Abstract
A tracheoesophageal fistula (TEF) is an abnormal connection between the trachea and esophagus, which is an intractable clinical challenge associated with a poor prognosis and a high mortality. We report the successful closure of a complex, large-diameter TEF in a male patient in his late 60s following radical esophagectomy, with initial surgical and esophagoscopic interventions considered unfeasible. A tracheal stent was placed to cover the fistula as a bridging therapy, followed by definitive closure using an over-the-scope clip (OTSC). The tracheal stent was removed at the 5-month follow-up, and no serious complications were observed during or after endoscopic management. For complex TEFs that are not amenable to surgical repair or esophagoscopic closure, tracheal stent placement serves as a bridging therapy, thereby providing a window for subsequent interventions. The combined strategy of tracheal stenting with the OTSC provides a minimally invasive and highly effective treatment option for complex TEFs through multidisciplinary collaboration.
Introduction
Tracheoesophageal fistula (TEF), an abnormal connection between the trachea and esophagus arising from diverse etiologies, is an intractable clinical challenge associated with poor prognosis. 1 Under these circumstances, aspiration pneumonitis and malnutrition further complicate fistula repair. Video-assisted thoracoscopic surgery or open thoracotomy is recommended for TEF closure when the clinical conditions permit a major reoperation. 2 Endoscopic closure of fistulae signifies major progress in TEF management, particularly for critically ill patients who are inoperable, with a success rate exceeding 95%.3–5 The primary goal of endoscopic therapy is to interrupt the flow of luminal content through the fistula. Various endoscopic techniques have been described for TEF closure, including stenting,3,5 endoclips,6–8 atrial septal defect occluders, 9 endobronchial one-way umbrella-shaped valves, 10 argon plasma coagulation, 11 suturing, 12 submucosal dissection, 13 biosynthetic meshes, 14 fibrin glue, 8 platelet-rich plasma, 11 and mesenchymal stem cells. 15 Endoscopic approaches offer minimally invasive, well-tolerated, and cost-effective procedures, and provide a safe interventional strategy with the potential to improve the quality of life in patients with TEF. 4
Here, we report the successful closure of a complex, large TEF after esophagectomy in which surgical intervention and esophagoscopy were initially deemed infeasible. This case underscores the importance of a multidisciplinary collaborative strategy, which offers a minimally invasive and highly effective treatment option for complex TEFs. Signed informed consent for the treatment was obtained from the patient. The reporting of this study conforms to CARE guidelines. 16
Case presentation
An Asian male in his late 60 s with a history of type 2 diabetes mellitus presented to Jining No.1 People's Hospital (located in Jining, Shandong Province, P.R. China) in October 2024 complaining of fever, progressive dysphagia, and aspiration cough. Five months before presentation, the patient had undergone robot-assisted (Da Vinci system) thoracoabdominal radical esophagectomy (modified Ivor Lewis approach) with intrathoracic esophagogastrostomy. Histopathological examination confirmed esophageal squamous cell carcinoma (Figure 1). Based on the 8th edition of the American Joint Committee on Cancer/Union for International Cancer Control (AJCC/UICC) staging system, the tumor was staged as T1bN0M0. 17 An anastomotic leak was first detected on an upper gastrointestinal contrast study 15 days postoperatively, and this leak failed to resolve with conservative management. Chest computed tomography (CT) indicated severe lung infection. Bronchoscopy revealed a 12 × 10 mm fistula on the inner wall of the orifice of the right intermediate bronchus (Figure 2(a)). Under endoscopy, after injecting methylene blue dilution through the esophagus, a large amount of the methylene blue leaked into the tracheal lumen, indicating a TEF (Figure 2(b)).
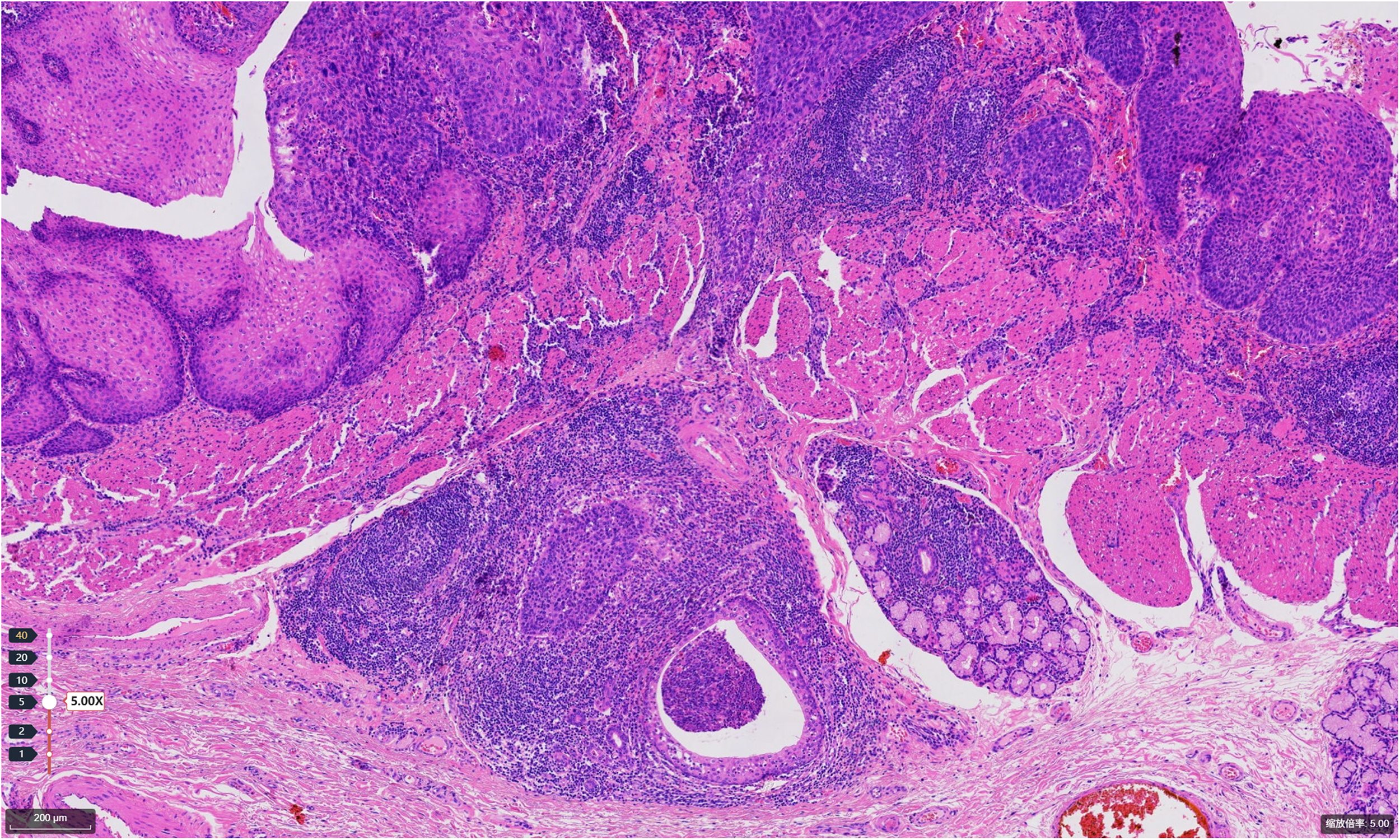
Histopathological examination of hematoxylin and eosin (H&E)-stained sections confirmed esophageal squamous cell carcinoma. Microscopically, irregular nests and trabeculae of atypical squamous epithelial cells are seen infiltrating into the submucosa, accompanied by a prominent lymphocytic infiltrate within the stromal compartment. The tumor cells display marked nuclear atypia. Scale bar: 200 µm.

Endobronchial image of the TEF. (a) The bronchoscopic view shows the fistula on the tracheal side (red arrow), with a maximal diameter of 12 × 10 mm. (b) After injecting methylene blue dilution through the esophagus, a large quantity of the methylene blue leaked into the tracheal lumen from the fistula (red arrow). (c) A silicone tracheal stent was deployed to cover the fistula, with the distal bifurcation positioned at the origin of the right intermediate bronchus. (d) At the main carina, the Y-stent was deployed with its main body in the right main bronchus; a black asterisk denotes the superior edge of the stent. (e) One-month follow-up after stent deployment showed granulation tissue formation at the inferior edge of the stent, as indicated by a black asterisk. TEF, tracheoesophageal fistula.
The patient presented with a low-positioned fistula, leading to significant pulmonary aspiration and severe infection. Immediate therapeutic intervention was imperative to close the fistula and alleviate the aspiration. Treatment options for managing such large, complex TEFs include surgical procedures and nonsurgical (endoscopic) techniques. Notably, surgeons did not recommend reoperation for this type of unstable TEF. Despite significant therapeutic challenges, uncertain outcomes, and limited feasibility of endoscopic intervention in this case, it remained a potential option for selective application as needed. Therefore, for this patient, a staged, minimally invasive endoscopic strategy was developed. Tracheal stent placement was proposed as an initial treatment option for fistula closure. Because of the proximity of the fistula to the orifice of the right middle/lower lobe bronchi and the progressive distal narrowing of the right intermediate bronchus, the procedure entailed significant technical challenges. After precise measurements, a modified silicone stent (16 × 15 − 13 × 25 − 13 × 8 mm) (Novatech, Aubagne, France) was successfully deployed under rigid bronchoscopies (Figure 2(c), 2(d)). After confirming that the stent was in a good position, a diluted methylene blue solution was injected, which revealed no leaked contrast from the esophagus to the trachea. Concurrent supportive management, including nasogastric tube placement, was implemented. Recognizing the inherently palliative character of airway stenting for managing large-diameter fistulas, and the potential for long-term complications such as insufficient fistula occlusion and airway narrowing secondary to granulation tissue overgrowth. Consequently, we planned to close the esophageal-side fistula using an over-the-scope clip (OTSC). However, the patient declined this intervention initially, as his symptoms had improved substantially after stent placement. To optimize long-term outcomes, a follow-up endoscopy was scheduled 1 month later. Bronchoscopy performed 1 month post-stent revealed a small amount of contrast medium extravasation and granulation tissue hyperplasia (Figure 2(e)), at which point the OTSC was strongly recommended once more. With the goal of achieving a complete clinical cure, the patient consented to undergo the OTSC. Esophagoscopy revealed a narrow anastomosis and a fistula next to the anastomosis (Figure 3(a), 3(b)). The stenosis was dilated, and a nasogastric tube was placed. The surrounding mucosa of the fistula was first electrocoagulated using hot biopsy forceps, followed by complete closure with an OTSC (Ovesco Endoscopy, Tübingen, Germany) (Figure 3(c)). Although contrast radiography on postoperative day 1 revealed minimal residual leakage, no spilled contrast was observed after 1 month of supportive therapy, enabling the patient to resume oral feeding and remove their nasogastric tube. Closure of the fistula was confirmed through sequential endoscopic and radiological assessments at 4 and 5 months postoperatively. The tracheal stent was extracted with grasping forceps under direct tracheoscopic visualization at 5 months, and closure of the original fistula was observed, with slight scarring of the adjacent tissues (Figure 4(a)). Contrast radiography performed 24 h after stent removal showed no evidence of contrast extravasations (Figure 4(b)). The patient maintained favorable outcomes during the 6-month post-removal follow-up period.
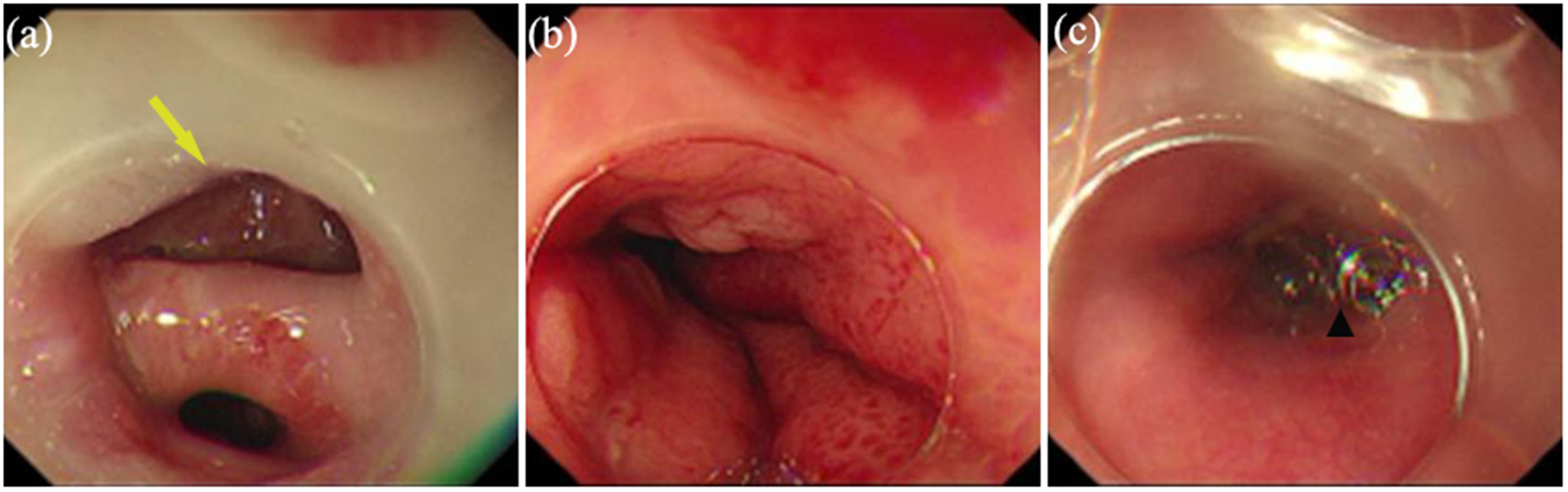
Endoscopic findings in the esophagus. (a) The esophagoscopic view shows the fistula on the esophageal side (yellow arrow). (b) Esophagoscopic view of the narrow anastomosis. (c) Esophagoscopic visualization confirmed successful fistula closure with the over-the-scope clip (black triangle).

Imaging findings after tracheal stent removal. (a) The bronchoscopic view of the original fistula with slight scarring of the adjacent tissue within the white dashed circle. (b) Contrast radiography performed 24 h after stent removal showed no evidence of contrast extravasation.
Discussion
TEF remains the most feared complication after esophagectomy, with a poor prognosis and high associated mortality. 18 The primary challenge in managing the present case stemmed from the complex nature of the fistula, which was not amenable to surgical repair or esophagoscopic closure, compounded by its unfavorable anatomical location at the orifice of the right intermediate bronchus. A single-center European study had previously established that fistulas located in the right main bronchus are associated with worse survival outcomes compared to fistulas occurring in other locations. 5 Taking these factors into account, we initially deployed a tracheal stent to cover the fistula, which established a temporary barrier and interrupted the flow of luminal contents. While conventional straight stents were considered unsuitable because of risks of migration and right upper lobe obstruction in this anatomical configuration. In this case, we ultimately selected a small silicone Y-shaped stent, and the edge of the stent only exceeded the fistula by approximately 1 cm, thus ensuring effective closure, while reducing mechanical irritation to the distal airway. Compared to metallic stents, silicone stents provide robust luminal support and are modifiable prior to implantation. Moreover, silicone stents exhibit superior biocompatibility, making them well-suited for long-term internal applications and enabling easier removal, thereby creating conditions for subsequent endoscopic interventions. 19 Notably, the fistula was located at the orifice of the right intermediate bronchus, and the diameter of the side branch of the Y-stent used to close the fistula was approximately 2 mm larger than that of the right middle segmental bronchus. Consequently, long-term deployment of a tracheal stent in this location carries a high risk of complications, including granulation tissue proliferation, pulmonary atelectasis, luminal stenosis, and compromised sealing effectiveness. This thus necessitates the use of combined approaches involving mechanical closure of the fistula tract. Recognizing the anticipated extended survival of the patient, concerns regarding long-term stent-associated complications, and the inherently palliative character of airway stenting in the context of a large-diameter fistula, we proposed a sequential approach involving initial airway stenting followed by an OTSC to enable definitive closure. However, owing to the initial informed refusal of the patient, the OTSC procedure was ultimately delayed for 1 month following the initial stent implantation. Postoperatively, contrast radiography and bronchoscopic reexamination were continuously performed to ensure the stability of the fistula closure. In this case, the fistula closed 5 months after endoscopic intervention, which is consistent with the observations presented in a previous report. 20 This protocol prevents fistula recanalization resulting from premature stent removal, while avoiding complications such as granulation tissue hyperplasia caused by prolonged compression resulting from delayed stent extraction.
The management of TEF following esophagectomy presents significant clinical challenges. Selecting the endoscopic technique is based on the size, number, and location of the fistula and the condition of the mucosa surrounding the fistula. Clip deployment is preferentially used for fistulas < 1 cm in size, whereas endoscopic stents are primarily used for large fistulas (>1 cm) at initial intervention. 20 The OTSC system achieves fistula closure principally through the potent mechanical compression provided by its clipping device. When tissues surrounding the fistula are excessively friable or heavily fibrosed, as in most fistulas, the clip cannot achieve complete closure of the fistula. In a study of 3025 patients, its application for fistulas yielded the lowest overall success rate among all records: only 55.8% (347/622). 21 The primary objective of stent placement is to cover the fistula and provide a temporary barricade to the region. This approach is especially applicable to large fistulas, providing high procedural success rates, prompt symptom relief, and significant improvement in quality of life. 5 Primary stent-related complications include migration, granulation tissue formation, patient discomfort, and retained secretions. In the present case, as the fistula had been epithelialized for 5 months, simple closure techniques or stent placement alone were not sufficient to close the fistula. Therefore, our therapeutic strategy integrated both an airway stent and OTSC. The OTSC is used to close the fistula on the side of the esophagus, which prevents additional leakage of liquids or gases and forms a dual protection mechanism with the tracheal stent. Notably, the tracheal stent provides a physical barrier, and the endoclip promotes tissue healing. This combined therapeutic approach can avoid the risk of fistula nonhealing after tracheal stent-only placement and relieve issues such as delayed healing and inadequate fistula occlusion associated with clip-only application.
Conclusion
For complex TEFs that are not amenable to surgical repair or esophagoscopic closure, tracheal stent placement serves as a bridging therapy, thereby providing a window for subsequent interventions. The combined strategy of tracheal stenting with the OTSC provides a minimally invasive and highly effective treatment option for complex TEFs through multidisciplinary collaboration.
Footnotes
Ethics approval
This was a case report using anonymous patient data. This study protocol was reviewed and approved by the Ethics Committee of Jining No.1 People's Hospital (No. 2025-IIT-K099).
Consent for publication
The patient provided written informed consent for publication of the details of his medical case and any accompanying images.
Author contributions
Ran-ran Mo drafted the original manuscript and designed the overall structure of the case report. Li-ran Zhang performed the initial revision of the manuscript. Yan-fang Si conducted an in-depth discussion on the clinical aspects and provided valuable comments. Cui-xia Bian critically reviewed the manuscript and made substantial revisions. All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R&D Program of Jining (Grant No. 2024YXNS164).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All the data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author on reasonable request.
Statement
We have de-identified all patient details.
