Abstract
Objective
To estimate the prevalence of human papillomavirus (HPV) infection among males in Jinshan District, Shanghai, China.
Methods
Study design: single-center, retrospective cross-sectional study. Setting: Tinglin Hospital, Jinshan District, Shanghai, China. Participants: 116 males aged 22–71 years from outpatient, inpatient, and routine physical examination settings. Outcomes: overall HPV prevalence, age-specific prevalence, and genotype distribution (high-risk and low-risk).
Results
We first describe the overall and age-specific prevalence, followed by genotype distribution and the pattern of single versus multiple infections. A total of 116 males aged 22 to 71 years old were enrolled for investigation. The overall HPV infection rate was 31.90% (37/116). The infection rates of different HPV subtypes in various age groups were listed as follows: 12% (3/25) for the 22–30 age group, 27.59% (8/29) for the 31–40 age group, 50% (18/36) for the 41–50 age group, 16.67% (3/18) for the 51–60 age group, and 62.5% (5/8) for the over 60 age group. Among the infected individuals, 27 out of 116 (23.28%) suffered from single subtype infections, while 8 individuals (6.90%) endured dual infections, and 2 individuals (1.72%) had three or more subtypes detected. High-risk HPVs accounted for 30.17%, with the highest infection rates observed in HPVs including HPV-53, HPV-58, HPV-52, HPV-16, and HPV-31. In comparison, low-risk HPVs accounted for 12.93%, with the highest infection rates seen in HPVs including HPV-6, HPV-81, and HPV-11. Apart from that, HPVs such as HPV-18, HPV-59, HPV-73, HPV-70 and HPV-83 were not detected in any of the specimens.
Conclusion
Males are predominantly infected with a single-subtype HPV infection. The infection rate is highest in the age group of 60 and above, while the infection rate is the lowest in the age group of 22–30 years. It is essential to formulate corresponding strategies and screening plans for the elderly over 60 years to lower or prevent the risk of HPV infection.
Keywords
Introduction
Human papillomavirus (HPV) is a common sexually transmitted infection and a small double-stranded DNA virus. 1 More than 200 HPV genotypes have been found, which are divided into high-risk HPV (HR-HPV) and low-risk HPV (LR-HPV)2–4 according to potential to cause cancer in humans; HR-HPV can give rise to cervical cancer, vaginal cancer, vulvar cancer, anal cancer, oropharyngeal cancer, and penile cancer5–9; It can also affect the skin and genitals. 10 HPV infects both cutaneous and mucosal epithelia, leading to distinct clinical manifestations at the skin and anogenital sites. High-risk HPV infection in men has also been associated with infertility, as supported by several epidemiological studies.11–13 As pointed out by the World Health Organization, about 630 million people worldwide are infected with HPV, with an estimated prevalence rate of 9–13%. 38 The health community has recognized the significant impact of HPV on men's health. 14 As validated by relevant literature, HPV infection accounts for approximately 5% of the global cancer burden. 15 It is particularly noteworthy that there are few studies on male HPV infection.11–17 Nonetheless, Previous epidemiological studies have reported geographical variations in HPV infection patterns, underscoring the importance of region-specific data.18–20 HPV DNA testing is widely recognized as a reliable method for detecting infection.21,22 In such case, these findings underscore the need to strengthen male health care and implement targeted screening programs.
To inform male health guidance and research and development (R&D) of related vaccines in this region, we analyzed HPV test results from outpatients, inpatients, and routine physical examinations at Tinglin Hospital in Jinshan District, Shanghai, China.
Methods
Data collection
This single-center, retrospective cross-sectional study included 116 men aged 22–71 years who underwent HPV testing at Tinglin Hospital (Jinshan District, Shanghai) between January 2023 and April 2025, including outpatients, inpatients, and routine physical examinations. Participants were grouped into five age categories (22–30, 31–40, 41–50, 51–60, and >60 years; n = 25, 29, 36, 18, and 8, respectively).
Participants were consecutively recruited, meaning that all eligible men who underwent HPV testing at our hospital during the study period were included without selective sampling.
Inclusion and exclusion criteria
Inclusion criteria: (i) male patients aged 22–71 years; (ii) attended outpatient, inpatient, or routine physical examination services at Tinglin Hospital; (iii) provided penile specimens adequate for HPV DNA testing; and (iv) provided written informed consent.
Exclusion criteria: (i) insufficient or invalid specimens on quality control; (ii) prior treatment that could compromise HPV DNA detection (e.g. recent topical antiviral therapy at sampling sites); and (iii) missing essential demographic or laboratory information.
Instruments and reagents
The instruments used in this study were:
Golden Dean Automatic Nucleic Acid Extraction and Purification Instrument (EB-1000; Hangzhou Dean Biotechnology, China).
Real-time fluorescence quantitative PCR system (MA6000; Suzhou Array Biotechnology, China).
The reagents included:
HPV nucleic-acid typing assay (PCR melting-curve method).
Nucleic-acid extraction/purification kits.
A 23-genotype HPV typing kit.
Corresponding quality-control materials, all from Hangzhou Jindian Biotech.
The 6 low-risk HPV types were HPV-6, HPV-11, HPV-42, HPV-70, HPV-81, and HPV-83.
The 17 high-risk HPV types were HPV-16, HPV-18, HPV-31, HPV-33, HPV-35, HPV-39, HPV-45, HPV-51, HPV-52, HPV-53, HPV-56, HPV-58, HPV-59, HPV-66, HPV-68, HPV-73, and HPV-82.
Specimen collection and preservation
HPV specimens were all collected by clinical doctors who had attained the professional title of attending physician or above at our hospital. First, a moist HPV-specific brush was used to obtain a sufficient amount of exfoliated cell samples from the dorsal and ventral sides of the penis, the inner and outer surfaces of the coronal sulcus, the glans, and the distal part of the urethra. All specimens were collected from penile sites; no anal or oropharyngeal samples were obtained. The head of the cervical brush was put into the cell preservation solution, the brush handle was broken along the crease of the brush handle, and the cap of the cell preservation solution tube was screwed tightly. The samples were well labeled, and the cell preservation tube was kept in an upright position. Once the specimens were collected, they were sent for testing as promptly as possible. When stored at room temperature, they were not kept for more than 48 h. If they were stored in a refrigerator at 2–8°C, the storage period did not exceed 7 days, and the testing must have been completed within 1 week.
Ethics approval and informed consent
This study was conducted in accordance with the Declaration of Helsinki (1964) and its later amendments, most recently revised in 2024. The protocol was approved by the Ethics Committee of Tinglin Hospital, Jinshan District, Shanghai (approval No. TLYY-KYKT-2024-001) on 2024-03-15. All participant data were de-identified prior to analysis. Written informed consent was obtained from all participants prior to specimen collection. The reporting of this study conforms to the STROBE guidelines. 23
HPV nucleic acid extraction
After the HPV specimens were thoroughly vortexed and left to stand for 1–2 min, a 100–1000 microliter pipette was used to aspirate 400 microliters of the sample, which was then added into the nucleic acid extraction or purification reagent plate.
During each batch of HPV sample detection, quality control products (negative, weak-positive, and positive) with the same batch number as the reagents were randomly added into the wells of the nucleic acid extraction or purification reagent plate. Subsequently, the nucleic acid extraction or purification reagent plate was placed on the Jindi Safe Automatic Nucleic Acid Extraction and Purification Instrument (EB-1000) for automatic nucleic acid extraction.
The HPV reagents were taken out from a-20°C refrigerator and allowed to dissolve at room temperature (repeated freezing and thawing were not permitted). After that, they were strictly prepared according to the actual instructions and added into the wells of the nucleic acid amplification plate.
Once the nucleic acid extraction was completed, the nucleic acid extraction solution was sequentially added into the amplification plate wells. Then, a 0.2 mL PCR plate with a semi-skirt edge, which was transparent, enzyme-free, pyrogen-free, and had a transparent heat-sealing film, was used to cover the wells. The plate was then moved to a uniform heat-sealing film instrument (set at 165°C) for film sealing.
After that, the sealed amplification plate was placed into a microplate centrifuge (HL-25 W) and centrifuged for 3 min. Following centrifugation, the amplification plate was put into a real-time fluorescent quantitative PCR instrument (MA6000) for the amplification reaction.
HPV nucleic acid amplification and analysis of results
Amplification parameters and results were determined in strict accordance with the kit instructions. Quality control: For the negative quality control wells, there should be no Tm value in the amplification results of Plate A, while the internal control should exhibit a Tm value in the ROX channel of Plate B. For the weak-positive and positive quality controls, they should show positive results with Tm values at their corresponding positions in both Plate A and Plate B. Only when these conditions are met can the experiment be considered valid; otherwise, the experiment is deemed invalid.
Operation of the experiment personnel
All HPV nucleic acid testing personnel were qualified, having completed the training program on clinical gene amplification testing organized by the National Health Commission or the Shanghai Clinical Laboratory Center and passing both theoretical and practical assessments.
Statistical analysis
We used IBM SPSS version 25 (IBM Corp., Armonk, NY, USA) in the statistical analysis. The χ2 test was used for all age groups, and p < 0.05 was considered statistically significant. Fisher's exact test was applied when the expected counts were less than 5.
Results
Human papillomavirus infection among males at different ages
Among 116 penile HPV test specimens from males aged 22–71, a total of 37 positives were detected, yielding an overall penile HPV infection rate of 31.9%. As illustrated in Table 1, the 41–50 and >60-year age groups had the highest HPV infection rates. A statistically significant difference was observed between the 41–50 and 22–30-year age groups, whereas differences among the 22–30, 31–40, and 51–60-year age groups were not significant. The HPV infection rate of males differed significantly across age groups (χ2 = 15.60, P < 0.05). The distribution of HPV subtype infection rates across age groups is presented in Figure 1.
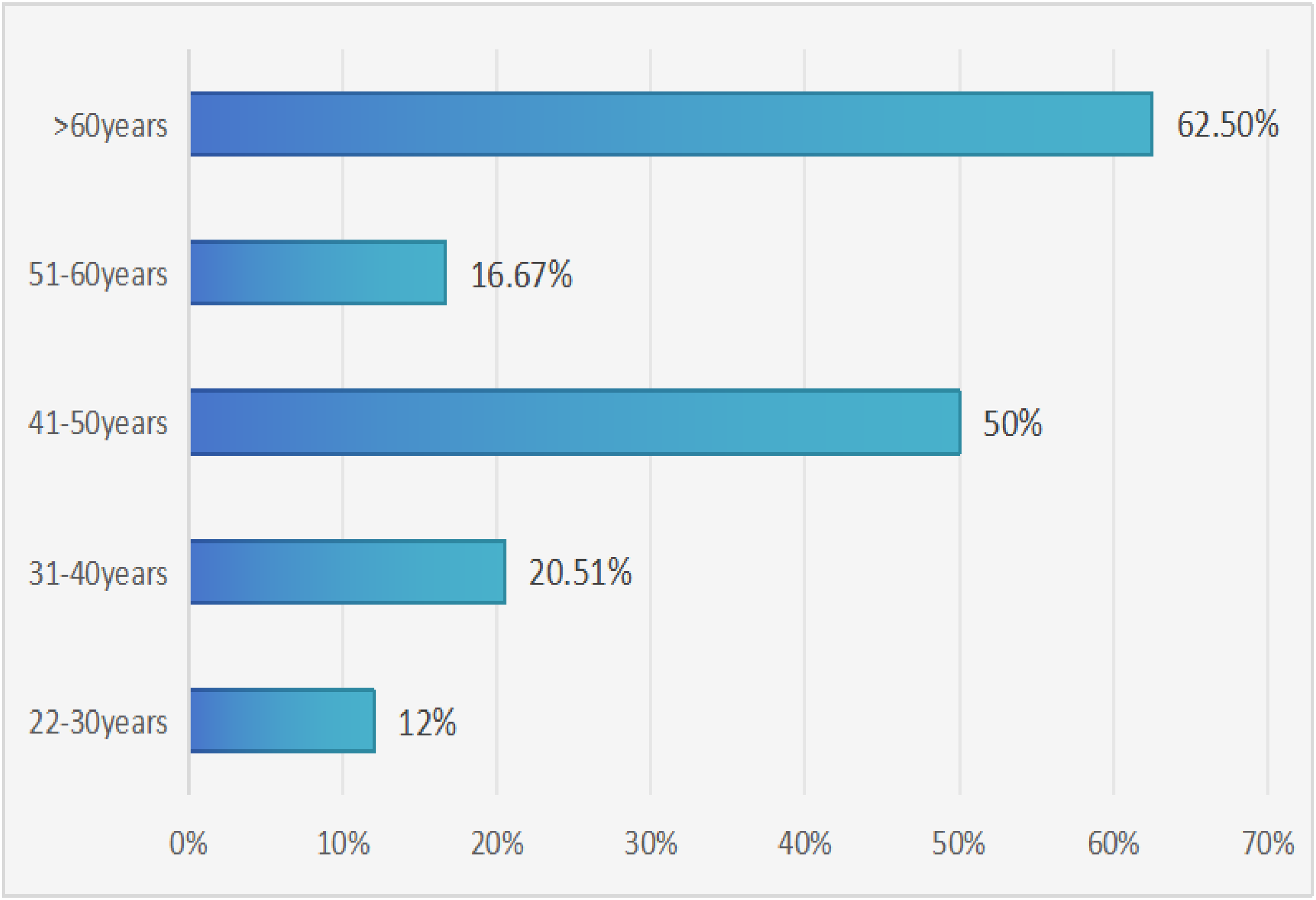
Age-specific HPV infection rate in males. x-axis: Infection rate (%); y-axis: Age group (years).

Distribution of high-risk HPV genotypes among infected males. Colors indicate individual genotypes (HPV-16, −31, −33, −35, −39, −45, −51, −52, −53, −56, −58, −66, −68, −82). Genotypes not detected but included in the panel: HPV-18, −59, −73.

Distribution of low-risk HPV subtypes among infected males.
Human papillomavirus infection among males at different ages.

Distribution of high- and low-risk HPV subtypes among males
The assay panel targeted 17 high-risk and 6 low-risk genotypes (23 total). In this cohort, 14 high-risk (HPV-16, −31, −33, −35, −39, −45, −51, −52, −53, −56, −58, −66, −68, −82) and 4 low-risk (HPV-6, −11, −42, −81) genotypes were detected.
Subtypes included in the panel but not observed in any sample were high-risk HPV-18, HPV-59, HPV-73 and low-risk HPV-70, HPV-83. The most frequent detected genotypes were HPV-53, HPV-58, HPV-52, HPV-16, and HPV-31 (Figures 2 and 3).
Distribution of single or multiple HPV infections among males at different ages
In this study, the overall male HPV infection rate was 31.9% (37/116). Most infections were single infections (23.28%, 27/116), followed by dual infections (6.90%, 8/116), and triple-or-more infections (1.72%, 2/116).
The age-specific distribution was as follows:
22–30 years: 12% (3/25), all single infections. Dual and triple infections were not observed.
31–40 years: 20.69% (6/29) single infections; 6.90% (2/29) dual infections. No triple infections were observed.
41–50 years: 36.11% (13/36) single infections; 11.11% (4/36) dual infections; 2.78% (1/36) triple infections.
51–60 years: 11.11% (2/18) single infections; 5.56% (1/18) dual infections. No triple infections were observed.
≥60 years: 37.5% (3/8) single infections; 12.5% (1/8) dual infections; 12.5% (1/8) triple infections.
These results demonstrate that single infections predominated across all age groups, while dual and triple infections were relatively rare.
Discussion
In this study, 116 male HPV specimens aged 22–71 years were collected between January 2023 and April 2025. The overall prevalence of HPV infection in males was 31.90%. The reported prevalence of male HPV in Europe was 30.9%, 24 while the global combined prevalence was 31%, 17 both of which are consistent with our findings. For example, Bruni et al. conducted a systematic review and meta-analysis of studies on genital HPV in men worldwide and reported an overall prevalence of approximately 31%, with substantial between-study heterogeneity driven by population and sampling differences. This aligns with our observed prevalence (31.9%) in Jinshan District and supports that site-specific sampling (penile swabs only) may yield lower estimates than multi-site protocols. By contrast, studies from China reported higher rates: 53.67% in Tianjin, 25 52.45% in pooled national data, 26 and 57.3% in Shandong Province. 27 Research from Brazil, Mexico, and the United States indicated an overall prevalence of 65.2%, 28 and studies in Rwanda found a persistence rate of 47.5% for high-risk penile HPV. 29 Considering China's geographic complexity and climatic diversity, our results suggest that the overall infection rate in Jinshan District was lower than those reported in other regions.
As suggested by relevant literature, the prevalence and incidence of male genital HPV vary according to geographic location.30–34
In our study, HPV infection was observed across different age groups: 12% (3/25) in the 22–30 age group, 27.59% (8/29) in the 31–40 age group, 50% (18/36) in the 41–50 age group, 16.67% (3/18) in the 51–60 age group, and 62.5% (5/8) in the >60 age group. The highest prevalence of HPV infection was found in older males, which may be associated with a decline in immunity. Additionally, the infection rate was the lowest (12%) in the 22–30 age group. Some literature studies have shown that the infection rate is relatively low among young males. 35
Statistical analysis of this data showed that the top five high-risk HPV subtypes were HPV-53, HPV-58, HPV-52, HPV-16, and HPV-31, respectively; it has been reported in relevant literature 29 that the highest percentage of people were infected with HPV-66, HPV-52, and HPV-16; and the highest percentage of people were infected with HPV-16, HPV-52, HPV-51, HPV-58, and HPV-18 36 in Beijing, China (HPV infections); the result was different from this study. Some of the HPV subtypes were genetically detected with homogeneity, however, our data showed that the HPV subtypes that MALE were not infected with were high-risk HPV-18, HPV-59, and HPV-73; our study also showed that the top three low-risk HPV subtypes were HPV-6, HPV-81, and HPV-11; the literature showed that the most common subtypes were HPV 6, HPV11 26 ; the literature reported that the most common subtypes were HPV11 26 ; HPV-6 was reported to be at the top of the list in relevant literature. 37
In this study, according to the distribution of males being infected by HPV subtypes single, dual and triple and above were 23.28%, 6.90% and 1.72% respectively; our research findings suggested that males being infected by HPV subtypes were predominantly mono-infected, followed by dual infections, and triple and above infections were less common. It has been shown that males are most commonly infected by HPV subtypes singly 36 and it has also been reported in relevant literature that single HPV infection in males is more common than multiple infections. 26
This study is advantageous for us to understand the current overall prevalence of HPV in China, but there are some limitations. Above all, our research design focuses on examining male HPV specimens in a single area of Jinshan District, Shanghai. The number of specimens is relatively small, and future research will need to add more specimens for verification results. Aside from that, this study provides evidence for the prevention of HPV infection and related diseases. In addition, this study solely focuses on the incidence and type distribution of HPV infection in Jinshan District, Shanghai, failing to take into consideration other influencing factors such as sexual behavior and immunity.
A key limitation is that sampling was restricted to penile sites; therefore, the findings reflect penile HPV only.
Generalization to other anogenital or oropharyngeal sites should be made with caution.
Conclusion
In this research, males in Jinshan District, Shanghai, China, were at the lowest risk of being infected by HPV subtypes in young people and at the highest risk in the older age groups. The prevalent genotypes of HPV subtypes in males in this area were HPV-53, HPV-58, HPV-52, HPV-16, and HPV-31, and the most common low-risk genotypes were HPV-6, HPV-81, and HPV-11. Our findings highlight that elderly men (>60 years) are at the highest risk, while young men (22–30 years) show the lowest prevalence. These results underscore the importance of targeted prevention strategies, including vaccination and regular screening, particularly for older male populations.
Footnotes
Abbreviations
Acknowledgements
We thank Shanshan Chen and Jing Chen for their help with the completion of this manuscript.
Human ethics and consent to participate declarations
The study was approved by the Ethics Committee of Tinglin Hospital, Jinshan District, Shanghai (No. TLYY-KYKT-2024-01). All participant data were de-identified prior to analysis. Written informed consent was obtained from all participants.
Consent to participate
All participants agreed.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Author contributions
Tao Xiao wrote the manuscript; Shanshan Chen and Jing Chen conducted statistical analysis of the data.
Funding
This research was supported by the Hospital- level Project Fund of Tinglin Hospital, Jinshan District, Shanghai, (grant number TLYY-KYKT-2024-01).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data cannot be made public owing to patient privacy issues.
