Abstract
Cancer treatment is performed differently depending on location of the tumor and the patient's condition. Among various cancer treatment techniques, microwave ablation (MWA) is a method that uses temperature elevation as its mechanism. It uses a microwave coaxial antenna (MCA), and microwaves emitted from the MCA are absorbed by the tissue, causing tissue temperature to rise. In this study, the slot design within the MCA was modified, and therapeutic effects of each slot design were analyzed. A dual-slot antenna was selected, and microwave energy absorption and corresponding temperature distribution were analyzed numerically based on length ratio between slots, spacing between slots, and input power. Furthermore, based on this, the Arrhenius damage integral was calculated to quantitatively assess the thermal damage to the tumor and surrounding normal tissue. Finally, thermal damage conditions for dual-slot design and input power were presented, which inflicted thermal damage throughout the entire tumor while simultaneously minimizing thermal damage to surrounding normal tissue. The study confirmed that as the area occupied by the slot increases, the input power required to inflict thermal damage on the entire tumor decreases. The conditions that simultaneously inflicted thermal damage on the entire tumor while minimizing thermal damage to surrounding normal tissue were calculated to be ψl = 2.0, lsd = 1.5 mm and Pin = 16 W. The results indicate that while extending the upper slot enhances the ablation performance, varying the lower slot length does not show a consistent trend, and the therapeutic effect is more sensitive to input power than to geometric modification. At these conditions, the degree of damage to surrounding normal tissue was confirmed to be 62.1%. This suggests that the accuracy and efficiency of future MWA procedures can be improved.
Keywords
Introduction
Microwave ablation (MWA) is a cancer treatment technique that is based on the mechanism of temperature elevation through microwave emission.1–3 The temperature increase within tissue during MWA is induced by a microwave coaxial antenna (MCA).4–6 Microwaves emitted from the MCA cause water molecules within the tissue to vibrate through dipole rotation and ionic conduction mechanisms, thereby raising the temperature.7,8 Compared to other methods, MWA has the advantages of causing less bleeding and being able to remove tumors located relatively deep within the body.9,10 Since thermal damage generally occurs when tissue temperature reaches 43 °C or higher, maintaining the tumor temperature above 43 °C during MWA is critically important.11,12
MWA is being researched in various fields such as bioengineering, electrical and electronic engineering, and heat transfer. Within the heat transfer field, there is a strong focus on analyzing temperature rise and its distribution due to microwave absorption within tissues. Radosevic et al. 13 analyzed the differences between continuous and pulse modes of microwave emission during MWA. They calculated the electromagnetic-thermal coupled problem through numerical analysis and identified the coagulation zone in each mode. The results showed that a broader coagulation zone generally occurs in the short pulse mode compared to the continuous mode. Shamekhi et al. 14 analyzed the temperature distribution within tissue when using MCA with various slot designs. They analyzed three commonly used slot designs and a newly proposed micro-cut slot (MCS) design. Based on transient Maxwell's electromagnetic equations, the extended Darcy-Brinkman equation, and local thermal non-equilibrium equations for porous medium approximation, the effective ablation zone was identified for various tumor shapes. The results showed that the slot design generating the optimal ablation zone differs depending on the tumor shape. Furthermore, the newly proposed MCS design was found to minimize reverse heating while achieving high uniformity in the temperature distribution within the tumor. Wei et al. 15 proposed a novel antenna design featuring a tapered-tip symmetric dipole structure with a sleeve. The research showed that when this new antenna design was applied, the ablation zone exhibited a nearly spherical shape. Furthermore, increasing the input power resulted in a broader ablation zone and enhanced sphericity. Additionally, numerous researchers have conducted studies on MWA from a heat transfer perspective.16–19
Existing MWA studies analyzed from a heat transfer perspective have primarily focused on verifying the temperature distribution within cancer tissue resulting from changes in the internal slot design. They alter slot designs and input power to measure the degree of microwave energy absorption within the tissue, calculate the resulting temperature distribution, and assess therapeutic efficacy. However, these studies analyze results with a focus on the tumor itself, and research quantitatively quantifying thermal damage to surrounding normal tissue remains insufficient. Furthermore, they merely analyze results for each case without proposing conditions that yield optimal efficacy. Kim and Kim 16 and Kim and Kim 20 analyzed the therapeutic effects in MWA using antennas with a single slot through numerical analysis. These studies analyzed therapeutic efficacy based on the temperatures at which two cell death mechanisms, apoptosis and necrosis, occur. However, currently, apoptosis is only known to occur within a specific temperature range, and research on quantitative metrics, such as the degree to which that temperature must be maintained for cell death, remains insufficient. Accordingly, this study modified the internal slot design of the dual-slot antenna and presented the optimal therapeutic effects for each slot design. Numerical analysis was performed to calculate the distribution of microwave energy absorption within the medium and the temperature distribution as a function of various slot length ratios, spacing, and input power. Furthermore, based on these results, the Arrhenius damage integral was applied to calculate thermal damage not only to the tumor but also to surrounding normal tissue. Conditions were proposed that simultaneously inflict thermal damage to the entire tumor while minimizing thermal damage to surrounding normal tissue.
Materials and methods
Electromagnetic wave behavior analysis
The behavior of the electromagnetic waves emitted after antenna insertion was calculated using the Helmholtz electromagnetic wave equation in Equations (1) to (3).
21
For dielectric properties, they vary depending on each tissue type, microwave frequency, and temperature. 22 Accordingly, in this study, the dielectric properties of the tissue were defined as functions of temperature, separately for tumor and liver tissue, as shown in Equations (6) to (9). 23
The MCA used for treatment consists of a dielectric, catheter, and slot. The dielectric properties were adopted from values in prior research and are summarized in Table 1. 24
Dielectric properties of dielectric, catheter, and slot. 24

Temperature distribution analysis
When microwaves are emitted by inserting an MCA into tissue, the energy is absorbed by the tissue, which results in a temperature increase. In this study, the temperature behavior within the tissue was analyzed using the Pennes bioheat equation in Equation (10). 25
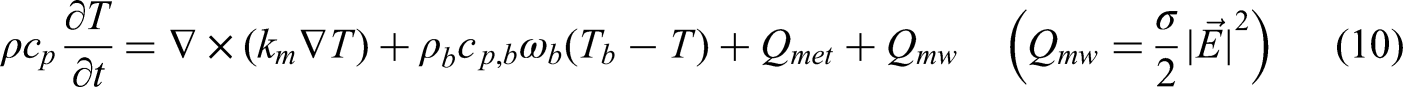
Where ωb, T, t, km, ρ, cp denote blood perfusion rate, temperature, time, thermal conductivity, density, and specific heat, respectively. Qmet represents the heat generated by metabolism and has different values for each tissue. Qmw denotes heat generation due to microwaves and is calculated based on the tissue's electrical conductivity and the magnitude of the electric field.
The thermal properties of liver tissue are also expressed as functions of temperature.
26
They are expressed as functions of Wa, representing the remaining tissue water content, as shown in Equation (11). Based on the calculated Wa, the tissue's density, specific heat, and thermal conductivity can be calculated as functions of Wa, as shown in Equations (12) to (14).

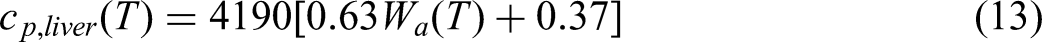

The thermal properties of tumor tissue were set to fixed values, with density, specific heat, and thermal conductivity selected as 1040 kg/m3, 3960 J/(kg·K), and 0.57 W/(m·K), respectively. 24
Thermal damage analysis
Biological tissues sustain thermal damage as temperature increases. Accordingly, this study analyzed this phenomenon using the Arrhenius thermal damage integral (Equation (15)). 27

Where Ω, R, A, Ea represent the Arrhenius damage integral value, ideal gas constant, frequency factor, and activation energy, respectively, with A set to 7.39 × 1039 s−1 and Ea to 2.58 × 105 J/mol. 28 The Ω value calculated via the Arrhenius thermal damage integral indicates the degree of irreversible thermal damage in tissue. When the calculated Ω ≥ 1, it indicates a 63% probability of cell destruction, and Ω must reach 4.63 for the probability of cell destruction to reach 99%. 29 Based on this, the Arrhenius thermal damage ratio (ϕArrh) and the Arrhenius thermal damage ratio of normal tissue (ϕN) were used to calculate the degree of thermal damage in the tumor and surrounding normal tissue. 30 ϕArrh represents the ratio of the volume with Ω ≥ 4.63 to the total tumor volume. In the case of normal tissue, the goal is to minimize thermal damage, so the area where Ω ≥ 1 in the surrounding normal tissue was calculated, and this can be confirmed through ϕN (Equations (16) and (17)). Additionally, for normal tissues, only a distance equivalent to 50% of the tumor radius was included in the calculation range.


Numerical modeling and conditions
This study simulated the implementation of MWA targeting tumors occurring within liver tissue using three-dimensional numerical analysis modeling using COMSOL Multiphysics 6.3. Schematic of the liver tissue, tumor, and MCA, which are the subjects of the numerical analysis, are shown in Figure 1.

Numerical modeling geometry and antenna configuration.
A tumor with a diameter of 20 mm was assumed to have developed at the center of liver tissue with a diameter of 60 mm and a height of 80 mm, and the situation where an MCA was inserted into the center of the tumor was simulated. The MCA consists of a catheter, outer conductor, dielectric, inner conductor, and slot, with diameters set at 1.79 mm, 0.92 mm, 0.67 mm, and 0.27 mm, respectively. The MCA was assumed to be inserted at a point 1 mm from the bottom of the tumor. The slot was divided into an upper slot and a lower slot, with the lower slot positioned 2 mm from the tip of the MCA. Since this study verified results by varying the length of each slot (upper slot length, lower slot length) and the spacing between slots (lsd) in the dual-slot antenna, the length ratio of each slot was defined as shown in Equations (18) and (19).


ψl represents the ratio of upper slot length to lower slot length, while ψu represents the ratio of lower slot length to upper slot length. The reference slot length was fixed at 1 mm, and slot lengths ranging from 0.5 to 2.0 were selected. Additionally, the distance between slots was varied and denoted as lsd.
Finally, in this study, the temperature distribution within the tissue was calculated by varying the length ratio between each slot (ψl, ψu), the distance between slots (lsd), and the input power (Pin). The microwave exposure time was fixed at 600 s, and ψl and ψu were set in 0.5-unit increments from 0.5 to 2.0, respectively. The lsd was set from 0.5 mm to 1.5 mm in 0.5 mm increments, and Pin was set from 0 W to 20 W in 0.5 W increments. Results were analyzed for a total of 861 cases, and each condition is summarized in Table 2.
Numerical analysis conditions.

Results and discussion
Mesh independence analysis
First, a mesh independence test was performed on established numerical analysis model. The tested case involved a slot design with an upper slot length and lower slot length both equal to ψ = 1, where lsd = 1 mm, with 5 W applied. Temperature results were analyzed. The results were shown as a function of radial distance from tumor center (T(r, z = 40)) and as a function of time at 3 mm from the tumor center (T(3,40)).
Figure 2 shows the mesh independence results. As seen in Figure 3 (a) and (b), results showed an error of 3% for all mesh counts. However, differences were clearly present below a certain mesh count, and temperature differences below the 10−3 order were observed starting from approximately 110,000 meshes. Accordingly, this study set the total mesh count to 112,784 and proceeded with the analysis.

Mesh independence results. (a) Radial distance. (b) time.

Validation result. (a) schematic of validation modeling. (b) Validation of temperature changes over time.
Additionally, validation was performed between the results of Aziz et al. 31 and the numerical analysis model constructed in this study. This model inserted an MCA into the center of a liver tissue with a radius of 30 mm and a height of 80 mm. A dual-slot antenna was used, operating at a frequency of 2.45 GHz with an input power of 10 W. The validation was performed by comparing the temperature results at points 5 mm, 10 mm, and 15 mm radially from the bottom, each located 20 mm from the bottom. The comparison confirmed excellent agreement with the results of Aziz et al., 31 with the average RMSE calculated as 0.23 for each point. This confirmed the validity of the numerical analysis model constructed in this study.
Absorbed power analysis
Depending on the slot design within the antenna, the degree of microwave energy absorption within the tissue varies. Accordingly, the degree of microwave energy absorption within the tissue was analyzed for various dual-slot designs. Due to space limitations, changes in ψl were only graphically represented for the case where lsd = 1 mm, and changes in lsd were only graphically represented for the case where ψu = 1.5.
Figure 4 shows the degree of microwave energy absorption in tissue for various ψl values when Pin is 14 W and lsd is 1 mm. The white line in the graph indicates the tumor boundary. As ψl increases, the length of the upper slot increases. Consequently, the range of microwave energy within the tumor was observed to widen. If the range of microwave energy absorption within the tumor can be controlled to be even wider, it is judged that maximizing the length of the upper slot would be more advantageous, as this would induce a temperature rise over a broader area within the tumor.
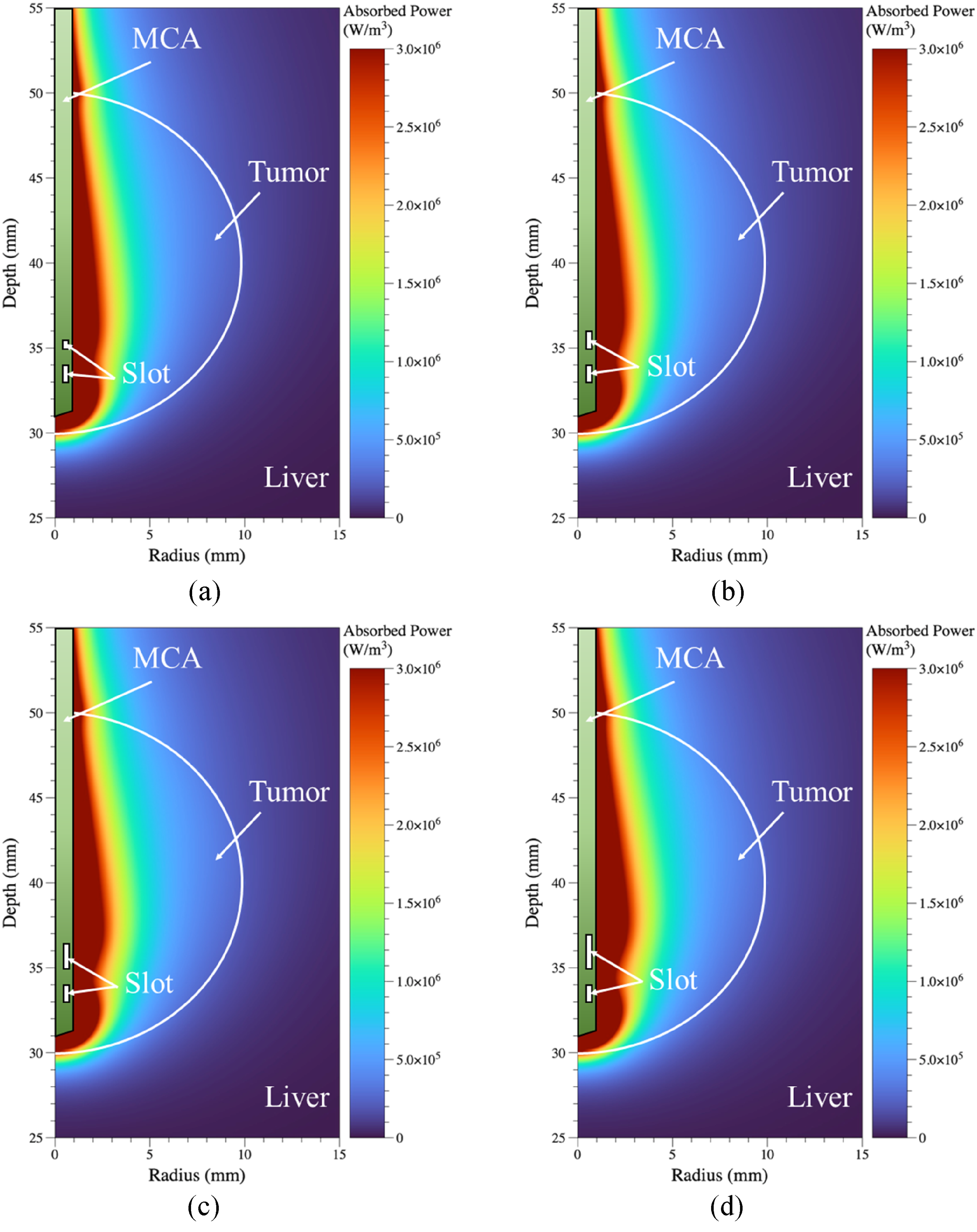
Microwave energy absorption for various ψl (Pin = 14 W, lsd = 1 mm). (a) ψl = 0.5. (b) ψl = 1.0. (c) ψl = 1.5. (d) ψl = 2.0.
Figure 5 shows the degree of microwave energy absorption in tissue for various lsd values when Pin is 14 W and ψu is 1.5. Similarly, the white line in the graph indicates the tumor boundary. As lsd increases, the position of the upper slot rises, confirming an increase in the range of microwave energy absorption within the tumor. Therefore, increasing lsd is also considered more advantageous as it can induce a broader range of temperature elevation within the tumor. However, if the range of temperature elevation within the tumor expands, temperature elevation in the surrounding normal tissue may also occur over a wider area. Consequently, it is necessary to carefully monitor the temperature distribution within the tissue.

Microwave power absorption for various lsd (Pin = 14 W, ψu = 1.5). (a) lsd = 0.5 mm. (b) lsd = 1.0 mm. (c) lsd = 1.5 mm.
Temperature distribution analysis
In previous Section, the distribution of microwave energy absorption within tissue was shown for various dual-slot antenna slot designs. Changes in slot design alter the range of heat generation within tissue. Accordingly, this section presents the temperature distribution within tissue for different slot designs. Due to space limitations, only the same cases as in previous Section are shown graphically in this Section.
Figure 6 shows the temperature distribution within the tissue for various ψl values when the Pin power is 14 W, the lsd is 1 mm, and the exposure time t is 600 s. The black line in the graph indicates the tumor boundary. The red line indicates the point where the Arrhenius damage integral Ω equals 1, and the blue line indicates the point where Ω equals 4.63. As mentioned earlier, when Ω ≥ 1, cell destruction is considered to occur with a 63% probability in the tissue, and when Ω ≥ 4.63, cell destruction is considered to occur with a 99% probability. The region within the red and blue line in the graph corresponds to this area. As shown in the graph, increasing ψl expands the region within the tumor where Ω ≥ 4.63 and the region in normal tissue where Ω ≥ 1. This is thought to be due to the increased area of microwave energy absorption within the tissue, as confirmed in previous Section. When quantitatively identifying the areas within the tumor where Ω ≥ 4.63, the percentages of tumor volume were found to be 83.7% at ψl = 0.5, 88.4% at ψl = 1.0, 92.0% at ψl = 1.5, and 94.8% at ψl = 2.0. Additionally, the ablation area in typical MWA does not occur spherically but rather in a droplet shape. Accordingly, analyzing from the aspect ratio perspective of the ablation area, regions with Ω ≥ 4.63 were confirmed at ψl = 0.5 to 4, ψl = 1.0 to 4.1, ψl = 1.5 to 4.2, and ψl = 2.0 to 4.25. However, as the degree of thermal damage within the tumor increased, thermal damage also increased in the surrounding normal tissue. These results will be discussed in greater detail in the Section on the Arrhenius thermal damage ratio, which quantitatively calculates the thermal damage to the entire tumor.

Temperature distribution for various ψl (Pin = 14 W, lsd = 1 mm, t = 600 s). (a) ψl = 0.5. (b) ψl = 1.0. (c) ψl = 1.5. (d) ψl = 2.0.
Figure 7 shows the tissue temperature distribution for various lsd values when Pin = 14 W, ψu = 1.5, and t = 600 s. As lsd increases, the position of the upper slot rises. Consequently, the range of microwave energy absorption within the tumor increases, leading to a broader temperature rise. This means that at the same input power, the area of heat generation increases, leading to a greater degree of thermal damage within the tumor. Quantitatively, as lsd increased from 0.5 mm to 1.5 mm, the degree of thermal damage within the tumor was confirmed to be 88.2%, 91.9%, and 94.7%, respectively. From an aspect ratio perspective, analyzing the ablation zone (Ω ≥ 4.63), the values were confirmed to be 4.12, 4.1, and 3.89 respectively as lsd increased. However, when comparing the results in Figures 6 and 7, the differences in therapeutic effects caused by variations in the upper and lower slot ratios and the slot-to-slot distance are not easily distinguishable in the visualized results. This is mainly because all cases were simulated under the same input power condition. Nevertheless, a general trend can be observed in both figures: the therapeutic effect tends to improve as the total area occupied by the slots increases. Depending on each slot design condition, however, the input power required to induce thermal damage at all points within the tumor varies accordingly. As a result, this relationship does not hold uniformly across all configurations or input powers. In particular, as will be discussed later in Figures 8 and 9, when only the lower slot length is modified, the increase in the total slot area does not always lead to an enhanced therapeutic outcome. And also, while it is important to induce thermal damage at all points within the tumor during treatment, as the extent of thermal damage within the tumor increases, thermal damage eventually occurs in the surrounding normal tissue as well. Accordingly, the following section analyzes the thermal damage to both the tumor and surrounding normal tissue in all cases to present the conditions for achieving optimal therapeutic efficacy.

Temperature distribution for various lsd (Pin = 14 W, ψu = 1.5, t = 600 s). (a) lsd = 0.5 mm. (b) lsd = 1.0 mm. (c) lsd = 1.5 mm.
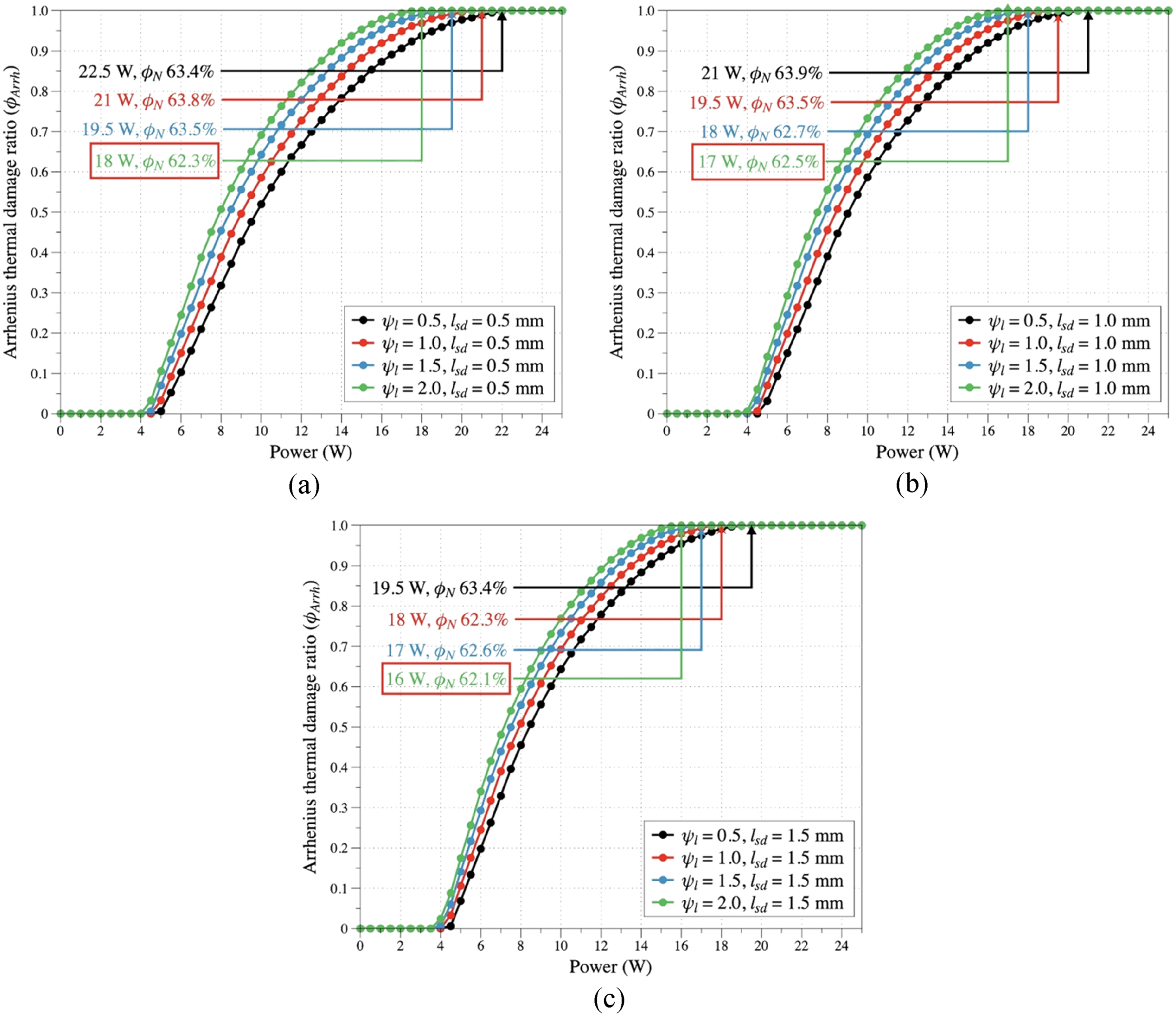
Arrhenius thermal damage ratio (ϕArrh) for various ψl and lsd. (a) lsd = 0.5 mm. (b) lsd = 1.0 mm. (c) lsd = 1.5 mm.

Arrhenius thermal damage ratio (ϕArrh) for various ψu and lsd. (a) lsd = 0.5 mm. (b) lsd = 1.0 mm. (c) lsd = 1.5 mm.
Quantitative thermal damage analysis
When the temperature of tissue rises excessively, thermal damage occurs. In the case of tumors, thermal damage must be induced, but for surrounding normal tissue, thermal damage must be minimized. Accordingly, this section analyzed the degree of thermal damage to the tumor using ϕArrh and quantitatively analyzed the thermal damage to the surrounding normal tissue using ϕN to determine the conditions for achieving optimal therapeutic efficacy.
Figures 8 and 9 are graphs analyzing ϕArrh for ψl and lsd, and ψu and lsd respectively, and analyzing ϕN at the point where ϕArrh begins to reach 1. The case marked with a red box in the graph indicates the case with the smallest ϕN among each set of cases. Generally, it was confirmed that as ψl and ψu increase, Pin at which ϕArrh begins to reach 1 decreases. This is because increasing ψl and ψu increases the heat generation area within the tumor, allowing the temperature causing thermal damage to be reached even with a relatively low Pin applied. Similarly, as lsd increases, the thermal generation area also increases. Consequently, Pin at which ϕN begins to reach 1 was observed to decrease. However, when analyzing the results from the perspective of ϕN at the point where ϕArrh begins to reach 1, no clear trend emerged in the results based on increases or decreases in ψl, ψu, and lsd. This is thought to be because Pin required for ϕArrh to reach 1 differs under each condition, and the heat generation area at each ψl, ψu, and lsd also varies. Overall, thermal damage to surrounding normal tissue was confirmed to occur at approximately 62 to 64% across all cases. Synthesizing the results, the slot design that simultaneously induces thermal damage at all points within the tumor while minimizing damage to surrounding normal tissue was identified as having ψl = 2.0 and lsd = 1.5 mm. At this design, the degree of damage to surrounding normal tissue was confirmed to be 62.1%.
Additionally, Figure 10 shows the tissue temperature and damage distribution conditions at each slot length ratio where all locations within the tumor are ablated while minimizing thermal damage to surrounding normal tissue.

Temperature and damage distribution of optimal conditions for each slot design (expose time = 600 s). (a) ψl = 2.0, lsd = 1.5, Pin = 16 W, ϕN = 62.1%. (b) ψu = 2.0, lsd = 1.0, Pin = 17 W, ϕN = 62.2%.
Analysis of Figure 10 revealed that the final damage to surrounding normal tissue was calculated at 62.1%. Typically, in actual treatment, resection is performed with a margin of 5 to 10 mm. However, if treatment is performed based on the conditions proposed in this study, damage to the lower part of the tumor would be limited to a maximum of 4 mm, and damage to the lateral sides would be limited to a maximum of 2 mm. However, damage to the upper part of the tumor was confirmed to extend up to approximately 15 mm due to the formation of a teardrop-shaped ablation zone. Furthermore, the overall aspect ratio of the ablation zone was calculated to be 1.54. This is because the thermal damage threshold for the tumor was set at a line with Ω = 4.63, while the threshold for surrounding normal tissue was set at a line with Ω = 1.
Additionally, the results were compared with previous studies. Brace et al. 32 optimized dual-slot antenna geometry based on electromagnetic performance and spherical SAR distribution. In contrast, our study focuses on treatment-oriented optimization by evaluating temperature fields and Arrhenius-based tissue damage to determine conditions that achieve complete tumor ablation. Radmilović-Radjenović et al. 33 provided a broad review of computational models for microwave ablation, focusing on general modeling frameworks and patient-specific simulations rather than optimization of a specific antenna design. In contrast, the present study performs a detailed parametric optimization of a dual-slot coaxial antenna and identifies treatment conditions that ensure complete intratumoral ablation while minimizing normal-tissue injury. Ibitoye et al. 34 evaluated a dual-slot antenna with a floating metallic sleeve through combined simulation and ex vivo experiments, focusing on its ability to enlarge the ablation diameter and improve lesion sphericity compared with a monopole antenna. In contrast, the present study does not rely on a sleeve structure but instead performs a systematic parametric optimization of slot length ratios, inter-slot spacing, and input power to identify conditions that achieve complete tumor ablation with minimal collateral damage.
Conclusions
This study analyzed the performance of MWA on tumors originating within liver tissue through numerical simulation. It simulated the insertion of a dual-slot antenna into the tumor's center, analyzing results by varying the dual-slot length ratio, the distance between slots, and the input power. Electromagnetic wave behavior within the tissue was analyzed using the Helmholtz electromagnetic wave equation, while temperature distribution analysis utilized the Pennes bioheat equation. Finally, based on the calculated temperature results, the Arrhenius thermal damage ratio for both the tumor and normal tissue was determined to quantify the occurrence of thermal damage occurrence.
The study confirmed that as ψl and ψu increased, the Pin required for ϕArrh to reach 1 decreased. This is because the increased area of heat generation within the tumor due to the rise in ψl and ψu allows the temperature necessary for tissue death to be achieved even at lower Pin values. Furthermore, for surrounding normal tissue, thermal damage of approximately 62 to 64% was observed when all cases were combined. The final conditions that maximized therapeutic efficacy—inducing thermal damage at all points within the tumor while minimizing thermal damage to surrounding normal tissue—were identified as ψl = 2.0, lsd = 1.5 mm and Pin = 16 W. Overall, the ablation efficiency improved with the upper slot extension, but not necessarily with the lower slot variation, and the input power was found to play a more dominant role than the geometric configuration. Based on the results analyzed in this study, it is anticipated that the accuracy and efficiency of MWA can be enhanced in future applications.
Footnotes
Abbreviation
Ethical committee/institutional review board (IRB) approval
No experiments were performed that required IRB or Ethical committee approval.
Authors contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea, (grant number NRF-2022R1A2C2012470).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available on request.









