Abstract
Objective
Since late 2023, human parvovirus B19 (B19V) has been erupting continuously in multiple countries. Although transfusion-transmitted infections remain rarely documented, donations harbouring high-titre, undetected virus could still breach current pathogen-reduction processes and pose a potential threat to the safety of blood and blood products. To evaluate local risk, this study first determines the immunoglobulin G (IgG) seroprevalence of B19V and its geographic distribution among voluntary blood donors in Shaanxi Province.
Methods
In this cross-sectional study, from August to December 2024, an enzyme-linked immunosorbent assay was used to detect the IgG antibody against B19V in sampled batches of qualified blood samples from five municipal central blood stations in Shaanxi Province, and the distribution characteristics of samples were analysed from multiple perspectives.
Results
In Shaanxi Province, the B19V IgG antibody positivity rate was 24.95% (533 out of 2136), and increased with age (p < 0.001). Positive cases showed significant spatial clustering (p < 0.001), with a higher number of positive cases in Central Shaanxi and a lower number of positive cases in Northern and Southern Shaanxi. The infection risks were significantly different between northern and southern regions (odds ratio [OR]: 0.592, 95% confidence interval [CI]: 0.448–0.782, p < 0.001). Compared to type A blood, individuals with type B blood have a lower risk of previous infection (OR: 0.718, 95% CI: 0.549–0.940, p < 0.05).
Conclusions
The previous infection rate of B19V is high among blood donors in Shaanxi Province, influenced by age, blood type, and geography. Given that B19V screening is not yet part of routine donor testing in China, we recommend that blood or blood components intended for immunocompromised, high-risk recipients be tested for B19V before transfusion, so as to reduce the risk of transfusion-transmitted infection in an economical and efficient manner. Future studies should focus on the long-term dynamics of B19V infection and the effectiveness of screening programs in reducing transmission risks.
Keywords
Introduction
Human parvovirus B19 (B19V) is the only known small single-stranded linear DNA virus that infects humans within the genus Erythrovirus of the Parvovirinae subfamily. Respiratory droplets are the primary mode of transmission, but the virus can also be transmitted through blood transfusions or blood products. It is widely distributed worldwide. 1 The clinical manifestations of B19V infection are closely related to the host's age, haematological status, and immune condition. Healthy individuals often exhibit asymptomatic or mild, nonspecific symptoms after infection, including erythema infectiosum (fifth disease) in children and arthritis in adults. However, in immunocompromised individuals, B19V infection may lead to viral reactivation and cause severe complications. For instance, immunodeficient patients and organ transplant recipients are at risk of developing chronic anaemia. Patients with blood disorders, such as sickle cell anaemia, may experience an aplastic crisis, resulting in acute anaemia. Infection during pregnancy may lead to foetal hydrops or even foetal demise. Additionally, certain cases of nephropathy, hepatitis, neurological diseases, myocardial injury, and some autoimmune diseases have also been associated with B19V infection.2–6
An asymptomatic blood donor during the acute phase of infection may have a B19V viral load in their blood ranging from 1011 to 1014 copies/ml, which is sufficient to highly contaminate a pooled blood supply. 7 Moreover, although modern plasma-derived products are manufactured under stringent virus-reduction protocols, B19V’s minute, non-enveloped structure endows it with exceptional resistance to filtration, inactivation, and pasteurization. Should a high-titre donation go undetected or manufacturing deviations bypass multiple clearance steps, the virus can still break through and cause transmission.8–10 Therefore, international organizations and governments are deeply concerned about the safety of virus-contaminated blood products. Although B19V is not currently a mandatory screening target in blood establishments in most countries, the World Health Organization 11 has, since 2005, recommended in its Recommendations for the Production, Control and Regulation of Human Plasma for Fractionation that source plasma be screened for B19V DNA by nucleic-acid testing. Subsequently, studies have shown that B19V infection is typically not triggered when the viral load is below 105 IU/ml. Therefore, regulatory agencies such as the US Food and Drug Administration, 12 the European Medicines Agency, 13 and the Plasma Protein Therapeutics Association 14 issued policies or adjusted standards in 2009, 2011, and 2013, respectively, to recommend B19V DNA testing for pooled plasma and to require that the viral load should not exceed 104 IU/ml.15–18
Since late 2023, significant and continuous outbreaks of B19V have been reported across Europe, including in the United Kingdom. These outbreaks represent a serious clinical threat to immunocompromised individuals and pregnant women.19–21 Therefore, it is particularly important to be alert to the potential harm of this phenomenon to blood and blood products, and to quickly monitor its epidemic situation and regional distribution in China. 22 While certain regions in China have initiated screening for B19V antibodies or nucleic acids, there is a lack of pertinent documentation and technical guidelines for parvovirus surveillance at the national level. Additionally, essential data on the rates of antibody and nucleic acid positivity in different regions are currently inadequate.
Based on this, this study aims to provide both empirical data and a theoretical framework for targeted blood-screening strategies. Enzyme-linked immunosorbent assay (ELISA) was first used to determine the prevalence of B19V IgG antibodies in Shaanxi Province, which is located in the hinterland of China and is an important transportation hub in Northwest China. The distribution characteristics of the samples were analysed from multiple perspectives, including age, sex, education level, occupation, ABO blood type, and spatial distribution.
Materials and methods
area overview
Located in northwest China, Shaanxi Province straddles the Yellow and Yangtze River basins and covers 205,600 km2. At the end of 2023, its permanent population was about 39.52 million. The terrain is high in the north and south and low in the middle. The Beishan upland and the Qinling Range divide the province into three natural regions: the loess plateau in the north (40% of the land, 8.22 out of 20.56), the Guanzhong Plain in the centre (24%, 4.94 out of 20.56), and the Qinling–Daba mountains in the south (36%, 7.40 out of 20.56). 23
Study samples
This was a cross-sectional survey of voluntary blood donors from five municipal central blood stations in Shaanxi Province (Xi’an, Ankang, Hanzhong, Yan’an, and Yulin) conducted from August to December 2024. The detailed inclusion and exclusion criteria are as follows:
Inclusion criteria: (1) Being in good health and meeting the health standards for blood donors as specified in the Health Examination Requirements for Blood Donors (GB18467-2011) 24 ; (2) having complete basic information, including donor code, blood collection date, blood collection address, date of birth, sex, occupation, education level, and ABO blood type; (3) donating whole blood.
Exclusion criteria: The exclusion criteria included individuals with unqualified test results for transfusion-transmitted disease markers, the detection items included alanine aminotransferase (ALT), hepatitis B virus DNA (HBV DNA), hepatitis B surface antigen (HBsAg), hepatitis C virus RNA (HCV RNA), antibody to hepatitis C virus (anti-HCV), human immunodeficiency virus RNA (HIV RNA), HIV antigen/antibody, and antibody to Treponema pallidum (anti-TP).
Conducted in accordance with the Helsinki Declaration (1975, as revised in 2024), this study received approval from the Ethics Committee of Xi’an Central Blood Station (approval no. 202507, dated 11 April 2025). Immediately after collection, all data were de-identified by removing names, ID numbers, and donation codes; only anonymous serial numbers were retained, precluding any possibility of individual identification. Written informed consent was obtained from every participating blood donor. The reporting of this study conforms to Strengthening The Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 25
Reagents and instruments
We collected 5 ml of venous blood from each blood donor and stored it in EDTA-K2 anticoagulant tubes. After centrifugation, the plasma was used to detect markers of transfusion-related infectious diseases. Specifically, we assessed the levels of ALT using the fully automated biochemical analyser (Suzhou Beckman Coulter AU680, Lot No. AUZ3075). We detected HBV DNA, HCV RNA, and HIV RNA using the STAR automated nucleic acid extractor and ABI 7500 PCR instrument (Suzhou Huayimei, Lot No. MA20240502). We also tested for HBsAg (Solin Italy, Lot No. E056910; Shanghai Kehua, Lot No. 202402051), anti-HCV (Solin Italy, Lot No. E071310; Zhuhai Lizhu, Lot No. 202404081), HIV Ag (Beijing Wantai, Lot No. H20240508B; Shanghai Kehua, Lot No. 202405041), and anti-TP (Xiamen Xinchuang, Lot No. 2024057513; Beijing Wantai, Lot No. N20240514B) using the fully automated ELISA workstation (Shenzhen Aikang Uranus 368, Yantai Osborne FAME24/20).
The eligible samples, post the aforementioned tests, underwent ELISA detection for B19V IgG antibody using the Beijing Beier Company kit (Lot No. 20240701). The procedures strictly adhered to the guidelines provided by the manufacturer for the instrument and kit. The microplate reader (LabServ K3 Touch) was set to 450/630 nm; when quality-control criteria were met, samples with an absorbance (optical density) ≥ the kit's cut-off value were considered positive. As reported by the manufacturer, this assay exhibits 100% sensitivity and specificity (positive and negative concordance 10 out of 10 against corporate reference panels), precision of coefficient of variance ≤ 15%, and clinical agreement of 99.62% (vs. CE-marked/China-approved reference ELISA, n = 1050), with no cross-reactivity to common viral pathogens.
Statistical analysis
We performed statistical analysis on the distribution characteristics of the samples based on age, sex, education level, occupation, ABO blood type, and regional information using SPSS 27.0 software. Non-normally distributed measurement data were presented as the median (M) with interquartile range (Q1, Q3), while count data were expressed as the number of cases or frequency (%). Group comparisons were made using the χ2 test, and the Kruskal–Wallis test was applied to ordinal data. Logistic regression analysis identified factors influencing B19V infection. The significance level was set at α = 0.05, with p < 0.05 indicating a significant difference. Forest plots were generated using GraphPad Prism 10.1.2 software.
Additionally, using ArcGIS 10.8 and GeoDa 0.95i software, we analysed the spatial correlation of B19V-positive cases in Northern, Central, and Southern Shaanxi based on blood collection addresses and created maps. The Local Indicators of Spatial Association (LISA) cluster map intuitively displays the spatial distribution of B19V-positive donors and their relationships with surrounding areas. The LISA significance map highlights regions with statistically significant clustering.
Results
Overall positivity rate and baseline analysis in Shaanxi donors
The study included 2136 qualified blood samples, with the sample inclusion process shown in Figure 1. The participants comprised 1506 males (70.51%) and 630 females (29.49%), with ages ranging from 18 to 59 years. The median age was 35 years, with the interquartile range (Q1, Q3) being 26 to 44 years.
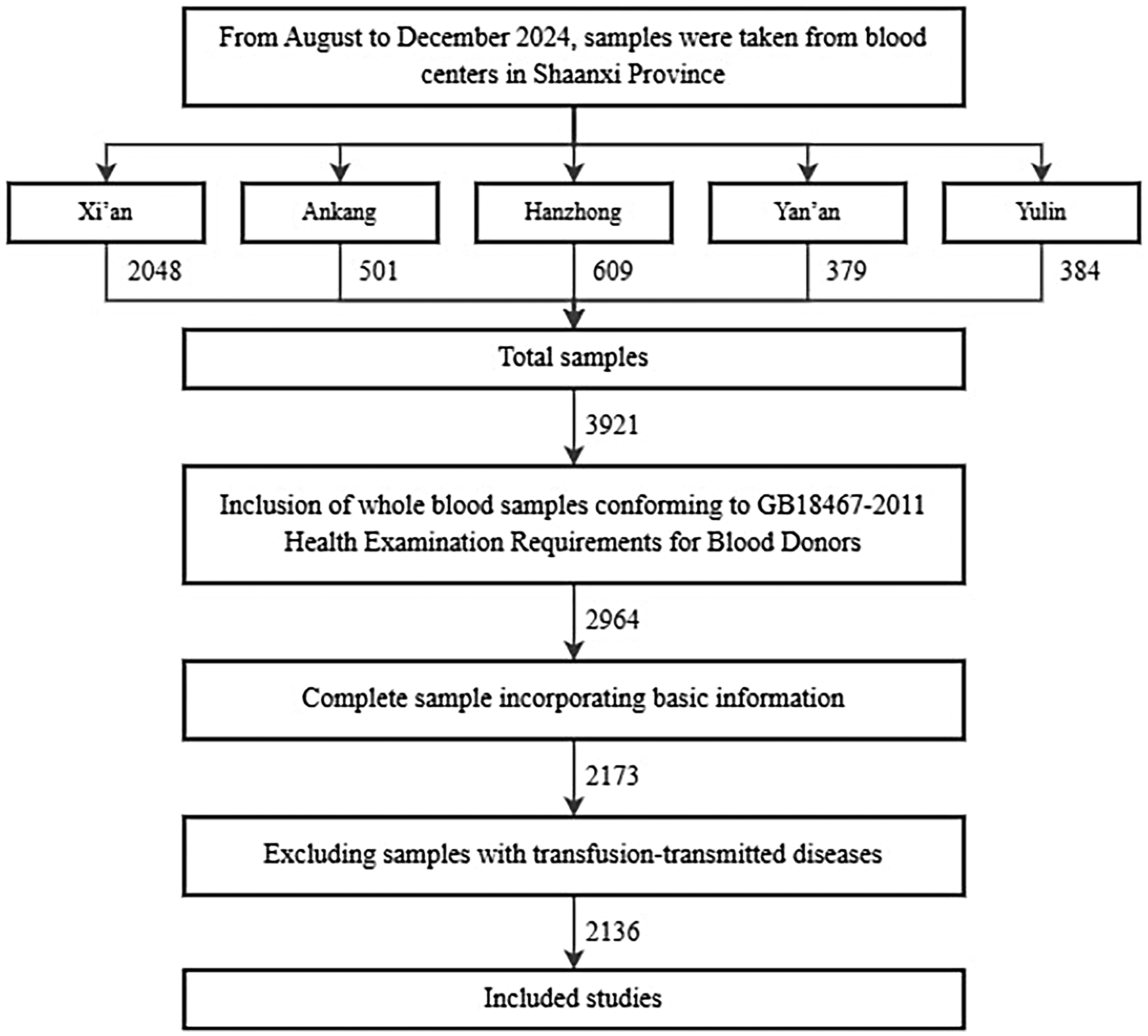
Inclusion process of research samples. This figure illustrates the screening process of samples collected from five municipal central blood centres in Shaanxi Province. We ensured that the included samples were from healthy individuals based on the “Health Examination Requirements for Blood Donors” (GB18467-2011) standard and the results of tests for markers of transfusion-transmitted diseases. Additionally, we limited the blood donation type to whole blood (with a donation interval of ≥6 months) to ensure that no blood from the same source was collected during the study period.
A total of 533 samples tested positive for B19V IgG antibodies, yielding a positive rate of 24.95% (533 out of 2136). The positive rate of B19V IgG was significantly different across age groups (Kruskal–Wallis test, p < 0.001) and regions (χ2 test, p = 0.037), and increased progressively with age. There is no significant difference in the positive rate of B19V IgG antibody among blood donors with different sex, occupation, education, and blood type (Ps > 0.05), as detailed in Table 1.
Comparison of basic data of blood donors in Shaanxi Province (n = 2136).

IgG: immunoglobulin G.
Note. ‘*’ indicates significance at the 0.05 level; ‘†’ indicates significance at the 0.001 level.
Logistic regression of B19V in Shaanxi donors
Univariate logistic regression analysis showed that age, education, blood type, and regional differences were all statistically significant (p < 0.05). Subsequently, these statistically significant factors were further included in the multivariate logistic regression analysis.
The results indicated that the risk of previous infection gradually increased with age (p < 0.001). Compared with the (18–30) age group, the OR (95% CI) of [30–40], [40–50], and [50–60] age groups were 1.527 (1.151–2.026), 2.341 (1.744–3.143), and 2.782 (1.996–3.877), respectively. In addition, compared with blood donors of type A, individuals with type B have a lower risk of previous infection (OR: 0.718 (0.549–0.940), p < 0.05). The risk of infection also differed between Southern Shaanxi and Northern Shaanxi (OR: 0.592 (0.448–0.782), p < 0.001), as detailed in Figure 2.

The left side of the figure represents the results of the univariate logistic regression analysis, where four groups of factors, including age, education level, blood type, and region, show statistical significance. These factors were included in the multivariate logistic regression analysis shown on the right side of the figure, which indicates that age, blood type, and region remain significant. In the figure, the asterisk (*) denotes significance at the 0.05 level, and the dagger (†) denotes significance at the 0.001 level.
Global spatial autocorrelation analysis
Based on the blood collection address information, we have analysed the distribution of positive cases among blood donors in the three regions of Northern Shaanxi, Central Shaanxi, and Southern Shaanxi. For details, please refer to the regional section in Table 1.
Subsequently, we created a global Moran's I scatter plot using GeoDa software (see Figure 3) and employed the Monte Carlo method to test the significant differences in the number of positive cases across regions. The results showed that Moran's I = −0.8654, E(I) = −0.5000, standard deviation = 0.3491, Z = −1.0591, and p < 0.001.
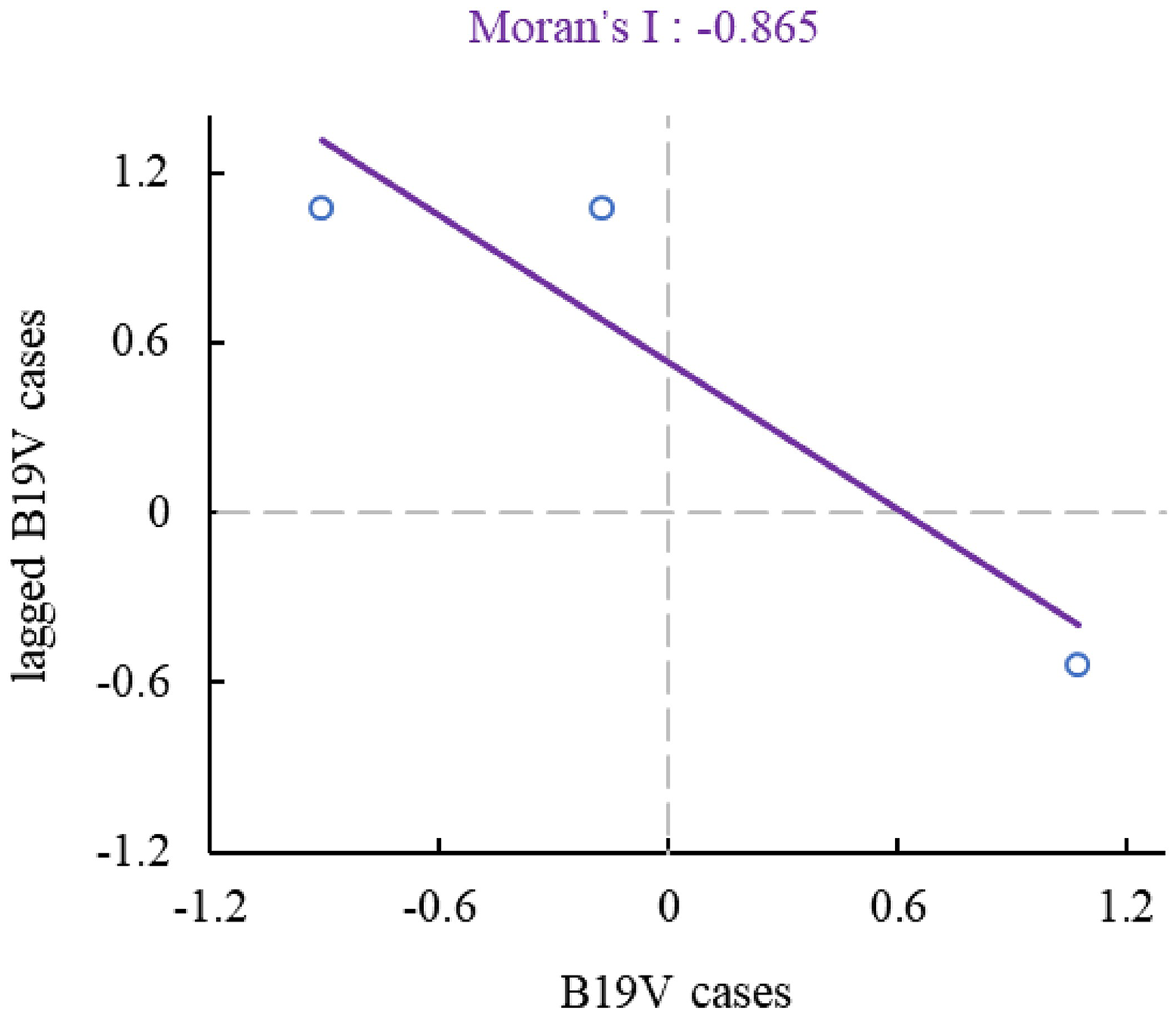
Global Moran's I scatter chart of the number of blood donors with B19V IgG antibody positive in the counties of Shaanxi province. This figure uses the standardized variable values of each region (Northern Shaanxi, Central Shaanxi, and Southern Shaanxi) as the horizontal axis and the lagged variable values (i.e. the average of each region's variable values with the neighbouring regions’ variable values) as the vertical axis to plot a Global Moran's I scatter chart, reflecting the spatial correlation of the number of blood donors with B19V IgG antibody positivity in these regions. The Moran's I value is −0.865, indicating significant spatial negative autocorrelation (i.e. areas with high values tend to be surrounded by areas with low values, and vice versa).
Local spatial autocorrelation analysis
The results illustrated in the LISA cluster map (Figure 4(a)) and the LISA significance map (Figure 4(b)) revealed distinct spatial patterns. Central Shaanxi exhibited a ‘high–low’ spatial aggregation at the 0.001 significance level, while northern and Southern Shaanxi showed a ‘low–high’ aggregation at the same significance level.

LISA cluster map (a) and LISA significance map (b) of the number of B19V positive cases in Shaanxi. The local spatial clustering pattern of the number of blood donors positive for B19V IgG antibodies in different regions of Shaanxi Province (Map A) shows that Central Shaanxi is a high-low cluster. Northern Shaanxi and Southern Shaanxi are low-high clusters. Map B is the significance map, indicating the level of spatial significance of each region.
Discussion
According to the literature, B19V infection usually occurs as sporadic cases or small-scale clusters, so it initially received limited attention and was largely neglected in terms of surveillance and prevention. However, since late 2023, nine European Union/European Economic Area countries have reported unusually high levels of B19V infection on EpiPulse, the European infectious disease surveillance portal, especially for pregnant women and young children. 21 For example, Germany 26 has seen a significant increase in B19V DNA positivity between December 2023 and March 2024 in screening blood pools of donors, and France has reported a similar trend in the number of primary B19V infections. 27 During the same period, the incidence of B19V in Central Europe peaked in April 2024, 33 times higher than in 2009. 28 IgM positivity in blood rose from <3% to 10% of all ages in the United States, with IgM antibodies detected in children aged 5 to 9 rising from 15% to 40%. 29 A hospital in Milan, Italy, reported only seven cases of B19V infection per year in pregnant women before 2023, compared with 59 cumulative cases from January to July 2024. 30 Multinational outbreaks across several continents suggest that B19V infection rates are continuing to rise. At the same time, the virus has good physical and chemical tolerance because of its small diameter (about 25 nm) without a lipid envelope. Its resistance to filtration, inactivation, and pasteurization – and the resulting risk of transmission – poses a major challenge for maintaining the safety of blood products. 8 As a key quality attribute of blood products, the control of B19V has drawn increasing attention from international organizations and governments. Countries such as Germany, Austria, and the Netherlands have launched B19V screening programs. 28
Although there is no relevant literature report on the B19V outbreak in China, we still need to pay close attention to and be vigilant about B19V infection in the future. At present, some areas in China have gradually begun to conduct B19V antibody or nucleic acid testing, and the Pharmacopoeia of the People's Republic of China (2020 edition) has recommended that B19V be included in the scope of testing. 22 But no specific requirements have been established for screening blood products for the virus. The laboratory diagnosis of B19V rests on two complementary approaches: molecular biology and serology. The former focuses on active infection, for which nucleic-acid testing is the first-line choice; the latter underpins clinical interpretation and epidemiological surveys by qualitatively detecting B19V-specific IgM and/or IgG in serum or plasma, routinely using platforms such as immunofluorescence, enzyme immunoassay, and chemiluminescence. 15 IgG antibodies – markers of previous infection – usually appear 2 to 3 weeks after infection and can persist at high titres for months or longer. However, most domestic screening studies on B19V IgG have been conducted in the central and southern regions. We therefore investigated the prevalence of B19V infection in Shaanxi Province and conducted a seroprevalence and spatial analysis.
This research shows that the total positive rate of B19V IgG among voluntary blood donors in Shaanxi Province was 24.95% (533 out of 2136 samples), which was significantly lower than the infection data in Europe for late 2023 to early 2024. 1 Compared with other regions in China, the results lower than Zhengzhou 33.0% (345 out of 1046) 31 and Chongqing 36.68% (405 out of 1104), 32 and higher than Xiamen 16.79% (181 out of 1078) 33 and Shangqiu 7.35% (5268 out of 71,693). 34 In terms of age structure, the rate of previous infection gradually increased with age (p < 0.001), presumably because people are generally susceptible to the virus, and B19V IgG antibody can persist for life, which leads to the cumulative increase of the antibody-positive rate with age. In terms of regional structure, the results show that there is a significant global negative spatial autocorrelation in Shaanxi Province (p < 0.001), which indicates positive cases are primarily concentrated in Central Shaanxi, while Northern and Southern Shaanxi have relatively fewer cases.
The authors speculate that the high concentration of positive cases in the Guanzhong region is mainly due to its dense tourist attractions, concentrated educational resources, and abundant employment opportunities, which attract a large influx of people 35 and thereby facilitate the spread of B19V. Blood-type analysis showed type B individuals to be at lower infection risk (OR: 0.718 (0.549–0.940), p < 0.05). Another team, Healy et al. 36 observed the same pattern in a B19V DNA screen of 167,123 German blood donors: among 22 currently infected cases, group B was exactly 0, the lowest of the four types. Although that study could not perform reliable parameter estimation because of the absence of B-type cases, the two independent datasets both point to the same conclusion – individuals with blood type B are markedly under-represented in both ‘current’ and ‘previous’ infections, suggesting that blood group B may inhibit B19V infection. The authors speculate that the observed pattern may arise because α1,3-galactosyltransferase-catalysed conversion of the H antigen to the B antigen elongates the glycan chain and alters membrane topology and micro-domain organization, thereby partially masking or functionally modifying the major B19V receptor Gb4Cer and creating inter-individual differences in viral adhesion efficiency. This hypothesis remains to be experimentally validated. Besides, there is no significant difference in sex, occupation, and education (Ps > 0.05).
Conclusions
In conclusion, the previous infection rate of B19V is high among blood donors in Shaanxi Province and is influenced by age, blood type, and geographical factors. This study provides important data support and a theoretical basis for formulating personalized clinical blood transfusion strategies and blood product safety policies. Future research should focus on the long-term dynamics of B19V infection and the practical effectiveness of screening programs in reducing transmission risks. Given that B19V screening has not yet been incorporated into the routine donor testing system in China, we recommend implementing pre-transfusion B19V testing for blood or blood components intended for immunocompromised patients, organ transplant recipients, and other high-risk populations, so as to reduce the risk of transfusion-transmitted infection in an economical and efficient manner.
Supplemental Material
sj-pdf-1-sci-10.1177_00368504261436108 - Supplemental material for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study
Supplemental material, sj-pdf-1-sci-10.1177_00368504261436108 for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study by Boya Zhao, Xiaoyue Chu, Fangyao Chen, Hailong Chen, Yuqi Jin, Dan Zhang, Yongheng Pan, Wei Cao, Jin Wang, Hanshi Gong, Ping Chen, Xiaomin Sun, Deng Pan and Chaofeng Ma in Science Progress
Supplemental Material
sj-pdf-2-sci-10.1177_00368504261436108 - Supplemental material for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study
Supplemental material, sj-pdf-2-sci-10.1177_00368504261436108 for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study by Boya Zhao, Xiaoyue Chu, Fangyao Chen, Hailong Chen, Yuqi Jin, Dan Zhang, Yongheng Pan, Wei Cao, Jin Wang, Hanshi Gong, Ping Chen, Xiaomin Sun, Deng Pan and Chaofeng Ma in Science Progress
Supplemental Material
sj-pdf-3-sci-10.1177_00368504261436108 - Supplemental material for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study
Supplemental material, sj-pdf-3-sci-10.1177_00368504261436108 for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study by Boya Zhao, Xiaoyue Chu, Fangyao Chen, Hailong Chen, Yuqi Jin, Dan Zhang, Yongheng Pan, Wei Cao, Jin Wang, Hanshi Gong, Ping Chen, Xiaomin Sun, Deng Pan and Chaofeng Ma in Science Progress
Supplemental Material
sj-pdf-4-sci-10.1177_00368504261436108 - Supplemental material for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study
Supplemental material, sj-pdf-4-sci-10.1177_00368504261436108 for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study by Boya Zhao, Xiaoyue Chu, Fangyao Chen, Hailong Chen, Yuqi Jin, Dan Zhang, Yongheng Pan, Wei Cao, Jin Wang, Hanshi Gong, Ping Chen, Xiaomin Sun, Deng Pan and Chaofeng Ma in Science Progress
Supplemental Material
sj-pdf-5-sci-10.1177_00368504261436108 - Supplemental material for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study
Supplemental material, sj-pdf-5-sci-10.1177_00368504261436108 for Seroprevalence and spatial distribution of immunoglobulin (IgG) antibodies against human parvovirus B19 in voluntary blood donors from Shaanxi Province, China: A cross-sectional study by Boya Zhao, Xiaoyue Chu, Fangyao Chen, Hailong Chen, Yuqi Jin, Dan Zhang, Yongheng Pan, Wei Cao, Jin Wang, Hanshi Gong, Ping Chen, Xiaomin Sun, Deng Pan and Chaofeng Ma in Science Progress
Footnotes
Acknowledgements
We sincerely appreciate the vigorous support provided by the Yulin, Yan'an, Ankang, and Hanzhong Central Blood Stations for this work.
Author contributions
Boya Zhao: Validation, investigation, visualization, and writing – original draft; Xiaoyue Chu: Conceptualisation, supervision, funding acquisition, and writing – review & editing; Fangyao Chen: Software, formal analysis, visualization, and writing – review & editing; Hailong Chen: Software, investigation, and project administration; Yuqi Jin: Validation, investigation, and visualization; Dan Zhang, Yongheng Pan, and Wei Cao: Resources, methodology, and data curation; Jin Wang and Hanshi Gong: Resources and funding acquisition; Ping Chen: Validation, and investigation; Xiaomin Sun, and Deng Pan: Resources, and data curation; Chaofeng Ma: Conceptualisation, funding acquisition, methodology, and writing – review & editing.
Ethical approval
This study was approved by the Ethics Committee of the Xi’an Central Blood Station (No. 202507).
Consent to participate
All blood donors included in this study have provided written informed consent.
Map approval statement
The local map has been reviewed and approved by Shaanxi Surveying and Mapping Geographic Information Bureau (approval number: Shaanxi S (2027) No. 7).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Chaofeng Ma was supported by (1) Key Research and Development Program of Shaanxi Province (No. 2024SF-YBXM-272), (2) Xi'an Elite Talents Program (No. XAYC220046), and (3) Shaanxi Province Health Scientific Research Innovation Ability Promotion Plan (No. 2025YF-12). Xiaoyue Chu was supported by (1) Xi'an Science and Technology Planning Project (No. 23YXYJ0013), (2) China Blood Transfusion Association Weigao Fund (No. CSBT-WG-2024-04), (3) Key Research and Development Program of Shaanxi Province (No. 2025CY-YBXM 138), and (4) Health Research Project of Shaanxi Province in 2022 (No. 2022D050). Jin Wang was supported by the Xi'an Health and Family Planning Commission Research Project (No. 2023yb48). Hanshi Gong was supported by the China Blood Transfusion Association Shengxiang Fund (No. CSBT-SX-2023-02).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the results of this study are not made public because of information that may damage the privacy of the participants. For further enquiries, please contact the correspondent directly.
Supplemental material
Supplemental material for this article is available online.
