Abstract
Objective
To establish a predictive model based on ultrasound (US) radiomics to determine whether the ablation zone of papillary thyroid microcarcinoma (PTMC) disappears within 24 months after microwave ablation (MWA).
Study design
Retrospective study.
Methods
This study enrolled 201 PTMC patients who underwent MWA in Affiliated Beijing Friendship Hospital of Capital Medical University between January 2013 and September 2020. All patients were followed up at 1 h, 1 month, 3 months, 6 months, 12 months, and 24 months after MWA. Radiomics features were extracted from the preoperative US images, and a Rad-score was constructed. Univariate and multivariate logistic regression analyses were used to screen out the independent clinical factors associated with the disappearance of the ablation zone after MWA, and a radiomics nomogram was established to predict whether the ablation zone of PTMC disappeared within 24 months after MWA. The performance of the model was validated in the testing cohort.
Results
75.6% of patients achieved the ablation zone disappeared within 24 months after MWA. The disappearance rate was significantly associated with MWA energy and baseline lesion volume (p < 0.05). The US radiomics nomogram integrated Rad-score, MWA energy, and baseline lesion volume. In the testing cohort, the area under the curve of this nomogram outperformed that of the clinical model and the radiomics model (0.772 vs 0.714 and 0.679, respectively).
Conclusions
The nomogram based on US radiomics can reliably identify whether lesions of PTMC will disappear within 24 months after MWA. The nomogram is useful for screening optimal candidates for MWA and may assist clinicians and patients in choosing the best treatment option between MWA and surgery.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) is one of the most common subtypes of primary thyroid malignancies with a significantly increasing incidence worldwide over the past decade, and it has a stably high survival rate.1,2 According to the World Health Organization (WHO), papillary thyroid microcarcinoma (PTMC) is defined as papillary thyroid carcinoma with a tumor diameter ≤1 cm. 3 In recent years, with the widespread application of high-frequency ultrasound (US) and fine needle aspiration biopsy (FNAB) an increasing number of PTMC cases were diagnosed.4,5 Until a decade ago, surgery remained the standard treatment for PTC. 6 PTC has low malignancy and favorable prognosis. However, surgical complications such as hypoparathyroidism and recurrent laryngeal nerve injury, as well as thyroid hormone replacement therapy, still have an impact on the quality of patients’ life.7,8 Therefore, the 2023 National Comprehensive Cancer Network (NCCN) Thyroid Cancer Guidelines recommend active surveillance as viable option for patients who are evaluated by ultrasonography or computed tomography (CT) with the absence of cervical lymph node metastases in the central or lateral neck regions and have suspicious lesions ≤1 cm in diameter.9–11 However, the implementation of active US surveillance for PTMC is confronted with certain barriers, including patient preference, anxiety about living with cancer, lack of professional teams to evaluate disease progression, and variations in health system policies.12,13
In recent years, US-guided thermal ablation, such as radiofrequency ablation (RFA), laser ablation (LA), and microwave ablation (MWA) has emerged as an alternative to surgery and active US surveillance for patients with PTMC. Compared with surgery, US-guided thermal ablation offers superior safety and minimal invasiveness, and patients have shorter recovery time after treatment. Over the past decades, numerous studies have demonstrated encouraging outcomes regarding the safety and efficacy of thermal ablation in patients with inoperable PTC and low-risk PTC.14–17 And the complete disappearance rate of PTC lesions after MWA ranges from 40.9% to 71.0%.14,18 One of the concerns for clinicians and patients is whether the ablation zone of PTMC will disappear within a specific timeframe after ablation. Anxiety about the failure of lesion disappearance after ablation may lead to unnecessary secondary surgeries for patients. 19 Therefore, predicting whether the ablation zone will disappear within 24 months after MWA is crucial to identify the optimal candidates for this treatment. Patients with anticipated poor post-ablation responses may be advised to choose surgery or other supplementary treatments. In this study, we developed an US radiomics-based predictive model to evaluate whether the ablation zone of PTMC disappeared within 24 months after MWA and validated its predictive performance.
Materials and methods
Patients
This study enrolled patients with PTMC diagnosed by preoperative US-guided fine needle aspiration (FNA) biopsy who underwent MWA in Affiliated Beijing Friendship Hospital of Capital Medical University between January 2013 and September 2020. This study received ethical approval from the Institutional Review Board of Affiliated Beijing Friendship Hospital of Capital Medical University (Approval Number: 2022-P2-390-01, 29 November 2022), with informed consent waived due to its retrospective nature. All procedures of this study were in accordance with the 1975 Declaration of Helsinki (2024 revision). And all patient details were de-identified.
The inclusion criteria were: (1) PTC confirmed by FNA; (2) the maximum diameter of the tumor ≤ 10 mm; (3) no evidence of extrathyroidal extension (based on clinical or imaging findings); 20 (4) no evidence of lymph node metastasis or distant metastasis (based on ultrasonography or CT); and (5) no history of neck irradiation.
The exclusion criteria were: (1) aggressive subtype of PTC suggested by preoperative biopsy; (2) incomplete clinical data or loss of follow-up.
After exclusions, 201 patients who underwent MWA were included in this study (Figure 1).

Showed the enrollment procedure.
The dataset contained patients’ demographics, preoperative US images of lesions, volumes of lesions/ablation zones at baseline, 1 h, 1, 3, 6, 12, and 24 months after MWA, MWA energy delivered per lesion and duration of procedure. Preoperative US examinations were performed within one week before MWA.
Region-of-interest (ROI) delineation and US radiomics feature extraction
ROIs were manually delineated on preoperative US images using MRIcroGL v1.0 (http://www.mccauslandcenter.sc.edu/mricrogl/). All delineation procedures were conducted by sonographers with ≥10 years’ experience in thyroid US examination. The boundaries of each PTMC lesion were delineated in both the transverse and sagittal planes of the lesion, respectively, with exclusion of hemorrhagic, necrotic, and cystic zones. The images and ROI masks of the lesions were exported. US radiomics features were extracted using PyRadiomics (version 3.0.1; Computational Imaging & Bioinformatics Lab, Harvard Medical School, Boston, MA, USA; https://pyradiomics.readthedocs.io).
Features selection and the radiomics score construction
The min–max normalization method was used to normalize radiomics features both in the training cohort and the independent testing cohort. Then, the K-nearest neighbor (KNN)-based missing value padding method was used to predict the probable value of the missing data by referencing KNN data from the training cohort.
US radiomics feature selection: Initially, correlation analysis was performed in the training cohort to explore associations with the disappearance of the ablation zone within 24 months after MWA. The nonsignificant radiomics features with p > 0.05 were excluded via unpaired t-test. Second, least absolute shrinkage and selection operator (LASSO) regression was employed to identify the optimal radiomics features. The radiomics score (Rad-score) was constructed by weighting each optimal feature with its regression coefficient from the LASSO model.
Clinical factors selection: Univariate logistic regression analysis of clinical factors was performed, followed by multivariate logistic regression analysis. Clinical factors with p < 0.05 in the multivariate logistic regression analysis were selected as clinical independent predictors of the disappearance of the ablation zone within 24 months after MWA.
Model establishment
The radiomics model was developed based on the Rad-score. The clinical model was constructed using the logistic regression with the independent clinical predictors. The combined model was established by integrating the Rad-score and the clinical independent predictors. An US radiomics nomogram based on the combined model was constructed in the training cohort.
Assessing the performance of the US radiomics nomogram predictive model
The predictive performance of these three models was validated by US examination findings at 24-month after MWA. The performance was evaluated using the area under the curve (AUC) of the receiver operating characteristic (ROC) curve analysis, sensitivity and specificity. Then, the performance of the US radiomics nomogram was validated in the independent testing cohorts. Calibration curves were used to evaluate the calibration ability of the three models. Decision curve analysis (DCA) was employed to determine the clinical application utility of the predictive model by the net benefits at different threshold values in the independent testing cohort.
Statistical analysis
Continuous variables with a normal distribution were presented as mean ± standard deviation and analyzed using unpaired t-test, while continuous variables without a normal distribution were expressed as median (P25, P75) [M (P25, P75)] and analyzed with the Mann–Whitney U test. For correlation analysis, the Pearson correlation coefficient was used for normally distributed features, whereas the Spearman rank correlation coefficient was used for non-normally distributed features. Categorical variables were compared using the chi-square test. Unpaired t-tests, Mann–Whitney U tests, chi-square tests, Fisher's exact tests, as well as univariate and multivariate logistic regression analyses, LASSO feature selection, model development and nomogram construction were performed using Python (version 3.8; Python Software Foundation, Wilmington, DE, USA; https://www.python.org). All statistical significance levels were two sided, with p < 0.05.
Results
Clinical characteristics
The characteristics of patients and PTMC lesions at baseline and 24 months after treatment are shown in Table 1. In this study, all patients were followed up for at least 24 months after MWA. A total of 147 patients (73.13%) were followed up for 24 months after treatment, while 54 patients (26.87%) were followed up for more than 24 months after treatment.
Baseline characteristics of 201 MWA-treated patients.

Changes in lesions at baseline and after MWA during follow-up
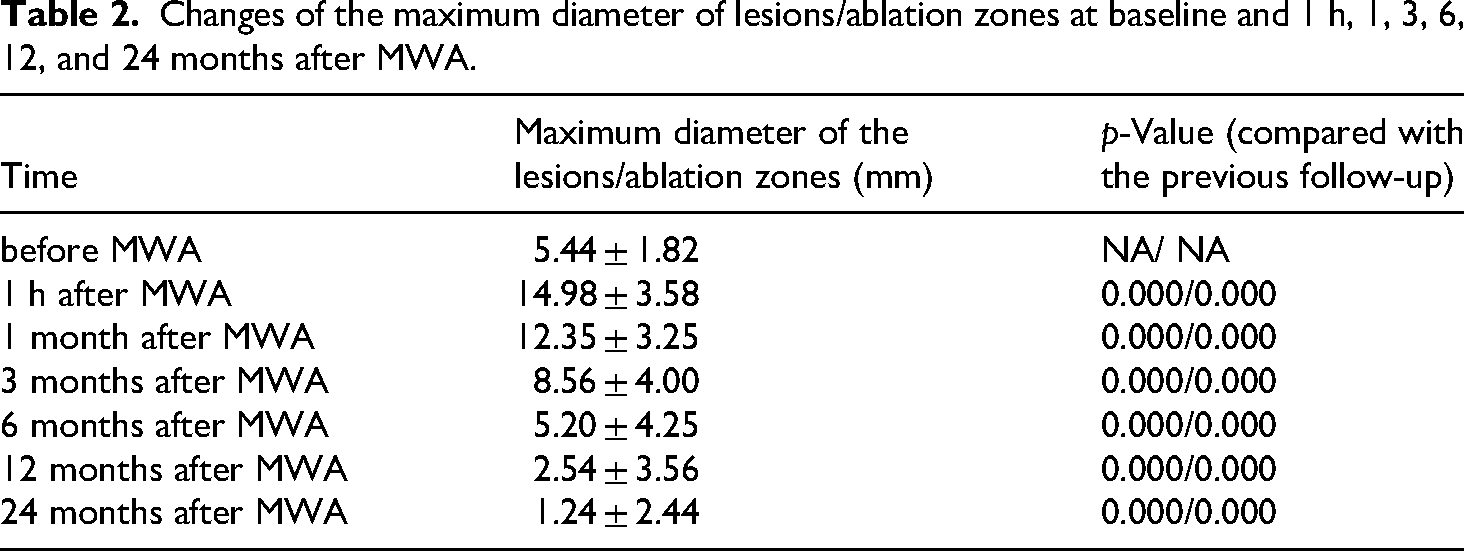
The changes in the maximum diameter and volume of lesions/ablation zones at baseline and at 1 h, 1, 3, 6, 12, and 24 months after MWA are shown in Tables 2 and 3.
Changes of the maximum diameter of lesions/ablation zones at baseline and 1 h, 1, 3, 6, 12, and 24 months after MWA.
Changes of the volume of lesions/ablation zones at baseline and 1 h, 1, 3, 6, 12, and 24 months after MWA.

Analysis of related factors with ablation zone disappearance within 24 months after MWA
The characteristics of patients with and without the disappearance of the ablation zone within 24 months after treatment are shown in Table 4. There were significant differences in baseline lesion maximum diameter, baseline lesion volume, duration of procedure, MWA energy delivered, and MWA energy of unit volume. However, there were no significant differences in sex, age, or volume of ablation zone.
Characteristics of patients with or without ablation zone disappearance within 24 months after MWA treatment.

MWA energy and baseline lesion volume were identified as independent predictors of ablation zone disappearance within 24 months after MWA by multivariate logistic regression (p < 0.05).
Selection of US radiomics features
A total of 851 features were extracted from the ROIs of US images in transverse and sagittal planes of each lesion, respectively. In the training cohort, 475 radiomics features with no statistical significance according to the unpaired t-test were excluded. Subsequently, 10 optimal radiomics features were selected using the LASSO regression model with 10-fold cross-validation (Figure 2). The 10 selected radiomics features are as follows:

The least absolute shrinkage and selection operator (LASSO) screening process for feature selection. (a) This was used to reduce the dimension of the grouping characteristics. (b) Ten features corresponding to the minimum error. (c) The weighted values of the final selected radiomics features.
Transverse: original_firstorder_Kurtosis, original_firstorder_RobustMeanAbsoluteDeviation, original_glcm_ClusterProminence, original_glcm_ClusterShade, original_glcm_Imc, original_glszm_GrayLevelVariance, original_glszm_ZoneVariance,
Sagittal: original_firstorder_RobustMeanAbsoluteDeviation, original_glcm_DifferenceVariance, original_glszm_GrayLevelVariance.
Model development and validation
A total of 160 PTMC patients who underwent MWA between January 2013 and April 2020 in Affiliated Beijing Friendship Hospital of Capital Medical University were enrolled as the training cohort. Based on the results of univariate and multivariate logistic regression analyses, three models were established in the training cohort: the clinical model (incorporating MWA energy and baseline lesion volume), the radiomics model (based on Rad-score), and the combined clinical and radiomics model (integrating MWA energy, baseline lesion volume, and Rad-score). A total of 41 PTMC patients who underwent MWA between May 2020 and September 2020 in Affiliated Beijing Friendship Hospital of Capital Medical University were recruited as an independent testing cohort to compare the diagnostic efficacy of the above three models. Validation in the independent testing cohort showed that the AUC, sensitivity, and specificity of the combined model were significantly superior to those of the clinical model and radiomics model (AUC: 0.772 vs 0.714 and 0.679; sensitivity: 0.929 vs 0.929 and 0.821; specificity: 0.692 vs 0.615 and 0.385, respectively) (Table 5). Thus, the combined model exhibited superior performance in predicting ablation zone disappearance within 24 months after MWA (Figure 3). The combined model was selected for nomogram construction. Therefore, a radiomics nomogram incorporating these three predictors (MWA energy, baseline lesion volume, and Rad-score) was developed (Figure 4). The calibration curve of the nomogram (based on combined model) exhibits no overall shift or significant divergence. Compared with the curves of the other two models, it shows smaller deviations from the ideal diagonal across the range of predicted probabilities, indicating good agreement between the predicted probabilities and actual outcomes (Figure 5). The DCA demonstrated that the combined model yielded the greatest overall net benefit among the tested models when the threshold probability for clinicians or patients ranged from 0.1 to 0.8, and it was more beneficial than either “the treat-all” or “treat-none” strategy (Figure 6). Collectively, our nomogram showed robust performance in the independent testing cohort.

Receiver operating characteristic (ROC) curves for the radiomics model, the clinical model and the combined clinical and radiomics model.
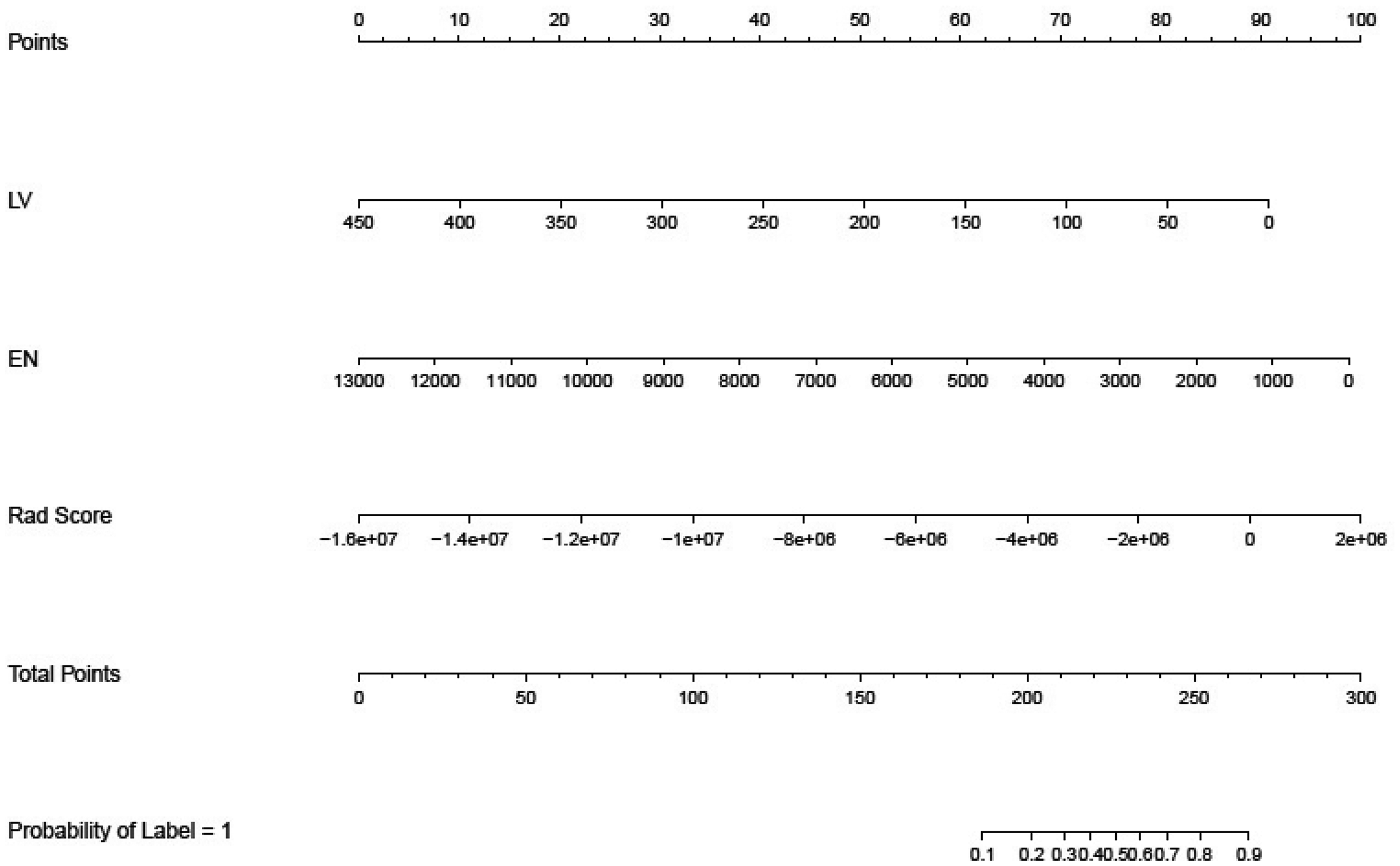
Nomogram visualization. The nomogram presents the developed model for predicting the disappearance of the ablation zone within 24 months after MWA. MWA energy (EN), baseline lesion volume (LV), and Rad Score.

The calibration curve of the nomogram. The calibration curve shows whether the nomogram has goodness-of-fit. The diagonal line represents the ideal prediction. The closer the calibration curve is to the diagonal line, the better the model's performance is.
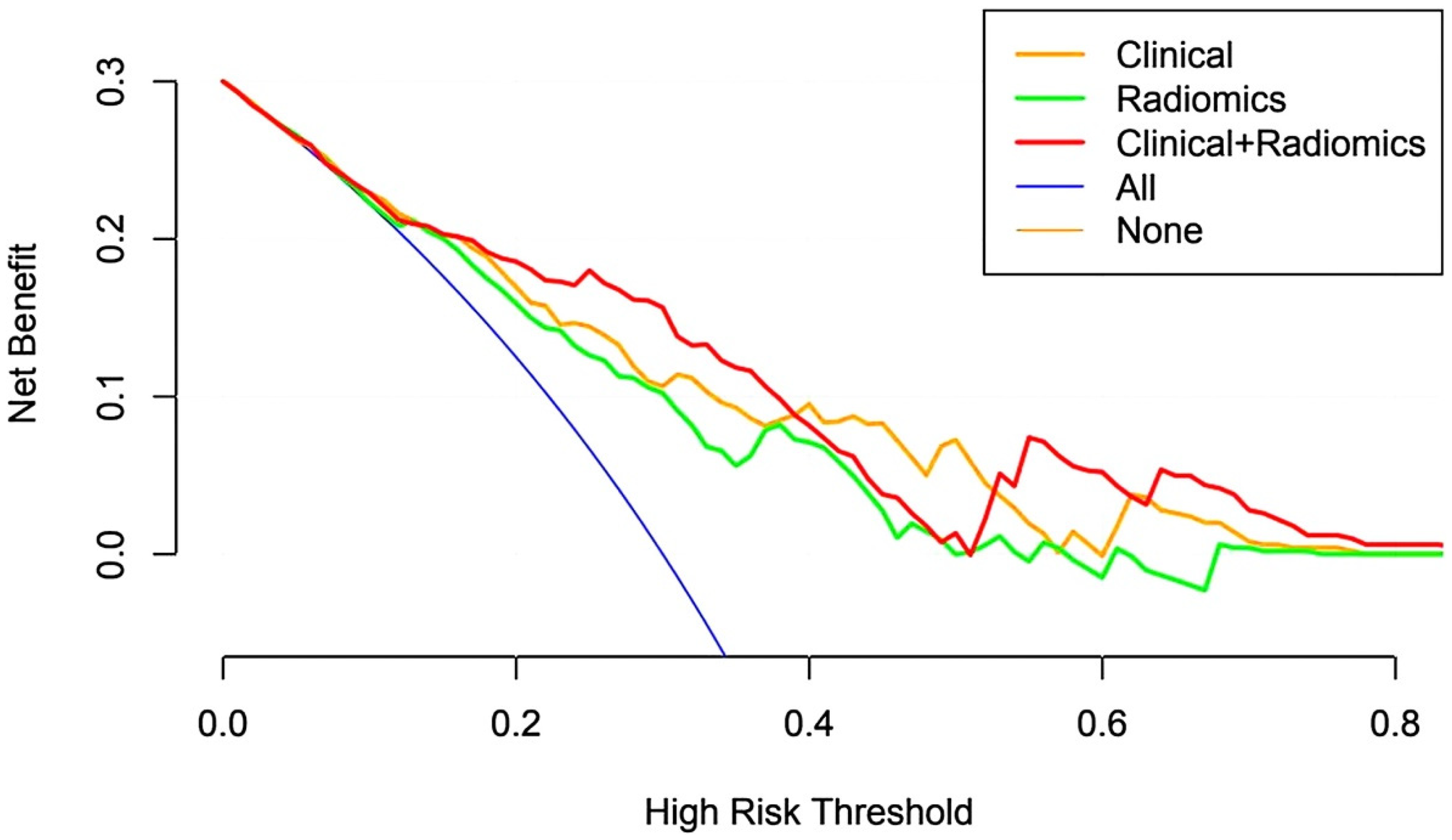
Decision curve analysis of the nomogram. The y-axis indicates the net benefit; the x-axis indicates the threshold probability.
Performance of different models in predicting ablation zone disappearance within 24 months after MWA.
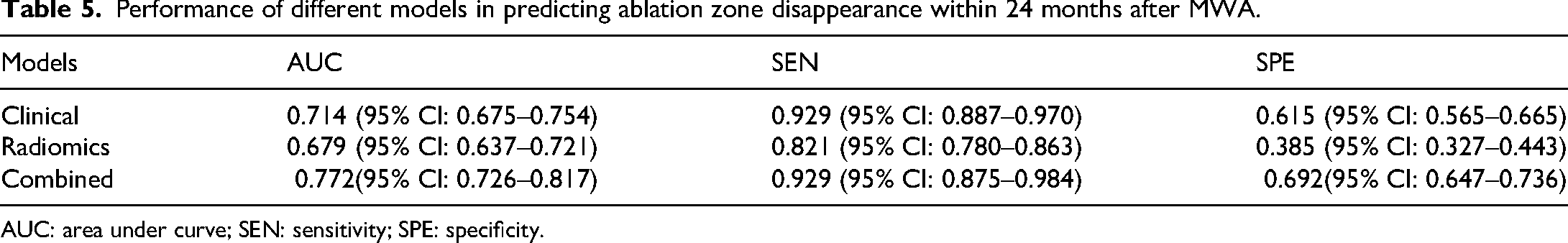
AUC: area under curve; SEN: sensitivity; SPE: specificity.
Discussion
PTC is the most common endocrine malignancy, with a significantly increasing incidence over the past decade worldwide.2,21,22 PTMC maintains a consistently high survival rate and also a marked upward trend. 23 Currently, controversy abounds with the options of treatment strategy for PTMC. In recent years, thermal ablation has emerged as a new potential therapeutic alternative for patients with PTMC. Numerous studies have demonstrated the efficacy and safety of thermal ablation in managing patients with inoperable PTMC and low-risk PTC.14–17 The disappearance rate after thermal ablation was 66% (95% CI, 52%–81%) at 12 months of follow-up and increased to 79% (95% CI, 65%–94%) at the end of follow-up. 24 Consequently, whether ablation zones will disappear within a specific timeframe after PTMC ablation is a key concern for both clinicians and patients. It may induce patients anxiety that ablation zones cannot disappear or ablation zones disappear after a long time. And that may even lead to unnecessary secondary surgical interventions.
The main finding of this study is that the combined model integrating MWA energy, baseline lesion volume, and Rad-score could enhance the accuracy of predicting ablation zones disappearance within 24 months following MWA for PTMC. Therefore, we obtained a novel tool for identifying optimal candidates for MWA. When clinicians and patients have to decide between MWA and surgical resection for PTMC, the predictive outcomes of the combined model may assist in choosing the appropriate treatment strategy.
Choi 25 systematically reviewed and analyzed 11 recent studies on thermal ablation for PTMC (including 4 on MWA, 4 on RFA, and 4 on LA) and reported that the complete disappearance rate of PTMC was 57.6% (95% CI, 35.4–79.8%); the recurrence rate was 0.4% (95% CI, 0–1.1%); the average volume reduction rate (VRR) was 98.1% (95% CI, 96.7–99.5%); and the incidence of major complications was only 0.7% (95% CI, 0–1.5%), all of which were non-life-threatening voice changes. These findings demonstrate the efficacy and safety of thermal ablation in the treatment of PTMC.
Our team has attempted to use MWA for treatment of patients with low-risk PTC since 2013. All patients underwent strict preprocedural evaluation. In this study, 160 of 201 lesions (79.6%) completely disappeared. Among them, 152 lesions (75.6%) disappeared within 24 months after MWA. Some lesions disappeared as early as 3 months after MWA. Only 4.0% (8/201) lesions disappeared beyond 24 months of follow-up.
By comparing the clinical characteristics and MWA procedural parameters between patients with and without ablation zone disappearance within 24 months after MWA, we identified significant differences between the two groups in baseline lesion maximum diameter, baseline lesion volume, the duration of procedure, MWA energy delivered, and MWA energy of unit volume (p < 0.05). Logistic regression analysis showed that MWA energy and baseline lesion volume were independent predictors of ablation zone disappearance within 24 months after MWA. A study by Cho revealed a correlation between disappearance of lesions after MWA and preoperative lesion diameter. 26 Lesions with preoperative maximum diameter ≤5 mm are more likely to disappear than those with preoperative maximum diameter ≥5 mm. A meta-analysis of thermal ablation for PTMC showed that the mean VRR of RFA was 99.3%, whereas the VRRs of MWA and LA were slightly lower (95.3% and 88.6%, respectively). That may be affected by the different energy outputs generated by various ablation modalities. 25 However, Teng's study27,28 showed that there was no significant correlation between ablation zone disappearance and delivered energy. A study by Yue 29 showed that the lesion disappearance rate was significantly higher in the group with moderate energy and shorter ablation time than in the group with high energy and longer ablation time (78.1% vs 22.2%). In addition, these inconsistent results across above studies might be related to interoperator variability in procedural practices, and also differences in the ablation equipment used, follow-up duration, and US image interpretation among different researchers.
Radiomics is a more precise, objective, and effective approach that can improve the accuracy of traditional imaging diagnosis. It extracts hundreds of quantitative features from medical images and conducts self-training and self-learning based on pathological outcomes to assist in clinical diagnosis and decision-making. Currently, studies based on clinical and radiomics models have been conducted to predict tumor treatment responses, such as those for lung cancer and prostate cancer. Most predictive models have achieved favorable predictive performance. 30 In our study, the AUC and specificity of the combined clinical and radiomics model were significantly superior to those of the clinical model in predicting the disappearance of the ablation zone within 24 months after MWA. It was suggested that the disappearance of the ablation zone after MWA may be correlated with the US features of the lesions. US radiomics features extracted in the study were analyzed. It was found that most of these features differences in the internal echoes of ROIs are challenging for radiologists to distinguish quantitatively.
To provide an useful tool for radiologists, we developed a radiomics nomogram based on the combined clinical and radiomics model. In the independent testing cohort, the AUC of the nomogram was 0.772 (95% CI: 0.726–0.817), which exhibited superior predictive performance to the clinical model and the radiomics model (0.714 [95% CI: 0.675–0.754] and 0.679 [95% CI: 0.637–0.721], respectively). Subtle variations in images and physicians-related subjective factors have a substantial impact on the identification and assessment of lesion characteristics. The application of the radiomics nomogram may provide greater standardization and efficiency in clinical practice. The DCA demonstrated that within the threshold probability range of 0.0–0.8, the net benefit of the combined model was consistently superior to that of the radiomics model and the clinical model. This finding suggests that patients with a predicted probability of 0.1–0.8 are potential candidates for MWA. If this method is clinically applied, it may improve the efficiency and accuracy of clinicians in predicting whether ablation zone will disappear within a specific timeframe after PTMC ablation.
There are some limitations of this study: (1) This is a single-center retrospective clinical study, which was ethically driven to maximize patient benefit. In this study, we enrolled patients with single lesion distant from the thyroid capsule to reduce the risk of recurrence and metastasis of PTMC after MWA. In the future, multicenter large-sample prospective studies will be conducted to assess the safety and efficacy of MWA for low-risk PTC; (2) All cases in this study were followed up for at least 24 months. However, due to the low malignancy, slow progression and high rate of occult cervical lymph node metastasis of PTC, longer-term follow-up is required; (3) The factors affecting the disappearance of the ablation zone included in this study are limited. In the future, we will integrate multimodal US imaging with clinical, molecular pathological, immunohistochemical features and other relevant characteristics to establish a more robust predictive model; (4) We hypothesized a linear relationships between predictors and the outcome's log-odds in this study, applying LASSO regression for feature selection and logistic regression to develop the model. However, Oka 31 emphasized that the complexity of medical imaging and the variability in disease response to treatment may lead to non-linear data. That may induce model bias and reduce generalization ability. In the future, we will employ non-parametric statistical methods to optimize our model.
Conclusion
In conclusion, MWA is a novel therapeutic modality that can effectively and safely treat low-risk PTC. A radiomics nomogram for predicting the disappearance of the ablation zone within 24 months after MWA in PTMC patients was established based on the US radiomics signature and clinical risk factors. The main findings of this study provide a novel approach to identifying the optimal candidates for MWA treatment. MWA can induce significant and sustained volume reduction of PTMC lesions, thus representing a valid alternative to surgery. Our US radiomics nomogram may provide comprehensive information regarding whether the ablation zone will disappear within 24 months after MWA in PTMC patients. This will assist in formulating clinical treatment strategies for PTMC patients. Our study also demonstrated that the ablation zone is more prone to disappearance when smaller lesions are chosen before MWA and MWA energy is properly controlled.
Footnotes
Acknowledgements
We would like to thank Professor Linxue Qian for his valuable guidance on study design and manuscript revision.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by QW, YZ, YL, and ZW. The first draft of the manuscript was written by QW and ZW, and all authors commented on previous versions of the manuscript. All authors contributed to the article and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
