Abstract
Objective
Tachycardia is common in pediatric intensive care and may contribute to hemodynamic instability when persistent despite adequate resuscitation. Data on β-blocker therapy in critically ill children are limited. Our objective was to evaluate the safety and hemodynamic effects of propranolol in critically ill pediatric patients with persistent sinus tachycardia.
Methods
Retrospective cohort study of children aged 1 month to 18 years who received propranolol in a tertiary-care pediatric intensive care unit between March 1, 2019, and March 1, 2024. The inclusion criteria were heart rate ≥2 standard deviations above age norms that persisted despite adequate resuscitation and conventional treatments. Patients with primary cardiac disease or a history of β-blocker use were excluded. Propranolol was administered orally or via a nasogastric tube at a dose of 1 mg/kg/day for persistent sinus tachycardia despite normothermia, adequate sedation-analgesia, and standard hemodynamic support. The primary outcome was the change in heart rate over 48 h. Secondary outcomes included trends in blood pressure, lactate and glucose levels, vasoactive–inotropic score (VIS), and adverse events.
Results
Among 93 patients (median age, 19 months), propranolol caused a significant reduction in heart rate within 6 h (mean decrease, ∼30 bpm; p < 0.001), with stabilization by 24 h. Systolic (SBP) and diastolic blood pressures (DBP) increased modestly (SBP +7 mmHg; DBP +3 mmHg, p < 0.001), and lactate levels declined significantly at 6 h (p = 0.003). The VIS did not change significantly. Adverse events were rare (hypotension 2.2%, bradycardia 1.1%).
Conclusions
In this large pediatric cohort, propranolol was well tolerated and linked to positive hemodynamic and metabolic effects. These results support cautious use in carefully selected critically ill children and emphasize the need for future prospective trials.
Keywords
Introduction
Critically ill patients often exhibit excessive sympathetic activation and autonomic dysregulation due to elevated circulating catecholamines. Persistent tachycardia after adequate resuscitation may indicate ongoing sympathetic overstimulation and has been identified as an independent predictor of mortality in ICU populations.1–9 Whether primary or secondary in origin, tachycardia is a common clinical finding in critically ill patients and often signals underlying hemodynamic compromise.
Initial management of tachycardia requires prompt identification and correction of reversible causes, such as anemia, hypovolemia, dystonia, hypoxemia, pain, or pharmacologic adrenergic stimulation. In the ICU setting, persistent tachycardia is rarely benign, especially when coupled with hemodynamic instability. It may contribute to myocardial ischemia, increased myocardial energy demand, and maladaptive structural remodeling, ultimately impairing cardiac function and reducing end-organ perfusion. When tachycardia persists despite correction of precipitating factors, pharmacologic intervention becomes necessary.
Beta-adrenergic blockers (β-blockers) represent a cornerstone therapy for blunting sympathetic overactivity and are well-established in the management of various rhythm disturbances. Their mechanism involves competitive inhibition of catecholamine binding to β-adrenergic receptors, attenuating the downstream physiological effects of sympathetic stimulation.
Clinically, β-blockers are indicated for systemic arterial hypertension, tachyarrhythmias, and heart failure. 1 By reducing afterload, they lower myocardial oxygen consumption and improve perfusion efficiency and stroke volume. Beyond their cardiovascular effects, β-blockers have attracted interest for their potential pleiotropic benefits in critically ill patients, including modulation of inflammatory cytokine release, reduction of metabolic dysregulation, and immunomodulatory activity.10–12 Additionally, β-blockade may provide protection against catecholamine-induced myocardial injury and enhance ventricular remodeling and contractile function.2,3
In pediatric populations, especially in managing severe burns, β-blockers—particularly propranolol—have been shown to reduce heart rate (HR) without causing significant hypotension, decrease the hypermetabolic response, lower inflammatory cytokine levels, and reduce insulin resistance, lipolysis, proteolysis, cardiac workload, and bone loss.4,5 Despite these potential benefits, some studies have found no significant difference in sepsis incidence or mortality rates among burn patients treated with propranolol. 6
Despite these theoretical advantages, current consensus guidelines offer limited direction on using β-blockers in critically ill populations. Concerns still exist about their negative inotropic effects and potential to worsen hypotension, especially in patients with impaired cardiac function. There are case reports that have documented, under conditions of hypoperfusion due to sepsis or hemorrhage, propranolol administration may trigger unopposed alpha-adrenergic stimulation, leading to splanchnic vasoconstriction and potentially resulting in intestinal ischemia. 13 As a result, intensivists face complex clinical decisions regarding the initiation, titration, and continuation of β-blocker therapy in unstable patients. While data supporting β-blocker use in critically ill adults has expanded, evidence in pediatric populations remains limited.
Based on the statement in the Cardiovascular Monitoring and Cardiovascular Drug Therapy chapter under the β-blocker section of A Practical Handbook on Pediatric Cardiac Intensive Care Therapy 7 “only very conditionally do we share the view that children [including neonates] are dependent on a high HR to maintain cardiac output. In our experience, HR >150/min is usually not beneficial,” and supported by a concurrently published, albeit rare, adult studies, β-blockers have been introduced in our unit for the management of persistent tachycardia since 2019. Over time, based on perceived clinical benefit and the absence of adverse hemodynamic effects, the practice became more standardized for certain patients, although no formal written protocol was established. The present study was designed to retrospectively assess this evolving practice and its outcomes, aiming to provide preliminary data to guide future prospective studies.
In this study, we present our clinical experience with β-blocker therapy in critically ill children with persistent sinus tachycardia, focusing on hemodynamic outcomes and safety parameters.

Flowchart of patient selection.
Methods
Time and patient selection
We retrospectively reviewed all 2862 patients admitted to the pediatric intensive care unit (PICU) of Istanbul University, Istanbul Faculty of Medicine, between March 2019 and March 2024. Among these, 136 patients who received propranolol during their PICU stay were identified, and their medical records were examined in detail. Forty-three patients were excluded based on predefined exclusion criteria, leaving 93 patients for the final analysis (Figure 1).
Settings and data collection
The study was conducted in our 14-bed PICU, which admits all medical and noncardiac surgical patients, excluding burn cases. The unit is equipped to provide the full spectrum of advanced intensive care interventions, including extracorporeal membrane oxygenation.
In our hospital's electronic medical record (EMR) system, imaging studies, blood gas results, complete blood counts, and biochemical parameters are stored digitally. Medication orders, vital signs, and arterial blood gas measurements are also recorded on paper charts by physicians and nurses. For this study, patient data were collected through a thorough review of both the EMR and paper chart records.
Clinical data collected included peak HR, systolic blood pressure (SBP), diastolic blood pressure (DBP), and vasoactive–inotropic score (VIS) 8 ; propranolol timing and dosage; and lactate and glucose levels at 0, 6, 12, 24, and 48 h after the initiation of the drug. Additionally, the Pediatric Risk of Mortality-III (PRISM-III) 9 and Pediatric Logistic Organ Dysfunction 2 (PELOD-2) 10 scores for all patients were calculated and recorded.
This study was conducted in accordance with the principles of the Declaration of Helsinki (1975), as revised in 2024. The study was conducted in accordance with the Declaration of Helsinki and was reviewed and approved by the Institutional Ethics Committee of Istanbul University, Istanbul Faculty of Medicine (Date: 7 March 2025, Decision No: 05, File No: 2025/295). All patient data were fully de-identified prior to analysis to ensure confidentiality and protect patient privacy.
Inclusion and exclusion criteria
Patients were eligible for inclusion if they met all of the following criteria:
Persistent sinus tachycardia, defined as a HR ≥2 standard deviations (SDs) above age-specific norms before starting propranolol, that persists despite standard management following international treatment guidelines (e.g. Surviving Sepsis Campaign).
11
Standard management comprised fluid resuscitation, use of vasoactive therapy when needed, ventilatory support, sedation, antipyretics, analgesia, and blood transfusions as appropriate. Only patients who received propranolol specifically for this condition were included. age between 1 month and 18 years; and initiation of propranolol treatment during the PICU stay.
Exclusion criteria included: (i) known contraindications or intolerance to β-blockers, (ii) sinus bradycardia (HR below the age-specific lower limit of normal), (iii) second-degree atrioventricular block (Mobitz type II) or third-degree atrioventricular block, (iv) administration of propranolol for primary cardiac disease as the main reason for PICU admission (e.g. congenital heart disease, heart failure, myocarditis, or arrhythmia), (v) use of propranolol for hypertension, and (vi) administration of propranolol prior to PICU admission for any indication.
Sinus tachycardia was confirmed in all patients by continuous bedside electrocardiographic monitoring prior to propranolol initiation. The decision to start β-blocker therapy was made at the discretion of the attending intensivist. In our study, propranolol was given enterally (by mouth or through a nasogastric tube) at a total daily dose of 1 mg/kg, divided into three doses every 8 hours; no intravenous administration or continuous infusion was used. The primary goal was to lower the HR to the upper limit of the age-specific normal range. The continuation of therapy and subsequent dosing were determined by the attending PICU specialist based on HR assessments at each scheduled dose.
The final cohort consisted of 93 patients who received propranolol specifically for persistent sinus tachycardia despite normothermia, appropriate analgesia and sedation, and standard hemodynamic support measures, including fluid resuscitation, vasoactive therapy, and respiratory support.
At the start of the study period, sepsis was defined according to the 2005 International Pediatric Sepsis Consensus Conference (IPSCC) criteria. 14 From early 2024 onward, the updated Phoenix criteria were adopted. 15 Consequently, sepsis diagnoses were retrospectively confirmed using both sets of criteria. Septic shock was defined based on current international guidelines. 12
Primary and secondary outcomes
The primary outcome was the reduction in HR within the first 48 h of propranolol therapy. Secondary outcomes included: (i) hemodynamic stability throughout the treatment course, (ii) PICU mortality, and (iii) lengths of stay in both the PICU and the hospital. Hemodynamic stability was evaluated by monitoring for bradycardia, hypotension, and hypoglycemia as safety outcomes.
Bradycardia was defined according to age-specific pediatric critical care standards: <80 bpm for newborns (0–3 months), <75 bpm for infants/young children (4 months–2 years), <60 bpm for children aged 2–10 years, and <50 bpm for adolescents (>10 years). Hypotension was defined based on Pediatric Advanced Life Support thresholds
16
: SBP <70 mmHg for neonates up to 1 month and infants aged 1–12 months, SBP <70 + (2 × age in years) mmHg for children aged 1–10 years, and SBP <90 mmHg for adolescents >10 years. Hypoglycemia was defined as blood glucose <70 mg/dL.
Definition of adequate resuscitation
Adequate resuscitation was defined according to established pediatric critical care guidelines (e.g. Surviving Sepsis Campaign), incorporating both clinical and laboratory criteria. Patients were considered adequately resuscitated when they demonstrated: (i) normalization or near-normalization of serum lactate levels, (ii) restoration of age-appropriate mean arterial pressure without ongoing fluid bolus requirements, and (iii) capillary refill time ≤2 s in the absence of peripheral vasoconstriction. In our cohort, most patients were normotensive with lactate values approaching the normal range prior to propranolol initiation.
Statistical analysis
All data were entered into a dedicated research database and analyzed with IBM SPSS Statistics, version 30.0 (IBM Corp., Armonk, NY, USA). The Kolmogorov–Smirnov and Shapiro–Wilk tests were used to evaluate the normality of continuous variables.
Normally distributed data (e.g. HR, SBP, DBP) are reported as mean ± SD, whereas non-normally distributed data are presented as medians with interquartile ranges (IQR: 25th–75th percentile). Categorical variables are described as counts and percentages.
Within-group comparisons of pre- and posttreatment values were conducted using paired t-tests for parametric variables and the Wilcoxon signed-rank test for nonparametric variables. Analyses were performed for the entire cohort and for predefined subgroups, including patients receiving vasoactive support and those diagnosed with sepsis. For comparisons between more than two independent groups in subgroup analyses (e.g. subgroup analysis by infection source), one-way analysis of variance (ANOVA) was used for normally distributed variables. All statistical tests were two-tailed, and a p-value <0.05 was considered statistically significant.
Results
A total of 93 pediatric patients (median age: 19 months, IQR: 8–96) who received propranolol in the PICU for persistent sinus tachycardia were included. Sinus tachycardia was confirmed in all patients, and no supraventricular or ventricular arrhythmias were present at propranolol initiation. Of these, 41.9% were male. The median PRISM-III and PELOD-2 scores were 5 (IQR: 2–11) and 4 (IQR: 3–6), respectively. Nearly one-third (29%) were admitted postoperatively, including pediatric surgery (n = 14), neurosurgery (n = 8), orthopedic surgery (n = 4), and plastic surgery (n = 1) cases.
Sepsis was diagnosed in 46.2% of patients based on the Phoenix criteria and in 76.3% according to 2005 IPSCC-based sepsis definitions. Among those with sepsis, the most common sources were pneumonia/lower respiratory tract infection (n = 37), bloodstream infection (n = 18), intra-abdominal/gastrointestinal infection (n = 6), bone and soft tissue infection (n = 5), central nervous system infection (n = 3), and urinary tract infection (n = 2). Mechanical ventilation was required in 58.1%, and 34.4% received vasoactive therapy (Supplemental Figure 1). The microorganisms identified in patients diagnosed with sepsis are listed in Supplemental Table 1.
Propranolol was initiated at a median of 26 h (IQR: 8–72) after PICU admission, at a median dose of 1 mg/kg/day and continued for a median of 96 h (IQR: 60–192). Bradycardia and hypotension were observed in 1.1% and 2.2% of patients, respectively; no cases of bronchospasm or hypoglycemia (blood glucose level <70 mg/dL) were reported.
The median length of hospital stay was 20 days (IQR: 12–34), while the median PICU length of stay was 10 days (IQR: 5–20). There were no statistically significant differences in either hospital or PICU LOS between patients with sepsis and those without sepsis (p = 0.244 and p = 0.237, respectively).
The overall mortality rate in the cohort was 2.2% (n = 2). Both nonsurvivors were female patients who presented with shock and required vasoactive support. One patient was a two-month-old infant with underlying immunodeficiency and hemophagocytic lymphohistiocytosis, with mild left ventricular hypertrophy and mild mitral regurgitation on echocardiography. The second patient was a 12-year-old child admitted with septic shock following liver transplantation, with Escherichia coli isolated from abdominal fluid cultures. The duration of PICU stay prior to death was 41 and 8 days, respectively (Table 1).
General characteristics and PICU treatments of the study population.

PICU, pediatric intensive care unit; IQR, interquartile range; SIRS, systemic inflammatory response syndrome; GI, gastrointestinal; LOS, length of stay; PRISM-III, Pediatric Risk of Mortality III; PELOD-2, Pediatric Logistic Organ Dysfunction-2.
Heart rate response
A significant reduction in peak HR was observed within 6 h of starting propranolol in all patients (mean decrease: ∼30 bpm; p < 0.001), with levels stabilizing after 24 h. These patterns were consistent across subgroups, including patients with sepsis and those on vasoactive support. In patients with sepsis (n = 71), propranolol caused a steady and significant drop in HR from a baseline of 164 ± 20 bpm to 125 ± 20 bpm by 24 h after starting the medication (p < 0.001), without affecting blood pressure. Similar trends appeared in the vasoactive subgroup (n = 32), where HR declined significantly within the first 6 h (p < 0.001) and stayed stable afterward. No clinically significant bradycardia or hypotension was observed in either group.
Blood pressure and blood glucose trends
Systolic and diastolic pressures increased significantly within 6 h posttreatment in all groups (SBP: mean increase ∼7 mmHg; DBP: ∼3 mmHg) (p < 0.001), with values stabilizing over the next 48 h. Mean glucose values at each time point are provided in Table 2, and no episodes of hypoglycemia occurred in the study cohort (Table 2 and Figure 2).
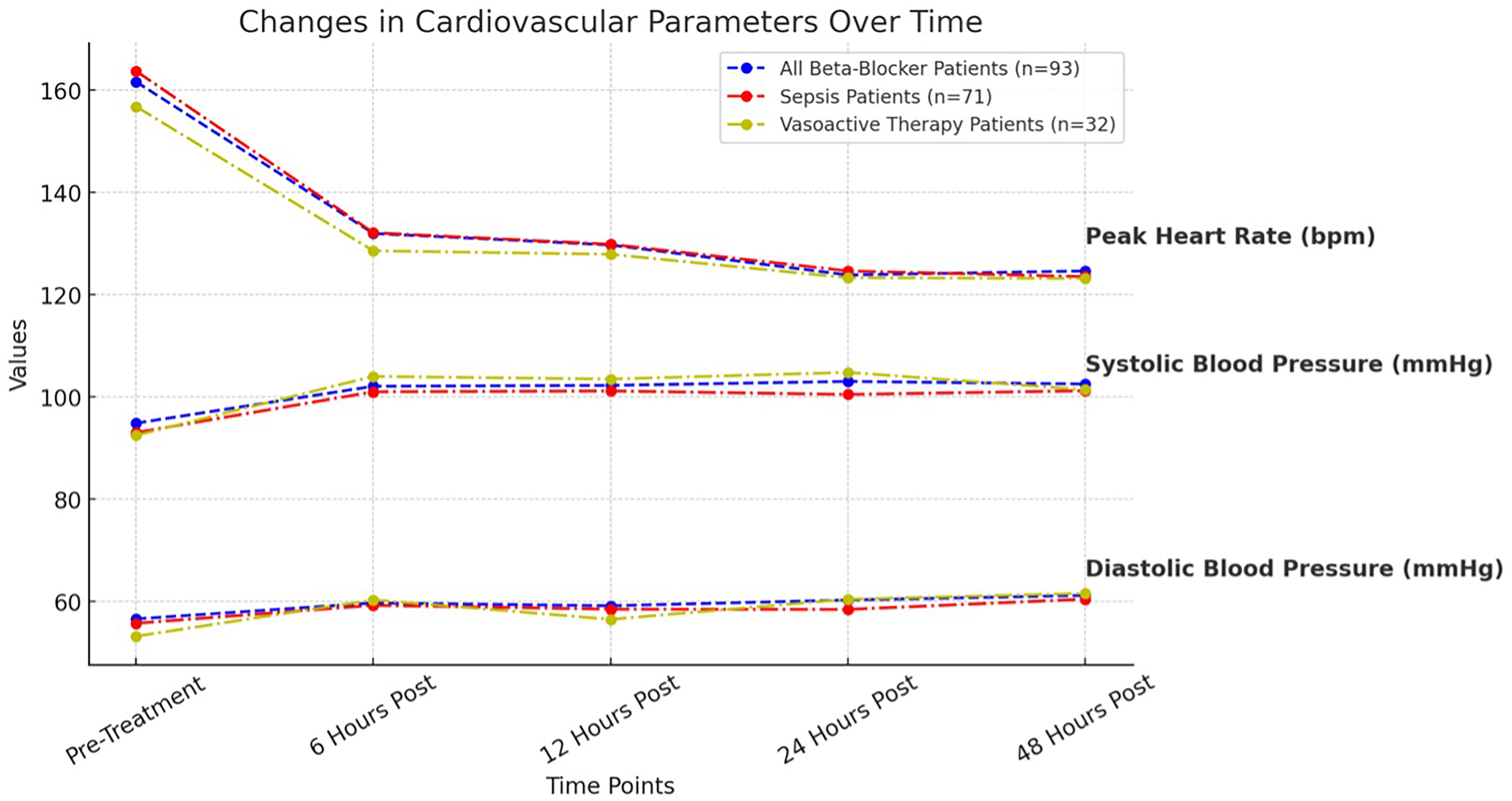
Changes in peak heart rate, systolic blood pressure, and diastolic blood pressure over time in patients receiving β-blockers, patients with sepsis, and patients receiving vasoactive therapy before and after treatment. HR, heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Changes in peak heart rate, systolic and diastolic blood pressure, bicarbonate, and lactate levels in all patients receiving β-blockers (n = 93).
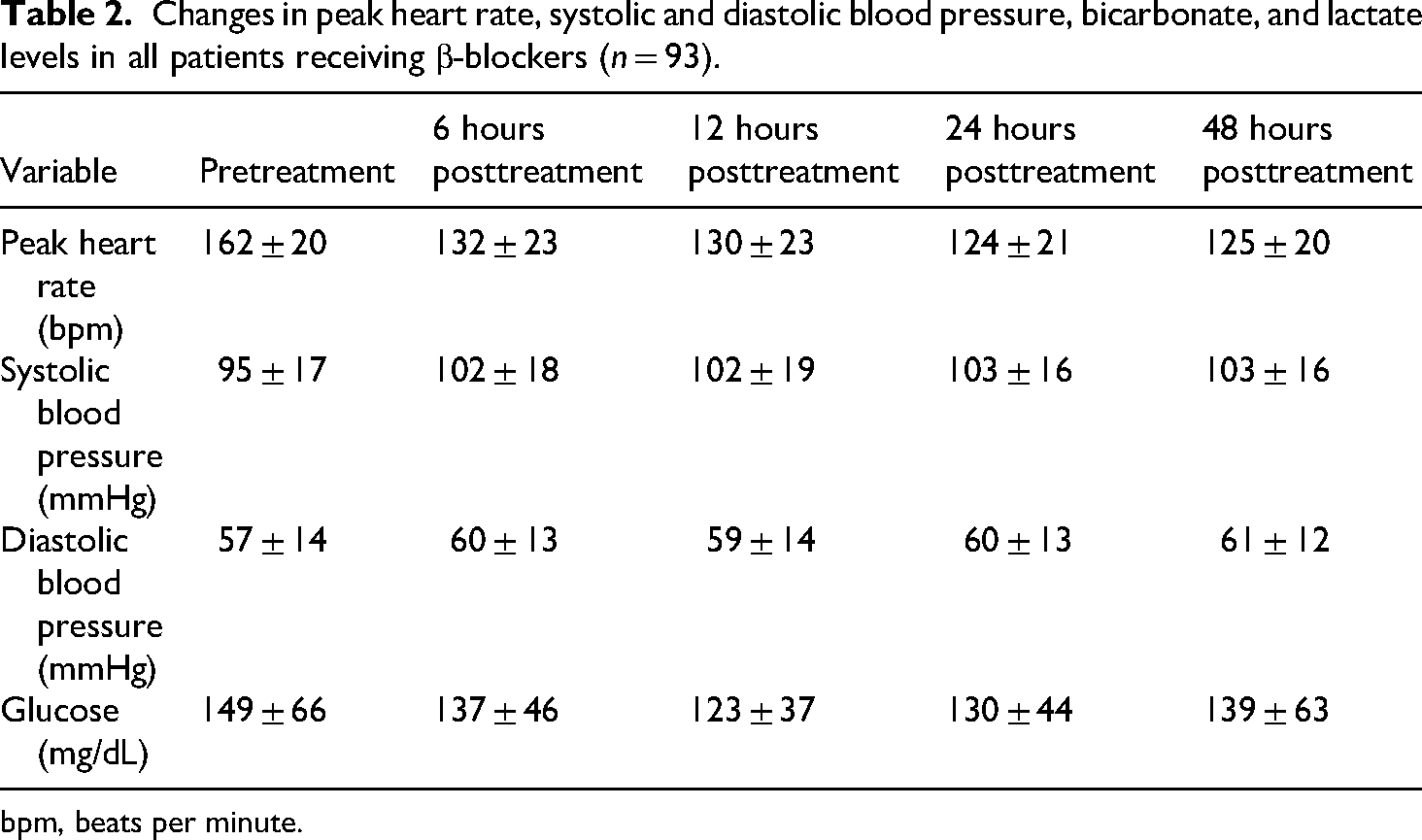
bpm, beats per minute.
Lactate dynamics
Lactate levels dropped significantly after starting propranolol, especially within the first 6 h. In the total cohort (n = 93), median lactate decreased from 1.7 mmol/L (IQR: 0.9–2.6) at baseline to 1.2 mmol/L (IQR: 0.9–1.9) at 6 h (p = 0.003), remaining stable afterward. Among patients with sepsis (n = 71), median lactate levels modestly declined from 1.7 mmol/L (IQR: 0.9–2.7) at baseline to 1.2 mmol/L (IQR: 0.9–2.0) at 6 h, with a continued downward trend through 48 h (1.3, IQR: 0.9–1.8), though these changes were not statistically significant. In patients on vasoactive therapy (n = 32), lactate levels did not change significantly (decreased from 1.8 mmol/L (1.0–2.9) at baseline to 1.3 mmol/L (0.9–2.5) at 6 h). As shown in Figure 3, early reductions were more prominent in the overall group and sepsis subgroup, but a slight rebound in lactate levels was observed after 24 h (Table 3 and Figure 3).

Changes lactate levels over time in patients receiving β-blockers cohort and subgroups before and after treatment.
Changes in lactate levels in all patients receiving β-blockers, patients with sepsis and patients receiving vasoactive therapy.

IQR, interquartile range.
Vasoactive–Inotropic Score
Among patients receiving vasoactive therapy, the difference in VIS between pretreatment and 6 h after propranolol initiation (16.1 vs. 15.5, p = 0.097) was not statistically significant. Similarly, numerical increases observed between 6–12 h (15.5 vs. 19.75, p = 0.318) and 12–24 h (19.75 vs. 20.00, p = 0.914) did not reach statistical significance. However, the decrease in VIS between 24 and 48 h posttreatment was statistically significant (20.0 vs. 10.0, p = 0.006) (Table 4).
Changes in vasoactive–inotropic score in patients receiving vasoactive therapy (n = 32).

IQR, interquartile range.
Subgroup analysis by age groups
For additional characterization of our cohort, subgroup analyses were performed according to age, following commonly used classifications in the pediatric critical care literature (0–6 months, 6–11 months, 1–5 years, 6–10 years, and ≥11 years). These analyses demonstrated statistically significant heterogeneity in illness severity across age groups: PRISM-III (p = 0.008), PELOD-2 (p = 0.006), and PELOD-mortality scores (p = 0.034) differed significantly, with the highest severity indices observed in the 0–6-month and ≥11-year groups. Hemodynamic parameters likewise showed age-dependent variation. Baseline SBP and DBP values were significantly different between groups (both p < 0.01), and this pattern persisted at several early follow-up time points (6, 12, 24, and 48 h). Peak HR also demonstrated significant between-group variation at baseline and throughout the first 48 h, consistent with expected developmental cardiovascular differences. In contrast, metabolic markers showed no meaningful age-related differences: lactate concentrations did not differ significantly at baseline or any subsequent time point; similarly, glucose, bicarbonate, potassium, and pH values were comparable across age categories—with the exception of a small but statistically significant baseline difference in pH (p = 0.002), which was not present thereafter. Treatment-related parameters, including propranolol dose (mg/kg/day), timing of initiation, and duration of therapy, did not differ across age groups, nor did VISs or PICU length of stay. Together, these findings indicate that although younger infants and older children exhibited higher severity-of-illness scores and distinct baseline hemodynamic profiles, age did not appear to modify metabolic responses or treatment exposure in our cohort (Figure 4).
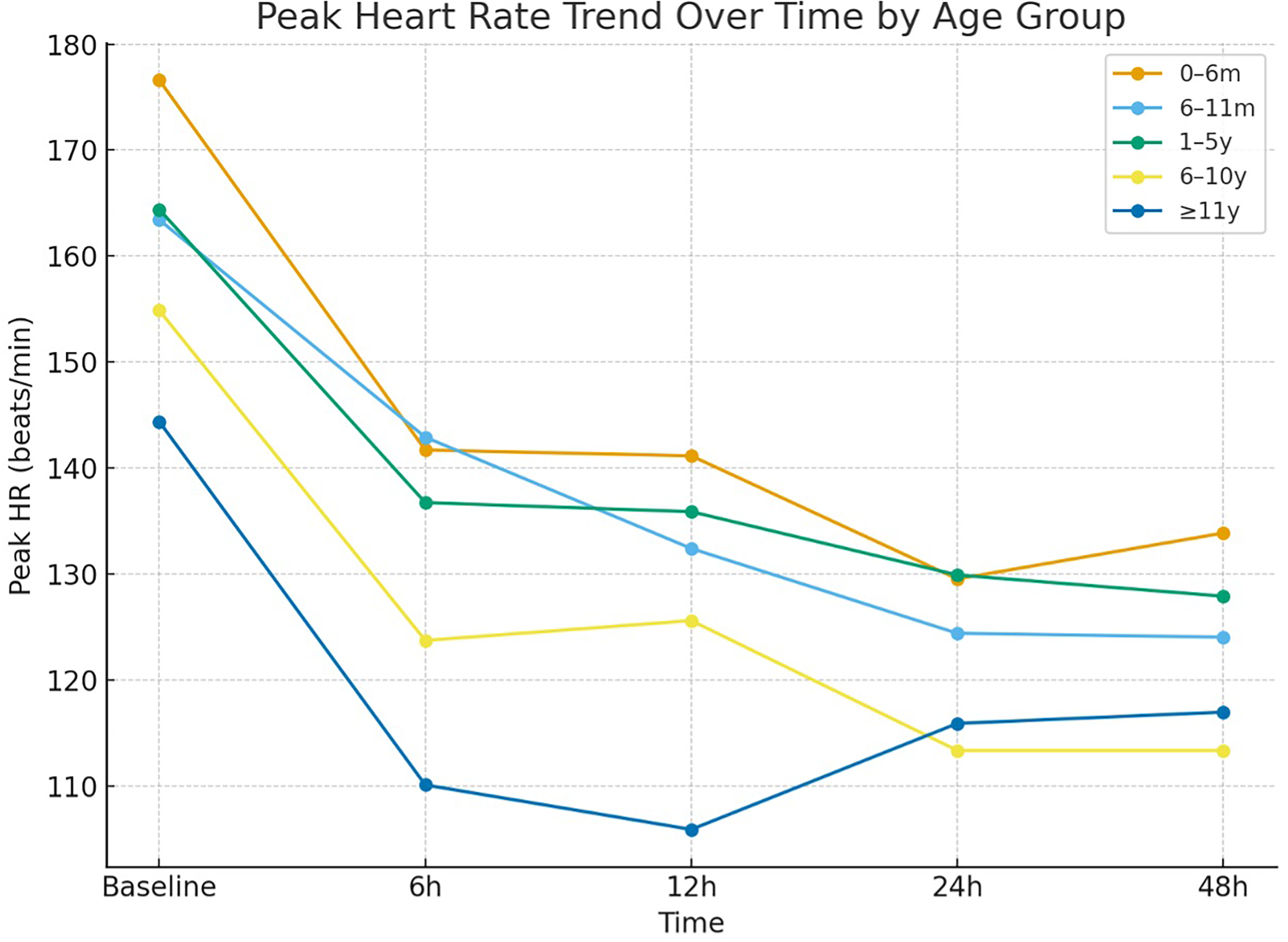
Peak heart rate over time by age groups. HR, heart rate.
Subgroup analysis by infection source
To explore whether the clinical profile and physiological response to propranolol differed by infection source, we performed a subgroup analysis within the sepsis cohort. Consistent with prior pediatric sepsis literature, patients were categorized into six groups: pneumonia/lower respiratory tract infection (n = 37), bloodstream infection (n = 18), intraabdominal / gastrointestinal infection (n = 6), central nervous system infection (n = 5), urinary tract infection (n = 3), and bone and soft-tissue infection (n = 2). Illness-severity indices demonstrated significant between-group variation for PELOD-2 (p = 0.004), whereas PRISM-III (p = 0.367) and PELOD-mortality scores (p = 0.379) did not differ significantly across infection sources. Baseline peak HR values were broadly similar among groups, although patients with pneumonia/lower respiratory tract infection, urinary tract infection, and central nervous system infection exhibited slightly higher means at presentation. Time-trend evaluation from baseline through 48 h demonstrated parallel declines in HR across all infection categories, with no statistically significant differences at any subsequent time point (all p > 0.05). Systolic blood pressure and DBP trajectories also displayed comparable patterns across subgroups, and ANOVA results showed no significant group effects for any time point. Similarly, lactate concentrations at baseline and during follow-up did not differ between infection sources (all p > 0.05), indicating a uniform metabolic response profile. Treatment-related variables—including propranolol dose, initiation time, and duration—were largely similar between groups, except for a modest difference in total duration of therapy (p = 0.034). Collectively, these findings suggest that while PELOD-2 scores varied by infection source, hemodynamic and metabolic responses to propranolol were broadly consistent across the major etiological categories of sepsis in this cohort (Figure 5).

Peak heart rate over time by infection source. HR, heart rate.
Discussion
This study is among the few to systematically assess the hemodynamic effects of β-blocker therapy—specifically propranolol—in critically ill pediatric patients with persistent sinus tachycardia. Our findings demonstrate that, when applied in a carefully selected PICU population, propranolol is associated with a favorable safety profile and results in significant and sustained HR reduction. Notably, these changes occurred without hemodynamic deterioration (e.g. hypotension) and were accompanied by stabilization or modest improvement in blood pressure and lactate levels.
The stress response in critical illness is characterized by excessive catecholamine release, which—if sustained—can result in cardiotoxicity and vascular dysregulation. 17 Experimental studies suggest that β1-adrenergic signaling plays a central role in this process, causing substantial cardiomyocyte loss and functional impairment.13,14,18 These findings reinforce the hypothesis that β1-adrenergic receptor signaling is the principal mediator of catecholamine-induced cardiotoxicity. Beta-blockers may offer systemic protection by attenuating the effects of excessive catecholamine stimulation. In septic shock, inducible nitric oxide synthase (iNOS) and the resulting overproduction of nitric oxide (NO) contribute to hypotension and vascular hyporesponsiveness. 15 Experimental models have shown that esmolol infusion suppresses iNOS expression in vascular tissue19,20 and simultaneously upregulates α1-adrenergic receptor mRNA expression. 19 Since downregulation of α1-receptors is a key factor in sepsis-related vasoplegia, β-blockers—despite lacking direct action on vascular β1-receptors—may help restore vascular tone and improve vasopressor responsiveness by indirectly reversing α1-receptor suppression.
In pediatric populations, most studies have focused on burn patients. For example, Herndon et al. demonstrated that long-term propranolol therapy in children with burn injuries significantly reduced HR, and this reduction persisted at one year post-burn. Although they reported a slight decrease in mean arterial pressure, there was no increase in the incidence of hypotension. 21 Jeschke et al. showed that propranolol combined with recombinant human growth hormone attenuated the inflammatory cascade and reduced hypermetabolism in severely burned pediatric patients. The authors reported that this regimen reduced inflammatory mediators such as TNF, IL-6, IL-8, and cortisol, while lowering HR, preserving lean body mass, and avoiding significant hypotension or bronchospasm. 22 In another study, Jeschke et al. found that propranolol significantly reduced serum tumor necrosis factor and interleukin-1β levels compared with controls, without increasing the incidence of infection or sepsis. 6
Consistent with these findings, our study also demonstrated that propranolol reduced HR in pediatric patients with tachycardia refractory to conventional treatment. Hypotension occurred in only two patients, one of whom developed hypotensive shock secondary to pneumothorax—an event unrelated to β-blocker therapy. Among the 32 patients receiving vasoactive therapy, VIS values showed only mild fluctuations from baseline to 48 h posttreatment, with no statistically significant overall change (an initial, nonsignificant rise in the first 24 h followed by a significant decline between 24 and 48 h). Compared with baseline, VIS at 48 h remained statistically unchanged. The relatively small number of patients on vasoactive support in our cohort may have contributed to the lack of statistical significance. Considering the absence of a control group and the limited number of patients receiving vasoactive therapy, the most reliable conclusion from our data is that propranolol did not increase vasoactive requirements. However, larger randomized controlled trials are warranted to better determine whether β-blocker therapy influences vasoactive support needs.
In pediatric burn populations, propranolol is most frequently used at a dose of 4 mg/kg/day, 4 although some studies have reported varying regimens. Ojeda et al., in an effort to determine the most effective and safe dose across different pediatric age groups, titrated propranolol to achieve at least a 15–20% reduction in HR. The authors reported target doses of 5.2 ± 2.8 mg/kg/day for ages 0–3 years, 4.2 ± 1.8 mg/kg/day for ages 4–10 years, and 2.9 ± 1.4 mg/kg/day for ages 11–18 years. 23 Martinez et al. reported that in the presence of vasopressor infusion and endogenous catecholamine release from shock, β-adrenergic blockade with propranolol could lead to severe splanchnic vasoconstriction from unopposed α-adrenergic activity, potentially resulting in intestinal ischemia. 5 In our cohort, the absence of lactate elevation in any subgroup suggests that splanchnic perfusion was not compromised. Furthermore, our use of lower propranolol doses compared with those reported in the burn literature may have contributed to the low incidence of adverse effects. A slight rebound in lactate levels was observed after 24 h in both the overall cohort and the sepsis subgroup. Several factors may account for this finding. First, lactate dynamics in critically ill children are known to be influenced by the natural progression of the underlying disease, including fluctuations in tissue perfusion, inflammatory burden, and metabolic stress, particularly during the early phases of critical illness. Second, lactate levels may vary in response to ongoing therapeutic interventions, such as adjustments in vasoactive support, fluid resuscitation, or mechanical ventilation, which can transiently affect oxygen delivery and utilization. Additionally, physiologic variability and the timing of lactate measurements relative to clinical interventions may contribute to modest increases observed at later time points. Given the observational nature of the study and the lack of a control group, this lactate rebound cannot be attributed specifically to propranolol administration. Rather, it likely reflects the complex and dynamic metabolic milieu of critically ill pediatric patients. Further prospective studies with standardized lactate monitoring protocols are needed to better characterize lactate trajectories in this setting.
A meta-analysis involving 2103 critically ill adult patients across 11 studies demonstrated a significant mortality benefit for those treated with β-blockers (risk ratio: 0.65; 95% CI: 0.53–0.79; p < 0.0001) compared to controls. 24 In a study by Zhou et al., critically ill adult patients with acute heart failure were categorized based on β-blocker use: Newly Started, Withdrawn, Continued, and Not Treated. Both the Newly Started and Continued groups had significantly lower in-hospital mortality compared to the Not Treated group (OR: 0.45, 95% CI: 0.34–0.61; and OR: 0.53, 95% CI: 0.41–0.69, respectively; both p < 0.001). Conversely, β-blocker withdrawal was associated with increased mortality risk (OR: 2.59, 95% CI: 1.63–4.10; p < 0.001). 25
A randomized, single-center clinical trial previously demonstrated that continuous β-blocker infusion in adult patients with persistent tachycardia following fluid resuscitation led to improved cardiovascular performance—including reductions in HR, increased stroke volume, enhanced SVR and decreased lactate levels—while concurrently reducing vasopressor requirements without notable adverse effects. 26
Although β-blockers are generally associated with reductions in blood pressure, propranolol may lead to modest increases in systolic and diastolic pressures in selected critically ill children with high sympathetic tone. In hyperadrenergic states, excessive tachycardia shortens diastolic filling time and limits effective stroke volume. By reducing HR and attenuating adrenergic drive, propranolol may improve ventricular filling and stroke volume, resulting in modest improvements in blood pressure. 27 Our findings are consistent with prior observations in hyperadrenergic conditions, including pediatric sepsis, where HR control has been associated with improved hemodynamic efficiency.
In our study, we similarly observed a marked reduction in HR within the first 6–24 h following propranolol initiation, with stabilization thereafter. This pattern aligns with prior adult and preclinical studies indicating that β-adrenergic blockade blunts excessive sympathetic activation and enhances myocardial efficiency. Importantly, the observed HR reductions were achieved without precipitating clinically significant bradycardia or hypotension, reinforcing the notion that propranolol may have a safe and effective therapeutic window in selected pediatric critical care populations. In both the overall cohort and key subgroups (sepsis and vasoactive support), SBP and DBP showed modest but statistically significant increases at 6 h posttreatment.
These results challenge long-standing caution regarding β-blocker use in critically ill patients with marginal perfusion status. Furthermore, lactate kinetics provided supportive evidence of improved tissue oxygenation. Both in the overall cohort and among patients with sepsis, lactate levels significantly declined within 6 h and remained stable, consistent with enhanced oxygen delivery-utilization balance after HR modulation. Although changes in lactate among patients on vasoactive agents did not reach statistical significance, the observed numerical decreases and subsequent stabilization suggest potential clinical benefit, particularly in mitigating catecholamine-induced metabolic stress.
While no early changes were observed in the VIS, a statistically significant reduction emerged at 48 h. This delayed improvement likely reflects enhanced myocardial efficiency and afterload reduction secondary to HR control, ultimately facilitating vasopressor de-escalation. The decline in VIS further substantiates the safety and potential hemodynamic benefit of propranolol when carefully titrated and monitored in pediatric ICU settings.
In the present study, the median PICU and hospital LOS were 10 and 20 days, respectively. These values are comparable to those reported in previous pediatric sepsis and critical illness studies in which β-blocker therapy was not routinely used, where median PICU stays typically range between 7 and 14 days depending on illness severity and comorbid conditions. The absence of a significant difference in LOS between septic and nonseptic patients suggests that propranolol administration did not appear to prolong critical care or hospital stay in this cohort.
The observed overall mortality rate of 2.2% is lower than mortality rates reported in many pediatric sepsis and septic shock studies, which commonly range from 5% to 20%, particularly among patients requiring vasoactive support. Importantly, both deaths in our cohort occurred in patients with severe underlying conditions and advanced critical illness at presentation, including immunodeficiency, hemophagocytic lymphohistiocytosis, and post-liver transplantation septic shock.
While these findings may suggest that propranolol administration was not associated with increased mortality, causal conclusions cannot be drawn due to the observational design of the study and the absence of a control group. Nevertheless, the low mortality rate and favorable safety profile observed in this cohort support the feasibility of propranolol use in selected pediatric critical care populations and warrant further investigation in controlled studies.
This study has several limitations that should be considered. First, its retrospective, single-center design inherently restricts the generalizability of the findings and introduces potential selection and information bias. Although strict inclusion and exclusion criteria were applied, residual confounding—such as differences in underlying diagnoses, illness severity, and concurrent treatments—could not be entirely eliminated. Second, the decision to start propranolol was at the discretion of the attending physician, which may have caused treatment allocation bias. Third, although serial measurements of hemodynamic and metabolic parameters were taken, causality cannot be determined from these observational data. Fourth, the absence of a comparison group limits our ability to assess the relative effectiveness or safety of propranolol compared to other β-blockers or alternative HR control methods. Fifth, the lack of long-term follow-up data prevents conclusions about functional recovery or postdischarge morbidity and mortality.
We also recognize that HR reduction—though an expected effect of β-blockade—does not, on its own, serve as a definitive therapeutic endpoint. Without a control group, it is impossible to determine whether the observed reductions were solely due to propranolol or also related to clinical improvement. Our selection of the primary endpoint reflects the exploratory nature of this study and the limited pediatric data available in this field. Future prospective, controlled trials should include more clinically relevant outcomes, such as mortality, organ dysfunction scores, or duration of vasoactive support, to better define the therapeutic role of propranolol in critically ill children.
Finally, a major limitation is the inability to retrospectively confirm with certainty that all patients received optimal sedation and analgesia before starting propranolol. Although our unit routinely uses validated scales such as the Comfort B 28 to guide analgesia, these were not consistently documented in a structured format that could be easily extracted. As a result, while the clinical impression was that tachycardia continued despite adequate sedation and analgesia, this cannot be objectively confirmed for every patient.
Despite these limitations, our study has significant strengths. To our knowledge, it is one of the first and largest to systematically evaluate the hemodynamic and clinical effects of nonselective β-blockade—specifically propranolol—in critically ill pediatric patients with persistent sinus tachycardia. Patient selection was strict, with clear inclusion and exclusion criteria that minimized confounding from primary cardiac disease. The thorough collection of serial physiological parameters—including HR, blood pressure, lactate, and VIS—allowed for a detailed assessment of treatment response over time. Additionally, subgroup analyses of patients with sepsis and those receiving vasoactive support improve the relevance of our findings across various clinical scenarios. Importantly, propranolol was administered with close monitoring, without significant hemodynamic instability, highlighting its feasibility in a population traditionally considered high risk for β-blocker therapy.
Conclusion
In conclusion, the use of propranolol in carefully selected critically ill pediatric patients with persistent sinus tachycardia seems to be safe and may provide meaningful hemodynamic and metabolic benefits. These findings align with previous adult, pediatric, and preclinical data and offer important new insights into pediatric critical care. However, prospective multicenter randomized controlled trials are necessary to establish causality, determine optimal dosing and timing, and refine patient selection criteria. Such evidence will be vital for defining the role of β-blockade as a safe and effective adjunct therapy in pediatric intensive care settings.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261424393 - Supplemental material for Beta-blocker therapy in pediatric intensive care: Safety and efficacy of propranolol for persistent tachycardia
Supplemental material, sj-docx-1-sci-10.1177_00368504261424393 for Beta-blocker therapy in pediatric intensive care: Safety and efficacy of propranolol for persistent tachycardia by Emrullah Aygüler, Servet Yüce, Muhammed Susuz, Beste Günebakan and Demet Demirkol in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504261424393 - Supplemental material for Beta-blocker therapy in pediatric intensive care: Safety and efficacy of propranolol for persistent tachycardia
Supplemental material, sj-docx-2-sci-10.1177_00368504261424393 for Beta-blocker therapy in pediatric intensive care: Safety and efficacy of propranolol for persistent tachycardia by Emrullah Aygüler, Servet Yüce, Muhammed Susuz, Beste Günebakan and Demet Demirkol in Science Progress
Footnotes
Acknowledgements
The authors would like to thank the staff of our intensive care unit for their valuable support in data collection for this study.
Ethical approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki and was reviewed and approved by the Institutional Ethics Committee of Istanbul University, Istanbul Faculty of Medicine (Date: 7 March 2025, Decision No: 05, File No: 2025/295).
Authors’ contributions
Emrullah Aygüler and Demet Demirkol conceptualized the study. Muhammed Susuz and Beste Günebakan collected the data and contributed to the data analysis. Servet Yüce and Emrullah Aygüler prepared Figures 1–2 and Tables 1–4. Demet Demirkol supervised all study processes. All authors read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of supporting data
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
