Abstract
Sleep disordered breathing (SDB), including obstructive sleep apnea (OSA) and central sleep apnea (CSA), is a common comorbidity of patients with cardiovascular diseases, including heart failure and aortic stenosis (AS). In specific, studies report a high prevalence of SDB among patients with severe AS, ranging from 34% to over 90%. The pathophysiological relationship between SDB and AS is considered to be bidirectional, as both conditions can initiate mechanisms such as circulatory delay, fluid shift theory or inflammation and oxidative stress that further contribute to this interplay. This interaction between SDB and AS raises questions concerning a possible combined therapeutic approach. Indeed, encouraging results have emerged regarding the beneficial effects of transcatheter aortic valve intervention (TAVI) on SDB in patients with AS, with TAVI being associated with a reduction in the prevalence and severity of SDB and improvements of the apnea-hypopnea index (AHI). Thus, the aim of this narrative review is to summarize the evidence linking SDB with AS, emphasizing the combined effects of TAVI on both conditions. The available evidence from observational studies indicates that TAVI is associated with significant reduction in the severity of SDB, particularly central sleep apnea, accompanied by improvements in the AHI and sleep parameters following the intervention, while obstructive events appear less responsive to valvular correction. These findings highlight the close pathophysiological interaction between cardiac hemodynamics and sleep regulation and suggest that treatment of the valvular pathology may provide benefits extending beyond cardiovascular improvement, with important clinical implications for the comprehensive management of these patients.
Keywords
1. Introduction
Sleep disordered breathing (SDB) refers to a group of different syndromes mainly consisting of obstructive sleep apnea (OSA) and central sleep apnea (CSA), where in the presence of either obstructed or unobstructed airflow, respectively, patients experience apnea or hypopnea episodes during sleep. 1 Regarding the prevalence of these pathologies, it is estimated that CSA represents 5-10% of all SDB, with the rest being OSA phenotypes. 2 Sleep apnea is common comorbidity in patients with cardiovascular disease. In particular, OSA represents an important risk factor for arterial hypertension, coronary artery disease, atrial fibrillation and other cardiovascular pathologies.3–10 On the other hand, CSA has been associated with congestive heart failure (CHF) and is accompanied by an unfavorable prognosis.
Valvular heart disease (VHD) is an independent risk factor for SDB, and particularly for CSA, as patients with valvulopathies tend to have a higher apnea-hypopnea index (AHI). 11 Aortic stenosis (AS) is the most common valvulopathy among elderly, and its prevalence is expected to further increase due to the aging population. AS and SDB frequently coexist, with their coexistence rates ranging from 34% to 90%,12–19 based on variations in patient age, testing method (portable polygraphy vs full polysomnography) and threshold used for AHI. Regarding patients undergoing transcatheter aortic valve implantation (TAVI), Linhart et al. 15 report a SDB prevalence of 71% (25% OSA, 46% CSA). Interestingly, in contrast to the general population where OSA predominates, patients with severe AS undergoing TAVI appear to exhibit a higher prevalence of CSA. This finding is likely related to the hemodynamic impairment and circulatory delay associated with advanced valvular disease, which promote instability of ventilatory control during sleep and favor central rather than obstructive respiratory events. This distinction is important for understanding the differential response of SDB phenotypes to aortic valve intervention, which is further discussed below. Given the negative effects of sleep apnea on left ventricular remodeling, the therapeutic implications between AS and SDB are of great importance in order to improve both cardiovascular as well as sleep physiology.15,20–22 Considering that recent studies in various non-structural heart diseases showcase that treating the underlying cardiac condition can improve breathing during sleep,15,23,24 this narrative review aims to summarize and critically discuss the available evidence regarding the coexistence of AS and SDB, as well as the impact of interventional management of AS on SDB. Moreover, this review will synthesize current knowledge on the pathophysiological interaction between these conditions and to evaluate the clinical implications of aortic valve intervention on sleep physiology.
2. Methods
For this narrative review, a structured literature search was conducted in June 2025 independently by two reviewers to identify available studies investigating the association between SDB and AS, as well as the impact of interventional management of AS on sleep parameters (Figure 1). The primary database used for the search was PubMed/MEDLINE. Additional screening of references from relevant articles was performed to identify further eligible studies, as well as identify suitable studies for the documenting the pathophysiology and clinical association of both conditions. The search was performed using combinations of the following keywords and Medical Subject Headings (MeSH) terms: “aortic stenosis” AND “sleep apnea”, “aortic stenosis” AND “sleep disordered breathing”, “TAVI” OR “TAVR” AND “sleep apnea”, “transcatheter aortic valve implantation” AND “sleep”, “valve replacement” AND “central sleep apnea”, “valvular heart disease” AND “sleep apnea”. No restrictions regarding publication date were applied. Only articles published in English were considered. For describing studies that assessed the effect of TAVI in SDB, the following criteria were used: 1) original clinical studies evaluating patients with AS or VHD and documented SDB, 2) studies assessing the effect of transcatheter or surgical aortic valve intervention on sleep parameters (e.g., AHI, SDB phenotype, sleep quality), 3) observational studies (prospective or retrospective), cohort studies, and case series, 4) studies reporting objective sleep assessment through polygraphy or polysomnography. Studies referring to the management of SDB in other cardiovascular pathologies, the effect of SDB treatment in cardiovascular disease, the interaction and common pathophysiology behind AS and SDB were also identified based on the aforementioned search and also through identifying suitable articles using the snowballing method. The search strategy yielded a limited number of studies directly addressing the interaction between AS, SDB, and aortic valve intervention (five studies). This narrative review was conducted and revised taking into consideration the quality principles described in the Scale for the Assessment of narrative review articles (SANRA).
25
Flow diagram of the literature search and study selection process.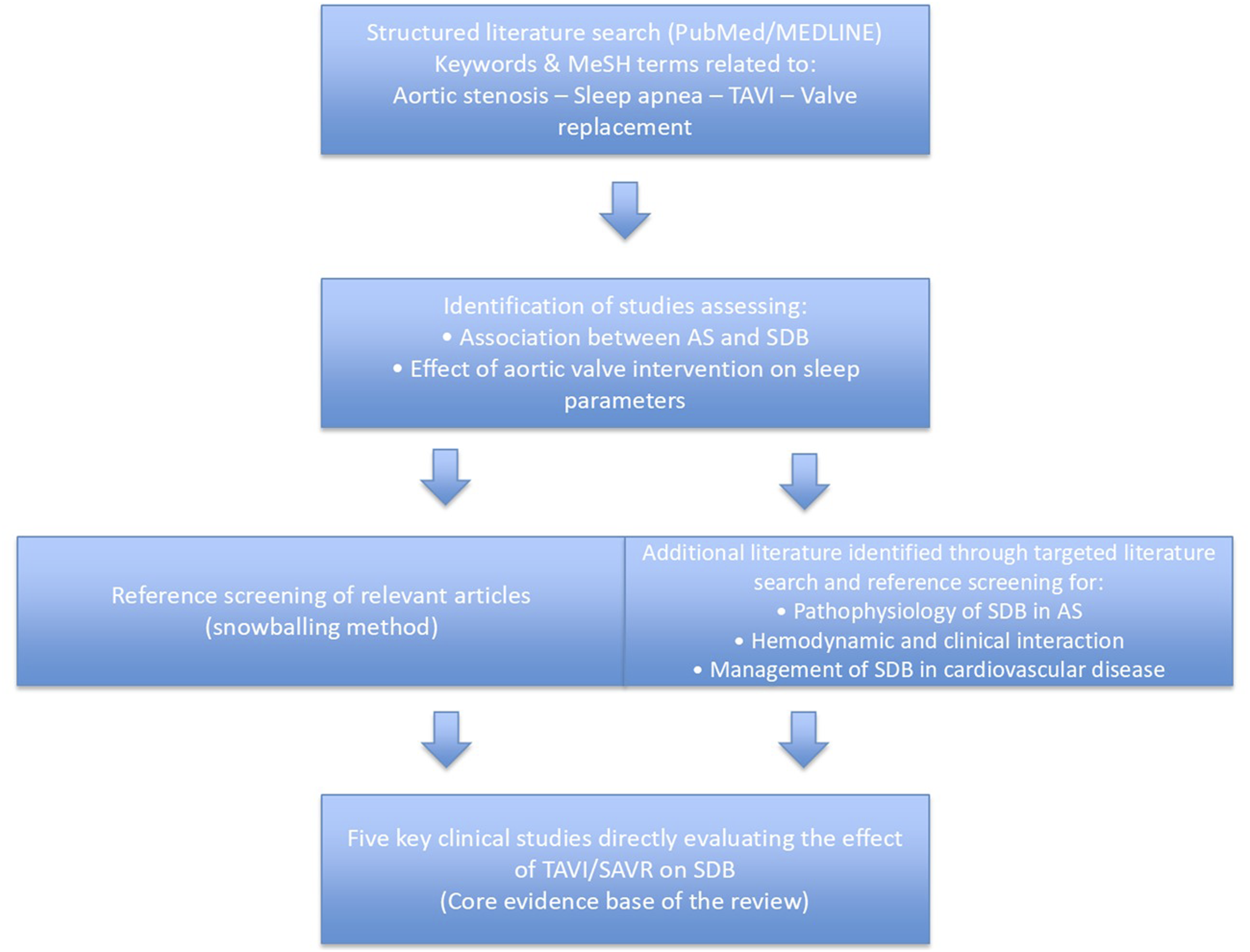
3. Pathophysiology: Circulatory delay & fluid shift
AS is characterized by limited systemic blood flow during systole due to a stenotic aortic valve, leading to pressure overload. With the progression of the disease, the physiological adjusting mechanisms of left ventricular hypertrophy and dilation that aim to support systemic circulation fail to maintain the proper, onward direction of the cardiac output, which are followed by a reduction of the cardiac output and backward increase of pressure in the left atrium and the pulmonary vasculature. 26 These events lead to pulmonary congestion and a subsequent imbalance in gas exchange (hypoxemia) that, in combination with repetitive stimuli arising from the vagal juxtacapillary receptors, gradually cause hyperventilation. Subsequently, the arterial partial pressure of carbon dioxide (CO2) falls below the apnea threshold, resulting in cessation of breathing and, thus, apneic episodes. An effective feedback system restores breathing when the arterial partial pressure of CO2 returns to the eupneic threshold. These mechanisms are particularly present, and even overactive, in chronic conditions such as heart failure, leading to the development of hypopneic or apneic episodes during sleep in such patients and therefore the clinical manifestation of sleep apnea.
Another mechanism contributing to hypopnea in patients with AS and heart failure is the prolongation of the circulation time. In particular, the duration of the ventilation period is determined by the time required for the blood to reach the peripheral chemoreceptors after passing through the pulmonary circulation, known as circulation time. Normally, chemoreceptors in the carotid bodies and brainstem sense blood CO2 and O2 levels timely to adjust breathing frequency. However, patients with severe AS exhibit prolonged circulation time as a consequence of reduced cardiac output, defined as circulatory delay,27–32 leading to delayed chemoreceptor sensing of blood gas composition changes. In parallel, the feedback system restoring breathing after apneic episodes is also deferred. This theory may better explain the relationship between the duration of the respiratory cycle and the severity of heart failure in patients with CSA. 33 Furthermore, in this setting, there is also an increased chemosensitivity to CO2, triggering excessive breathing, with the recurrent drop of the arterial partial pressure of CO2 below the apnea threshold perpetuating this vicious cycle.27–30 Studies have also emphasized on the connection between circulatory delay, apnea duration, and ventilation duration, indicating that a progressively less effective feedback system may result in longer cycle durations and thus prolonged episodes. 31 Interestingly, this theory has differences among SDB phenotypes. In OSA, the duration of the episodes is also related to pulmonary congestion. The longer circulation time causes delayed control of the blood gas partial pressures, resulting in longer respiratory cycles. Moreover, in CSA, the severity of heart failure has been shown to correlate with both apnea duration and the overall length of the respiratory cycle. In contrast, in OSA, heart failure severity appears to be associated with prolongation of the respiratory cycle and the ventilation phase, but not with the duration of the obstructive apnea itself. 34
Other theories explaining the pathogenesis of SDB, such as the fluid shift theory, can also be related to cardiac function and its deterioration, particularly in patients with established valvulopathies. In specific, the shift of fluids from the lower extremities to the neck during sleep may cause or aggravate upper airway obstruction and exacerbate pulmonary congestion. The resulting peripharyngeal edema and increased pharyngeal collapsibility lead to a higher likelihood of OSA and hypopnea episodes.35–39 Moreover, the negative intrathoracic pressure observed during OSA episodes increases venous return (i.e. right ventricular preload), while pulmonary vasoconstriction due to hypoxemia lead to right ventricular afterload increase. Consequently, the right ventricle dilates, resulting in leftward displacement of the interventricular septum during diastole and difficulty in left ventricular filling. This combination leads to a reduction in stroke volume and cardiac output, with a simultaneous decrease in coronary blood flow and oxygen delivery. Simultaneously, the present negative intrathoracic pressures in such episodes further increase left ventricular wall stress causing an increase in its afterload and a rise in myocardial oxygen consumption. These changes, when chronic, compromise myocardial function and contractility and frame a condition that, without timely diagnosis and treatment, leads to myocardial remodeling and eventually to heart failure40–43 (Figure 2). Pathophysiological interaction between aortic stenosis and sleep-disordered breathing.
Inflammation and oxidative stress also appear to play a crucial role in the relationship between cardiovascular disease and SDB. These mechanisms are well-established risk factors of cardiovascular disease, including AS.44–46 However, recent evidence also support their role in sleep apnea, characterizing SDB as a low-grade chronic inflammatory disease, 47 particularly in relation to tumor necrosis factor-alpha (TNF-a) and interleukin-6 (IL-6) expression. More specifically, particularly in OSA, TNF-α expression is upregulated due to sleep fragmentation and chronic intermittent hypoxia through the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathway, contributing to excessive sleepiness, cognitive deficits, and mood disturbances.48–51 Similarly, IL-6 release is also promoted, with studies demonstrating that adults with OSA and excessive daytime sleepiness exhibit higher IL-6 levels, which generally decrease following continuous positive airway pressure (CPAP) therapy. 47 Finally, hypoxia per se during the apnea episodes increases formation of oxygen free radicals, resulting in pro-inflammatory environment.52–57 Therefore, inflammation is a shared risk factor in cardiovascular and sleep disorders that may explain the increased rates of coexistence among these pathologies, as well as propose a new therapeutic target that, through inflammatory system inhibition, improves both sleep and cardiovascular physiology.
Finally, repeated apnea and hypopnea episodes, combined with arterial oxygen desaturation and hypercapnia, lead to activation of the sympathetic nervous system. This sympathetic overactivity contributes to episodes of arterial hypertension and tachyarrhythmias. Conversely, increased vagal tone, triggered by pulmonary congestion through stimulation of the juxtacapillary receptors, may result in episodes of bradyarrhythmias and hypotension.58,59
4. Combining treatment: Does it work?
4.1. The role of CPAP in cardiovascular disease and implications for AS
The use of CPAP in patients with AS could hold significant therapeutic potential for both SDB and AS, considering its role in altering proper oxygenation during sleep and altering cardiovascular hemodynamics. It has been described that, especially for CSA, pressure overload due to an existing valvular or myocardial pathology backwards increases pulmonary pressures and propagates pulmonary congestion. Importantly, investigators have identified a correlation, particularly for CSA, with the development of pulmonary hypertension due to cardiac maladaptation to increased pressures, which is associated with increased pulmonary capillary wedge pressure and lower partial pressure of CO2 value, thus setting the link between pulmonary congestion and CSA as explained by arterial hypocapnia. 19 Despite CPAP could, therefore, alleviate the poor oxygenation parameter and improve sleep breathing, the hemodynamics of AS should be taken into consideration when applying a positive airway treatment. As positive airway therapy would expectedly increase intrathoracic pressure, it would therefore impact preload, afterload and left ventricular wall stress. To start with, an increased intrathoracic pressure would increase venous resting pressure and subsequently reduce venous return and left ventricular preload. This change could alter cardiac output, especially considering that it is preload-dependent in patients with a AS and thus worsen systemic perfusion. Concurrently, positive airway treatment could decrease LV transmural pressure and therefore LV afterload and myocardial wall stress, which could improve myocardial energetics and decrease oxygen consumption. However, these alterations have unknown clinical importance, to date, especially in patients with decompensated AS, which would be more sensitive to acute alterations of hemodynamic status. Therefore, in the absence of studies exploring the effect of such therapies on the hemodynamics and clinical outcomes of AS, the use of positive airway treatment in such patients, despite holding potential, should be cautiously treated taking into consideration that small changes of extracardiac pressures could result in clinically meaningful changes of intracardiac hemodynamics, with either beneficial or not patient outcomes, depending on each patients physiology, comorbidities and homeostasis.
Multiple studies have analyzed the effects of CPAP therapy, commonly used in patients with SDB, on cardiac patients with the resulting data being conflicting.60–62 The conflicting results of CPAP therapy in cardiovascular populations may be partly explained by differences in patient selection, adherence to therapy, and the underlying pathophysiological substrate of the disease. While OSA is primarily a disorder of upper airway collapsibility, CSA is more closely related to cardiac performance and ventilatory control instability. Consequently, the response to positive airway pressure may vary significantly depending on whether the predominant mechanism is obstructive or central.
Notably, randomized studies in patients with coronary artery disease or acute coronary syndromes did not report any significant benefit of routine CPAP prescription in patients with SDB), with the exception of the RICCADSA trial, that reported a benefit in a sub-analysis following adjustment for baseline comorbidities and compliance with treatment.60–62
On the other hand, in patients more similar to AS patients, i.e. heart failure patients without, however, valvular heart disease, the CANPAP trial showed benefit in 258 patients with HF and CSA using CPAP. In specific, in patients with severe central sleep apnea and heart failure, positive airway treatment resulted in significant reductions of apnea and hypopnea episodes (-21+/-16 vs. -2+/-18 per hour, P<0.001) and norepinephrine levels (-1.03+/-1.84 vs. 0.02+/-0.99 nmol per liter, P=0.009), in parallel with significant increases in left ventricular ejection fraction (LVEF) (2.2+/-5.4 % vs. 0.4+/-5.3 %; P=0.02) and six-minute walking distance (20.0+/-55 vs. -0.8+/-64.8 m, P=0.016). 63 Similar results have been found by other investigators in HF patients with CPAP treatment, noting a more marked improvement in patients with a LVEF>30%. 64
Unfortunately, as aforementioned, until today, no studies have directly assessed the effect of CPAP devices on AS hemodynamics or patient outcomes, therefore limiting the interpretation of data extrapolated from other pathologies into the AS phenotype. In contrast, the interventional management of AS seems to have a positive effect on SDB. TAVI, developed over twenty years ago, is a technique applied in high surgical risk patients with severe AS. It is a minimally invasive procedure that can significantly and relatively promptly improve the patient’s hemodynamic status. 65 The effectiveness of TAVI is now unquestionably documented and validated by a large number of studies, even in other phenotypes of patients such as lower-risk patients, those with asymptomatic severe AS or patients with acute heart failure, where data also show benefit.66–71 Therefore, TAVI can be safely used in patients over 70 years of age, in patients at high surgical risk or deemed unsuitable for surgery, as well as in patients with risk factors not considered in conventional scoring systems.72,73
4.2. The effect of aortic valve intervention on SDB
Studies assessing the effects of transcatheter aortic valve implantation and surgical aortic valve replacement in sleep disordered breathing.

AHI: apnea-hypopnea index, TAVI = transcatheter aortic valve implantation, ESS: Epworth sleepiness scale, SpO2: oxygen saturation, HR: heart rate, AS: aortic stenosis, SDB: sleep disordered breathing, CSA: central sleep apnea, OSA: obstructive sleep apnea, LVEDP: left ventricle end diastolic pressure, SPAP: systolic pulmonary artery pressure, PSQI: Pittsburgh sleep quality index, EQ-5D-5L: EuroQol 5-Dimension 5-Level questionnaire, SAVR: surgical aortic valve replacement, HF: heart failure, VHD: valvular heart disease, EEG = electroencephalography, EOG = electrooculography, EMG = electromyography.
Dimitriadis et al. 16 also included 79 patients undergoing polygraphy before TAVI, with the follow-up being completed by 62 of them undergoing a second polygraphy 21 days after the procedure. Among these patients, 58% had OSA and 36% CSA. The AHI was significantly higher among CSA patients. Similarly with the previous study, CSA patients with optimal procedural results had a significant reduction in central respiratory events (AHI 39.6 ± 19.6-23.1 ± 16.0/h, p = 0.035), while no changes were detected in OSA (AHI 18.8 ± 13.0-20.25 ± 13.4/h, p = 0.376). Importantly, the investigators also showed that moderate to severe post-procedural regurgitation was associated with the development of CSA in patients without prior SDB or in patients with isolated OSA, along with worsening of CSA episodes in patients with prior history, therefore showcasing the effect of suboptimal hemodynamics even after intervention in sleep breathing regulation. As with other studies in this field, the AHI cut-off values used for SDB classification (Table 1) should be considered when interpreting these findings, as differences in diagnostic thresholds may affect both the reported prevalence and the apparent response to intervention.
Another study by Linhart et al. 15 aimed to evaluate the impact of pre-TAVI SDB in clinical outcomes. In this study, 140 patients underwent polygraphy and were assessed using the Epworth sleepiness scale (ESS) prior to TAVI. Ninety-nine patients had SDB (27% mild, 23% moderate, 21% severe), of which 64 (46%) had CSA and 35 (25%) had OSA, with SDB phenotype being independent of left ventricular function. Patients with OSA had predominantly mild SDB and patients with CSA mostly had severe SDB. The authors showed that 1- and 2-year survival rates (74.4% and 71.3%, respectively) did not differ significantly among patients without SDB or those with OSA and CSA (p = 0.81), with no difference being also noted based on the severity of each SDB. This outcome comes in contrast to the well-established negative effect of SDB in non-valvular cardiac disease; however, it indicates that, despite the improvement of SDB post-TAVI, with the available data SDB cannot be considered as an independent risk factor for adverse events in AS.
Despite no apparent effect on mortality being found, Lorenzoni et al. 74 aimed to analyze the effect of TAVI on sleep quality and quality of life using the Pittsburgh Sleep Quality Index (PSQI) and the EuroQoL (EQ-5D-5L) instruments, respectively. Twenty-seven patients completed the questionnaires on the day of discharge and one month later during follow-up. Patients were categorized according to the PSQI score, where a global PSQI score ≤5 defined “good sleepers” and >5 defined “poor sleepers”. At discharge, seventeen patients were classified as “poor sleepers” and ten as “good sleepers” based on these objective criteria. The global PSQI evaluation revealed a significant improvement at follow-up (p = 0.007), although a high incidence of postprocedural insomnia was noted. Small positive changes were detected in the Self-care and Usual activity domains of the EQ-5D-5L, while no correlation was observed between EQ-5D-5L scores and sleep quality. This study was limited by the small sample size and the lack of preprocedural sleep assessment.
Finally, the impact of surgical aortic valve replacement (SAVR) on SDB has only been roughly delineated by Abe et al. 21 In their observational study, 54 received surgical replacement of the aortic valve either for AS or aortic regurgitation (41 patients underwent SAVR, while 13 received aortic and mitral valve surgery). Polysomnography was used for this study before and after the procedure (during follow-up). Interestingly, following surgical treatment, the AHI was significantly reduced (40.3±14.4 vs 31.9±21.1, p = 0.002), especially for CSA (9.0±12.0 vs 1.0±2.9, p<0.001), but not for OSA (11.0±9.2 vs 11.9±12.1, p = 0.712). Unfortunately, the study was limited by the brief and variable follow-up period, performed 14.6±6.5 days after the procedure, and the inclusion of patients with various and mixed types of VHD undergoing different surgical procedures.
To summarize, a consistent finding across the available studies is that while the central component of SDB shows marked improvement following aortic valve intervention, the obstructive component remains largely unchanged. This observation is particularly important from a pathophysiological perspective. It suggests that OSA may often be a pre-existing condition related to upper airway mechanics, whereas the central component develops or worsens with the progression of AS and cardiac dysfunction. In this context, the increasing predominance of CSA in patients requiring TAVI may be viewed as a manifestation of advanced hemodynamic impairment and circulatory delay. Therefore, correction of the underlying structural cardiac disease, rather than isolated treatment of sleep apnea, can represent a fundamental approach to restoring ventilatory stability during sleep.
5. Discussion and future perspectives
SDB is a well-established risk factor for cardiovascular disease and increased mortality compared to patients with a normal breathing pattern, 75 while as described, it presents with high prevalence among patients with severe AS, with or without heart failure. 15 The effect of SDB treatment in patients with cardiovascular disease is complex, despite established therapies. In OSA, the use of CPAP may improve left ventricular ejection fraction, sympathetic nervous system hyperactivation, and blood pressure control, while also reducing cardiovascular events, and has been recommended in the 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. 76 However, its true clinical benefit remains debatable, especially considering the suboptimal patient adherence, with non-compliance rates ranging between 29–83%, 77 which has been also reported in the large, randomized studies of CPAP use in cardiovascular disease. On the other hand, therapeutic strategies for patients with CSA remain challenging, in the presence of conflicting data, with studies reporting both benefit and deterioration of outcomes in patients with CSA and reduced ejection fraction.78,79 These observations are mainly influenced by the results of the SERVE-HF study, that showed an increase of all-cause and cardiovascular mortality with adaptive servo-ventilation (ASV) therapy. 79 Importantly, the CANPAP study, while also showing no mortality benefit in patients using CPAP, identified a transplant-free survival benefit in patient with CSA in whom the AHI fell below 15 events/hour while on CPAP. 80 Moreover, using ASV, the ADVENT-HF study showed no harm in patients using the treatment, as ASV had no effect on the primary composite outcome of all-cause mortality, first admission to hospital for a cardiovascular reason, new onset atrial fibrillation or flutter, and delivery of an appropriate cardioverter-defibrillator shock (hazard ratio [HR] 0.95, 95% CI 0.77-1.18; p=0.67) or the secondary endpoint of all-cause mortality (88 deaths in the control group vs. 76 in the ASV group; 0.89, 0.66-1.21; p=0·47), despite improving sleep parameters. 81 Similar results have been also found by smaller studies.82–85 Therefore, based on the previously described results, it is crucial to not only focus on the treatment of SDB, but also analyze the underlying cardiovascular pathology and physiology in order to properly apply treatment strategies that are both safe and effective for this population.
Analyzing the treatment of SDB under the spectrum of the underlying cardiovascular pathologies and, thus, the appropriate cardiovascular interventions for each is crucial, especially for patients with CSA. As established thus far, despite OSA can be a pre-existing to the cardiovascular pathology comorbidity, CSA is largely influenced by cardiovascular hemodynamics and as described by AS management. Supporting this observation are most of the aforementioned studies that noted an improvement of CSA episodes after both surgical and transcatheter aortic valve replacement, but not a similar improvement for OSA. Therefore, for patients with SDB, considering the cardiovascular comorbidities that add to their pathophysiology along with oxygenation treatment is important, as alleviating an important pathophysiological contributor to CSA (in this case, AS) by using available treatment (i.e. TAVI) could further enhance patient outcomes and improve symptom management, on top of sleep apnea directed treatment, for both the cardiovascular as well as the sleep disorder.
Despite the low number of studies performed so far in this context, the existing data provide promising information regarding the beneficial role of interventions improving cardiac hemodynamics and function in extracardiac comorbidities. The exact mechanisms of such effects remain unclear; however, improved hemodynamics and alleviations of increased pressures may explain such benefits. Namely, as shown by Linhart et al. 12 a strong correlation was present between the immediate hemodynamic improvement of patients and the LVEDP, which could be further investigated as a marker for its potential effect on SDB. It is therefore possible that the overall successful management of heart failure and its valvular comorbidities in these patients led to an improvement of their respiratory profile; however, it is unclear whether the subsequent remission of sleep apnea episodes was a direct result of the intervention or due to an overall improvement of patients’ cardiovascular status.
This narrative review summarizes the limited but consistent body of evidence investigating the interaction between AS and SDB, and particularly the effects of aortic valve intervention on sleep physiology. Compared with previous literature focusing primarily on the coexistence of SDB and cardiovascular disease, this review emphasizes the distinct pathophysiological mechanisms linking AS with both OSA and CSA and highlights how correction of valvular hemodynamics may differentially affect SDB phenotypes. By integrating data from studies using both polygraphy and full polysomnography, this review also underscores how differences in diagnostic modality and AHI thresholds influence the reported prevalence and severity of SDB, an aspect often overlooked in earlier reports. As the isolated effect of interventional therapy in patients with AS and SDB remains to be determined, further research and studies are necessary, in order to provide data that will help to better understand the impact of transcatheter and surgical interventions for AS on sleep parameters, as well as on the modification of the obstructive or CSA phenotype. Moreover, the phenotype of patients with SDB who may benefit the most from these interventions should be more highlighted, along with evidence showing benefits on adverse events, survival or quality of life following the improvement of both cardiac and sleep conditions. Such results will provide new information about the impact of aortic valve replacement on sleep physiology and could even contribute to the expansion of indications for aortic interventions, especially in patients with pre-existing AS and severe SDB that could directly benefit from the intervention.
This review has several limitations. First, the available evidence is derived from a small number of observational studies with limited sample sizes and heterogeneous follow-up protocols. Second, variability in diagnostic methods (polygraphy versus polysomnography) and differences in AHI thresholds across studies may affect the comparability of results. Third, the narrative nature of this review does not allow for quantitative synthesis of data. Finally, most studies focus on short-term changes in sleep parameters after valve intervention, while long-term clinical implications remain insufficiently explored.
6. Conclusion
SDB is highly prevalent among patients with severe aortic stenosis, with a predominance of CSA compared to the general population. Interventional treatment of AS, particularly with TAVI, is associated with a significant reduction in the severity of SDB, mainly affecting central events, while obstructive events appear less responsive to valvular correction. Thus, improvement of valvular hemodynamics may lead to meaningful changes in sleep physiology, extending the benefits of aortic valve intervention beyond traditional cardiovascular outcomes. Further prospective studies are warranted to clarify patient profiles most likely to benefit, the optimal timing of intervention, and the potential impact on long-term endpoints and quality of life.
Footnotes
Acknowledgments
The authors acknowledge the use of AI-assisted tools exclusively for English language editing and proofreading.
Author contributions
Conceptualization: A.P., K.D.; Literature search and data curation: A.P., E.A., N.P., E.B., E.D.; Writing – original draft preparation: A.P., E.A., N.P., K.M., K.C., G.T.; Writing – review and editing: A.P., E.A., N.P., D.K., N.B, K.A.; Supervision: K.D., K.T. All authors have read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
