Abstract
Objective
To assess the feasibility of three-dimensional (3D) stereophotogrammetry as a tool in pediatric outpatient assessment, in exploring associations between facial morphometric parameters of children aged 2–17 years, and their risk of sleep-disordered breathing (SDB) as evidenced by their Pediatric Sleep Questionnaire (PSQ) score.
Methods
A cross-sectional study of 180 pediatric patients (aged 2–17 years) recruited consecutively from a tertiary pediatric otolaryngology clinic. 3D facial images were acquired using stereophotogrammetry, and patients’ SDB risk was evaluated using the validated PSQ. Linear regression models assessed associations between PSQ scores and demographic and facial morphometric variables. Geometric morphometric (GMM) analysis was performed to assess differences in facial morphology between subjects at high and low risk for SDB.
Results
33.9% of patients scored at high risk for SDB. No statistically significant associations were found between PSQ score and any 3D facial morphometric parameter. PSQ score was not associated with sex, age, age×sex interaction, or body mass index. Geometric morphometric analysis found no difference in facial shape between those at high risk for SDB and those at lower risk.
Conclusions
Stereophotogrammetry may be successfully applied in a specialist pediatric outpatient clinic as a non-invasive and low-resource method for measuring 3D facial features. No linear or angular measurements assessed in this study were found to have a strong correlation with PSQ score. These findings suggest that 3D external facial soft-tissue measures may not add discriminatory value beyond symptom-based screening in this tertiary clinic setting, and that facial morphology alone is unlikely to be a sufficient screening tool for pediatric SDB. Screening should primarily be based on patient signs and symptoms, and consideration of internal anatomical factors.
Keywords
Introduction
Sleep-disordered breathing (SDB) is a collection of conditions that ranges from snoring, to partial upper airway obstruction, to obstructive sleep apnea (OSA). The prevalence of habitual snoring and OSA are estimated at 3.2–12% and 0.7–2.9%, respectively, in the general pediatric population.1–3 However, the burden of these disorders may be even larger as breathing issues are commonly under-reported by parents. 4 The negative sequelae of pediatric SDB result from frequent arousals from sleep and possible blood oxygen desaturation. The consequences of pediatric SDB include neurocognitive dysfunction and effects on behaviour, cardiopulmonary complications, metabolic dysfunction, enuresis, failure to thrive, and negative impact on quality of life. 5
The exact relationship between SDB and altered facial morphology, and whether one predisposes the other, remains unknown. Facial alterations reported in the pediatric SDB literature include mandibular retrognathia, narrow upper airway space, and increased facial height.6,7 The persistence of mouth breathing in SDB may be due to nasal obstruction, such as the case with adenoid hypertrophy. Obstruction of the upper airway has been hypothesized to alter facial growth and increase anterior face height through postural changes of the mandible and associated soft tissues to facilitate oral respiration.6,8 While recent meta-analysis of published data has noted statistically significant differences in facial morphology of SDB populations, the clinical relevance of the very small magnitude of these alterations has been questioned. 9
Most previously published studies assessing craniofacial shape in relation to sleep have used anthropometric measurements or radiographs. However, there are well-known limitations to these techniques that can be overcome with recent advancements in three-dimensional (3D) imaging. 10 Stereophotogrammetry allows 3D representation of facial features to be obtained without exposure to radiation or invasive procedures. 11 The 3dMD stereophotogrammetry system (3dMD, Atlanta, GA, USA) rapidly acquires accurate soft-tissue facial morphology, which is of particular use in pediatric populations.
Geometric morphometrics (GMM) is a method utilizing landmarks to describe shape. GMM can be used to describe shape variation between individuals and to identify patterns of variation of interest. This overcomes several disadvantages of traditional morphometrics, including difficulties with shape analysis as linear distances are highly correlated with size, and generally do not take into account the geometric configuration of the landmarks relative to each other. 12 GMM also allows the whole of a shape to be analyzed and reduces an element of bias, as traditional morphometrics typically only considers a specific part of a shape as certain linear measurements and angles are arbitrarily selected. 12
The reliable triaging of patients with SDB for treatment or definitive diagnosis with polysomnography remains a challenge in the clinical practice of otolaryngology. Implementation of efficient and effective use of GMM in patient assessments remains untested, despite the potential to ultimately improve the management of this vulnerable patient population. The aims of this study were: (1) to assess the feasibility of using 3dMD stereophotogrammetry for pediatric research in a clinical setting; (2) to identify possible correlations between risk of sleep-disordered breathing in children aged 2–17 years, as measured by the PSQ, and their three-dimensional facial morphometric parameters.
Materials and methods
This was a prospective cross-sectional study. The reporting of this study conforms to STROBE guidelines. 13 Patients were recruited consecutively. Children between the ages of 2–17 years presenting to the Pediatric Otolaryngology Head and Neck Surgery outpatient clinic at BC Children’s Hospital, Vancouver, British Columbia, Canada, between July 2017 and January 2020, were invited to participate in the study. Exclusion criteria included a history of orthodontic treatment, tonsillectomy and/or adenoidectomy, or the presence of congenital craniofacial abnormalities/syndromes, including cleft lip and/or palate. Written informed consent for participation was obtained from the parents or guardians, and assent from all children above the age of 7.
This research was approved by the Research Ethics Board of the University of British Columbia and BC Children’s Hospital, Vancouver, Canada (ethics certificate number H17-01292, approval date: June 26, 2017. This research was conducted in accordance with the Declaration of Helsinki (1975, revised 2024). All patient data were de-identified prior to analysis; no personally identifying information is reported in this manuscript.
Basic demographic data including age, sex, BMI, and ethnicity were collected at time of recruitment. BMI data were available for a subset of 80 participants only; the reason for missing BMI data was that height and weight measurements were not routinely collected for all patients presenting to the outpatient clinic during the study period. All analyses involving BMI are therefore reported with the available complete-case sample (n = 80), and sensitivity analyses using the full cohort (n = 180) without BMI are also presented where applicable. Parents or guardians were asked to complete the validated Pediatric Sleep Questionnaire (PSQ). 14 The questionnaire consists of 22 symptom items with domains targeting sleepiness, snoring, and inattention/hyperactivity. Each question is answered by “yes”, “no”, or “don’t know”. The parent or guardian was instructed to answer “yes” if they believed that the participant elicited the behaviour more than half of the time. PSQ scores were calculated by dividing the sum of “yes” answers by the total number of questions answered. A score of 0.33 or higher indicated a high risk for sleep-disordered breathing.
The 3dMD imaging system was calibrated as per manufacturer’s recommendations a minimum of one time at the start of each day when it was being used for the study. Subjects were placed in an examination chair at a fixed distance from the 3dMD imaging system. Participants were asked to sit facing forwards in natural head position, with face relaxed and mouth closed in a neutral position. Any physical objects such as glasses, hats, or headbands that obstructed the head or face were removed. If the subject was unable to sit in the examination chair by themselves, they would sit on the lap of their parent or guardian.
A landmark template consisting of 19 landmarks (Figure 1), based on that used by Jayaratne et al.,
15
was generated utilizing the 3dMD Vultus software (3dMD, Atlanta, GA, USA). Landmarks were manually identified for each subject by a single investigator who was blinded to subject identity and PSQ score. Identification of landmarks for each subject was performed twice, on separate occasions. Landmarks were exported in .txt file format with their respective x, y, z coordinates. This .txt file was opened in Microsoft Excel and converted to the appropriate format for further analyses. An analysis template (Table 2) consisting of 48 linear and angular measurements based on the 19 landmarks was also generated using the 3dMD Vultus software. The 48 measurements for each subject were exported into an Excel spreadsheet. Each subject had two sets of 48 measurements based on landmarking on two separate occasions. These two sets of data were used to test for intra-rater reliability. Figure 1 was created by the authors using the 3dMD Vultus software and is original to this study. No artificial intelligence tools were used in any aspect of this research. Example of a 3-dimensional image with facial landmarks labelled, created by the authors using 3dMD Vultus software (3dMD, Atlanta, GA, USA) and original to this study. Nasion (N): midpoint on the soft tissue contour of the base of the nasal root at the level of the frontonasal suture. Glabella (G): smooth part of the forehead above and between the eyebrows. Gnathion (GN): most inferior midpoint on the soft tissue contour of the chin. Stomion Upper (STO_U): midpoint of the lower border of the upper lip. Stomion Lower (STO_L): midpoint of the upper border of the lower lip. Subnasale (SN): midpoint on the nasolabial soft tissue contour between the columella crest and the upper lip. Tragion Point (T): located at the upper margin of each tragus. Sublabiale (SL): most posterior midpoint on the labiomental soft tissue contour that defines the border between the lower lip and the chin. Exocanthion (EX): soft tissue point located at the outer commissure of each eye fissure. Endocanthion (EN): soft tissue point located at the inner commissure of each eye fissure. Alare (AL): most lateral point on each alar contour. Alar Curvature Point (AC): located at the facial insertion of each alar base. Crista Philtri Point (CPH): located at each crossing of the vermilion line and the elevated margin of the philtrum. Cheilion (CH): point located at each labial commissure. Pogonion (PG): most anterior midpoint of the chin. Labiale Superior (LS): midpoint of the vermilion line of the upper lip. Labiale Inferior (LI): midpoint of the vermilion line of the lower lip. Pronasale (PRN): most anterior midpoint of the nasal tip. Mid-Endocanthion Point (MEN): midpoint of endocanthion points.
The threshold for statistical significance was set at P < 0.05 for all tests. All variables measured had their Pearson correlation with the PSQ score calculated to identify possible associations. To determine the possible predictive value of each variable, LASSO (least absolute shrinkage and selection operator) regression analysis (with 10-fold cross-validation for the penalty term) was performed. Predictor variables were standardized (mean = 0, standard deviation = 1) prior to LASSO modelling to facilitate comparison of regression coefficients. Additionally, the association of age, sex (and their interaction), and BMI with PSQ score was assessed separately via linear regression. A sensitivity analysis omitting BMI was conducted using the full sample (n = 180) to assess whether the restriction to complete-case BMI data materially influenced findings.
Intra-rater reliability was tested by intraclass correlation coefficients (ICC). All images were landmarked twice on separate occasions by the same investigator. ICC estimates were calculated using SPSS statistical package version 23 (SPSS Inc., Chicago, IL) based on a two-way mixed-effects model. 16 Values less than 0.5 are indicative of poor reliability, values between 0.5 and 0.75 indicate moderate reliability, values between 0.75 and 0.9 indicate good reliability, and values greater than 0.90 indicate excellent reliability. 16
Generalized Procrustes analysis (GPA) 17 and subsequent analyses were performed with R software. Landmark coordinate data (x, y, z coordinates) were exported manually for each landmark and for each subject from 3dMD Vultus. The coordinate data were compiled into a Microsoft Excel spreadsheet formatted for R software. Procrustes shape coordinates were obtained via GPA. Principal component analysis (PCA) was performed to describe the shape variation within the dataset with principal components and their corresponding eigenvalues. ANOVA was performed for each principal component to determine whether sex, PSQ score, and sex×PSQ interaction had an effect on facial shape. MANOVA (Procrustes ANOVA) was then performed for the principal components describing 90% of the shape variation in the dataset. Allometry was removed via linear regression prior to analyses subsequent to GPA to remove the confounding variable of age-associated growth.
Results
Subject demographic data.

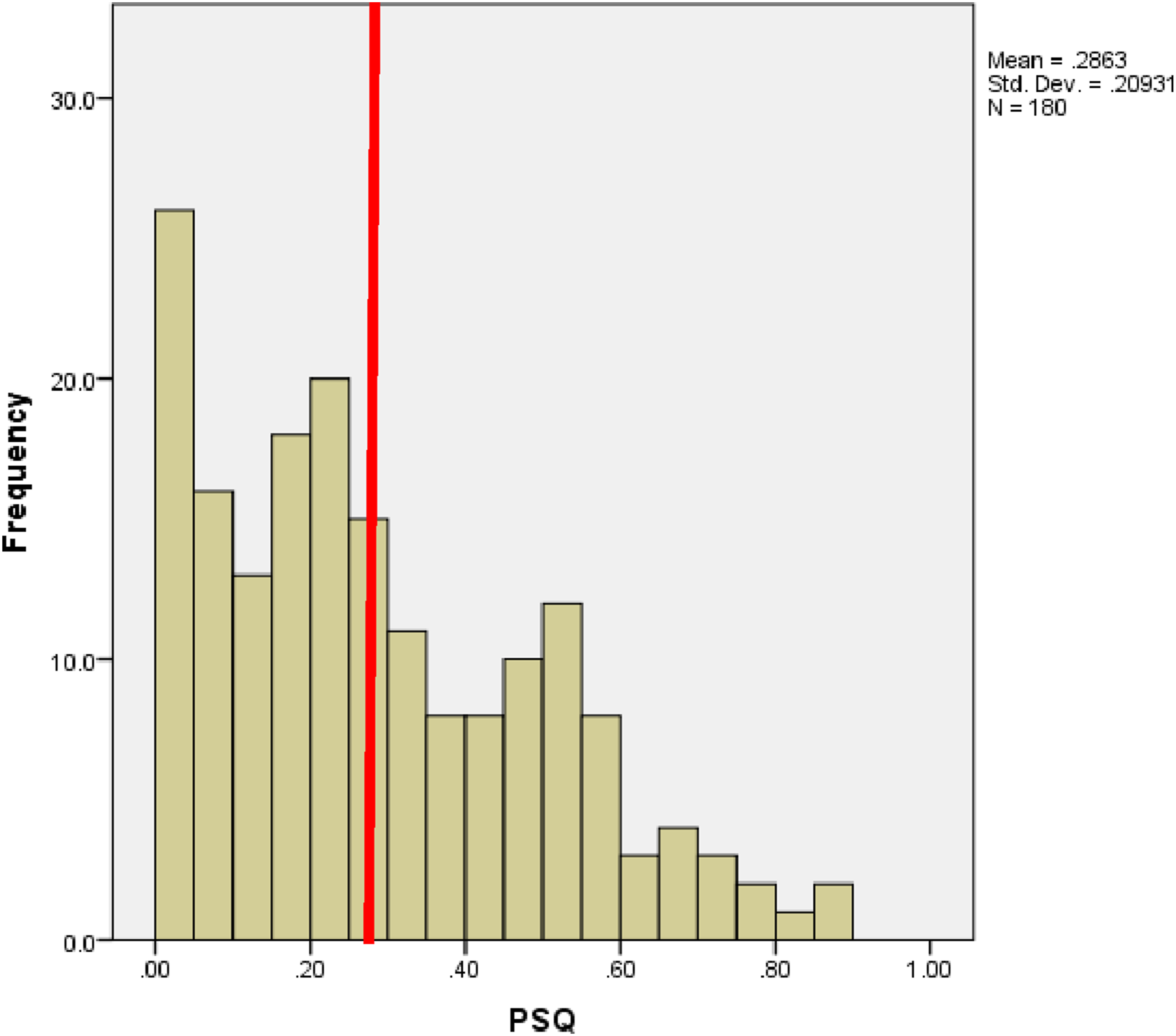
Distribution of subject PSQ scores (n = 180). The red vertical line indicates the threshold PSQ score of 0.33, above which subjects are classified as high risk for sleep-disordered breathing. Mean = 0.286, SD = 0.209.
Regarding feasibility, stereophotogrammetry was successfully integrated into clinic workflow with minimal disruption as imaging was conducted during downtime while waiting for the attending physician. Image acquisition and consent typically required fewer than 15 minutes per patient as performed by clinical research staff and was generally well accepted by patients and their families.
Intra-examiner reliability was assessed from measurements obtained from two separate landmarking datasets by the same investigator. All measurements showed good (ICC 0.75–0.90) or excellent (ICC > 0.90) intra-rater reliability except for philtrum width (ICC = 0.61), which showed moderate reliability.
Definition of facial anthropometric measurements used for analysing 3-dimensional images, and Pearson correlation with PSQ score.

Note. All correlations were non-significant (p > 0.05).
Abbreviations: N, nasion; GN, gnathion; STO_U, stomion upper; STO_L, stomion lower; SN, subnasale; T_R/T_L, right/left tragion; SL, sublabiale; EX_R/EX_L, right/left exocanthion; EN_R/EN_L, right/left endocanthion; AL_R/AL_L, right/left alare; AC_R/AC_L, right/left alar curvature point; CPH_R/CPH_L, right/left crista philtri point; CH_R/CH_L, right/left cheilion; PG, pogonion; G, glabella; LS, labiale superior; LI, labiale inferior; PRN, pronasale; MEN, mid-endocanthion point; PSQ, Pediatric Sleep Questionnaire; BMI, body mass index; R, right; L, left.
A linear regression model was fit including age, sex, their interaction, and BMI as predictors of PSQ score (n = 80, complete-case analysis). The regression coefficients for age (−0.009; p-value= 0.432), sex (−0.079; p-value= 0.210), their interaction (0.005; p-value= 0.538), and BMI (0.012; p-value= 0.067) were all statistically non-significant. This indicates no clinically meaningful change in PSQ score with sex, changing age, or changing BMI in this sample. A sensitivity analysis including all 180 subjects (excluding BMI) similarly found no significant association of age or sex with PSQ score, confirming that the restricted complete-case sample did not materially alter these conclusions.
Variables and regression coefficients from LASSO model, with correlation to PSQ score. All predictor variables were standardised (mean = 0, SD = 1) prior to modelling.
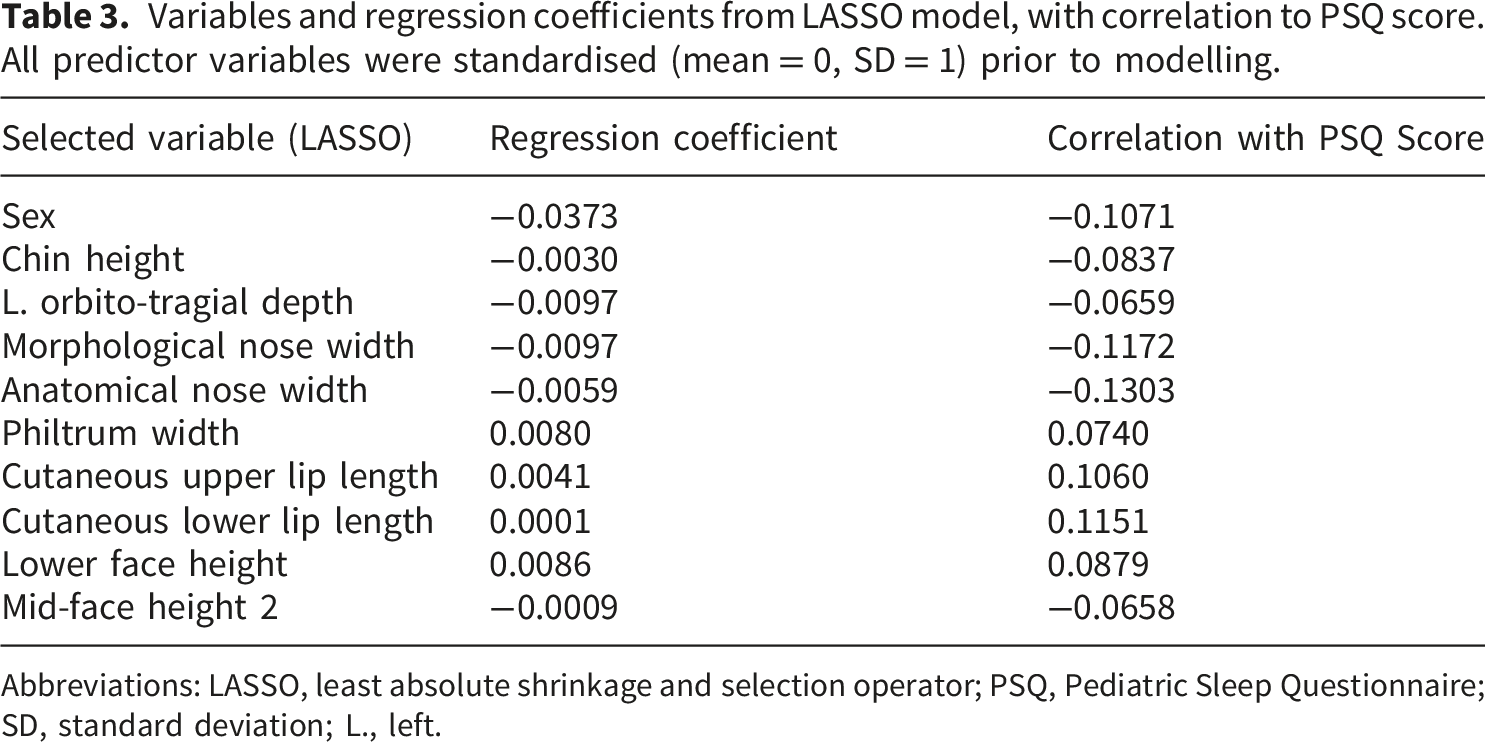
Abbreviations: LASSO, least absolute shrinkage and selection operator; PSQ, Pediatric Sleep Questionnaire; SD, standard deviation; L., left.
Procrustes ANOVA (MANOVA). Allometry removed; PC 1–21 included (explaining 90% of shape variation).
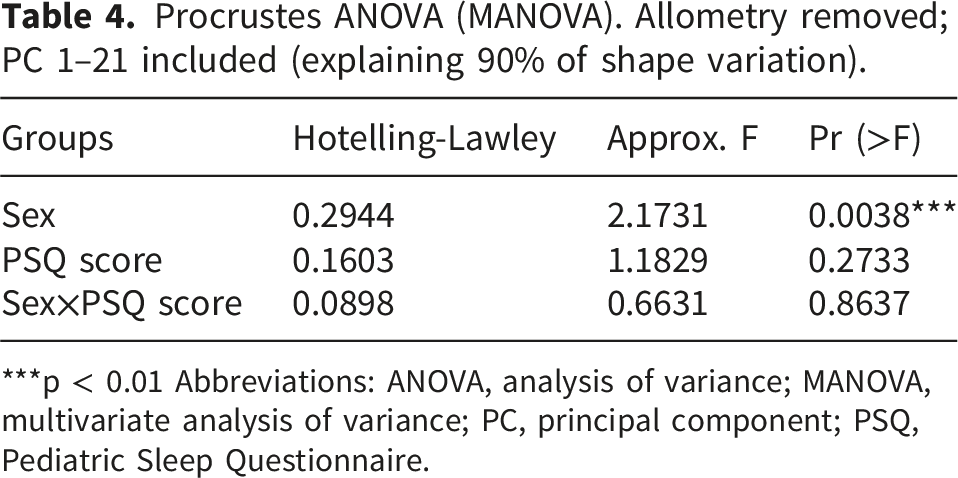
***p < 0.01 Abbreviations: ANOVA, analysis of variance; MANOVA, multivariate analysis of variance; PC, principal component; PSQ, Pediatric Sleep Questionnaire.
Discussion
The concept of sleep-disordered breathing playing a causal or predisposing role in craniofacial development remains controversial. Few studies utilizing three-dimensional imaging and investigating the relationship between pediatric SDB and facial morphology with geometric morphometrics currently exist. It was the aim of this study to utilize this relatively new imaging modality to aid in elucidating the relationship of pediatric SDB and facial morphology. This cross-sectional study included 180 young children from the Pediatric Otolaryngology Head and Neck Surgery outpatient clinic at a tertiary care hospital. Detailed assessment using both traditional and geometric morphometric analysis found no associations between three-dimensional facial form and symptoms of sleep-disordered breathing based on PSQ score.
The inclusion of children across a broad age range (2–17 years) was a deliberate feature of the study design, intended to maximize recruitment and reflect the realistic clinical population presenting to a tertiary pediatric otolaryngology service. However, this age span encompasses considerable craniofacial growth and developmental variation. Pre-school children (2–5 years), school-age children (6–11 years), and adolescents (12–17 years) differ substantially in facial proportions, skeletal development, and physiological determinants of SDB. These developmental differences may have introduced heterogeneity that masked subtle craniofacial differences associated with SDB risk. The authors acknowledge that age-stratified analyses (e.g., pre-school, school-age, and adolescent subgroups) would have been informative; however, the overall sample size of 180 would have resulted in underpowered subgroup analyses, and this is therefore identified as a limitation and a direction for future research. Furthermore, while chronological age and BMI were included as covariates, other markers of somatic growth—such as height or standing height—were not systematically collected and were therefore unavailable for analysis. Future studies should consider including height-derived metrics or standing height to better account for growth-related variability in facial morphology.
The 22-item PSQ has been used in multiple studies to examine associations between sleep-disordered breathing and facial morphometry18,19 or post-treatment outcomes. 20 This validated questionnaire allows an examination of the risk of sleep disorders without placing patients through the process of overnight polysomnography. We found 33.9% of our participants were considered “high-risk” for SDB based on their PSQ score. This is much higher than other epidemiological evaluations in this field. A study examining 1,038 patients from general pediatric clinics found a high-risk prevalence of 11.1% using the same cut-offs. 21 A likely explanation for this higher prevalence is the patient population originating from a tertiary Pediatric Otolaryngology clinic. This difference was anticipated in the study design; the population was specifically chosen to increase the likelihood of demonstrating a correlation between facial variables and SDB. By design, therefore, this recruitment source likely significantly overestimates the prevalence of sleep disorders in a general pediatric population.
Importantly, this study demonstrates that stereophotogrammetry systems can be practically employed in specialist outpatient clinics and may be a useful option for screening of craniofacial features. The system was well accepted by patients and their families; this study did not impede clinic flow, and it typically took under 15 minutes to fully consent patients and obtain images. In a real-life context, without requiring a non-clinician to obtain informed research consent, this process would be substantially quicker.
There are currently only a limited number of studies investigating 3D facial morphology and its relationship with pediatric SDB. Ali et al. conducted a longitudinal cohort study investigating the relationship between prevalence of SDB and facial morphology in 15-year-old Caucasian children utilizing 3D surface laser scans. 22 They reported differences in facial parameters between children with and without SDB, namely an increase in face height in SDB children of 0.3 mm, a decrease in mandibular prominence of 0.9 degrees, and a decrease in nose prominence and width of 0.12 mm and 0.72 mm, respectively. These differences are relatively small and, while perhaps statistically significant, their clinical significance is questionable. In a large cross-sectional study of 3,433 young children assessing associations with PSQ and dentofacial morphology, Li et al. 23 found no significant correlation between SDB risk and lateral facial profile, mandible plane angle, constricted dental arch form, or the severity of anterior overjet and overbite. In the present study, no such associations were found between similar parameters and risk of SDB as evidenced by PSQ score.
Similar trends are found when the 2D facial morphology of children with PSG-diagnosed OSA is investigated. Hsueh et al. 24 reported on 58 children with mean age 7.4 years who had completed an overnight sleep study and underwent facial photogrammetry. Craniofacial photogrammetry revealed an increased mandibular inclination of approximately 3 degrees in children with moderate to severe OSA, which may be of minimal clinical significance; none of the other 35 angular and linear variables assessed were related to OSA status. Yuen et al. 25 undertook a very similar protocol in 90 pre-pubertal children with OSA of varying severities and noted differences of 2 degrees in the maxillary-mandibular relationship angle between healthy children and those with severe OSA. In contrast to these earlier works, the effect size of any such associations may be so small that they were not demonstrable with the methodological parameters of the present study.
Critically, it should be emphasized that this study assessed external soft-tissue facial morphology only. SDB is a multifactorial disease, and subjects with higher risk of SDB may have differences in internal anatomy—such as reduced upper airway muscle tonicity, adenotonsillar hypertrophy, or an inherently collapsible upper airway—that are not detectable by 3D surface measurements. The absence of associations in the current study therefore does not preclude a meaningful relationship between SDB and craniofacial structures when assessed using internal imaging modalities such as MRI or CT. Future studies incorporating both external soft-tissue and internal airway morphology would provide a more complete characterization of the craniofacial phenotype in pediatric SDB.
The framing of the conclusion warrants clarification. Because the PSQ is itself symptom-based, a conclusion that “screening should be primarily based on signs and symptoms” could appear circular in this context. More precisely, the present findings indicate that in this tertiary clinic cohort, 3D facial soft-tissue measures did not add discriminatory value beyond the symptom information already captured by the PSQ. Therefore, facial morphology alone is unlikely to serve as a useful standalone screening tool in this clinical context, and the PSQ or equivalent symptom-based instruments should remain central to screening practice.
We did not find any association between age, sex, nor their interaction, with PSQ score. Some studies have suggested that males are at greater risk of SDB, 26 whereas others have not referenced any difference between the sexes. 27 Lumeng and Chervin propose that sex differences in SDB prevalence may not emerge until after puberty, when hormonal differences that are hypothesized to influence airway anatomy develop. 28 Ali et al. found that pediatric SDB was not significantly associated with sex, 22 consistent with the results of the current study. BMI was not associated with PSQ score in our study; however, it should be noted that the prevalence of obesity in the sample was low at less than 2%, and BMI data were available for only 80 of 180 participants, which may have limited statistical power to detect any association.
To detect potentially small or complex differences between groups, we employed GMM. GMM has advantages over traditional morphometrics in that the geometric relationships among variables are retained during the analysis, often allowing very subtle variation in shape to be captured. The results from Procrustes ANOVA suggest that the 21 principal components explaining 90% of the variation within the dataset do not indicate a significant influence of PSQ score on facial shape. However, sex does influence facial shape, consistent with published literature.29,30 The sexual dimorphism seen through our analysis lends credibility to the accuracy of the landmarking of the 3D facial images.
With regards to the cause and effect of craniofacial morphology and SDB, skeletal pattern and vertical facial growth tendency may be more so genetically determined, with environmental effects possibly exacerbating a pre-existing condition, if at all. While dentofacial morphology clearly influences airway function, the degree to which this occurs in otherwise healthy, non-syndromic children may be overestimated. As noted by others, 31 the complex pathophysiology underlying OSA may preclude effective screening based on facial morphology and symptoms alone.
There are several limitations to this study. Although the PSQ has been validated and is frequently used to identify pediatric sleep disorders, the gold standard for diagnosis of SDB and OSA is polysomnography, which was not performed in this preliminary study due to its resource- and time-consuming nature. The results from this study may not be generalizable to other settings as recruitment was from a single tertiary Pediatric Otolaryngology centre, which may overestimate the prevalence of SDB relative to the general population. A larger sample size, particularly for subgroup analyses, would have provided greater statistical power to detect small associations. No formal a priori sample size or power calculation was performed. Post-hoc estimation indicates that with n = 180 and a two-tailed α = 0.05, the study had approximately 80% power to detect Pearson correlations of |r| ≥ 0.21 between PSQ score and individual morphometric variables. Associations of smaller magnitude — which may nonetheless carry clinical relevance — could therefore have gone undetected, and the null results should be interpreted with this limitation in mind. The inclusion of a broad age range (2–17 years) introduces considerable developmental heterogeneity and may have masked subtle age-specific craniofacial differences; age-stratified analyses were not performed due to insufficient subgroup sample sizes but should be considered in future work. The absence of height measurements precluded the use of growth-stage markers beyond age and BMI. BMI data were available for only 80 of 180 participants, limiting the power of analyses involving this variable, though sensitivity analyses using the full cohort without BMI yielded consistent conclusions. Other potentially relevant contributors to SDB—such as tonsil size, nasal resistance, or upper airway muscle function—were not captured by 3D surface imaging and represent important directions for future research. The potential for self-selection bias and parental over- or under-reporting of symptoms on the PSQ may have resulted in inaccuracies in establishing SDB risk.
Conclusions
This study suggests that stereophotogrammetry may be successfully applied in the setting of a specialist pediatric outpatient clinic as a non-invasive and low-resource method for measuring 3D facial features, with good patient and family acceptance and minimal impact on clinic flow.
A pediatric patient aged 2–17 years at risk of SDB may not display obvious external craniofacial attributes. In this tertiary clinic cohort, 3D facial soft-tissue measures did not appear to add discriminatory value beyond symptom-based screening. These findings suggest that facial morphology alone may be insufficient as a standalone screening tool for pediatric SDB in this context, and that screening should remain primarily based on patient signs and symptoms. Internal anatomical factors—including adenotonsillar hypertrophy, upper airway collapsibility, and muscle tonicity—likely play a more direct role in SDB pathophysiology and warrant further investigation.
Footnotes
Acknowledgements
The authors thank the patients and families who participated in this study, and the clinical staff at BC Children’s Hospital Pediatric Otolaryngology clinic for their support. No artificial intelligence tools were used in the writing of this manuscript.
Ethical considerations
This research was conducted in accordance with the Declaration of Helsinki (1975, revised 2024) and was approved by the Research Ethics Board of the University of British Columbia and BC Children’s Hospital, Vancouver, Canada (ethics certificate number H17-01292, date of approval: June 26, 2017).
Consent to participate
Written informed consent was obtained from the legally authorized representative/parent/guardian of all participants, and assent was obtained from children above the age of 7. All patient details have been de-identified in accordance with applicable privacy regulations.
Author contributions
Felix Y.H. Wu: project conceptualization, data collection, manuscript review and editing. Alice Q. Liu: methodology, data collection, manuscript review and editing. Siddharth R. Vora: methodology, analysis, manuscript review and editing. Neil K. Chadha: project conceptualization, methodology, manuscript review and editing. Benjamin T. Pliska: project conceptualization, methodology, analysis, manuscript draft preparation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the British Columbia Children’s Hospital Research Institute and a Biomedical Research Award from the American Association of Orthodontists Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
