Abstract
Background and Objectives
Tumor Endothelial Marker 8 (TEM8) is integral to angiogenesis, tumor microenvironment remodeling, and cancer cell proliferation, and its expression is upregulated in a variety of malignancies. Despite its potential as both a prognostic biomarker and a therapeutic target in triple-negative breast cancer (TNBC), the exact role of TEM8 in TNBC remains poorly understood. This study seeks to examine the expression of TEM8 protein in TNBC and explore its associations with clinicopathological characteristics and patient survival outcomes.
Methods
Clinical and pathological data from 118 patients diagnosed with TNBC via surgical pathology between January 2015 and December 2017 were retrospectively analyzed at Harbin Medical University Cancer Hospital. A total of 35 adjacent non-cancerous tissue samples were randomly selected from the tumor tissue samples of the 118 TNBC patients. Immunohistochemistry (IHC) was employed to assess the expression levels of TEM8 and CD31 proteins. The correlations between TEM8 expression and various clinicopathological features, as well as survival status, were analyzed.
Result
The positive expression rate of TEM8 in TNBC tumor tissues was 89.8% (106/118), significantly higher than 60% (21/35) observed in adjacent non-tumorous tissues (χ2 = 17.029,
Conclusions
TEM8 is highly expressed in TNBC tissues and is closely associated with angiogenesis, tumor proliferation, lymph node metastasis, and poor prognosis. TEM8 may serve as a potential prognostic marker, offering new insights for the diagnosis and therapeutic strategies in TNBC.
Introduction
TNBC is an aggressive molecular subtype of breast cancer associated with a poor prognosis, characterized by early onset, high invasiveness, and high risk of recurrence and metastasis. 1 The absence of hormone receptor and HER2 expression severely limits therapeutic options for TNBC. Standard chemotherapy remains the primary treatment option; however, the development of resistance frequently leads to distant metastasis and worsens prognosis. 2 Thus, there is an urgent need to identify novel therapeutic targets and prognostic markers for TNBC.
In the search for viable targets, Tumor Endothelial Marker 8 (TEM8/ANTXR1) has emerged as a promising candidate. This type I transmembrane protein exhibits a stark contrast between its high expression in tumors and low presence in normal tissues.3,4 TEM8 is selectively overexpressed in tumor cells and stromal components—such as endothelial cells, pericytes, and cancer-associated fibroblasts—while remaining scarcely detectable in most normal adult tissues, suggesting a wide therapeutic window. 5 Functionally, TEM8 acts as an active driver of tumor progression rather than a passive marker. It contributes to key cancer hallmarks, including tumor angiogenesis, extracellular matrix remodeling, and metabolic reprogramming, largely through modulating the Wnt/β-catenin pathway and collagen metabolism.6–9 Its pro-tumorigenic role is further corroborated by its overexpression in various malignancies (e.g. lung, gastric, and ovarian cancers), where it promotes proliferation, migration, invasion, and predicts poorer survival.8,10–12
Notably, in breast cancer, TEM8 expression is significantly higher in TNBC compared to luminal and HER2-positive subtypes and is associated with shortened recurrence-free survival.10,13 Mechanistic studies have revealed that TEM8 enhances vasculogenic mimicry and stemness in TNBC by activating the RhoC/ROCK1/SMAD5 signaling pathway, and its expression is regulated post-transcriptionally by factors such as miR-664a-3p and the ubiquitin-proteasome system. 13 Despite these insights, a significant translational gap remains. Current evidence is predominantly derived from mechanistic studies in cell lines or limited patient cohorts. Consequently, there is a conspicuous lack of comprehensive analyses correlating TEM8 protein expression with clinicopathological features in large, well-characterized TNBC cohorts. Fundamental questions regarding its independent prognostic value remain unanswered. To address this, our study aims to rigorously investigate TEM8 protein expression in a large TNBC cohort. We will comprehensively analyze its association with key clinicopathological parameters and, crucially, evaluate its independent prognostic value for patient survival. Our findings are expected to solidify the foundation for establishing TEM8 as a novel prognostic biomarker and a potential therapeutic target in TNBC.
Materials and methods
Patients and clinical samples
Patient cohort and ethical statement
This was a retrospective cohort study. Surgical specimens and clinicopathological data were collected from 118 consecutive TNBC patients diagnosed by postoperative histopathology at the Department of Breast Surgery, Harbin Medical University Cancer Hospital between January 2015 and December 2017. All patients were monitored for a minimum of 5 years, with examinations every 4–6 months for the first 5 years and annually thereafter, until death or the study's conclusion on May 30, 2023. Overall survival was designated as the primary endpoint for prognostic analysis.
Inclusion criteria were as follows: (1) Histopathologically confirmed invasive ductal carcinoma; (2) Postoperative IHC-confirmed triple-negative status; (3) Female patients; (4) Single primary tumor; (5) No preoperative adjuvant therapy; (6) No concurrent malignancies; (7) All patients received postoperative chemotherapy comprising 6–8 cycles of anthracycline-based and taxane-based agents. This study was approved by the Ethics Committee of Harbin Medical University Cancer Hospital (Harbin, China) (Approval No: KY2023-41) on April 12, 2023. All procedures were performed in accordance with the ethical standards of this committee and with the 1964 Helsinki Declaration and its later amendments. The requirement for obtaining written informed consent from participants was waived by the ethics committee.
Immunohistochemical evaluation
Immunohistochemical staining protocol
Immunohistochemical staining was conducted on all samples following established standard procedures. Formalin-fixed, paraffin-embedded tissue sections (4μm thickness) were deparaffinized using xylene, rehydrated through a series of graded alcohol solutions, and rinsed in phosphate-buffered saline (PBS). The sections were subsequently baked at 70°C for 90 minutes to facilitate tissue adhesion. Antigen retrieval was carried out using a high-pressure heat retrieval method with EDTA buffer, followed by overnight incubation at 4°C with a 1:200 dilution of the primary antibody. The specimens were then washed with PBS. For secondary staining, the sections were incubated with biotinylated secondary antibody for 30 minutes, followed by incubation with streptavidin-horseradish peroxidase complex. Signals were visualized using DAB for 10 minutes, and the slides were counterstained with hematoxylin for contrast. PBS was used as a negative control in place of the primary antibody, and known positive specimens were employed as positive controls. The TEM8 antibody (Rabbit polyclonal; RRID: AB_2056891; Proteintech, USA; Catalog: 15091-1-AP; Working dilution: 1:200) and CD31 antibody (Mouse monoclonal Clone: WM-59;RRID:AB_2935131;Proteintech,USA;Catalog:CL750-65268;Working dilution:1:500) were procured from Proteintech, while the DAB kit (catalog #ZLI-9018) was sourced from Zhongshan Jinqiao Biotech
Evaluation of standard markers (ER, PR, HER2)
ER and PR status were assessed using a two-step IHC method (Zhongshan-Bio Co.), with positivity defined as ≥1% of tumor cell nuclei showing staining 10. HER2 scoring followed established guidelines: scores of 0 or 1+ were negative; 3+ was positive. A score of 2+ was considered equivocal and required further FISH testing for confirmation, which was not included in this TNBC cohort. Triple-negative status was defined by the absence of ER, PR, and HER2 expression.
Evaluation of TEM8 expression
Immunohistochemically processed specimens were evaluated using a double-blind method, where two independent observers reviewed the slides to minimize bias and error. For each slide, 3–5 high-power fields (at ×400 magnification) were randomly selected, and semi-quantitative assessment was performed based on staining intensity and the percentage of positive cells within the cytoplasm. Staining intensity was graded as follows: 0 (no staining), 1 (weak staining, light yellow), 2 (moderate staining, yellow-brown), and 3 (strong staining, brown). The extent of reactivity (ranging from 0 to 100%) was scored as follows: 0 (no positive tumor cells), 1 (≤10% positive tumor cells), 2 (10–50% positive tumor cells), 3 (50–75% positive tumor cells), and 4 (>75% positive tumor cells). The cytoplasmic expression score was calculated by multiplying the intensity score with the extent of reactivity score. Scores were classified as follows: 0–2 (negative, −), 3–4 (positive, +), 5–8 (moderate, ++), and >9 (strong, +++). Total scores ≤4 were classified as low expression, while scores >4 were categorized as high expression.
Evaluation of MVD
Specimens were stained with CD31, and MVD was assessed using the Weidner method. Initially, within areas of tumor cell infiltration, five regions with the highest density of clearly stained microvessel endothelial cells and optimal background contrast were identified under low magnification. Each distinct blood vessel or isolated endothelial cell, separated from other vessels or tumor cells/tissues, was counted as a single microvessel. Microvessels were then counted at ×400 magnification in each of the five selected fields, and the average count was recorded as the MVD value.
Statistical analysis
All statistical analyses were performed using SPSS 27.0 for Windows (SPSS, Inc., Chicago, IL). Continuous variables are presented as mean ± standard deviation or median with interquartile range and were analyzed using the unpaired Welch's t-test or non-parametric rank sum test, as appropriate. Categorical variables are expressed as frequency (percentage) and analyzed using the chi-square test or Fisher's exact test. OS was estimated using the Kaplan–Meier method, and differences between survival curves were compared with the log-rank test. The effects of various variables on survival were assessed using both univariate and multivariate Cox regression analyses, with hazard ratios (HRs) and their 95% confidence intervals (CIs) reported for each marker. A p-value of less than 0.05 was considered statistically significant.
Results
Comparative analysis of TEM8 expression in TNBC and adjacent non-tumor tissues
IHC analysis revealed that TEM8 protein was primarily localized in the cytoplasm, presenting as brownish-yellow granular deposits, with no nuclear staining observed (Figure 1). According to the predefined scoring criteria (refer to Methods section), TEM8 expression was significantly higher in TNBC tissues than in adjacent nontumor tissues. The distribution of expression scores (Figure 1) demonstrated that adjacent tissues were predominantly found in the low-expression categories (0–2 points: 40.0%; 3–4 points: 54.3%), whereas TNBC tissues were primarily concentrated in the high-expression categories (3–4 points: 69.5%; ≥5 points: 20.3%). Using a cutoff score of ≥3 to define positive expression, the TEM8 positivity rate was significantly higher in TNBC tissues (89.8%, 106/118) compared to adjacent tissues (60.0%, 21/35), a difference that was statistically significant (χ2 = 17.029,
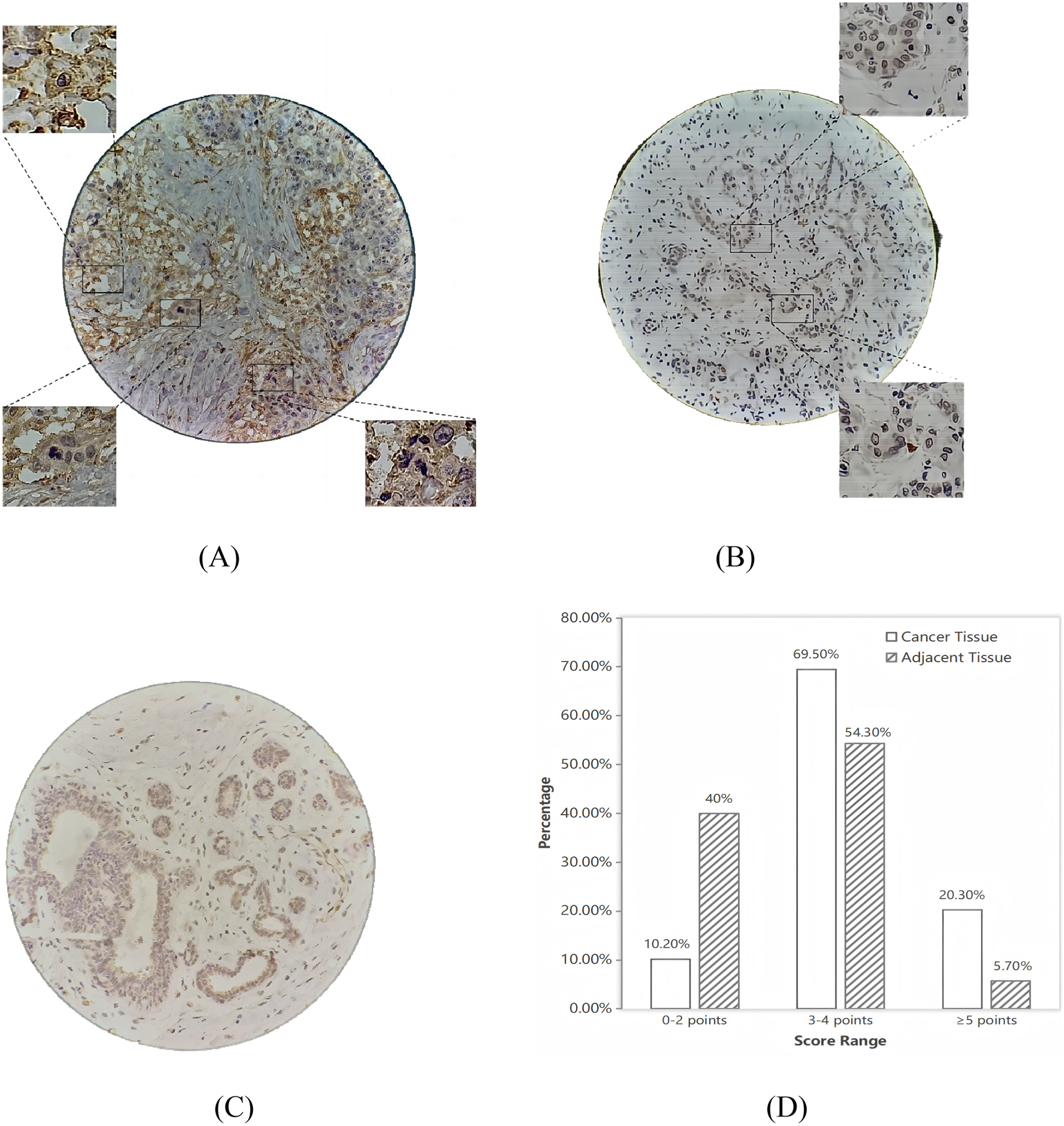
Expression and distribution of TEM8 in TNBC patient tissues. (A–C) Representative IHC images of TEM8 expression in TNBC tissues and adjacent non-tumor tissues (A: A case with high TEM8 expression. B: A case with low TEM8 expression. C: Adjacent normal tissue. 400×). (D) Distribution of TEM8 protein expression scores in 153 TNBC tissue specimens.
Expression of TEM8 in cancer tissues versus adjacent tissues of TNBC patients.

Association between TEM8 expression and MVD in TNBC
To further explore the relationship between TEM8 expression and MVD in TNBC, CD31 immunohistochemical staining was conducted on tumor tissue sections from 118 TNBC patients, followed by MVD quantification (Figure 2). The findings demonstrated that TNBC tissues exhibiting high TEM8 expression had significantly greater MVD values (26.33 ± 8.48) compared to those with low TEM8 expression (22.24 ± 7.37) (

Expression of CD31 in TNBC. (A, B: MVD status in high expression group specimens; C: MVD status in low expression group specimen; IHC, 400×).
Comparative analysis of CD31-positive microvessel density (MVD) in 118 TNBC patients stratified by TEM8 expression levels.

Statistical analysis was performed using Welch's t-test due to unequal variances between groups.
Association between TEM8 expression and clinicopathological features in TNBC
TEM8 expression was significantly associated with TNM stage and axillary lymph node metastasis (
Association between TEM8 expression levels and clinicopathological characteristics in 118 TNBC patients.

Notes: (a) TNM stage: The association between variables was determined to be statistically significant (
Kaplan–Meier survival analysis
Kaplan-Meier survival curves, stratified by TEM8 expression, are depicted in Figure 3. High TEM8 expression significantly affected overall survival (Log-rank χ2 = 5.521,

Kaplan-Meier survival curves of 118 TNBC patients stratified by TEM8 expression.
Univariate and multivariate analysis of prognostic factors in TNBC patients
Both univariate and multivariate survival analyses were conducted to assess the impact of TEM8 expression and clinicopathological characteristics on prognosis. Univariate analysis of OS using Cox regression identified TEM8 expression (
Univariate and multivariate cox regression analysis of prognostic factors in TNBC patients.
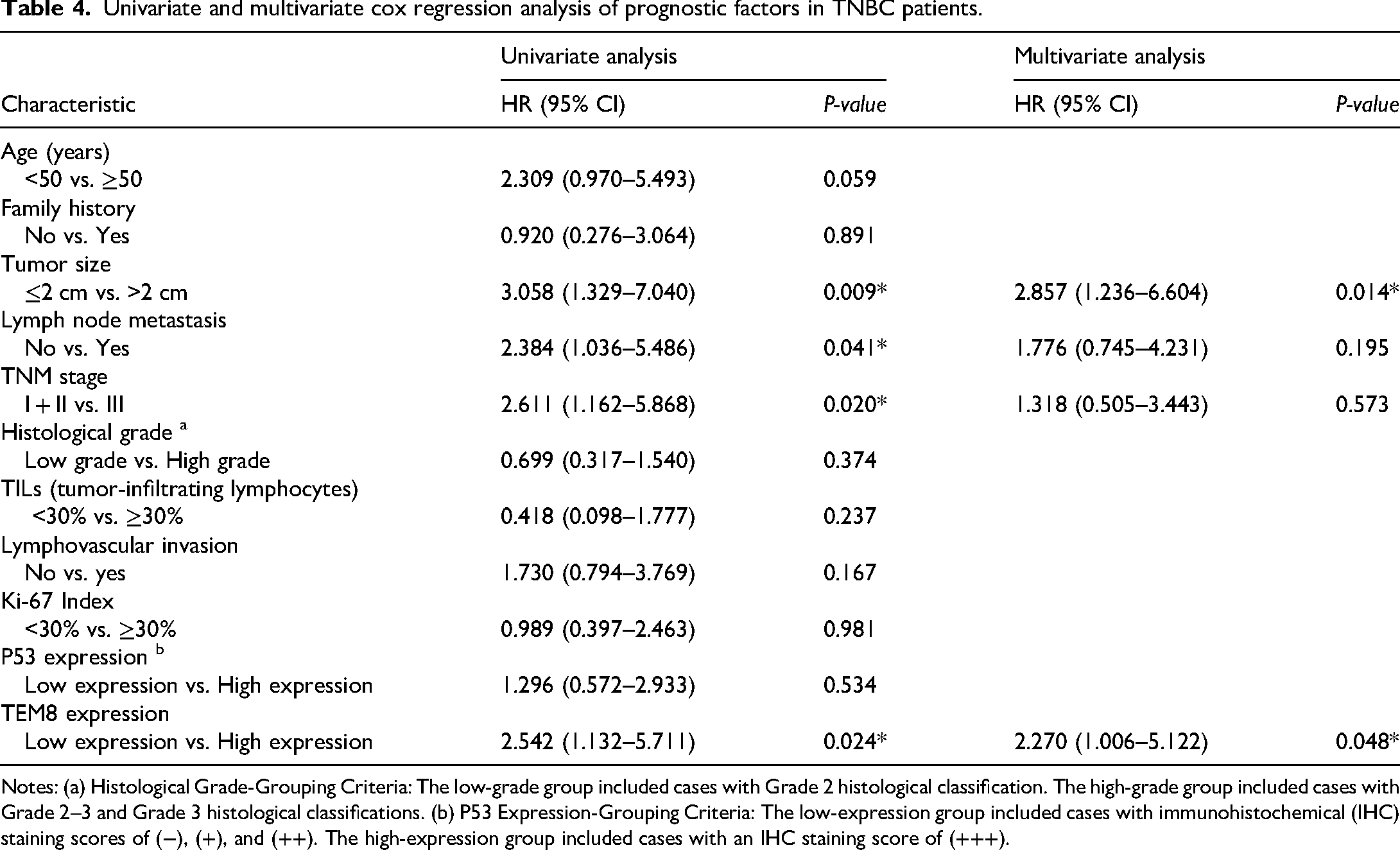
Notes: (a) Histological Grade-Grouping Criteria: The low-grade group included cases with Grade 2 histological classification. The high-grade group included cases with Grade 2–3 and Grade 3 histological classifications. (b) P53 Expression-Grouping Criteria: The low-expression group included cases with immunohistochemical (IHC) staining scores of (−), (+), and (++). The high-expression group included cases with an IHC staining score of (+++).
Discussion
Although immune checkpoint inhibitors combined with chemotherapy have become the standard of care for patients with PD-L1-positive advanced triple-negative breast cancer 14 (e.g. as demonstrated in the KEYNOTE-355 trial), the majority of patients still derive limited benefit, and primary or acquired resistance remains common. This underscores the urgent need to dissect the immunosuppressive tumor microenvironment and identify novel therapeutic targets. This strategy leverages synergistic interactions within the TME to achieve superior clinical outcomes.15–17 This is particularly relevant in TNBC, where angiogenesis and an immunosuppressive TME are critical drivers of tumor growth and metastatic dissemination. The tumor vasculature not only fulfills the obligatory nutrient and oxygen demands of rapidly proliferating cells but also constitutes a critical conduit for metastasis.18,19 Moreover, angiogenesis plays a central role in shaping the TME by influencing immune cell infiltration, promoting hypoxia, and facilitating tumor adaptation20–23 A key translational advantage of targeting the tumor vasculature lies in the relative genetic stability of endothelial cells, which are less prone to the acquisition of resistance compared to genomically unstable tumor cells. Consequently, identifying key nodal points that co-regulate both angiogenesis and immune evasion is paramount for developing next-generation therapies. Targeting the vascular niche presents a rational strategy to enhance the efficacy of immunotherapies and overcome the significant challenges of drug resistance and limited treatment options in TNBC.
The present study establishes TEM8 as a critical contributor to the aggressive phenotype of TNBC. We demonstrate that high TEM8 expression is significantly correlated with advanced disease stage, lymph node metastasis, increased tumor size, and elevated MVD. Moreover, TEM8 is an independent prognostic factor for worse overall survival. A pivotal finding of our research is the significant association between TEM8 expression and key T-cell exhaustion markers, providing a novel mechanistic link between TEM8 and the immunosuppressive TME. These findings position TEM8 not only as a biomarker but also as a promising therapeutic target, particularly within the compelling framework of combining anti-angiogenic and immunotherapeutic strategies. Our study situates TEM8 at the nexus of these two therapeutic pillars. The correlation between TEM8 expression and aggressive clinicopathological features mirrors findings across multiple malignancies, including lung, gastric, and ovarian cancers.10,24–26 This cross-cancer consistency underscores TEM8's fundamental role in tumor progression. The strong link to lymph node metastasis, consistent with prior observations in breast and colorectal cancers,10,25 suggests that TEM8 may facilitate metastatic spread via mechanisms such as enhanced endothelial adhesion or lymphangiogenesis.
A cornerstone of our findings is the role of TEM8 in angiogenesis. The significant increase in MVD associated with high TEM8 expression provides direct in vivo evidence of its pro-angiogenic function in TNBC. This aligns with established molecular mechanisms whereby TEM8, through its von Willebrand factor A (vWA) domain, interacts with collagen to activate the Wnt/β-catenin pathway, thereby promoting endothelial cell proliferation and vessel formation.6,27 Furthermore, TEM8 is known to drive vasculogenic mimicry via the RhoC/ROCK1/SMAD5 signaling axis. 13 From a therapeutic perspective, this reinforces the rationale for targeting TEM8 as an alternative or complementary approach to current VEGF-directed anti-angiogenic therapies. While previous research on TEM8 has primarily focused on its pro-angiogenic functions, a pivotal finding of our study is the significant correlation we revealed between TEM8 expression and key T-cell exhaustion markers. This provides the first direct link in clinical samples connecting TEM8 to T-cell dysfunction in TNBC, offering a novel immunological perspective on its function.
Although no statistically significant association was observed between TEM8 protein expression and tumor-infiltrating lymphocytes (TILs), a notable trend was observed: high TEM8 expression correlated with lower TIL levels in 95.8% of cases, consistent with prior research. Our findings align with the established role of TEM8 in immune evasion, which involves recruiting immunosuppressive cells to create an inhibitory microenvironment. Notably, the significant correlation between TEM8 expression and key T cell exhaustion markers (e.g. TIM-3, TIGIT, BTLA) suggests that TEM8 contributes to immune suppression not only by altering the cellular milieu but also by directly promoting T cell exhaustion. 28 Thus, the observed reduction in TIL infiltration in TEM8-high tumors likely reflects its pivotal function in driving T cell dysfunction and fostering an immunosuppressive niche, further supporting its role as a facilitator of immune evasion. Consequently, we propose a rational and compelling therapeutic hypothesis: targeting TEM8 may synergize with ICIs to overcome immune resistance in TNBC. This hypothesis is supported by the clinical success of combining anti-angiogenic agents with ICIs 29 (e.g. the camrelizumab, apatinib, and eribulin regimen achieved an ORR of 37.0% and median PFS of 8.1 months in heavily pretreated advanced TNBC) and is motivated by the limitations of current VEGF-targeted therapies, particularly the emergence of resistance. Given TEM8's role in co-regulating angiogenesis and immunosuppression, targeting it may represent a novel strategy that extends beyond VEGF inhibition, especially for tumors with an immune-desert phenotype or resistance to existing anti-angiogenic drugs. Preclinical proof-of-concept for this approach is strong, demonstrated by the ability of TEM8-specific oncolytic viruses to inflame the TME and the efficacy of TEM8-directed CAR-T cells.30–32 Thus, a TEM8-targeted agent could potentially remodel the TME, sensitizing tumors to subsequent ICI therapy and yielding potent synergistic antitumor effects.
The prognostic significance of TEM8, confirmed here as an independent risk factor for survival, is consistent with its role as a marker of aggressive disease and treatment resistance across numerous cancers, including glioblastoma and prostate cancer. This consolidates TEM8's profile as a multifaceted driver of tumor progression and a gatekeeper of therapy resistance.
Despite the valuable insights gained from this study regarding TEM8 expression and its correlation with clinicopathological parameters in TNBC, several limitations should be acknowledged. First, this study primarily utilized IHC to detect TEM8 expression. Although widely used in clinical research, IHC has limitations in sensitivity and quantification, which may affect the accuracy and reproducibility of the results. Second, the relatively small sample size may limit the generalizability and clinical applicability of the findings. Additionally, as a retrospective analysis, the study may be subject to selection and information biases, which could influence the outcomes. Future research should include a larger and more diverse patient population to enhance the representativeness and robustness of the findings. Furthermore, integrating advanced techniques such as high-throughput sequencing or quantitative PCR could offer a more precise understanding of TEM8 expression and its regulatory mechanisms. Importantly, investigating the specific roles of TEM8 in the tumor immune microenvironment and drug resistance could help clarify its contributions to cancer progression. Such investigations could pave the way for novel diagnostic and therapeutic strategies, offering substantial benefits for TNBC patients.
Footnotes
Acknowledgements
We thank all the patients for their participation in this study.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Harbin Medical University Cancer Hospital (Harbin, China) (Approval No: KY2023-41) on April 12, 2023. All procedures were performed in accordance with the ethical standards of this committee and with the 1964 Helsinki Declaration and its later amendments. The requirement for obtaining written informed consent from participants was waived by the ethics committee.
Author contributions
Furong Huang (first author): conceptualization, data curation, formal analysis, investigation, methodology, writing—original draft, writing—review and editing; Yang Liu (equal contributor): investigation, methodology, writing—original draft, writing—review and editing; Hong Ren: supervision and investigation; Yongdong Jiang and Huilei Qiu (corresponding authors): conceptualization, funding acquisition, resources, supervision, validation, writing—original draft, and writing—review and editing.
We understand that the corresponding authors are the sole contact for the editorial process. They are responsible for communicating with the other authors about progress, submissions of revisions, and final approval of proofs. We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Heilongjiang Postdoctoral Scientific Research Developmental Foundation (grant number LBH-Q20133).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are not publicly available due to privacy or ethical restrictions, but are available from the corresponding author on reasonable request.
