Abstract
Objective
Both subcutaneous adipose tissue (SAT) and neutrophil-to-lymphocyte ratio (NLR) are associated with the prognosis of hepatocellular carcinoma (HCC). Subcutaneous adipose tissue is an immunonutritional indicator, and NLR reflects the inflammatory status. The purpose of this study was to ascertain the validity of SAT/NLR as potential prognostic biomarkers in HCC patients who are undergoing liver resection.
Methods
This retrospective study encompassed the sequential enrollment of 682 patients diagnosed with HCC who underwent liver resection. The patients were categorized into high and low SAT/NLR groups using the median value, and forward stepwise logistic regression was utilized to ascertain independent predictors for one-year HCC recurrence. In order to minimize the influence of confounding variables, a propensity score matching (PSM) analysis was conducted between patients in high and low SAT/NLR groups. The Kaplan–Meier method was employed to assess and compare the recurrence-free survival (RFS) and overall survival (OS) between the two groups.
Results
The study divided patients into two groups based on their SAT/NLR levels: high SAT/NLR (≥35.34) and low SAT/NLR (<35.34) groups. Forward stepwise logistic regression analysis revealed that low SAT/NLR (p < 0.001), tumor size ≥50 mm (p < 0.001), alpha–fetoprotein level >200 ng/mL (p < 0.001), and presence of liver cirrhosis (p < 0.001) were significantly associated with one-year recurrence of HCC. Moreover, the results suggest that RFS and OS were significantly shorter in the low SAT/NLR group compared to the high SAT/NLR group, both before and after PSM (p < 0.05).
Conclusions
The preoperative biomarker SAT/NLR shows potential as a prognostic biomarker for one-year recurrence and prognosis in patients with HCC undergoing liver resection.
Introduction
Liver cancer is globally ranked as the sixth most lethal type of cancer, with hepatocellular carcinoma (HCC) accounting for over 80% of primary liver cancer cases. 1 Liver resection is widely recognized as the primary method for achieving curative treatment for HCC, as supported by multiple guidelines.2–5 Following the Barcelona Clinic Liver Cancer (BCLC) system, Western practice guidelines recommend liver resection for patients with HCC at a very early to early stage, while Asian practice guidelines limit its use to those with intermediate-stage HCC. Overall, patients with HCC undergoing liver resection exhibit varied outcomes, with five-year survival rates ranging from 35% to 70%6–8 and one-year recurrence rates ranging from 20% to 50%. 9 The observed variations can be ascribed to disparities in tumor burdens, histopathological status, immune response status, and nutrition status. In order to improve risk stratification and enhance prognostic accuracy, it is crucial to identify efficacious prognostic biomarkers for HCC patients undergoing liver resection.
In the realm of cancer progression, the significant effects of the inflammatory response and immunonutrition on the mechanisms of priming, proliferation, angiogenesis, and migration are widely recognized. 10 Various reports suggest that HCC fosters an environment marked by inflammation, immunosuppression, and nutritional inadequacies, both locally and systemically.11–15 These circumstances pose significant challenges that can adversely affect clinical outcomes and the efficacy of therapeutic interventions. For instance, studies have shown that HCC displays elevated levels of myeloid-derived suppressor cells in the tumor microenvironment, and HCC patients exhibit higher proportions of regulatory T cells in their peripheral blood.16–18 The neutrophil-to-lymphocyte ratio (NLR) is a marker of inflammation in peripheral blood, which can reflect the systemic immune and inflammatory status, and can be measured by blood routine, fast and convenient to obtain and low price. Elevated NLR indicates the relative increase of neutrophils, relative decrease of lymphocytes and immune imbalance. 19 Neutrophil-to-lymphocyte ratio has been reported to be associated with various inflammatory conditions,20,21 elevated NLR was reported in patients with cancer. 22 Neutrophil-to-lymphocyte ratio have been recognized as dependable prognostic markers for anticipating the clinical outcomes of HCC.16–18 In recent years, several studies revealed that altered body composition is associated with survival in several malignancies.23,24 Compared to commonly used anthropometry, such as body mass index or waist circumference, measurements on computed tomography (CT) scans allow an adequate differentiation of body composition components, including visceral and subcutaneous fat distribution, and are considered as the gold standard technique for assessing skeletal muscle mass.23,25 Moreover, existing scholarly literature has confirmed that subcutaneous adipose tissue (SAT) is a dependable marker of immunonutrition and possesses considerable prognostic significance for HCC patients.26,27 In brief, an increased NLR and a decline in SAT contribute to the promotion of tumor growth, while a decreased NLR and an augmentation in SAT exert a suppressive influence on tumor development. Consequently, the proposed biomarker SAT/NLR combines the advantages of both SAT and NLR, potentially leading to enhanced predictive precision.
In light of the indeterminate prognostic implications of preoperative SAT/NLR in HCC patients undergoing liver resection, this study aims to appraise the prognostic utility of SAT/NLR in this specific patient population and ascertain its suitability as an optimal biomarker for HCC prognosis.
Materials and methods
Study patients
This is a single-cohort, retrospective, and observational study. A total of 682 patients who underwent liver resection as initial therapy between August 2013 and November 2020 were consecutively enrolled in the survey. This retrospective observational study was approved by the institutional review boards of the Second Xiangya Hospital of Central South University and waived the requirement for informed consent as all data were analyzed retrospectively and anonymously. And this study was conducted in accordance with the ethical principles of the Declaration of Helsinki and STROBE 28 guidelines.
The flowchart of the study patients is shown in Figure 1. Patients were included when the following criteria were met (1) age >18 years; (2) preserved liver function; (3) Eastern Cooperative Oncology Group performance status of 0; (4) absence of macrovascular invasion or extrahepatic metastasis; and (5) only a single tumor present. Exclusion criteria were as follows: (1) absence of laboratory information within one week before surgery (n = 22); (2) absence of CT imaging within one month before surgery (n = 18); (3) incomplete follow-up data (n = 15); and (4) poor imaging quality (n = 5). In total, 622 patients were ultimately included and evaluated.

Diagram of the study population.
Data collection
The clinical data involved in this study including: age, gender, BCLC stage, Child–Pugh class, liver cirrhosis, portal hypertension, splenomegaly, satellite nodule, underlying liver disease, and tumor size. The laboratory data including: neutrophil, lymphocyte, platelet, and monocyte counts; serum albumin, total bilirubin, ALBI grade, and alpha–fetoprotein (AFP) levels. The calculated values of NLR, PLR, MLR, SII, and SIRI were obtained using the subsequent formulas: NLR = N/L, PLR = P/L, MLR = M/L, SII = N × P/L, SIRI = N × M/L. (Note: P = platelet count; N = neutrophil count; L = lymphocyte count; M = monocyte count.) The patients underwent CT scans up to one month prior to the surgical procedure. R0 resection was achieved in all patients with preserved liver function (Child–Pugh grade A classification, an indocyanine green 15-min retention rate of less than 30%, and a future remnant liver volume exceeding 40% of the standardized liver volume for patients with liver fibrosis or cirrhosis, or greater than 30% for those without liver fibrosis or cirrhosis) included in this study. Additionally, according to China Liver Cancer Staging, liver resection may be considered for patients presenting with splenomegaly and portal hypertension. 29 The CT images were subjected to independent evaluation by two radiologists, possessing 11 and 20 years of experience in abdominal imaging, respectively. The data collected at the level of the third lumbar vertebra included measurements of SAT, visceral adipose tissue (VAT), and total adipose tissue (TAT) expressed in square centimeters (Figure 2). In instances where the two radiologists disagreed on the interpretation of imaging characteristics, a consensus was reached to determine the final decision.

L3 vertebral level CT plain scan of a HCC patient (A), the red area represents subcutaneous fat deposition (B), the green area represents visceral fat deposition (C), and the blue area represents the total fat deposition (D). HCC: hepatocellular carcinoma; CT: computed tomography.
Recurrence evaluation and follow-up
The primary aim of this study is to assess the correlation between different SAT/NLR level and one-year recurrence rate of HCC after liver resection. In order to mitigate potential confounding and selection bias, propensity score matching (PSM) was employed. The primary clinical endpoint was recurrence-free survival (RFS), defined as the duration between liver resection and tumor recurrence. The secondary endpoint was overall survival (OS), defined as the duration between liver resection and death from any cause.
Statistical analyses
Categorical variables were assessed through the utilization of the χ2 test or the Fisher exact test. Continuous variables are reported as mean and SD if they satisfy normal distribution, and differences in means are compared using independent t-tests; if they do not satisfy normal distribution they are reported as median (interquartile spacing), and differences in medians are compared using Mann–Whitney U test. To identify the most effective marker, a forward stepwise logistic regression model was employed, incorporating both univariate and multivariate analyses. The comparison of one-year postoperative recurrence, RFS, and OS between the Low SAT/NLR and High SAT/NLR groups was conducted using the χ2 test and the log-rank test, both before and after matching. Statistical analyses were performed using the SPSS 26.0 statistical software, and a two-sided value of p < 0.05 denoted statistical significance.
Results
Baseline patients characteristics
A total of 622 patients were included in the study, with 528 males (84.9%) and 94 females (15.1%). The statistical power for this sample size was determined to be 0.99 using G-power software, employing a post-hoc power analysis with an effect size of 0.25, α error probability of 0.05, and n = 622. The prevalence of HBV infection in the study cohort was 76.0%, with BCLC stage A accounting for the majority at 92.0%. The median values for SAT and NLR were 35.34 cm2 (IQR: 22.39–55.31) and 2.42 (IQR: 1.81–3.25), respectively. Tables 1 and 2 present comprehensive baseline characteristics for both the entire sample and the two distinct groups.
Baseline clinical characteristics of patients in the total cohorts.

Data are presented as the mean ± SD.
Data are presented as median and IQR.
BCLC: Barcelona Clinic Liver Cancer; HBV: hepatitis B virus; ALBI: albumin-bilirubin index; AFP: alpha–fetoprotein; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; MLR: monocyte-to-lymphocyte ratio; SII: systemic immune inflammation; SIRI: systemic inflammation response index; PNI: prognostic nutritional index; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue areas; TAT: total adipose tissue areas.
Clinical characteristics of low SAT/NLR and high SAT/NLR cohort.

Data are presented as the mean ± SD.
Data are presented as median and IQR.
BCLC: Barcelona Clinic Liver Cancer; HBV: hepatitis B virus; ALBI: albumin-bilirubin index; AFP: alpha–fetoprotein; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; MLR: monocyte-to-lymphocyte ratio; SII: systemic immune inflammation; SIRI: systemic inflammation response index; PNI: prognostic nutritional index; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue areas; TAT: total adipose tissue areas.
Potential predictive factors for one-year recurrence
In the present study, a total of 230 patients (37.0%) experienced recurrence within a one-year period. The diagnostic efficacy of various biomarkers and their respective cutoff points were assessed using ROC curves. The median value of the potential biomarkers was established as the cutoff point for the ROC analysis. The area under the curve values for the PNI, MLR, NLR, PLR, SII, SIRI, SAT, VAT, TAT, and SAT/NLR were 0.539, 0.613, 0.629, 0.573, 0.607, 0.625, 0.676, 0.622, 0.666, and 0.709, respectively (Figure 3). In the univariate analysis, a significant correlation was observed between early recurrence of HCC and various factors, including liver cirrhosis (p = 0.004), presence of satellite nodule (p < 0.001), BCLC stage (p = 0.004), AFP level >200 ng/mL (p < 0.001), tumor size ≥50 mm (p < 0.001), NLR ≥ 2.42 (p < 0.001), MLR ≥ 0.23 (p < 0.001), SIRI ≥ 0.76 (p < 0.001), SII ≥ 368.53 (p < 0.001), SAT < 87.91 cm2 (p < 0.001), TAT < 173.32 cm2 (p < 0.001), and SAT/NLR < 35.34 (p < 0.001). Subsequently, a forward stepwise multivariate analysis was conducted using the significant risk factors identified in the univariate analysis. This analysis revealed that the presence of liver cirrhosis, tumor diameter ≥50 mm, AFP ≥ 200 ng/mL, and SAT/NLR < 35.34 was independent biomarkers that exhibited a correlation with early recurrence of HCC following liver resection. The odds ratios and 95% confidence intervals for these biomarkers are presented in Table 3.

Comparison of the AUCs for PNI, MLR, NLR, PLR, SII, SIRI, SAT, VAT, TAT, and SAT/NLR in predicting one-year recurrence of HCC after liver resection. SAT: subcutaneous adipose tissue; NLR: neutrophil-to-lymphocyte ratio; HCC: hepatocellular carcinoma; VAT: visceral adipose tissue; TAT: total adipose tissue; AUC: area under the curve.
Assessment of potential risk factors for 1-year HCC recurrence after liver resection.

BCLC: Barcelona Clinic Liver Cancer; HBV: hepatitis B virus; ALBI: albumin-bilirubin index; AFP: alpha–fetoprotein; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; MLR: monocyte-to-lymphocyte ratio; SII: systemic immune inflammation; SIRI: systemic inflammation response index; PNI: prognostic nutritional index; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue areas; TAT: total adipose tissue areas; HCC: hepatocellular carcinoma.
Impact of low and high SAT/NLR on the RFS and OS before PSM
Compared with low SAT/NLR group, the high SAT/NLR group had a significantly longer median RFS before PSM (high SAT/NLR, 46.3 months [95% CI: 33.7–58.8] vs. low SAT/NLR, 12.9 months [95% CI: 8.7–17.1]; p < 0.001) (Figure 4(a)) and OS (high SAT/NLR, 112.8 months [95% CI: 69.0–156.6] vs. Low SAT/NLR, 52.1 months [95% CI: 37.9–66.3]; p < 0.001) (Figure 4(b)).
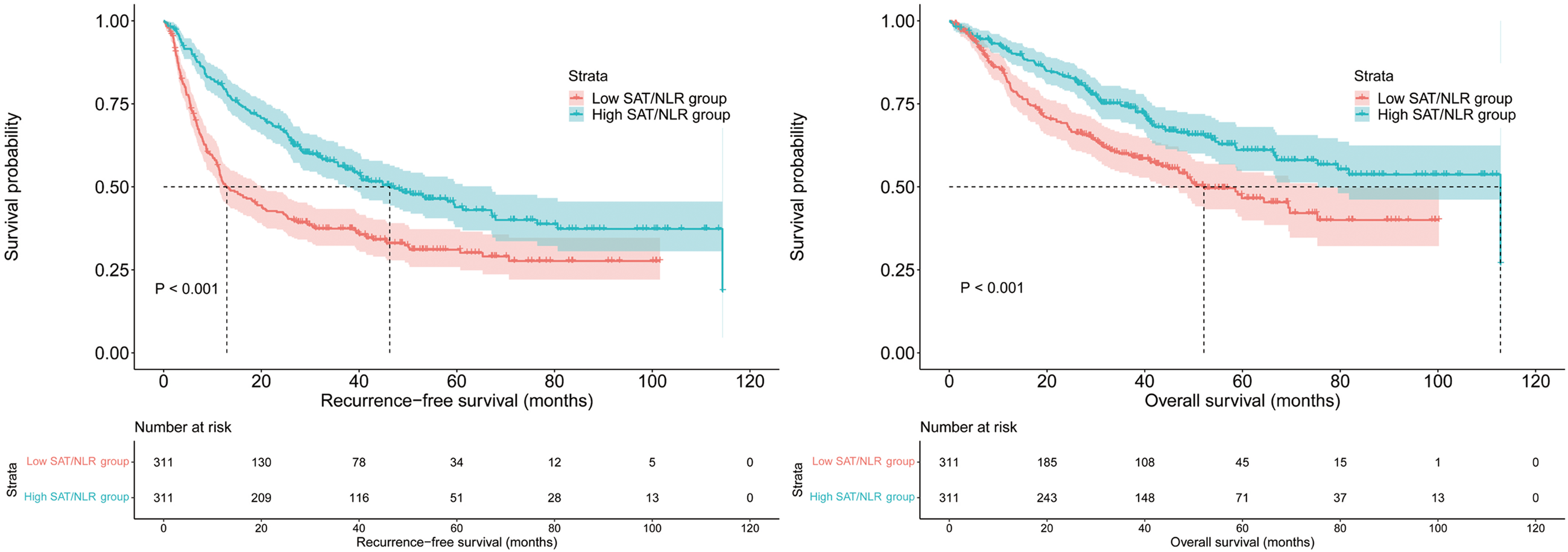
Log rank tests were used to compare RFS (a) and OS (b) between high SAT/NLR and low SAT/NLR groups before PSM. SAT: subcutaneous adipose tissue; NLR: neutrophil-to-lymphocyte ratio; PSM: propensity score matching; RFS: recurrence-free survival; OS: overall survival.
Impact of low and high SAT/NLR on the RFS and OS after PSM
To mitigate the influence of selection bias, PSM was conducted using a caliper value of 0.1, yielding a total of 266 patient pairs. The baseline characteristics of the matched patients exhibited no statistically significant disparities (refer to Table 4). Following the matching process, the high SAT/NLR group demonstrated a notably extended median RFS (high SAT/NLR 39.7 months [95% CI: 29.9–49.5] vs. low SAT/NLR, 13.5 months [95% CI: 8.4–18.6]; p < 0.001) (Figure 5(a)) and OS (high SAT/NLR 81.8 months [95% CI: 74.3–89.3] vs. Low SAT/NLR, 59.9 months [95% CI: 43.0–76.8]; p = 0.02) (Figure 5(b)).
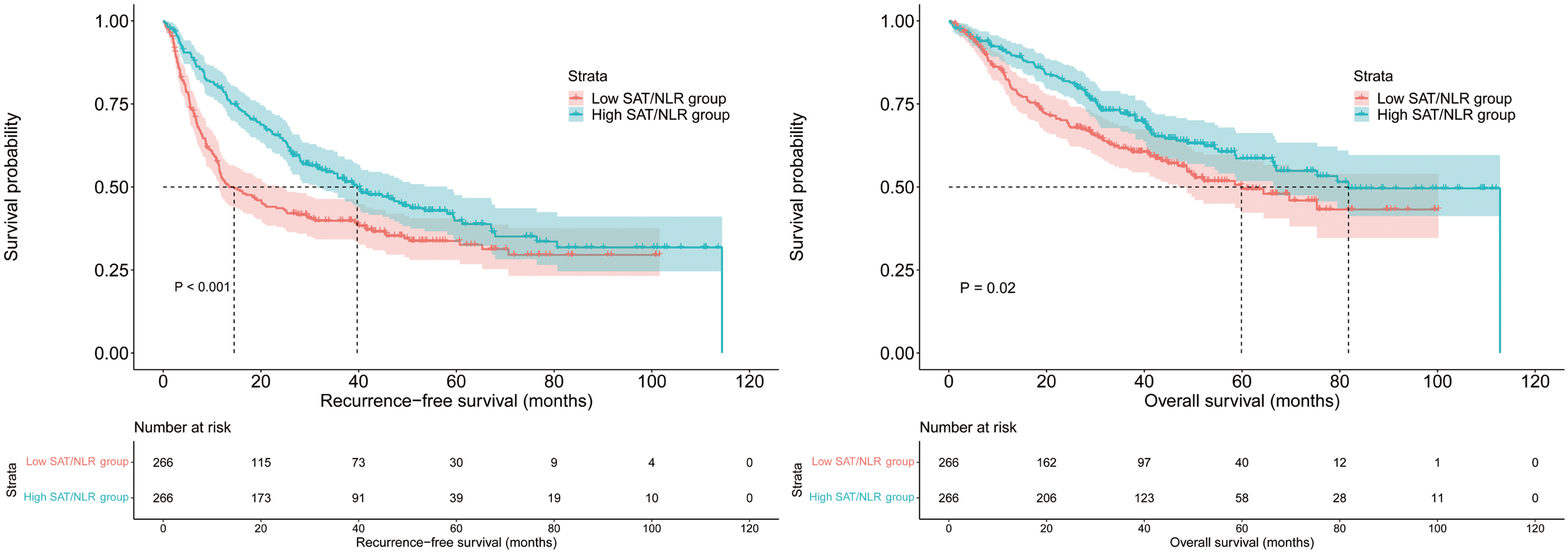
Log rank tests were used to compare RFS (a) and OS (b) between high SAT/NLR and low SAT/NLR groups after PSM. SAT: subcutaneous adipose tissue; NLR: neutrophil-to-lymphocyte ratio; PSM: propensity score matching; RFS: recurrence-free survival; OS: overall survival.
Clinical characteristics of low SAT/NLR and high SAT/NLR cohort after propensity score matching.

Data are presented as median and IQR.
BCLC: Barcelona Clinic Liver Cancer; HBV: hepatitis B virus; ALBI: albumin-bilirubin index; AFP: alpha–fetoprotein; SAT: subcutaneous adipose tissue; NLR: neutrophil-to-lymphocyte ratio.
Discussion
Hepatocellular carcinoma is a prototypical inflammation-associated cancer, with ∼90% of the HCC burden being associated with prolonged inflammation owing to viral hepatitis, excessive alcohol intake, NAFLD, or NASH. 1 The immune microenvironment plays a pivotal role in the pathogenesis of HCC. 1 In HCC, the presence of immune infiltrates is associated with a better prognosis, likely owing to more effective antitumor immunity. 1 This study included some peripheral blood inflammatory markers and body composition for studying the immune and nutritional status of patients. It has been shown that PNI is an independent predictor of survival in patients with early HCC after microwave ablation and in HCC patients receiving targeted therapy.12,30,31 Lower levels of PNI have been noted in other inflammatory conditions such as type 2 diabetes mellitus and infections.32,33 The results of this study results reported that PNI levels were lower in the low SAT / NLR group, but there was no significant association with HCC recurrence after liver resection. It has been shown that MLR can evaluate the prognosis of patients undergoing RFA and TACE.31,34,35 MLR is suggested as a disease marker in malignancy, functional bowel conditions, and gastrointestinal conditions.34,36 The results of this study results reported that MLR is increased in low SAT/NLR patients, in the univariate analysis, a significant correlation was observed between early recurrence of HCC and MLR ≥ 0.23(p < 0.05), but MLR is not an independent biomarkers that exhibited a correlation with early recurrence of HCC following liver resection. It has been found that high platelet count predicts poor prognosis in HCC patients undergoing TACE. 37 Also, platelet indices were linked with survival in intensive care population. 38 It has been shown that no significant difference in platelet values between low and high SAT/NLR groups. In our study, the univariate analysis and forward stepwise multivariate analysis revealed that the presence of liver cirrhosis, tumor diameter ≥50 mm, AFP ≥ 200 ng/mL, and SAT/NLR < 35.34 were independent biomarkers that exhibited a correlation with early recurrence of HCC following liver resection.
Currently, the therapeutic strategies for HCC primarily consider variables such as tumor burden, liver function, and performance status. However, the incorporation of patients’ inflammation and immune status into clinical guidelines is insufficient. In this study, we propose a novel index, the SAT/NLR ratio, which combines systemic inflammatory marker and adipose tissue quantity. The SAT/NLR ratio, which integrates the advantages of both SAT and NLR, exhibited superior efficacy in assessing one-year recurrence of HCC patients following liver resection, surpassing other inflammatory or nutritional indices. Typically, an elevated NLR indicates an intensified inflammatory response, while a reduced SAT signifies suboptimal nutritional status. Consequently, HCCs that display a low SAT/NLR often demonstrate a vigorous inflammatory reaction (high NLR). Additionally, due to their compromised nutritional state (low SAT), these HCCs are unable to adequately self-regulate, leading to the perpetuation of the inflammatory response. The prolonged existence of inflammation further undermines immune functionality, ultimately culminating in the recurrence, invasion, and metastasis of tumors. 39
The results of this study suggest that HCC patients with a high SAT/NLR prior to treatment have a better RFS and OS outcome following liver resection, in comparison to HCC patients with a low SAT/NLR. Previous scholarly works have consistently demonstrated a strong correlation between nutritional status, inflammatory response, and the advancement of malignant diseases.40–42 Wang et al. discovered that a heightened baseline NLR serves as a dependable prognostic indicator for individuals with HCC undergoing liver resection, while Sun et al. demonstrated a correlation between an elevated preoperative NLR and unfavorable prognosis in HCC patients receiving liver transplantation.41,43 Moreover, recent academic inquiries have unveiled the presence of a novel nutritional metric referred to as the adipose tissue index, which exhibits potential as a prospective prognostic marker for individuals afflicted with HCC. For instance, Ohara et al. have illustrated a substantial association between heightened radiodensity of SAT and an unfavorable prognosis among HCC patients, whereas Labeur et al. have demonstrated that a diminished adipose tissue index serves as a highly predictive factor for HCC patients undergoing sorafenib treatment.44,45 However, it is crucial to recognize the constraints associated with the aforementioned variables. The exclusive utilization of the NLR solely offers understanding into the inflammatory condition of the body, while the employment of the SAT solely reflects the nutritional status. Consequently, the amalgamation of both SAT (as a measure of nutrition) and NLR (as an indicator of inflammation) through the combined application of SAT/NLR has the potential to augment the predictive abilities in patients with HCC undergoing liver resection. The results obtained from both univariate and multivariate analyses provide evidence of a substantial correlation between elevated SAT/NLR and the one-year recurrence of HCC. Moreover, the Kaplan–Meier survival analysis reveals that low SAT/NLR continues to be an unfavorable biomarker for both OS and RFS, both prior to and following PSM.
The existing body of literature highlights a strong correlation between inflammation, nutrition, and the advancement of malignancies. In the context of tumor microenvironments, both tumor cells and immune cells possess the capability to generate diverse cytokines, including pro-inflammatory cytokines, growth factors, and chemokines, as part of the inflammatory process, all of which contribute to the progression of tumors.46,47 Neutrophils and lymphocytes, along with other blood components, play a significant role in initiating the inflammatory process. Peripheral blood neutrophils can be mobilized toward the tumor stroma and undergo differentiation into tumor-associated neutrophils. These neutrophils possess the capability to promote tumor progression and confer resistance to therapeutic interventions through various mechanisms.48,49 Neutrophils possess the ability to migrate toward the tumor microenvironment and release reactive oxygen species, thereby initiating genetic instability and inducing cellular DNA damage, ultimately contributing to the development of cancer and facilitating tumor advancement. 50 Lymphocytes have been found to have a significant impact on the development and advancement of tumors, as indicated by research showing a positive association between increased lymphocyte infiltration in tumors and enhanced survival rates among individuals with HCC. 51 Similarly, γδ T cells have demonstrated the ability to rapidly identify and react to various tumor antigens. 52 Also T-cell activation status has been proved to influence HCC recurrence. 53 Neutrophil-to-lymphocyte ratio, as a monitor of the systemic inflammatory response, reflects the dynamic changes in peripheral blood neutrophils and lymphocytes and the balance between the inflammatory response and the immune system. In contrast, the SAT complex serves as a reliable indicator of the body's nutritional status, subsequently affecting the efficacy of the immune surveillance mechanism and the timely detection of recurrent tumor cells.20,44,45 Overall, both the NLR and SAT exhibit associations with the prognosis of HCC. However, their individual responses are confined to either nutritional or inflammatory aspects. As a result, the utilization of both aforementioned indicators may potentially provide improved predictive effectiveness. Considering that an elevated inflammatory index denotes a strong inflammatory reaction, whereas a reduced SAT signifies inadequate nutritional condition, we have developed a novel composite indicator, SAT/NLR, which integrates these two measurements. This integration allows for the acquisition of a more reliable prognostic tool for HCC. The clinical significance of this study lies in the provision of a relatively convenient and non-invasive biomarker, specifically the combination of inflammation and nutrition status, for predicting the recurrence and long-term survival of HCC patients who have undergone liver resection. This biomarker has the potential to aid clinicians in identifying patients at higher risk of recurrence, thereby enabling personalized adjuvant therapy following radical surgical interventions.
Based on the aforementioned noteworthy findings, it is essential to acknowledge the inherent limitations present in this study. First and foremost, it is crucial to acknowledge that this study employed a retrospective design obtained from a single institution, making it susceptible to potential biases, particularly selection bias. Larger, multicenter studies are needed to confirm the results of the present study. Secondly, it is important to highlight that our study was conducted in a region with a high prevalence of hepatitis B virus, whereas hepatitis B is not the primary cause of HCC in Europe or America. Therefore, it is advisable to exercise caution when extrapolating our findings to the global population.
Conclusion
In summary, a decreased SAT/NLR ratio may be indicative of unfavorable prognosis in patients with HCC. Consequently, incorporating SAT/NLR as a biomarker may be practical in a clinical setting, enabling clinicians to develop more rational and personalized treatment strategies.
Footnotes
Author contributions
Shu-Xian Li and Bao Yan have made significant contributions to the acquisition, analysis, and interpretation of data, as well as the drafting and preparation of manuscripts. Tian-Cheng Wang has made substantial contributions to the design research and critical revision of manuscripts. All authors have read and approved the final version of the manuscript.
Availability of data and materials
Data are available upon reasonable request from Tian-Cheng Wang.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Considering the rigor of ethical standards in this study, ethical approval was sought from the institutional review boards of the Second Xiangya Hospital of Central South University and obtained ethical approval number.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Department of Science and Technology of Hunan Province (Grant Nos. 2021SK53528), the Natural Science Foundation of Hunan Province (Grant Nos. 2021JJ30979), and the Fundamental Research Funds for the Central Universities of Central South University (Grant Nos. 2023ZZTS0597).
