Abstract
Objective
To explore associations between Life's Essential 8 (LE8) scores of cardiovascular health and chest pain risk in adult in the USA.
Methods
This cross-sectional investigation utilized data from National Health and Nutrition Examination Survey study covering the period between 2005 and 2018. Life's Essential 8 scores were generated for each patient, and correlations between variables were probed with multivariate linear and logistic spline models. The participants are American adults over the age of 20.
Results
This study enrolled 15,619 American adults, including 4232 reporting chest pain. Multivariate regression analysis revealed that a 10-point increase in the total LE8 score was associated with a 14% drop in chest pain risk (odds ratio = 0.86, 95%CI: 0.83–0.90, p < 0.001). Analysis of each LE8 component revealed that increased dietary, nicotine exposure, sleep quality, BMI, and blood glucose score values were all independently related to decreased chest pain risk (all p < 0.05). In subgroup analyses, the LE8 exhibited a stronger protective effect in individuals under 60, those with low income, and those without diabetes (p for interaction < 0.05).
Conclusions
A significant association was found between LE8 scores and chest pain incidence in the US adults. Improvements in diet, smoking cessation, better sleep quality, glycemic control, and weight management may be key modifiable targets in the prevention of chest pain. These findings suggest that the LE8 scoring system may function as a valuable, integrated metric for assessing chest pain risk in clinical and public health contexts.
Introduction
Chest pain is one of the most frequently encountered symptoms in clinical practice and can stem from a wide spectrum of underlying conditions. These range from life-threatening cardiovascular emergencies, such as acute coronary syndrome (myocardial infarction), aortic dissection, and high-risk pulmonary embolism, to nonlethal yet quality-of-life-impacting disorders such as gastroesophageal reflux disease, pleurisy, and costochondritis.1,2 Such conditions are commonly present in both emergency department and outpatient settings.
Many modifiable clinical and lifestyle factors contribute to conditions that present with chest pain, such as tobacco use, obesity, poor dietary patterns, inadequate physical activity (PA), and insufficient sleep.3–5 These factors are believed to exert their effects primarily through mechanisms involving chronic inflammatory states, oxidative stress, and metabolic dysregulation. In response to the growing need for an inclusive evaluation of cardiovascular health (CVH), the American Heart Association devised the Life's Essential 8 (LE8) framework. This system quantitatively evaluates CVH across two core domains: health-related behavioral processes (diet, sleep, PA level, exposure to nicotine) and health factors (lipid and blood glucose, blood pressure, and BMI). 6 This system holds promise as a standardized predictive tool with potential utility as a means of predicting the risk of chest pain incidence.
Life's Essential 8 score-defined CVH has previously been investigated in the context of a variety of health-related outcomes, including longevity, biological aging, cognitive performance, cardiovascular disease (CVD), depressive symptoms, chronic kidney disease, cancer risk, and overall mortality.7–10 For example, Ma reported that associations between higher LE8 scores and enhanced life expectancy, 11 while Li demonstrated a negative link between the LE8 score and CVD and stroke risk, 12 and Shen identified a similar relationship with the risk of depression. 13 Despite this growing body of evidence, there has been no specific evaluation of associations between LE8 scores and chest pain. As such, there is a clear need to comprehensively determine how LE8 scores impact chest pain risk.
The cross-sectional multistage National Health and Nutrition Examination Survey (NHANES) survey of a representative population in the United States offers detailed, accessible health-related data collected through a series of standardized interviews, laboratory tests, and physical examinations. This cross-sectional study leveraged NHANES data to probe for potential associations between LE8 scores and chest pain among adults in the USA. These results may offer new information that will aid in the more effective prevention of chest pain in the clinic.
Methods
National Health and Nutrition Examination Survey investigation method
National Health and Nutrition Examination Survey employs a complex stratified, multistage, and probabilistic sampling design, aiming to ensure that the sample can represent the population of all age groups, races, genders, economic conditions, and geographical regions in the United States. This survey consists of multiple parts including detailed questionnaires, physical examinations, and laboratory tests, covering a wide range of aspects from basic demographic information to detailed dietary, lifestyle, disease history, and health behaviors. All relevant documents can be obtained in the following blog: https://www.cdc.gov/nchs/nhanes/about.
Data source
The NHANES study consists of an ongoing series of stratified, multistage, nationally representative surveys focused on the nutritional status and health of noninstitutionalized members of the civilian population in the USA. The NHANES Survey Methods and Analysis Guide provides full details regarding the procedures used for these surveys, which offer details regarding participant demographic, dietary, socioeconomic, and health-associated parameters collected in mobile examination centers. Approval for study protocols within the NHANES was granted by the Health Statistics Research Ethics Review Board of the US Center for National Food Safety. Additionally, all personally identifiable information in this study has been de-identified to protect participant confidentiality. This approval was contingent on attaining informed consent from all subjects, ensuring compliance with ethical standards.
Study design and subjects
Data were extracted from four NHANES cycles from 2005 to 2018. Exclusion criteria were applied to individuals < 20 years old (n = 39,749) or if they were missing data related to LE8 components (n = 12,763), chest pain (n = 8710), demographic characteristics (sex, education, ethnicity, whether married, or poverty-to-income ratio [PIR], n = 1528), alcohol consumption (n = 1128), or common diseases (CVD, diabetes, n = 1). In total, 15,619 participants were included in these analyses, of whom 4232 exhibited chest pain (Figure 1).

A flowchart detailing the participant selection process.
Life's Essential 8 score calculations
The LE8 initiative was recently introduced by the AHA as an update to the Life's Simple 7 system, resulting in a more effective quantification of CVH based on four health-related factors as well as four health behaviors to strengthen existing guidelines for the improvement of CVH among the general public. Details regarding the calculation of LE8 scores with NHANES data are provided in Table S1.
Dietary metrics were evaluated by the Healthy Eating Index 2015. This index was derived from data attained during initial 24-h dietary recall interviews with participants at the mobile examination center. Self-reported questionnaires served as the source of details regarding PA, sleep quality, diabetes, exposure to nicotine, and history of medication use. During physical examinations, the height, weight, and blood pressure of all participants were professionally measured. Samples of blood were collected in a central laboratory and used to analyze levels of lipids, plasma glucose, and hemoglobin A1c. It is important to note that all health factor scores in this study, including individual components of LE8, were constructed such that higher scores indicate closer alignment with the ideal or normal health status.
The comprehensive spectrum of CVH indicators, each ranging from 0 to 100, collectively contributed to the determination of the final CVH score. This cumulative score was ascertained by determination of the unweighted average of the individual indicators. The resulting total score was subsequently categorized into: high (80–100), medium (50–79), or low (0–49). Full details on the use of the LE8 scoring algorithm to evaluate adult NHANES data are provided in the online supplement and corresponding Presidential Advisory.
Outcome assessments
Chest pain was evaluated using the Rose questionnaire, with questions being answered by trained interviewers who conducted computer-assisted personal interviews (CAPI) at home. Data entry errors were minimized through consistency checks within the CAPI system, which also incorporated an online help screen that aided interviewers in defining key questionnaire-related phrases. Chest pain was assessed based on participant responses being asked, “Are you experiencing any pain or discomfort in your chest?” These responses were used to stratify subjects into individuals with and without pain. 10
Covariates
Covariates were selected based on earlier studies and included baseline age (60, ≥60 years), sex, race/ethnicity (Mexican American, non-Hispanic black, non-Hispanic white, other Hispanic, other), marital status (married, unmarried, cohabiting, other [including widowed, divorced, or separated]), education level (<high school, high school/equivalent, high school, or above), PIR (≤1.30, 1.31–3.50, and >3.50), alcohol intake (never [<12 lifetime drinks], former [≥12 lifetime drinks but no alcohol intake in the last year], mild [≤1 and ≤2 daily drinks in women and men, respectively], moderate [≤2 and ≤3 daily drinks in women and men, respectively], and heavy [≥3 and ≥4 daily drinks in women and men, respectively]), CVD history, and diabetes. A self-reported history of CVD was identified based on diagnoses of heart attack, stroke, angina pectoris, heart failure, or coronary heart disease, while the determination of prior CVD or diabetes was based on a questionnaire asking the patients whether they had been informed of these past conditions.
Statistical analysis
The NHANES Data Analysis Guidelines were used for all statistical analyses. This study was conducted strictly in accordance with the 1975 Heroctyl Declaration (revised in 2024). In addition, the reporting of this study conforms to STROBE guidelines. 14 Continuous and categorical data are, respectively, given as averages with standard deviations and proportions (weighted ratios, %). Categorical variables were analyzed using the Rao-Scott second-order corrected chi-square test, while continuous variables in complex survey-weighted samples were assessed via the Wilcoxon rank-sum test to evaluate differences across CVH levels. Chest pain was included as a binary dependent variable, and a multivariate linear regression approach was employed to evaluate correlations between LE8 or LE8 component scores and chest pain. The impact on the link between LE8 scores and chest pain was also assessed through analyses of different subgroups, estimating the interaction significance based on the corresponding p-value. “WTMEC2YR” was employed for weighting in the present analyses. R 4.3.5 and Free Statistics software (v2.0.0) were utilized to perform all analyses.
Results
Baseline characteristics
Specifics regarding participants’ inclusion and exclusion procedures are outlined in Figure 1.
Baseline participant characteristics are summarized in Table 1 according to whether or not they experienced chest pain. Significant differences in ethnicity, marital status, PIR, drinking status, CVD, diabetes, LE8 scores, PA, exposure to nicotine, sleep quality, BMI, blood glucose, and blood pressure scores were observed when comparing participant groups (all p < 0.05). Age did not differ significantly between groups. Lower CVH scores were evident in the group of individuals with chest pain. To verify the robustness of the research results, we conducted a sensitivity analysis based on populations with different baseline characteristics, and the analysis results were stable (Tables S4 and S5).
Baseline participant characteristics.

AHA: American Heart Association; CVD: cardiovascular disease; CVH: Cardiovascular Health Score; LE8: Life's Essential 8; NHANES: National Health and Nutrition Examination Survey; PA: physical activity; PIR: poverty income ratio; SD: standard deviation.
Participants self-reported their race and ethnicity.
Multiracial participants, as full race/ethnicity details are not provided by the NHANES.
Widowed, divorced, or separated individuals.
Congestive heart failure, stroke, heart attack, angina, and coronary heart disease.
Relationships between LE8 score values and chest pain
Relationships linking LE8 scores and chest pain were probed using multivariate logistic models. Model 1 did not receive adjustment, whereas Model 2 underwent adjustment for age, sex, ethnicity, education, and marital status, and Model 3 was further adjusted for PIR, alcohol intake, CVD, and diabetes (Table 2).
Relationships between LE8 scores and chest pain in multivariate regression analyses.

CI: confidence interval; CVD: cardiovascular disease; CVH: cardiovascular health; DM: diabetes; LE8: Life's Essential 8; PIR: poverty income ratio; OR: odds ratio.
The total CVH scores were logged as a continuous variable, denoting a per-10-point escalation. Subsequently, these scores underwent classification into three discernible groups: low (0–49), moderate (50–79), and high (80–100).
OR <1 indicates a reduced risk of chest pain.
Model 1: Crude model. Model 2: Adjusted for age, sex, ethnicity, education, and marital status. Model 3: Model 2 with further adjustment for PIR, alcohol intake, CVD, and DM.
Multivariate logistic regression yielded an odds ratio (OR) for chest pain among individuals with a high CVH score of 0.63 (95%CI, 0.52–0.77) (Table 2). Higher diet, nicotine exposure, sleep quality, BMI, blood lipids, and blood glucose LE8 component scores were also associated with a reduction in chest pain risk (Table 3). Subgroup analyses demonstrated that the negative relationship between LE8 scores and chest pain remained stable irrespective of whether patients had CVD (Table S3).
Relationships between LE8 components and chest pain.
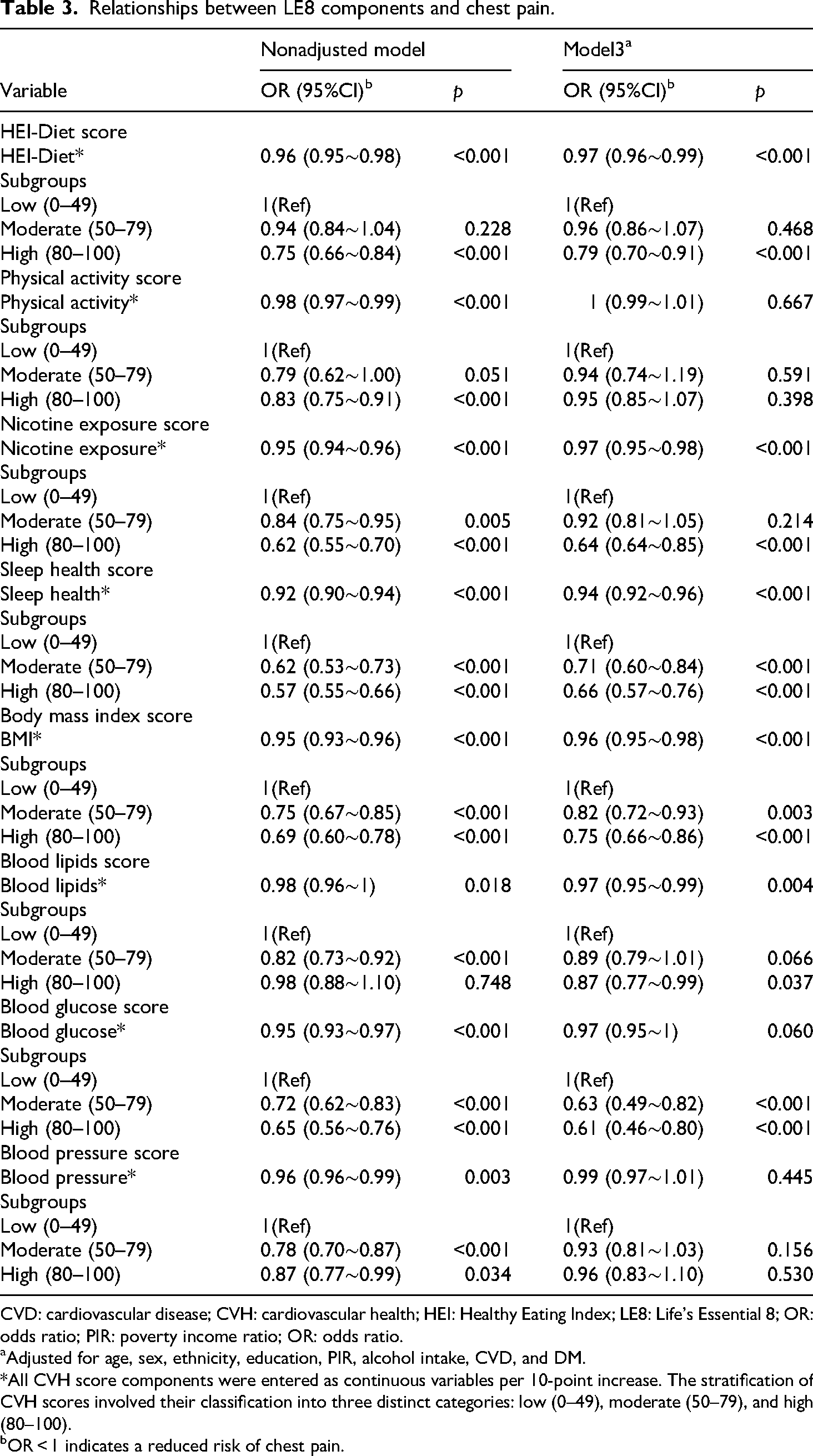
CVD: cardiovascular disease; CVH: cardiovascular health; HEI: Healthy Eating Index; LE8: Life's Essential 8; OR: odds ratio; PIR: poverty income ratio; OR: odds ratio.
Adjusted for age, sex, ethnicity, education, PIR, alcohol intake, CVD, and DM.
*All CVH score components were entered as continuous variables per 10-point increase. The stratification of CVH scores involved their classification into three distinct categories: low (0–49), moderate (50–79), and high (80–100).
OR < 1 indicates a reduced risk of chest pain.
Logistic curve fitting (Figure S1) indicated a marked nonlinear association between total LE8 scores and chest pain occurrence (p for nonlinearity < 0.01). Subgroup analyses, using an inflection point of 59.6 as a threshold, revealed a clear inverse relationship between CVH scores and chest pain risk above this score (Table 2, Table S1). Interaction effects for each subgroup are displayed in Figure 2. Overall, higher LE8 scores, reflecting better CVH, were consistently associated with reduced risk of chest pain. This inverse association remained robust across continuous exposure models (ORs < 1 with p < 0.001 across most strata). For example, the OR for men was 0.86 (95% CI: 0.82–0.91, p < 0.001), and for women, 0.86 (95% CI: 0.82–0.91, p < 0.001). However, subgroup analyses revealed significant heterogeneity in these associations. Age appeared to be a significant effect modifier, as a strong protective association was evident in those aged 40–59 years (OR = 0.83, 95% CI: 0.78–0.87, p < 0.001) than in those older than 59 years (OR = 0.93, 95% CI: 0.88–0.98, p = 0.01). Income, as measured by the PIR, also significantly modified the relationship. Individuals in the low-income group experienced substantially greater protective effects from high CVH (OR = 0.44, 95% CI: 0.28–0.68, p < 0.001) compared to those in the medium-to-high income group (OR = 0.67, 95% CI: 0.53–0.84, p < 0.001). Drinking status further stratified the relationship: among never-drinkers, LE8 score was not significantly associated with chest pain (continuous OR = 0.96, 95% CI: 0.84–1.09, p = 0.53; high CVH OR = 1.01, 95% CI: 0.52–1.97, p = 0.97). In contrast, significant associations were observed in individuals with current or past alcohol consumption (continuous OR = 0.86, 95% CI: 0.82–0.89, p < 0.001; high CVH OR = 0.60, 95% CI: 0.49–0.74, p < 0.001. Diabetes status also modified the association: among nondiabetic participants, high CVH was significantly protective (OR = 0.70, 95% CI: 0.57–0.87, p = 0.001), whereas the protective effect was diminished and statistically nonsignificant among those with diabetes (OR = 0.56, 95% CI: 0.25–1.26, p = 0.16). These findings suggest that comorbid conditions such as diabetes may attenuate the benefits of optimal CVH, and that individual characteristics including age, socioeconomic status, alcohol consumption, and metabolic health can substantially influence the strength of this association.

The association between Life's Essential 8 (LE8) scores and chest pain stratified according to patient characteristics. Adjustment was performed for all variables other than those being used for stratification (adjusted for age, sex, ethnicity, education, marital status, poverty income ratio [PIR], alcohol intake, cardiovascular disease [CVD], and diabetes).
Discussion
Using data from NHANES (2005–2018), this study investigated the association between CVH, assessed by the LE8 scoring system, and the prevalence of chest pain symptoms. The analysis revealed a nonlinear but generally inverse relationship such that as LE8 scores increased, the likelihood of experiencing chest pain significantly declined. Individuals classified as having high CVH were largely categorized as having low or very low chest pain risk, whereas those with lower CVH scores were at elevated risk. Importantly, the protective association was not only observed in overall LE8 scores but also in specific LE8 components. Notable among these were low nicotine exposure, high sleep quality, and favorable blood glucose levels, all of which were independently linked to substantial reductions in chest pain risk. This is the first study to have clarified the relationship between CVH and chest pain based on the utilization of LE8 scores as a measure of CVH. The OR for chest pain tended to decline with rising LE8 scores, consistent with higher scores being associated with greater protection against chest pain.
Multivariate regression analysis further supported the observed negative correlative relationship between LE8 scores and chest pain risk. When comparing individuals in the medium and high LE8 score categories to those with low scores, chest pain risk was reduced by 15% and 37%, respectively. Relative to participants in the lowest scoring groups, those with high glucose control, reduced nicotine exposure, and better sleep reported 39%, 36%, and 34% lower risks of chest pain, respectively. These results align closely with previous findings. 15 In addition, specific components of the LE8 score including a favorable blood lipid profile (indicated by a high hyperlipidemia score), adherence to a healthy diet (reflected in a high diet score), and maintaining a normal body mass index (captured by a high BMI score) were each independently associated with a lower likelihood of chest pain. Those in the high-scoring groups for these components experienced 13%, 21%, and 25% reductions in chest pain prevalence, respectively. Supporting evidence from Xu et al. 16 demonstrates that most LE8 components show significant correlations with atherosclerosis risk. Stratification by overall LE8 score revealed that those with moderate CVH were predominantly thought of as having a low risk for atherosclerosis, whereas over 90% of those with high CVH fell into the very low or low-risk categories. Since the majority of cardiogenic chest pain stems from atherosclerotic processes, these findings suggest that optimizing lifestyle-related health behaviors and biological factors such as lipid levels, body weight, and dietary habits can play a crucial role in modifying metabolic profiles and, in turn, substantially lowering the risk of chest pain.
Elevated blood glucose levels are critical in the deterioration of vascular health. Hyperglycemia contributes to the upregulation of endothelial adhesion molecules through oxidative stress pathways, leading to macrophage activation and pro-inflammatory cytokine release. These processes collectively promote vascular inflammation, endothelial dysfunction, and tissue fibrosis.17,18 Moreover, persistent hyperglycemia is a primary driver of increased expression of the GLUT1 glucose transporter and glycolytic enzymes in vascular cells, including endothelial cells, establishing a metabolic environment that favors atherogenesis.19,20 This mechanistic basis helps explain the paradoxical presence of elevated LE8 scores among diabetic individuals experiencing chest pain. While the complete mechanistic network remains under investigation, central pathways such as inflammation and oxidative stress have already been well established. Nicotine exposure, primarily through smoking, is another major contributor to chest pain, driven by both toxicological and inflammatory pathways. Cigarette smoke contains approximately 10¹⁵ free radicals and over 7000 chemical constituents, many of which are recognized toxins. These reactive species induce oxidative stress within the vasculature, leading to the uncoupling of eNOS and a subsequent reduction in NO bioavailability.21,22 Since NO is essential for maintaining vascular tone, inhibiting thrombosis, and regulating inflammation, its depletion impairs endothelial function.23,24 Oxidative stress, in this context, serves as both a catalyst for inflammation and an early pathological trigger for atherosclerosis development. 25 When myocardial oxygen demand surpasses supply, as during exertion or following plaque rupture that results in acute thrombus formation, clinical manifestations such as angina pectoris or acute coronary syndromes may occur. These events typically present as either exertional substernal pressure or sudden-onset, persistent chest pain at rest. 26 The elevated incidence of chest pain among smokers can be attributed to this pathological cascade. Additionally, mounting evidence links sleep disturbances to heightened pain perception, possibly mediated through immune and inflammatory responses. Both sleep and pain are accompanied by immune system activation, with cytokines including IL-1, IL-6, and TNF playing crucial roles in modulating both inflammation and neuropathic pain pathways. 27
Several pathological mechanisms underlying LE8 components also overlap with that driving noncardiogenic chest pain. For instance, in Type B aortic dissection, structural compromise of the aortic wall is often precipitated by atherosclerotic plaque erosion. Abnormalities in key LE8 metrics including elevated LDL-C, hyperglycemia, poor dietary patterns, and tobacco use interact synergistically to impair vascular integrity. Elevated LDL-C facilitates lipid core accumulation; advanced glycation end products reduce vascular elasticity; excessive sodium intake triggers renin–angiotensin–aldosterone system activation and increases blood pressure; and smoking activates matrix metalloproteinases that degrade collagen. Together, these factors promote endothelial dysfunction, medial layer degeneration, and intramural hematoma formation, all of which are key factors in the pathophysiology of aortic dissection.28–31 Furthermore, physical inactivity and obesity, both of which are integral to the LE8 scoring system, can enhance systemic inflammation and oxidative stress, marked by increased circulating levels of IL-6 and TNF-α, while also enhancing ROS production. These factors accelerate aortic remodeling and heighten the risk of noncardiogenic chest pain.32,33
These results offer valuable potential guidance regarding approaches to preventing and managing the risk of chest pain, but the study has some limitations. Most notably, its cross-sectional study design does not permit causal determinations. Additionally, the lack of differentiation between acute and chronic chest pain subtypes (cardiac emergencies vs. gastrointestinal causes) introduces ambiguity and may obscure distinct underlying pathophysiological mechanisms, thereby limiting the precision of clinical applications. Although statistical models adjusted for known confounding variables, the possibility of residual confounding, such as unmeasured genetic predispositions or environmental exposures, remains. Measurement limitations also warrant consideration. A single time-point LE8 assessment may not accurately capture long-term behavioral patterns, introducing regression dilution bias. Dietary metrics, derived from 24-h recall surveys, are subject to recall bias and may not reflect habitual intake. Moreover, mismatches in timing between dietary assessments and biomarker collection could further compromise data integrity. Lastly, the exclusion of individuals with incomplete data may have led to selection bias. Combined with the study's reliance on U.S.-based NHANES data, these factors may limit the generalizability of the results to broader international populations.
Conclusions
These results support a potential negative linear association between LE8 scores and the risk of chest pain in US adults. Improving LE8 component scores for dietary, sleep, nicotine exposure, blood glucose, diet, and BMI should be prioritized to potentially improve chest pain risk. These results suggest that LE8 scores offer value as a practical and effective composite indicator for the improvement of CVH that can be applied in clinical practice, potentially aiding efforts to identify risk factors associated with chest pain at an early point so as to minimize the burden of chest pain.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251383090 - Supplemental material for The relationship between Life's Essential 8 measures and chest pain among adults in the USA: A cross-sectional study
Supplemental material, sj-docx-1-sci-10.1177_00368504251383090 for The relationship between Life's Essential 8 measures and chest pain amon
Supplemental Material
sj-docx-2-sci-10.1177_00368504251383090 - Supplemental material for The relationship between Life's Essential 8 measures and chest pain among adults in the USA: A cross-sectional study
Supplemental material, sj-docx-2-sci-10.1177_00368504251383090 for The relationship between Life's Essential 8 measures and chest pain amon
Footnotes
Acknowledgements
The authors would like to thank the team of clinical scientists Huanxian Liu, Haoxian Tang from Shantou University Medical School, and Jie Liu from 301 Hospital for their comments and criticisms on the data analysis of the article.
Ethics statement
This study was approved by the Ethics Committee of the Second Hospital of Lanzhou University (Approval No.: D2022A-012). The address of the ethics committee: 82 Cuiyingmen, Chengguan District, Lanzhou 730030, Gansu Province, People's Republic of China.
Author contributions
Runze Song and Tao Liu (cofirst authors) were responsible for the research conception, NHANES data organization, complex sampling weight statistical analysis, and the first draft of the paper. Si Cai was responsible for completing the literature review, interpreting the NHANES data results, and revising the methodological section of the paper. Xuemei Zhou is responsible for conducting data validation to ensure the quality of NHANES dataset extraction. Jiming Zhang is responsible for providing epidemiological methodological guidance and support for statistical software. Yamin Zhang is responsible for obtaining financial support, managing project progress and ethical compliance, and assisting Liu Tao in writing the thesis. Dong Zhou (corresponding author) coordinated the entire research process, supervised the data usage norms of NHANES, and conducted the final review of the paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Health industry research plan of Gansu Province: Construction and application of informationized VTE risk early warning and assessment system (GSWSHL2021-023).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
All supplemental material mentioned in the text is available in the online version of the journal.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
