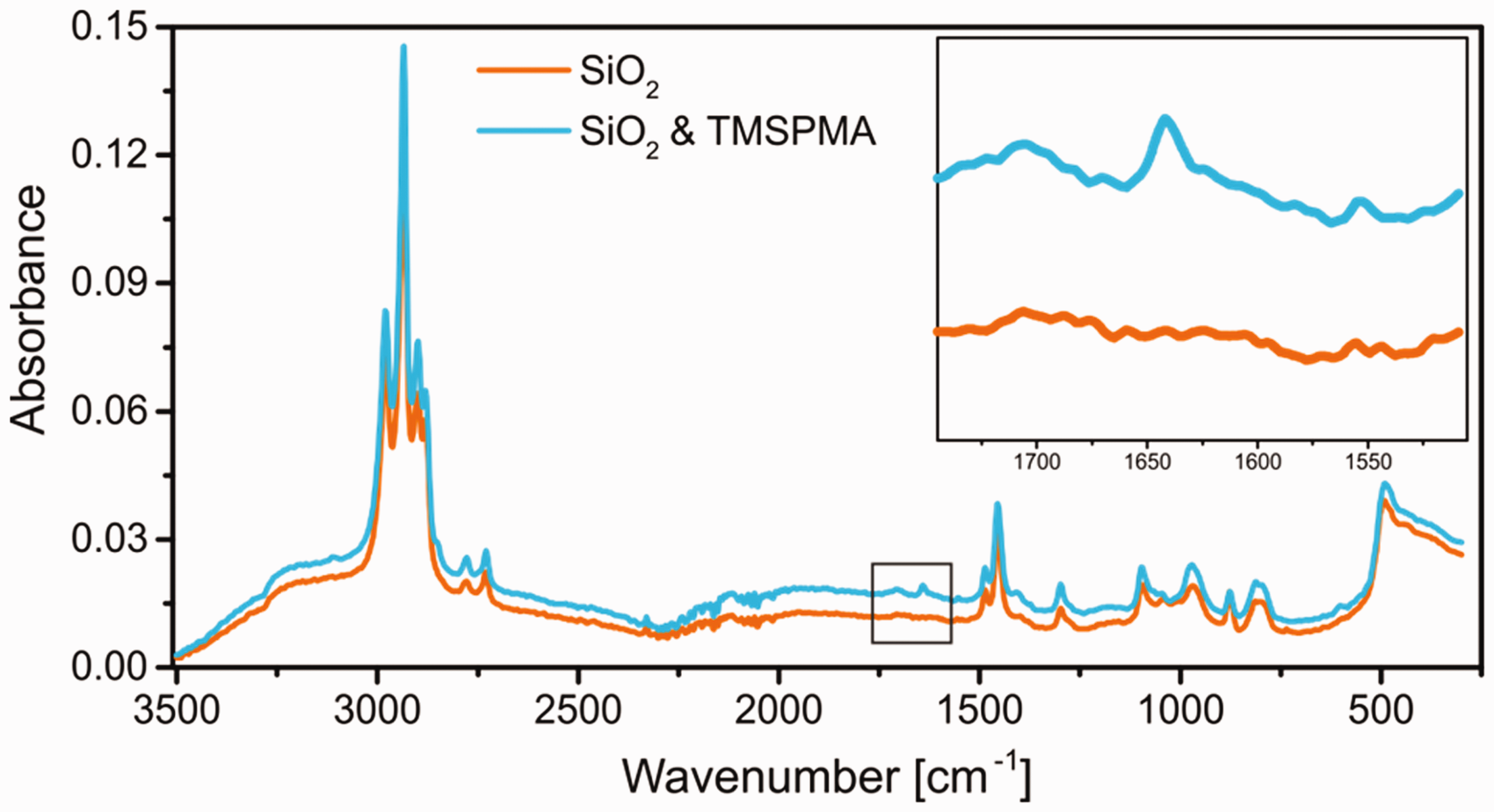
The measurements compare the IR spectra for silica nanoparticles with and without functionalization with TMSPMA. The almost identical spectra after treatment shows a new signal around 1641 cm−1, which can be attributed to the C=O stretching vibration introduced by the methacrylate group (see inset). 39 This proves indirectly the successful introduction of the double bonds, particularly for such a well-studied and common functionalization step.40–42
LCI modification
The last step for the production of highly adhesive coloring agents was the coupling of the anchor peptide LCI to the modified pigments. This reaction brought considerable analytical challenges, concerning the successful execution of the thiol-ene reaction. SEM images of the pigments after LCI functionalization could not visualize the attached LCI layer (compare Figure S4). As these experiments did not give absolute certainty about the successful coupling of the LCI to the modified pigment, the model system used previously in the shell growth step was further modified using N-acetylcysteine as a representative thiol coupling partner, using the same reaction conditions. The results of IR measurements are displayed in Figure 4. The spectrum of the coupled silica particles clearly shows two new signals: the large stretching vibration signal of the carbonyl group around 1600 cm−1 and a smaller signal at 3300 cm−1 for the corresponding amine group. The CH-stretching vibration of the acetyl group is not visible due to the strong underlying signal. This measurement validates the successful coupling of a small amino acid onto the silica surface using the thiol-ene reaction. Thus, the chosen chemistry is feasible for our approach. However, a further experiment was conducted to prove the successful coupling of the anchor peptide.
Infrared measurement of silica particles functionalized with trimethoxysilylpropylmethacrylate and afterwards coupled with N-acetylcysteine as a representative molecule containing a thiol group. Inset shows the magnification of the region between 1500 and 1700 cm−1.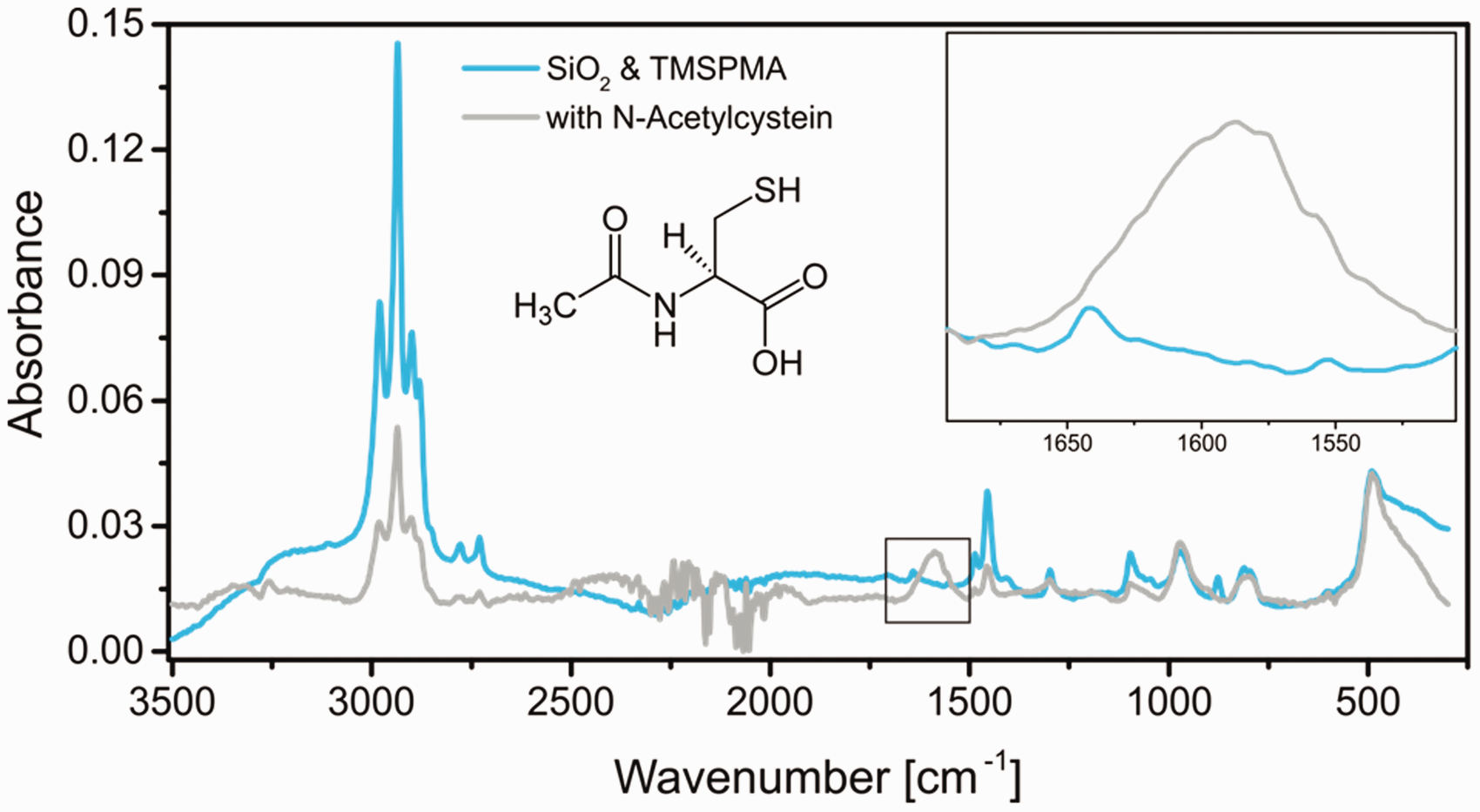
During the production of LCI it is possible to directly attach a green fluorescent protein (GFP) to the anchor peptide, which can be used to follow the successful coupling of our system. For this, the last modification step was reproduced while using the fluorescent labeled anchor peptide under the same reaction conditions. After thorough washing of the coupled LCI–pigment conjugates using purified water and repetitive centrifugation and redispersion, the samples were observed using a fluorescent microscope. Figure 5 shows the transmitting light and fluorescent channel of the obtained conjugates. The light microscope channel shows the Violet 19 pigment particles, which still possess their reddish color despite the multiple encapsulation steps with silanes. The fluorescent channel clearly shows a strong fluorescent signal of the pigments due to the successful coupling of the fluorescently labeled anchor peptide. The three obtained results, including the increase in particle size, the IR measurement and the coupling of a fluorescently labeled anchor peptide, indicate the successful binding of LCI to the encapsulated and modified pigment Violet 19 using the UV initiated radical thiol-ene reaction.
Anchor peptide conjugate images using: (a) microscope and (b) fluorescence microscope.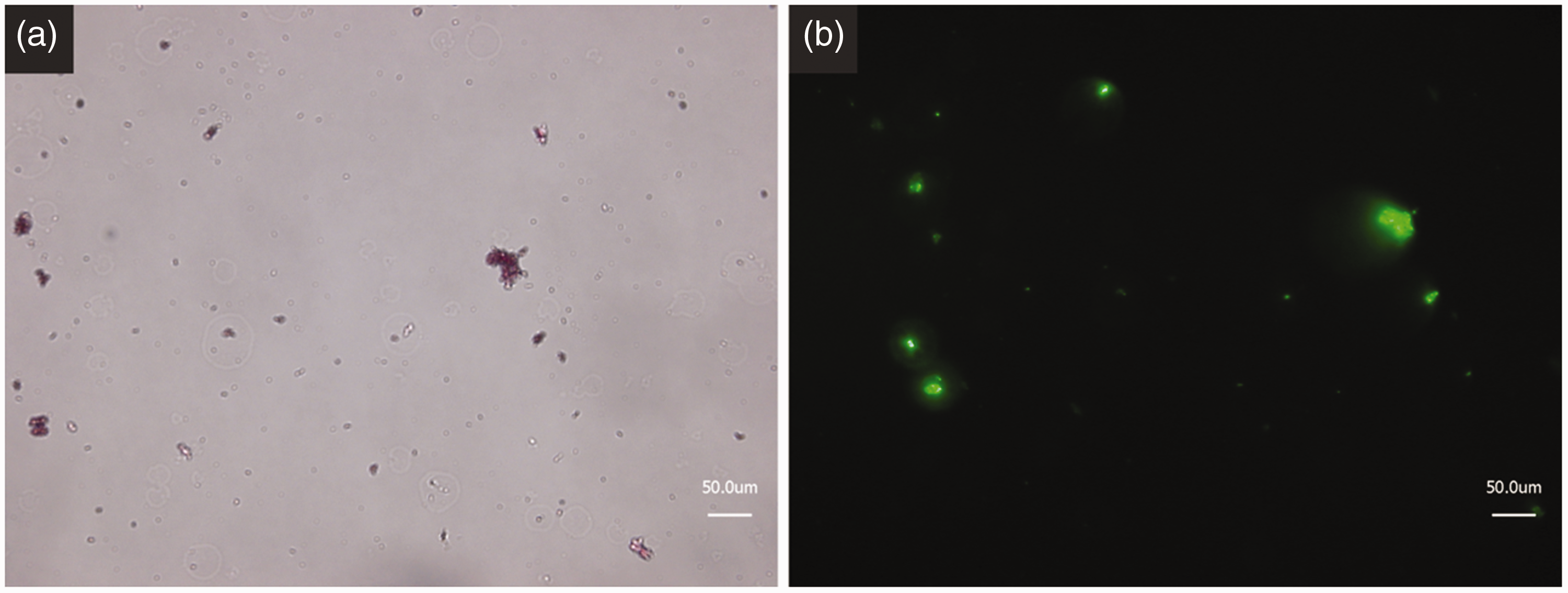
Adhesion of anchor peptides
Before testing the obtained conjugates on yarns and zippers consisting of polypropylene and polyester, the adhesive ability and stability during washing of the pure anchor peptide was evaluated. To measure the thickness of an applied LCI layer a very smooth surface was needed. Therefore, normal polypropylene samples could not be used for this experiment. As a replacement, polished silica wafers were functionalized using plasma treatment and chemical vapor deposition of OTES to simulate a polypropylene-like surface on the wafer (see Figure 6).
Top row: synthesis route of silica wafer model system for LCI attachment using air plasma treatment and the chemical vapor deposition of octyltriethoxysilane, to achieve a smooth and polypropylene-like surface. Bottom row: attachment and washing step for the anchor peptide LCI to the modified silica wafer.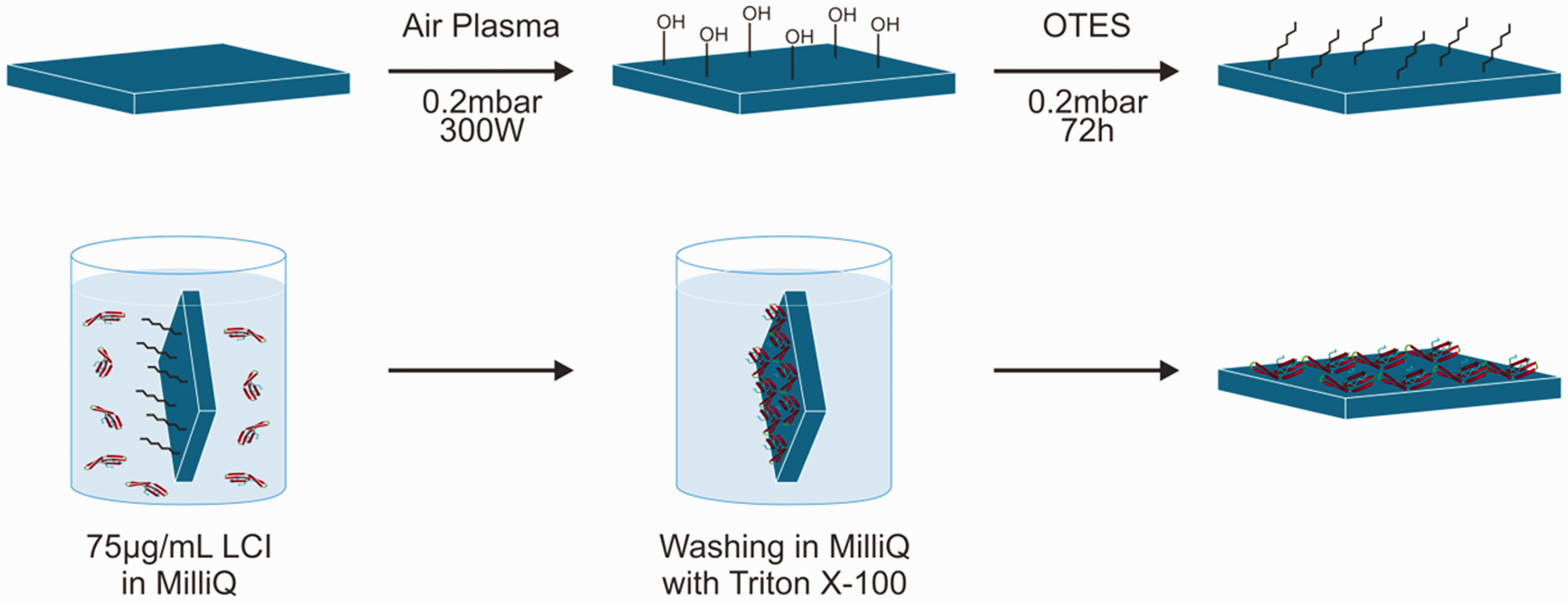
The successful modification of the silica wafer with OTES was investigated by contact angle measurement (see Figure S6) and surface roughness via AFM (see Figure S5). AFM results suggested a roughness after modification below 0.6 nm, which is sufficient for the intended experiment. In comparison with the untreated silica wafer with contact angle of 30°, measurements for the modified wafer showed a hydrophobic surface with a result of 102°, very close to the value for polypropylene surface with 103°.
The technique used for the deposition of the LCI anchor peptide with subsequent washing step is displayed in Figure 6. A diluted solution of the anchor peptide was prepared, and a modified silica wafer was submerged into a glass vessel. After this, the wafer was rinsed thoroughly with purified water and stored until it was completely dried under ambient conditions. The sample surface was scratched with a cannula before measuring the layer thickness with AFM. Figure 7(a) displays the film thickness after adhesion, rinsing and drying of the anchor peptide on the modified silica wafer. The images show a smooth and regular film on top of the modified silica wafer. This confirms the highly adhesive nature of LCI to hydrophobic surfaces. To test the stability of the LCI layer, the same sample was afterwards submerged into an aqueous solution containing a non-ionic surfactant (Triton X-100). The test wafer was again removed, rinsed with the surfactant solution followed by purified water and stored until completely dried under ambient conditions. The thickness measurements after treatment with surfactant solution are displayed in Figure 7(b). The surface shows nanoscaled irregularities inside the LCI layer, but overall the film is intact, and its thickness did not decrease during the washing step demonstrating the high adhesion ability of the anchor peptide LCI and the stability of the adsorbed LCI film during washing, which is an important property for a textile coloring agent. These results are also in good agreement with the observations made in previous studies concerning adhesion and washing stability for anchor peptides coupled with GFP.
19
Atomic force microscopy measurement of LCI film thickness (a) after deposition and (b) after the washing step using a Triton X-100 solution. Right side shows an overview picture and left side shows a high magnification picture of selected area on the scratch border and the corresponding cross-section graph made in the perpendicular direction to this border.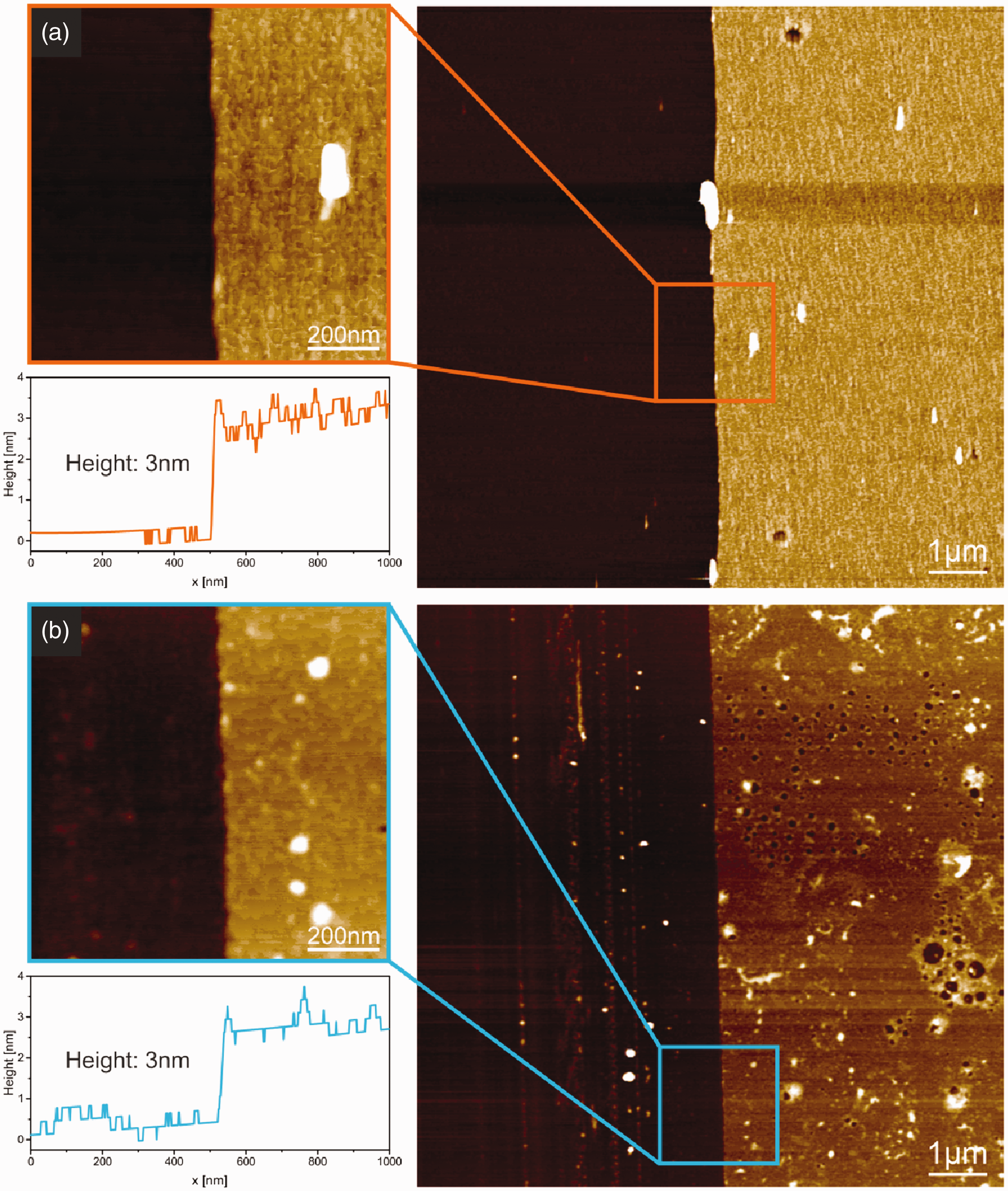
Dyeing of synthetic yarn
The synthesized peptide–pigment conjugates were tested for their dyeing ability as well as for the stability against washing and friction. These findings were compared with the results of the pristine pigment. To prepare the dyeing dispersions with the same absolute dye content, the amount of pure pigment inside the capsules had to be determined. To obtain these data, TGA measurements were taken for the pure and for the modified pigment after all shell growth steps were conducted (compare Figure S7). During this measurement a small amount of sample is heated continuously, starting at room temperature and reaching up to 600 ℃. The organic pigment does disintegrate during this procedure, leaving impurities and the surrounding silica shell. The difference between the residual weights of two samples was calculated to be 15 wt%, which gives the amount of introduced silica in the shells of the encapsulated pigment particles. With this information, comparable dyeing dispersions with 0.85 wt% pure pigment and 1.0 wt% peptide–pigment conjugates, respectively, were produced. The dyeing procedure is described in detail in the materials and methods section. The textiles used are washed to obtain equal starting conditions. Afterwards the textiles are submerged into 100 mL of each dyeing dispersion inside a dyeing apparatus for 150 min at room temperature. Next the textiles are rinsed with purified water and dried at ambient conditions. Finally, an additional washing step using a standard color detergent is conducted, again followed by drying at room temperature. Two different textile types were used for two different resistance experiments: a polypropylene yarn for a rubbing fastness test and a polyester zipper with a surrounding mixed fabric consisting of polypropylene and polyester for a washing fastness test. All results comparing the two dyeing dispersions are displayed in Figure 8.
Photographs of (a) polypropylene yarns after rubbing fastness test and (c) dyed zippers after washing fastness test, which were dyed with pristine pigment (left side) and LCI–pigment conjugate (right side). (b) Respective light microscope pictures of the polypropylene yarns after the rubbing fastness test.
The top row in Figure 8 shows the achieved dyeing results for the polypropylene yarn next to the textile on the left side which was used to rub over the yarn surface. Both polypropylene yarns show a strong red color, but the color intensity of the sample dyed with the conjugates is clearly higher. Almost no colorless spots are visible for this yarn. This is a very strong argument for the higher adhesion of the LCI modified pigment in comparison with the pristine pigment. However, both samples show a considerable transfer of pigments onto the second textile during the rubbing fastness test. In contrast to common dyeing processes, no high pressure was used during this procedure. At high pressure, pigments can penetrate the polypropylene yarns, reaching into the interstices between the fibers, and are therefore better protected against rubbing applied from the outside.15,43 In the case at hand, the pigments could not penetrate the fibers, which is clearly visible from the multilayer microscope images in the second row in Figure 8, which were taken after the rubbing fastness test. Pigments are almost exclusively visible inside the grooves between the fibers. Other pigments from the top surface were removed during the rubbing fastness test. Still, the polypropylene yarns do not show a reduced coloring effect at the rubbing position, leading to the theory that the pigments inside the groove do possess a sufficient dyeing ability. The third row displays the washing fastness test, which is conducted by wrapping an uncolored textile around the dyed zipper during the washing step using color detergent.
The pristine pigment clearly did not reach the color intensity of the modified pigment and the uncolored textile even showed clear signs of red staining due to dye transfer during washing. The adhesion of the peptide–pigment conjugate is significantly higher in comparison with the pure pigment, leading to a stronger dyeability and stability during washing. In comparison with the pure polypropylene fiber, the mixed zipper fabric demonstrates a lower coloration. This is understandable as it is a mixture of polypropylene and polyester and the anchor peptide was developed for strong hydrophobic surfaces. Although successful dyeing experiments have been conducted and a positive effect of the anchor peptide on the adhesion of the encapsulated pigment particles was established, the overall coloration intensity achieved is still not comparable with conventional industrial standards. A quantitative investigation concerning the color strength of the stained textiles was not possible due to the uneven coloration. However, there are important parameters which could provide access to a potential improvement. The pure pigment was used at a concentration of 0.85 wt%, which is still at the lower end of what is used in industry. Common dyeing processes are conducted at pigment concentrations between 0.01 wt% for very light colors and up to 3 wt% for fully dark colors. However, these concentrations include auxiliaries and compatibilizers which improve the overall dyeing process. Furthermore, these processes are conducted at high pressures and increased temperature. Additionally, the amount of applied peptide per encapsulated pigment concentration was kept at very low values. During the synthesis, 1 mg of LCI was utilized for the preparation of 125 mg of the encapsulated pigment. High-scale synthesis of the anchor peptide would reduce its cost dramatically and would give possibility to apply higher concentrations of LCI during the synthesis. This should in turn further improve the adhesion of the modified pigment. Also, the entire dyeing process could be improved. No adjustments concerning reaction time or mixing regime upon dyeing have been conducted.
Conclusion
In this work, a three-step synthesis of an adhesive staining agent for polypropylene fibers was presented. The organic coloring pigment Violet 19 was encapsulated in a first step using only TMPS, serving simultaneously as a shell building material as well as a surfactant, due to its attractive interaction with the pigment surface. By using TMSPMA in a second shell growth step, the necessary functional groups for a thiol-ene coupling reaction with the highly adhesive anchor peptide LCI were introduced. After successful synthesis of this peptide -pigment conjugate proven by SEM, light microscopy and DLS, its improved coloring capability in comparison with the pristine pigment was demonstrated by staining polypropylene fibers. In contrast to commonly utilized highly energy-intensive staining approaches, a simple mixing approach at ambient conditions was used, which could dramatically decrease the energy consumptions during the staining. The subsequent tests for discoloration upon washing and rubbing showed a better performance of polypropylene textiles colored using the new method than ones stained with the pristine pigment.
Footnotes
Supplemental material
Supplemental data can be found in the online version of this article. The supplementary materials give detailed descriptions of the production of pigment dispersions, DLS and zeta potential measurements, IR measurement of encapsulated pigments, TGA measurements and silica wafer functionalization.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
