Abstract
Objectives
The COVID-19-associated hyperinflammatory syndrome (cHIS) score has been proposed as a tool for identifying patients at risk of clinical deterioration. This study evaluated the predictive accuracy of the admission cHIS score for invasive mechanical ventilation and ICU mortality in critically ill COVID-19 patients.
Methods
We conducted a single-center retrospective observational study including 85 adults with laboratory-confirmed COVID-19 who were admitted to the ICU. The cHIS score was calculated on admission, and patients were stratified into two groups (<3 and ≥3). Associations with mechanical ventilation and ICU mortality were examined using ROC curve analysis and multivariable logistic regression, adjusted for age and comorbidity burden.
Results
An admission cHIS score ≥3 demonstrated moderate discrimination for ICU mortality (AUROC = 0.70; sensitivity = 0.76; specificity = 0.66) and mechanical ventilation (AUROC = 0.71; sensitivity = 0.73; specificity = 0.65). Higher cHIS scores were significantly associated with both outcomes in unadjusted analyses. After adjustment, the associations were attenuated and became borderline for mortality and mechanical ventilation, suggesting potential confounding by age and underlying comorbidities. Key inflammatory markers within the cHIS score—CRP, LDH, and D-dimer—showed the strongest individual associations with adverse outcomes. Kaplan–Meier analysis demonstrated significantly reduced survival probabilities in patients with cHIS ≥3.
Conclusion
The admission cHIS score reflects the degree of systemic hyperinflammation and is associated with greater illness severity, ICU mortality, and the need for mechanical ventilation. Although its predictive value diminishes after adjusting for age and comorbidities, the score remains a useful adjunct for early risk stratification in critically ill COVID-19 patients. Larger multicenter studies are needed to determine its independent prognostic utility.
Introduction
Severe COVID-19 is characterized by a dysregulated hyperinflammatory response driven by macrophage and T-cell activation, resulting in excessive release of cytokines such as IL-6, IL-1β, and TNF-α. 1 This immune overstimulation contributes to diffuse alveolar damage, endothelial injury, increased vascular permeability, and multiorgan dysfunction. Such systemic inflammation plays a central role in the development of acute respiratory failure and the need for advanced organ support in critically ill patients.2,3
To quantify this hyperinflammatory state, several scoring tools have been proposed, among which the COVID-19-associated hyperinflammatory syndrome (cHIS) score is one of the most widely used. The cHIS score incorporates clinical and laboratory markers of systemic inflammation and has been suggested as a tool for identifying patients at risk of disease progression. Since hyperinflammation is directly linked to lung injury, coagulopathy, and metabolic dysfunction, elevated cHIS scores may theoretically be associated with two major clinical outcomes in the intensive care unit (ICU): the need for invasive mechanical ventilation and mortality.4,5
Early identification of patients likely to require mechanical ventilation or at high risk of ICU mortality is clinically important. Accurate prognosis allows timely escalation of care, optimal allocation of ICU resources, and closer monitoring of high-risk patients. Determining whether the cHIS score can predict these outcomes may therefore assist clinicians in recognizing impending deterioration earlier in the disease course. 6
The aim of this study was to evaluate the predictive accuracy of the cHIS score for the need for mechanical ventilation and ICU mortality in critically ill COVID-19 patients.
Material and methods
Study design
This was a single-center, retrospective, observational study conducted in the ICU of the Tuzla University Clinical Center, a tertiary referral center in Bosnia and Herzegovina. All data were extracted retrospectively from the electronic medical records and paper charts of patients admitted during the study period. No experimental interventions were performed. The study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024. The University Clinical Center Ethics Committee approved it on 20 January 2021 (No. 02-09/2-82/20). The reporting of this study conforms to STROBE guidelines. 7 Since this study was a retrospective analysis with full anonymization of all collected data, individual informed consent was not required.
Participants
A total of 85 laboratory-confirmed critically ill and hospitalized COVID-19 patients who were ≥18 years of age were enrolled in this study. The patients’ body temperature, laboratory markers of hyperinflammation, length of stay, comorbidities, demographic data, and clinical outcomes (mechanical ventilation requirement after ICU admission, and mortality) were recorded.
The inclusion criteria were a positive polymerase chain reaction (PCR) test for SARS-CoV-2 and admission to the ICU. Patients who received immunomodulatory therapies before admission or who had incomplete medical records were excluded.
Data measurement
Hyperinflammatory response was evaluated through the cHIS score, which was calculated on admission to ICU. The following parameters were recorded: (1) fever (>38°C); (2) macrophage activation with ferritin ≥700 µg/L; (3) hematological dysfunction with neutrophil to lymphocyte ratio (NLR) ≥ 10 or hemoglobin ≤9.2 g/dL and platelet count ≤110,000 cells/L; (4) coagulopathy with D-dimer ≥1.5 µg/mL; (5) hepatic injury with lactate dehydrogenase (LDH) ≥ 400 U/L or aspartate aminotransferase (AST) ≥ 100 U/L; and (6) cytokinemia with interleukin-6 (IL-6) ≥ 15 pg/mL or triglyceride ≥150 mg/dL or C-reactive protein (CRP) ≥ 15 mg/dL. Each of the six parameters was credited with one point, and the sum of all parameters represents the cHIS score. Cut-off points were specified (CHIS score 3), and all the values greater than the cut-off point were taken to predict mortality.
Patients were divided into two groups: those with cHIS scores less than 3, and those with cHIS scores of 3 or higher. The groups were compared according to their clinical outcomes.
Statistical analysis
Descriptive statistics were used to report clinical and demographic characteristics, laboratory values, and clinical outcomes. Continuous variables were presented as median and interquartile range (IQR 25th–75th percentiles), and categorical variables as frequencies and percentages. The normality of the data was assessed using the Kolmogorov–Smirnov goodness-of-fit test. Comparisons between independent groups were performed using the Mann–Whitney
Primary analyses focused on the association between the cHIS score at ICU admission and two clinical outcomes: ICU mortality and the requirement for mechanical ventilation. Predictive performance was evaluated using receiver operating characteristic (ROC) curve analysis. The area under the curve was calculated, and its 95% confidence interval (CI) was estimated using DeLong's method. Optimal cut-off values were determined on the basis of the Youden index to maximize sensitivity and specificity.
To account for potential confounders, multivariable logistic regression models were used. Variables with established clinical relevance, age, and total number of comorbidities, were included as covariates alongside the cHIS score.
Separate logistic regression models were used for ICU mortality and mechanical ventilation, and the results were expressed as the odds ratios (OR) with 95% CIs.
All statistical analyses were conducted using the R (version 4.5.1), and statistical significance was indicated by
Results
Descriptive data
A total of 85 patients were enrolled in this retrospective study, comprising 28 females and 57 males. Patients’ ages ranged from 31 to 78 years, and they were categorized into five age groups (Figure 1). The difference in numbers between male and female patients was statistically significant (
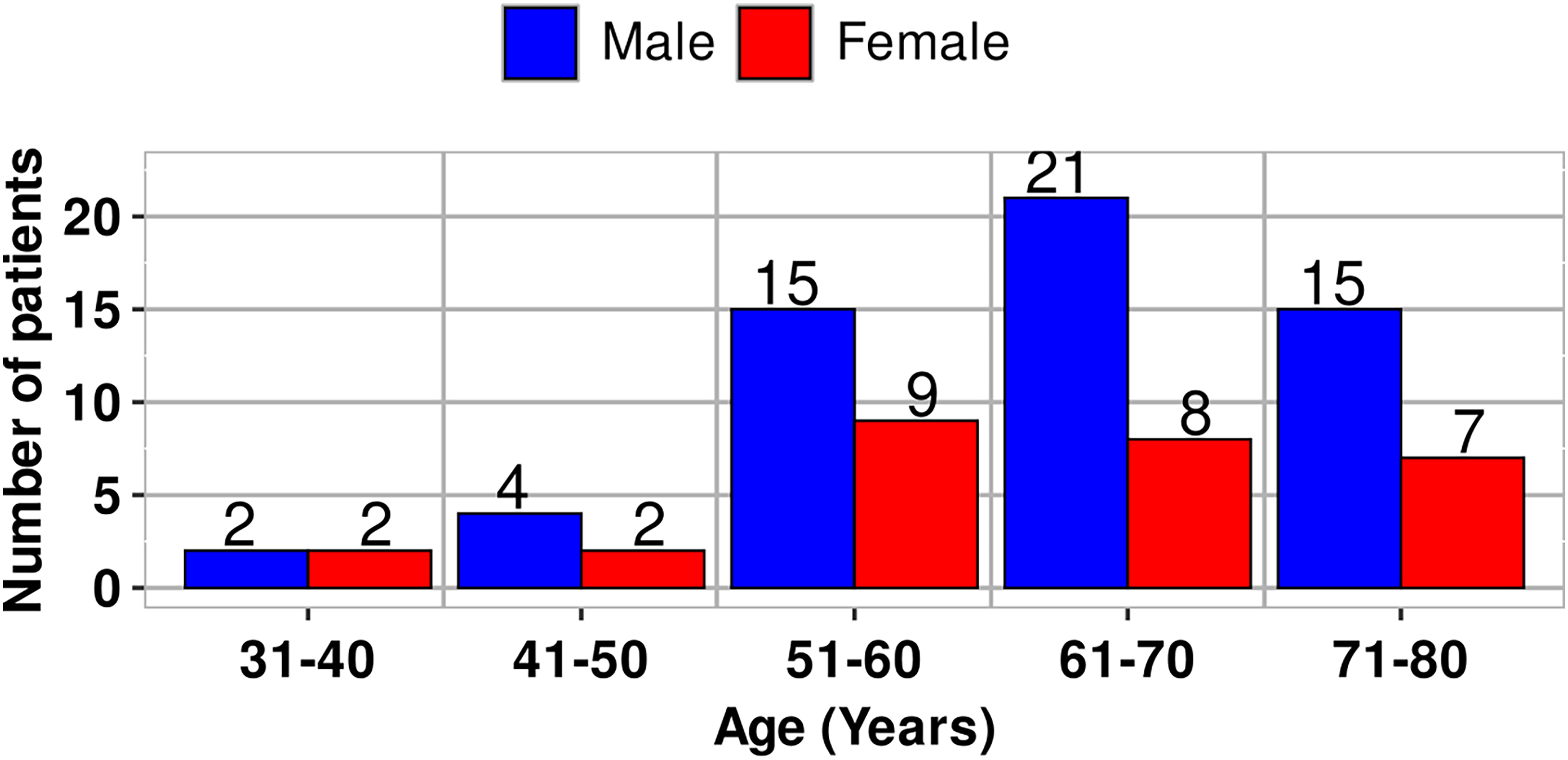
Age and sex distribution of patients.
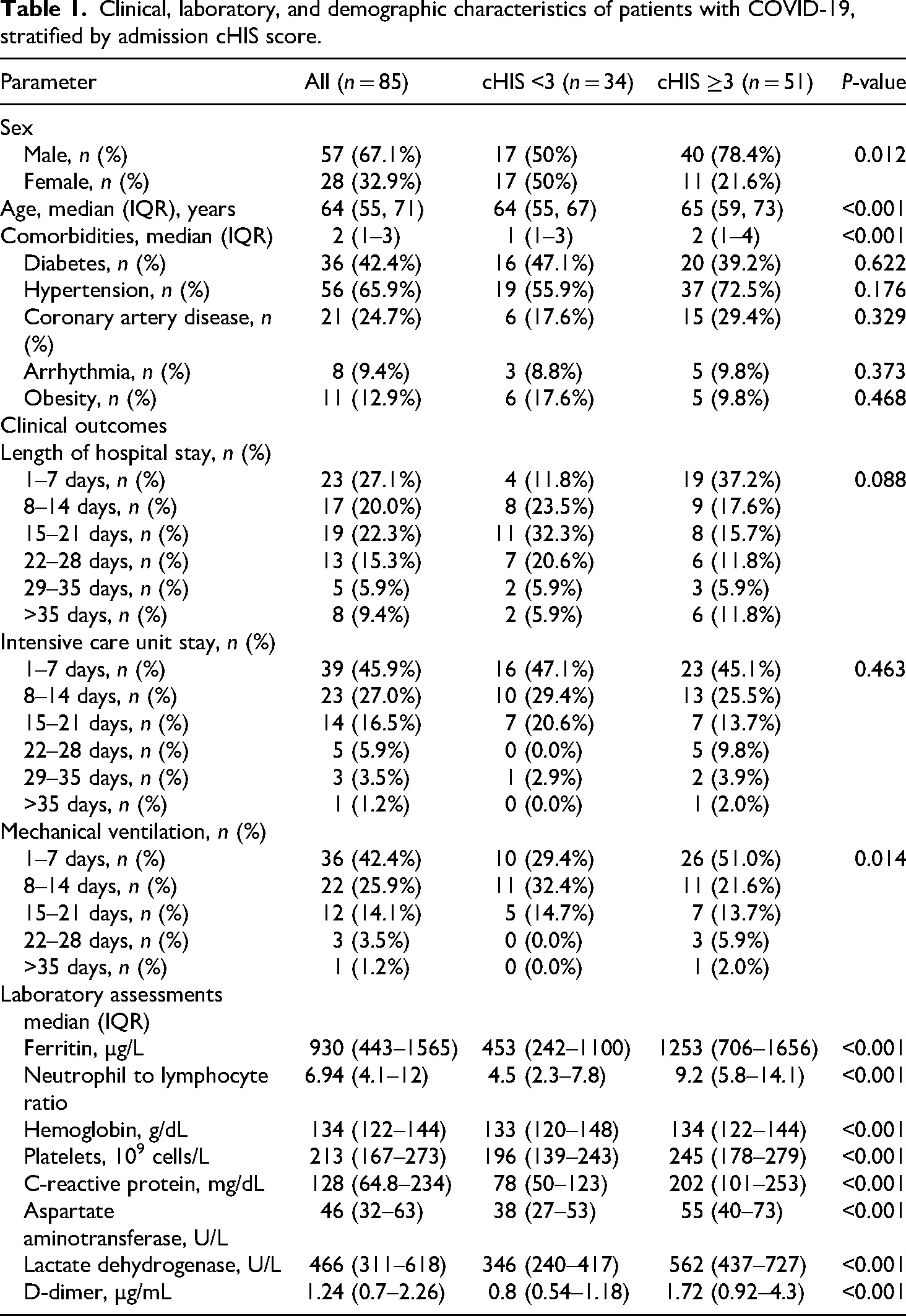
The cHIS scores for 85 patients were calculated. The clinical, laboratory, and demographic characteristics of patients with COVID-19 were stratified by admission cHIS score (Table 1). The median admission cHIS score was 3 (IQR 2–4). All data connected with two subsets of patients, those with cHIS<3 and the second classified group with cHIS ≥ 3, are given in Tables 1 and 2.
Clinical, laboratory, and demographic characteristics of patients with COVID-19, stratified by admission cHIS score.
Summary of length of hospital stay, intensive care unit stay, and mechanical ventilation of patients with COVID-19.
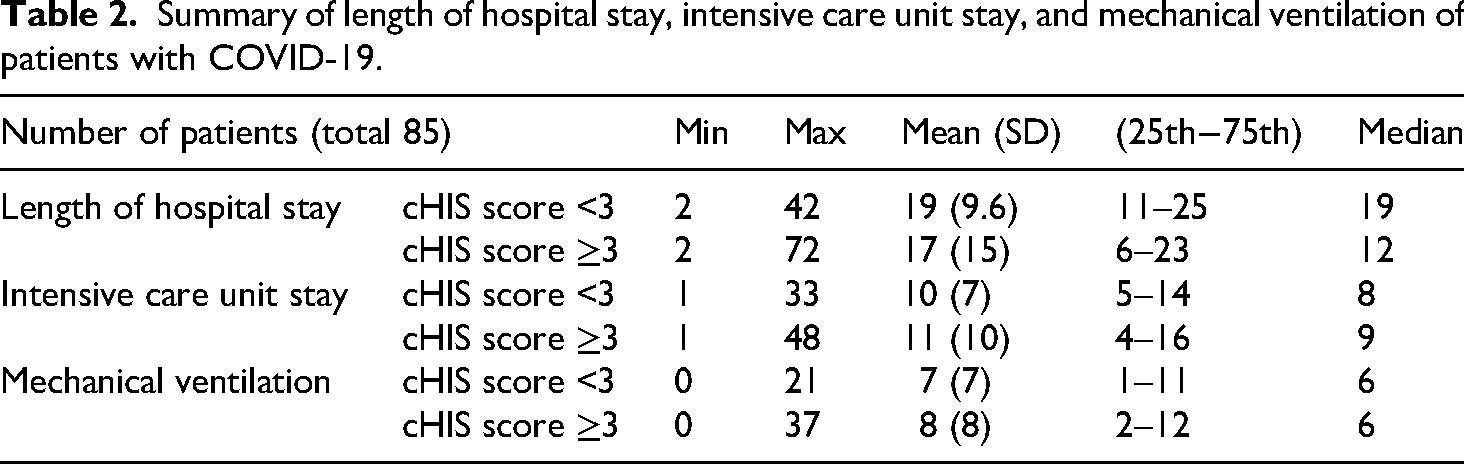
It is important to note that this shorter median length of hospital stay, ICU stay, and mechanical ventilation in the high cHIS group (Table 2) is a consequence of early mortality. When the analysis was restricted to survivors, this group required a longer median duration of hospital stay (27 days compared to 20 days), ICU stay (13 days compared to 8 days), and mechanical ventilation (6 days compared to 1 day) (Table 2).
In addition to the disparities in length of hospital and ICU stays, notable variances were also observed in the primary laboratory parameters of the cHIS score. Specifically, patients with a cHIS score of 3 or higher exhibited ferritin levels (measured in µg/L) nearly three times greater than those in the group with a score of less than 3 (Table 1).
Although the number of patients within the two classified groups is almost the same up to age 60, a greater difference is observed above age 61 in favor of patients with cHIS > 3 (Figure 2).

Age distribution of patients per cHIS score.
In Figure 3, median values and IQR are shown for the length of hospital stay, the ICU stay, and mechanical ventilation. Patients with a score of less than 3 had significantly different outcomes compared to those with a score of 3 or higher across several clinical endpoints. The median length of hospital stays for patients with a score of less than 3 was 19 days (IQR 11–25), while it was 12 days (IQR 6–23) for those with a score of 3 or more.

Box and Whisker plot for clinical outcomes.
Main results
The area under the receiver operating characteristic curve (AUROC) was used to assess the cHIS discrimination ability, while Youden Index Analysis was used to determine the optimal cut-off values, including the sensitivity and specificity of the cHIS score. On the basis of the Youden index, an admission cHIS score ≥3 had the best accuracy for ICU mortality, and the AUROC was 0.70 (95% CI 0.55–0.83, sensitivity = 0.76, specificity = 0.66). For mechanical ventilation, the cHIS score was also ≥3, and the AUROC was 0.71 (95% CI 0.57–0.81, sensitivity = 0.73, specificity = 0.65) (Figure 4).
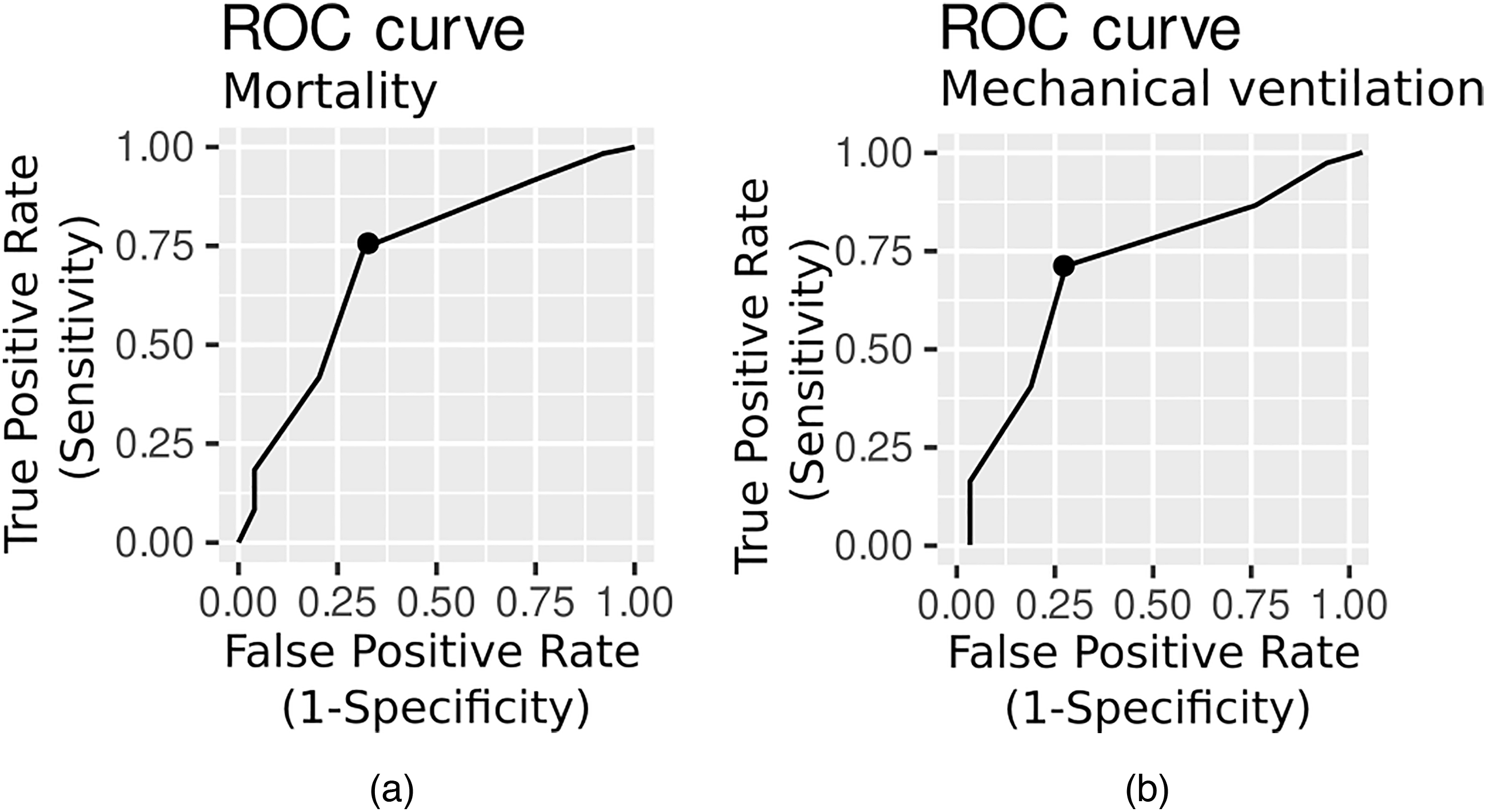
ROC curve for intensive care unit mortality (a) and mechanical ventilation (b).
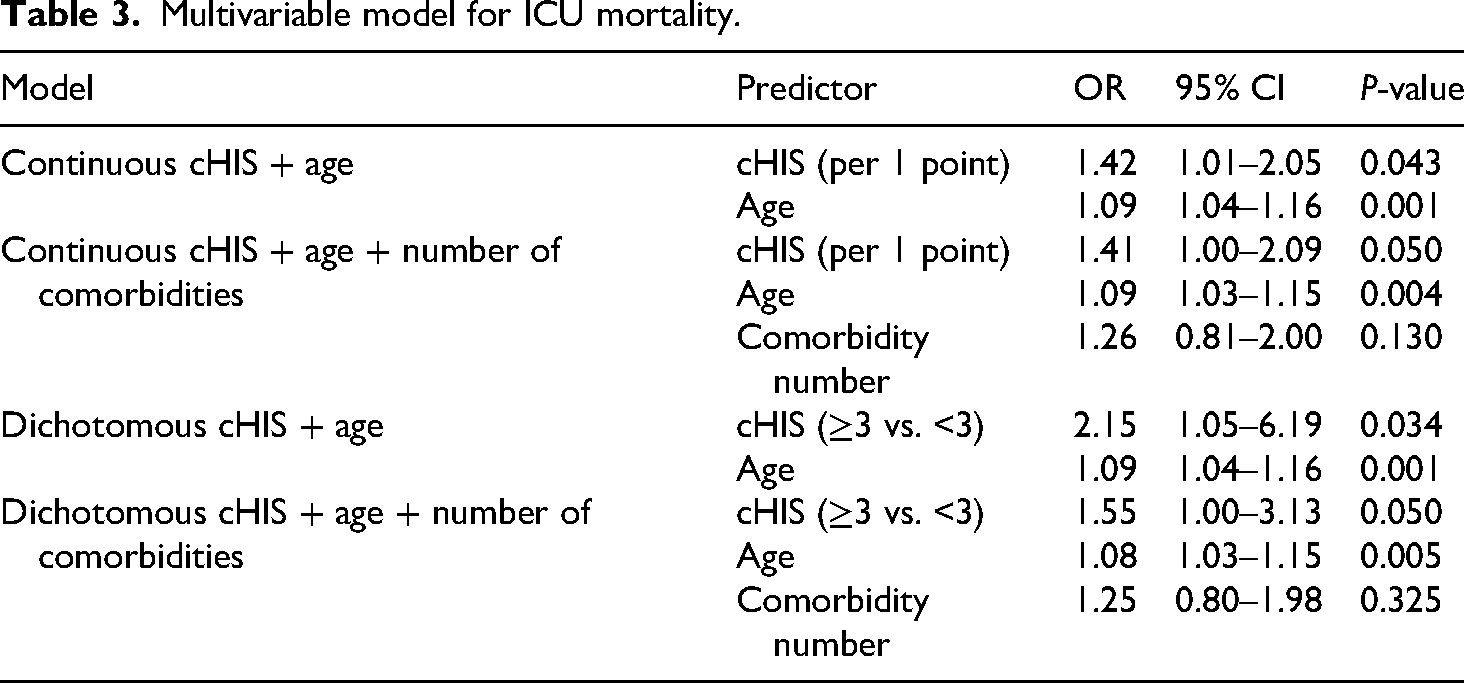
In bivariate analyses, the cHIS score demonstrated significant associations with both primary outcomes. When treated as a continuous variable (per 1-point increase), the cHIS score was significantly associated with ICU mortality (OR 1.45, 95% CI 1.05–2.13,
Using a predefined cut-off to define hyperinflammation, the dichotomous cHIS variable was significantly associated with ICU mortality (OR 4.92, 95% CI 1.31–23.9,
When adjusted for age and the number of comorbidities, the continuous cHIS score showed a borderline significant association with ICU mortality (OR 1.41, 95% CI 1.00–2.09,
For mechanical ventilation, the continuous cHIS score was a significant predictor in the fully adjusted model (OR 1.57, 95% CI 1.01–2.75,
Age was consistently and significantly associated with both outcomes, but the number of comorbidities was not independently predictive. A summary of these results is presented in Tables 3 and 4.
Multivariable model for ICU mortality.
Multivariable model for mechanical ventilation.

A post-hoc sensitivity analysis was used to evaluate the frequency and association of individual cHIS criteria with clinical outcomes. At a threshold of three or higher, the scale demonstrated exceptional sensitivity in predicting both mechanical ventilation and mortality. Concerning the laboratory parameters incorporated in the cHIS score, the preceding analysis yielded the following results: NLR (sensitivity = 0.70, specificity = 0.73, AUROC = 0.67), CRP (sensitivity = 0.61, specificity = 0.82, AUROC = 0.72), LDH (sensitivity = 0.24, specificity = 1.00, AUROC = 0.61), and D-dimer (sensitivity = 0.69, specificity = 0.73, AUROC = 0.71) were most specifically associated with mechanical ventilation. For mortality, CRP (sensitivity = 0.84, specificity = 0.59, AUROC = 0.74), LDH (sensitivity = 0.63, specificity = 0.94, AUROC = 0.80) and D-dimer (sensitivity = 0.67, specificity = 0.80, AUROC = 0.77) were the most important variables.
Patients with a cHIS score ≥3 had significantly worse survival probabilities than those with a cHIS score <3, according to the Kaplan–Meier survival analysis (log-rank test

The Kaplan–Meier survival function.
Discussion
The cHIS score was designed to estimate the degree of hyperinflammation in COVID-19 patients admitted to the hospital by calculating the maximum daily cHIS score. 4 Others evaluated the cHIS score on admission, focusing specifically on patients treated in the ICU. 6 Although the original description of the cHIS score proposes evaluating the daily maximum to capture evolving hyperinflammation, this approach may not always reflect a clearly defined stage of illness, and may be challenging to implement in busy ICU environments. Serial scoring would require repeated laboratory testing—particularly IL-6, ferritin, LDH, and D-dimer—which is not routinely feasible in many centers, especially during pandemic surges. For these reasons, our analysis focused on the admission cHIS score, which represents a pragmatic and clinically accessible approach. Admission values are routinely available, they reduce the burden of repeated testing, and still demonstrated good discriminatory ability for both mortality and mechanical ventilation in our cohort. While maximum daily scores may offer additional granularity, their practical applicability remains limited, and future studies should clarify whether serial cHIS monitoring provides superior prognostic value compared with a single admission assessment. High cHIS scores, low BMI, and the presence of chronic lung disease were important independent predictors of mortality across all cHIS scores, 8 but we also found that CRP, LDH, and D-dimer were predicting mortality.
The cHIS score shows acceptable discrimination for predicting ICU mortality and the need for mechanical ventilation in critically ill COVID-19 patients. The specified threshold values of the cHIS score vary across studies. One study 8 showed that a threshold score of 2 or more was predictive of both mortality and mechanical ventilation. In contrast, it was found that a cHIS score of 3 or more was associated with ICU mortality and invasive mechanical ventilation. 6 Further analysis 9 reported that a cHIS score of 2 or more was associated with higher odds of ICU admission, mechanical ventilation, and in-hospital mortality. It was demonstrated that a cHIS score of 3 or more was a better predictor of mortality, while a score of 2 or more was more predictive of the need for mechanical ventilation. 10 We found that the cHIS score could predict ICU mortality and the need for mechanical ventilation, with AUROC values of 0.70 and 0.71, respectively. In our study, the optimal cHIS cut-off of ≥3 was determined using the Youden index, which identifies the threshold that maximizes the balance between sensitivity and specificity within the internal dataset. As such, this cut-off is internally optimized and may not directly generalize to other patient populations. Our selection of ≥3 therefore represents the best-performing threshold in this specific cohort, but should be interpreted with caution and validated in external datasets.
Although the cHIS score showed good discriminatory performance, our adjusted analyses indicate that it may function primarily as a marker of systemic illness severity rather than as a fully independent predictor of mortality or mechanical ventilation. After adjustment for age and comorbidities, the associations between cHIS and the outcomes weakened, suggesting that the score may reflect the underlying disease burden and the global inflammatory state rather than exerting an independent prognostic effect. This interpretation is consistent with the biological function of the cHIS components, which capture features of advanced systemic inflammation. Therefore, while the cHIS score remains clinically useful for identifying high-risk patients, its predictive ability should be considered complementary to established risk factors rather than stand-alone.
Although regression models were adjusted for age and comorbidity burden, residual confounding remains possible, as older and more comorbid patients are more likely to exhibit elevated inflammatory markers associated with higher cHIS scores and to experience worse clinical outcomes.
However, this study has several limitations that should be acknowledged. First, the single-center design may limit the generalizability of our findings to other healthcare settings or patient populations. Second, the relatively small sample size (
Various attempts have been made to summarize clinical manifestations and laboratory findings, and facilitate better prediction of clinical outcomes.11–14 Among these efforts, the cHIS score has emerged as a promising tool for identifying patients at risk of poor outcomes. Early identification of high-risk patients using the cHIS score can facilitate timely interventions, such as immunomodulatory therapies or advanced respiratory support, potentially improving patient outcomes. Moreover, the cHIS score may also be valuable for other patients with hyperinflammatory syndromes, such as sepsis, septic shock, macrophage activation syndrome, systemic autoimmune diseases, or other viral infections.
However, this study has several limitations that should be acknowledged. First, the single-center design may limit the generalizability of our findings to other healthcare settings or patient populations. Second, the relatively small sample size (
Conclusion
The cHIS score provides a useful framework for identifying patients with marked hyperinflammation and greater overall illness severity. In our cohort, higher cHIS scores were associated with increased ICU mortality and mechanical ventilation; however, these associations were attenuated after adjustment for comorbidities and age. This suggests that the cHIS score may function more as a reflection of the underlying systemic inflammatory burden rather than as an independent prognostic tool. Nevertheless, the score remains clinically valuable for early recognition of high-risk patients and may complement existing clinical assessment strategies. Further multicenter studies with larger samples are needed to clarify its independent predictive role and to determine its utility across diverse critically ill populations.
Footnotes
Author contributions
JS: conception, methodology, validation, investigation, interpretation of data, writing, supervision; FIH: analysis, interpretation of data, writing; LM: acquisition, analysis; SS: acquisition, analysis; ES: acquisition, analysis; SD: conception, methodology, supervision, writing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
