Abstract
Objective
To compare long-term survival in early-stage breast cancer patients treated with different radiation therapy modalities.
Methods
Data was retrospectively derived from SEER database. We compared overall survival (OS), breast cancer specific survival (BCSS) and second primary malignancies (SPM) in early-stage breast cancer patients treated with postoperative radiotherapy (PORT) versus those treated neoadjuvant radiotherapy (NART) and intraoperative radiotherapy (IORT) after propensity score matching by 1:1.
Results
A total of 457,166 patients were included in this study. After matching, the 20-year OS of 1441 patients in NART cohort was lower than that in PORT cohort (p < 0.01), particularly in hormone receptor positive patients (p < 0.01). NART were dependent prognostic factors for 20-year OS [Hazard Ratio (HR):1.21, 95%CI: 1.06–1.38, p < 0.01). No significant difference in BCSS was observed between NART and PORT treatments. Additionally, patients undergoing NART had a lower risk of all SPM (p = 0.01) and second solid cancers (p = 0.02) but a comparable risk of second hematological malignancies (p = 0.55) than patients administered PORT. HR-positive was a risk factor for SPM. No OS, BCSS or SPM risk difference were significantly observed in the 2096 pairs of IORT and PORT groups.
Conclusion
Compared to PORT, NART and IORT don’t offer survival advantages for early-stage breast cancer patients. Altering the sequence of radiotherapy requires careful evaluation.
Keywords
Introduction
Globally, breast cancer is the most frequently diagnosed cancer and the leading cause of cancer-related death in women.1,2 The reduction in breast cancer mortality has been achieved through a multidisciplinary approach involving surgical oncology, radiation oncology, and medical oncology. 3 Following breast-conserving surgery (BCS), adjuvant radiotherapy (RT) after surgery (postoperative radiotherapy, PORT) to the tumor bed or the entire breast is the current standard of care for most patients. 4 Recent advancements in radiation oncology have introduced various RT modalities such as neoadjuvant radiotherapy (NART) administered before surgery or intraoperative radiotherapy administered during surgery (IORT). NART is particularly employed in locally advanced patients to downstage tumors, thus enhancing surgical feasibility and as a palliative measure to alleviate symptoms and improve quality of life. 5 It has been found that NART enables turn “cold” tumors into “hot” tumors, thereby enhancing their responsiveness to immunotherapy by immunogenic cell death and radiation in situ vaccine effect. 6 PORT, on the other hand, effectively controls residual but minimal tumor burden and reduces recurrence risk when the surgical margin is negative. 3 Despite the use of whole-breast radiation therapy, 90% of recurrences after BCS occur in the quadrant of initial lesion. 7 IORT offers precise targeting of the tumor bed, ensuring accurate irradiation and reducing the initiation time and duration of RT.8,9 Moreover, the alteration of cytokines in wound effusion after IORT can counteract the stimulatory effect of wound effusion on cell proliferation, invasion and migration. 10 Consequently, NART and IORT present potential alternatives to PORT for early-stage breast cancer patients undergoing BCS due to their radiobiological and immunological benefits.
Despite evidences supporting the role of RT in enhancing local control and long-term survival in BCS patients,11,12 recent studies indicate an increased risk of second primary tumors (SPM) in breast cancer patients receiving RT.13,14 Population-based analyses examining the long-term therapeutic effects of NART and IORT in early-stage breast cancer are limited. Thus, this study aims to assess the overall survival (OS) and breast cancer specific survival (BCSS) in early-stage breast cancer patients undergoing BCS who received NART or IORT stratified by hormone receptor (HR). Additionally, we sought to elucidate differences in SPM risk between patients treated with NART and IORT to provide evidence for optimizing prognosis and follow-up strategies.
Material and methods
Database and study population
We conducted a retrospective cohort study utilizing data from the Surveillance, Epidemiology, and End Results (SEER) Program database. To ensure sufficient long-term follow-up for 20-year survival and SPM analyses, our analysis incorporated all registry centers available since 1975, and duplicate entries across registries were removed. Given the retrospective nature of this study, various patient diagnosis times, and the highly skewed data distribution (98.42% PORT), we employed a propensity score matching (PSM) for outcomes comparisons between the IORT, NART, and PORT group. This study was performed in accordance with the National Cancer Institute (NCI)'s SEER limited-use data agreement. The SEER database was accessed using the SEER*Stat software provided by the NCI and the data was reorganized using a series of ASCII files to calculate OS, BCSS, and SPM for patients with primary breast cancer. All procedures involving human participants were conducted in accordance with the 1975 Helsinki Declaration as revised in 2024. All patient information was de-identified prior to analysis. Given its retrospective design and use of public data, the Ethics Committee of the National Cancer Center of China waived ethical approval. The reporting of this study followed the STROBE statement for observational studies. 15
Patient identification
Between 1975 and 2020, we identified all female patients diagnosed with unilateral localized breast cancer (Tis-T3; N0M0) who had no prior history of other cancers. These patients were categorized into three groups: the NART group, the IORT group, and the PORT group. All selected cases received radiation treatment and underwent BCS. Patients with non-breast epithelial malignant tumors or insufficient pathological types were excluded from the study. Figure S1 presents the flow of patient inclusion and the subsequent matching process in this study.
Study variables and study outcomes
We firstly extracted the following variables from the selected cohorts that included patient ID, sex, age, tumor behavior, year at diagnosis, race, laterality, pathological patterns, AJCC T Stages, type of surgery, radiation record, HR status, survival months and vital status. All tumor-related information was derived from the primary tumor. Optimal cutoffs for year of diagnosis were determined using X-Tile. 16 The year 2009 was selected as the optimal dichotomization point, which is consistent with previous reports 17 and corresponds to the transitional era of advanced radiotherapy technologies. 18 The last follow-up date is November 2020. The primary endpoint was OS, which was defined as the time from diagnosis to death or the last date of follow-up for review. BCSS is a secondary study endpoint calculated from the date of diagnosis until the date of death from breast cancer, with deaths from other causes treated as censored events. Survival outcomes included both 10-year and 20-year OS and BCSS, so patients who were alive at the end of 10 years or 20 years were censored for analysis respectively. SPM was the final outcome of interest, defined as a new and metachronous malignancy occurring after the initial diagnosis of breast cancer. After the PSM matching, information on the second tumor was extracted for the paired patients, including year of diagnosis, tumor site, tumor behavior, and laterality. Breast cancers occurring at the same site as the primary tumor were classified as recurrences. Only malignant tumors were considered in the assessment of SPM risk.
Statistical analysis
For PSM model, 19 propensity scores were calculated using a multivariable logistic regression model incorporating age, year of diagnosis, race, histological grade, and tumor size. A 1:1 nearest neighbor matching without replacement was performed with a caliper width of 0.2 standard deviation of the logit of the propensity score. Clinical materials of all pairs were complete, including follow-up records. Paired t-tests were used to compare continuous variables in matched cohorts, while the Cochran Q test was employed for categorical variables. Survival differences were estimated using Kaplan-Meier (KM) analysis. Multivariate Cox proportional hazards models were utilized to estimate hazard ratio, identifying factors associated with OS. Additionally, Fine-Gray's competing risk models were used to estimate the cumulative incidence of SPM over time following the diagnosis of breast cancer, and multivariable competing risk models were conducted to identify significant risk factors. All statistical analyses were performed using R version 4.3.0 software.
Results
Demographic and clinicopathological characteristics
The cohort comprised 455,133 early-stage breast cancer patients undergoing BCS. Group PORT represented 98.42% of the total study population, while Groups NART and IORT accounted for 0.58%, and 1.00% of the population, respectively. Detailed demographic and clinicopathological characteristics of the cohort are summarized in Table S1. Figure S2 shows the differences in the distribution of diagnosis years between patient groups before PSM. Following PSM with the PORT cohort, patients diagnosed before 1990 could not be successfully matched and were therefore excluded for survival analyses (Figure S3). Ultimately, 1441 patients from the NART cohort and 2096 patients from the IORT cohort were included in further survival analyses. There were no significant differences in histology, HR status, T stage, year of diagnosis, age, or race between the matched pairs (Table 1).
Baseline characteristics of the propensity score-matched cohorts.

Abbreviations: HR, hormone receptor; PORT, postoperative radiotherapy; NART, neoadjuvant radiotherapy; IORT, intraoperative radiotherapy.
Survival outcomes
The KM survival curve demonstrated that the 20-year OS of the PORT group was superior to that of the NART group. (Figure 1, p < 0.01). Subgroup analysis revealed that this difference persisted in the HR-positive subgroup (Figure 1, p < 0.01), while no statistically significant difference was observed between the PORT and NART groups in the HR-negative subgroup (Figure 1, p = 0.31). However, there was no significant difference in long-term survival over 20 years between the IOPT and PORT cohorts (Figure 1, p = 0.46). In addition, no significant difference in 10-year OS was observed between the NART and PORT groups (Figure S4, p = 0.13), or between the IORT and PORT groups (Figure S4, p = 0.58). Regarding BCSS, neither NART nor IORT treatments showed statistically significant differences compared to PORT treatment at the 20-year (Figure 1) and 10-year (Figure S4) follow-ups. The cumulative incidence of all SPMs (Figure 2A, p = 0.01) and second solid cancers (Figure 2B, p = 0.02) but not second hematologic malignancies (Figure 2C, p = 0.55) was significantly higher in NART-treated patients compared to PORT-treated patients. However, there was no statistically significant risk of developing all SPMs, solid or hematologic malignancies between IORT-treated and PORT-treated patients (Figure 2).
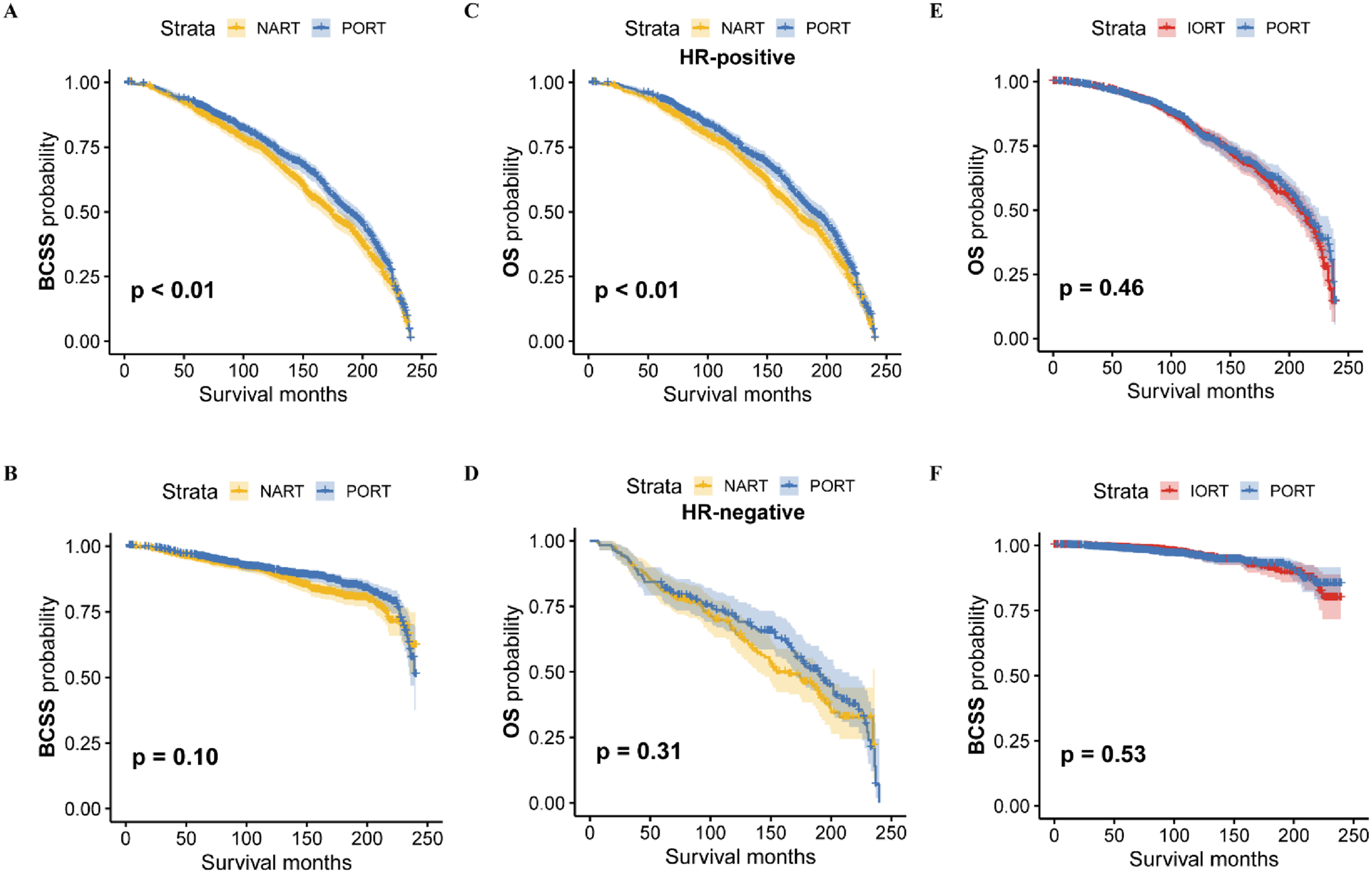
Comparison of 20-year survival curves for patients who underwent radiotherapy before, during, or after surgery. (A) and (B) Comparison of survival curves between cohort NART and PORT. (C) and (D) OS of cohort NART and PORT stratified by hormone receptor. (E) and (F) Comparison of OS and BCSS between the NART and PORT groups. Abbreviations: PORT, postoperative radiotherapy; NART, neoadjuvant radiotherapy; IORT, intraoperative radiotherapy; OS, overall survival; BCSS, breast cancer specific survival.

Cumulative incidence of second primary cancers. Abbreviations: (A) and (E) All second primary solid cancers (B) and (F) Second primary solid cancers. (C) and (G) All hematologic malignancies. Abbreviations: PORT, postoperative radiotherapy; NART, neoadjuvant radiotherapy; IORT, intraoperative radiotherapy.
Multivariate regression results
In the multivariate Cox proportional hazards model, HR status (positive or negative) did not meet proportional assumption and was therefore stratified into separate cohorts for KM curve (Figure 1) and Cox model analyses (Figure 3). The Hazard Ratio for 20-year OS was significantly higher among HR-positive patients treated with NART compared to those with PORT (Hazard Ratio:1.21, 95%CI: 1.06–1.38, p < 0.01). Additionally, patients staged with carcinoma in situ (Tis) had significantly better OS compared to patients with stage T1 tumors (Hazard Ratio: 0.71, 95%CI: 0.52–0.97, p = 0.03). Conversely, stage T2 tumors were associated with poorer long-term survival. Notably, patients treated more recently had a slightly lower hazard ratio for 20-year survival (Hazard Ratio: 0.66, 95%CI: 0.44–1.00, p < 0.05). The results of Fine-Gray's competing risk analysis indicated a lower risk of all SPMs (Hazard Ratio: 0.84, 95%CI: 0.72–0.97, p = 0.02) and solid carcinomas (Hazard Ratio: 0.84, 95%CI: 0.72–0.97, p = 0.01) in patients who received NART (Table 2). However, the risk for both all SPMs (Hazard Ratio: 1.25, 95%CI: 1.00–1.55, p < 0.05) and solid cancers (Hazard Ratio: 1.24, 95%CI: 1.01–1.53, p = 0.04) increased in patients with HR-positive tumors (Table 2).

Hazard Ratio of OS from a multivariate Cox proportional hazards mode in HR positive patients. Abbreviations: HR, hormone receptor; PORT, postoperative radiotherapy; NART, neoadjuvant radiotherapy.
Comparison of SPM risk between subgroups using Fine-Gray's competing risk analyses.

Abbreviations: HR, hormone receptor; PORT, postoperative radiotherapy; NART, neoadjuvant radiotherapy.
Discussion
Radiotherapy for cancer patients has undergone tremendous advances in technological innovations, biological and targeting strategies, and treatment delivery and optimization until now. 20 Beyond postoperative radiotherapy, the concepts of preoperative 21 and intraoperative radiotherapy 22 have been investigated to achieve better therapeutic outcomes and to meet the demands of personalized medicine. In this study, we employed a PSM model to minimize baseline differences and to investigate the long-term differences in overall survival (OS), breast cancer–specific survival (BCSS), and second primary malignancies (SPM) among the three radiotherapy modalities in early-stage breast cancer patients undergoing BCS. Although the study cohort spanned multiple decades, the final matched cohorts used for survival analyses exclusively included patients diagnosed from 1990 onward, as pre-1990 cases could not be successfully matched during PSM. This period coincides with the widespread adoption of CT-based radiotherapy planning, three-dimensional conformal radiotherapy, and subsequent intensity-modulated techniques, which substantially improved treatment precision and outcomes compared with earlier radiotherapy practices.20,23 Therefore, the results of this study predominantly reflect outcomes from the modern radiotherapy era and are relevant to contemporary clinical practice.
Our results showed that the cumulative incidence of SPM was lower in the NART group than in the PORT group, the 20-year OS of patients in the NART group is also significantly lower. It is worth noting that a lower 20-year OS can be observed in the NART with HR-positive group, but not in the NART with HR-negative group. This indicates that NART may not provide the same survival benefit in HR-positive patients as PORT. With respect to IORT, no significant differences in long-term OS, BCSS, or SPM risk were observed compared with PORT. This finding suggests that IORT may represent a viable therapeutic alternative in carefully selected patients, just given its comparable long-term survival outcomes.
The lower OS observed in the NART group compared to the PORT group, despite the lower SPM risk, aligns with previous studies. Zhang et al. 24 reported similar poor OS after an average follow-up of 82.5 months. Poleszczuk et al. 25 suggested that while NART may improve cancer-free survival, it does not necessarily enhance overall survival. Our study corroborates these findings, highlighting the complexity of NART's effects and the importance of precise patient selection and treatment timing.
NART has been shown to enhance immune response and downstage tumors, thus improving the feasibility of surgical intervention.26,27 It can trigger a robust immune response by exposing large amounts of tumor-associated antigens and stress proteins, an immune effect potentially absent in patients receiving PORT alone.24,28 This immune response might explain the lower SPM risk in the NART group. However, our results suggest that these potential benefits do not translate into improved long-term OS in HR-positive patients. Although generally considered homogeneous, NART may in fact trigger distinct immune responses depending on HR status. HR-positive breast cancers tend to have a lower immunogenicity due to their lower burden of malignant mutations and lower degree of differentiation. 29 The expression of immune checkpoint PD-L1 is markedly lower in HR-positive breast cancer compared with triple-negative breast cancer (TNBC) (53.10% vs. 84.40%). 30 These differences in the immune microenvironment may contribute to the observed variations in prognosis. In clinical trials, the objective response rate (ORR) to pembrolizumab was 12.00% in HR-positive breast cancer, whereas a higher ORR of 18.50% was observed in TNBC.31,32 The JAVELIN trial further confirmed these findings, reporting ORRs of 2.80% and 5.20% for avelumab in HR-positive breast cancer and TNBC, respectively. 33 Moreover, our SPM risk analysis provides a plausible explanation for the disparity in OS, as patients with HR-positive disease exhibited an increased risk of SPM. In addition, the interval between NART and subsequent surgery or systemic therapy may influence tumor regression and surgical complexity, thereby affecting long-term survival. 34 All of these underscore the complexity of integrating NART into the treatment regimen for HR-positive breast cancer patients and underscore the need for precise timing and coordination of treatment modalities.
Our study found no significant difference in long-term OS and SPM risk between the IORT and PORT groups. To our knowledge, this is the first study comparing SPM risk between IORT and PORT in breast cancer. The OS findings are consistent with existing literatures.35–37 Vaidya et al. 36 reported no statistically significant difference in OS, breast cancer mortality, distant disease-free survival and local recurrence-free survival between IORT and PORT groups after 8.6 years of median follow-up. This suggests that IORT could be a viable alternative to PORT offering reduced toxicity and improved cosmetic outcomes. IORT offers precise radiation delivery to the tumor bed during surgery, reducing the risk of damaging surrounding healthy tissues and minimize radiation exposure to other organs. 8 Patients undergoing IORT experience similar wound-related complication compared to those with PORT but have less radiotherapy toxicity, better cosmetic results and improved quality of life. 38 IORT might be beneficial in settings where access to postoperative radiotherapy facilities is limited or where patient compliance with prolonged treatment regimens is a concern.39,40
However, some studies have raised concerns about higher local recurrence rates with IORT. 41 In TARGIT-A clinical trial, the risk of local recurrence was 2.11% with TARGIT-IORT, compared with 0.95% with external beam radiotherapy at the five-year follow-up. 36 The 15-year incidence of ipsilateral breast tumor recurrence was 11.00% in patients treated with IORT. 37 Based on American Society for Radiation Oncology (ASTRO) guidelines, IORT alone is not recommended as a treatment option for patients with early-stage invasive breast cancer. Whole breast irradiation following IORT is particularly advised for patients with higher risk features. 42 Anderson et al. indicate IORT may be especially suitable for patients with early-stage breast cancer who have clear surgical margins and low risk of extensive microscopic disease. 43 Therefore, treating IORT should be tailored to patient and tumor characteristics.
This study has several limitations that should be acknowledged. The retrospective nature of the data analysis introduces inherent biases, limiting the ability to control for all confounding factors and potentially affecting the generalizability of the findings. The reliance on historical data without randomization and the potential for selection bias necessitate cautious interpretation of the results. Additionally, the study lacks detailed systemic treatment data, such as chemotherapy and endocrine therapy, which are critical components of comprehensive breast cancer management. The absence of this information means that the impact of these systemic treatments on survival outcomes could not be fully assessed, potentially influencing the observed differences between the treatment groups. Moreover, the standards for NART administration were not consistent across the datasets. Variations in radiation doses, timing, and protocols may have affected the outcomes, making it challenging to draw definitive conclusions about the efficacy of NART compared to PORT and IORT.
Given these limitations, future research should focus on prospective randomized controlled trials to validate these findings. Such trials would provide more robust evidence by minimizing biases and allowing for better control of confounding variables. Prospective studies would also enable the standardization of treatment protocols, ensuring more consistent and comparable results. Furthermore, future research should explore the impact of radiotherapy on cosmetic outcomes and quality of life, particularly for patients undergoing breast reconstruction. The effects of different radiotherapy modalities on these aspects are crucial for informed decision-making and patient counseling. Understanding how treatments influence cosmetic results and quality of life will help tailor therapies to individual patient preferences and improve overall satisfaction with breast cancer care.
Conclusion
This study provides a comprehensive evaluation of the long-term outcomes associated with different radiotherapy modalities in early-stage breast cancer patients undergoing BCS. The key findings suggest that while NART and IORT offer potential as alternative treatments, they do not surpass the efficacy of PORT in ensuring long-term survival, particularly in HR-positive patients. NART, despite a lower SPM risk, was associated with lower overall survival compared to PORT. Similarly, IORT showed comparable long-term OS and SPM risk to PORT, making it a viable alternative in specific clinical situations for its particularly beneficial advantages. The potential for higher local recurrence with IORT underscores the need for careful patient selection, guided by tumor characteristics and treatment goals. Given these findings, PORT remains the standard adjuvant radiotherapy for early-stage breast cancer patients due to its demonstrated reliability in improving long-term survival outcomes. Thus, optimizing the use of these radiotherapy modalities will enhance treatment outcomes and improve the overall care of breast cancer patients.
Footnotes
Acknowledgements
We are grateful to the participants involved in the Surveillance, Epidemiology, and End Results (SEER) Program database.
Ethical approval and informed consent statements
All patient data extracted and case lists constructed from the SEER database, are anonymous and shared, without containing any identifiers. For this reason, this study was exempted from the approval by Ethics Committee of the National Cancer Center of China.
Consent for publication
Not applicable
Author contributions
Dr Jing Wang, Dr Zhongzhao Wang, Dr Yi Fang takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Lingrui Li, Xiangyi Kong Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: Lingrui Li, Xiangyi Kong, Xiangyu Wang. Critical revision of the manuscript for important intellectual content: Zhuang Xiong. Statistical analysis: Lingrui Li.
This work was supported by the Natural Science Foundation of China (No. 82371842), the Natural Science Foundation of China (No. 82173328), the CAMS Innovation Fund for Medical Scienceshe CAMS Innovation Fund for Medical Sciences (CIFMS) (No. 2024-I2M-ZH-006), the (CIFMS) (No. 2024-I2M-C&T-A-004), the CAMS Innovation Fund for Medical Sciences (CIFMS) (No. 2024-I2M-C&T-B-057), the Beijing Hope Run Special Fund of Cancer Foundation of China (No. LC2022A19), Medical and Health Science and Technology Innovation project of Chinese Academy of Medical Sciences (No.2022-I2M-1-009), the Special Research Fund for Central Universities, Peking Union Medical College (No.3332025175), the Natural Science Foundation of China (No.82473205)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed in this study are available from the corresponding author on reasonable request.
