Abstract
Background
Local corticosteroid injections are widely used in the management of musculoskeletal conditions; however, the incidence and timeline of associated cutaneous complications, such as hypopigmentation and subcutaneous fat atrophy, remain poorly defined.
Objective
To prospectively evaluate the incidence, timing, and recovery of hypopigmentation and subcutaneous fat atrophy following corticosteroid injections into the dorsal wrist and elbow regions, and to assess the influence of sex, age, and skin shade on these outcomes.
Methods
Demographic data were collected, and patients were followed up monthly for 1 year. Patients with positive skin reactions were followed up for up to 18 months.
Results
A total of 45 injection cases were analyzed. After adjustment for potential confounders, hypopigmentation showed attenuated associations with injection region (adjusted odds ratio (OR) = 3.53, 95% confidence interval: 0.34–83.18) and sex (adjusted OR ≈ 0). Unadjusted findings indicated that hypopigmentation was initially more frequent after wrist injections compared to elbow injections and occurred only in women, but these effects diminished after adjustment. Subcutaneous fat atrophy occurred in three (6.7%) cases, all in women. Hypopigmentation developed 4–10 weeks after injection and fully resolved (mean 7.14 months, range 2–18), whereas two of three cases of fat atrophy showed incomplete recovery (P = 0.016 for recovery difference).
Conclusion
After adjustment for potential confounders, hypopigmentation was attenuated but remained more common following wrist injections and in women, while subcutaneous fat atrophy remained rare, with incomplete recovery in some cases. This study provides the first detailed timeline of onset and resolution for corticosteroid-induced hypopigmentation and fat atrophy, highlighting important patterns by injection site and sex, and offering guidance for clinicians in patient counseling and management.
Introduction
Local corticosteroid injections are widely used to treat various rheumatic and musculoskeletal disorders. 1 These injections often alleviate the symptoms and provide permanent relief in some instances. However, the risks associated with these injections are often underrecognized and challenging to quantify. A systematic review of the literature examining extra-articular corticosteroid injections revealed a reported incidence of major and minor adverse events ranging from 0% to 5.8% and from 0% to 81%, respectively. The authors concluded that accurately assessing the incidence of adverse effects following extra-articular corticosteroid injections presents significant challenges. 2
Local skin hypopigmentation and subcutaneous fat atrophy are among the well-documented complications of corticosteroid injections (Figure 1(a) and (b)). However, a literature review revealed a lack of definitive data on the incidence and timeline of these reactions. Many studies on extra-articular steroid injections do not indicate the frequency of these adverse effects, while those that do present a wide range of reported incidences, typically between 1% and 40%. 3 The limited number of prospective studies available offer inconsistent data, with reported rates varying from 31% to 79%.4,5
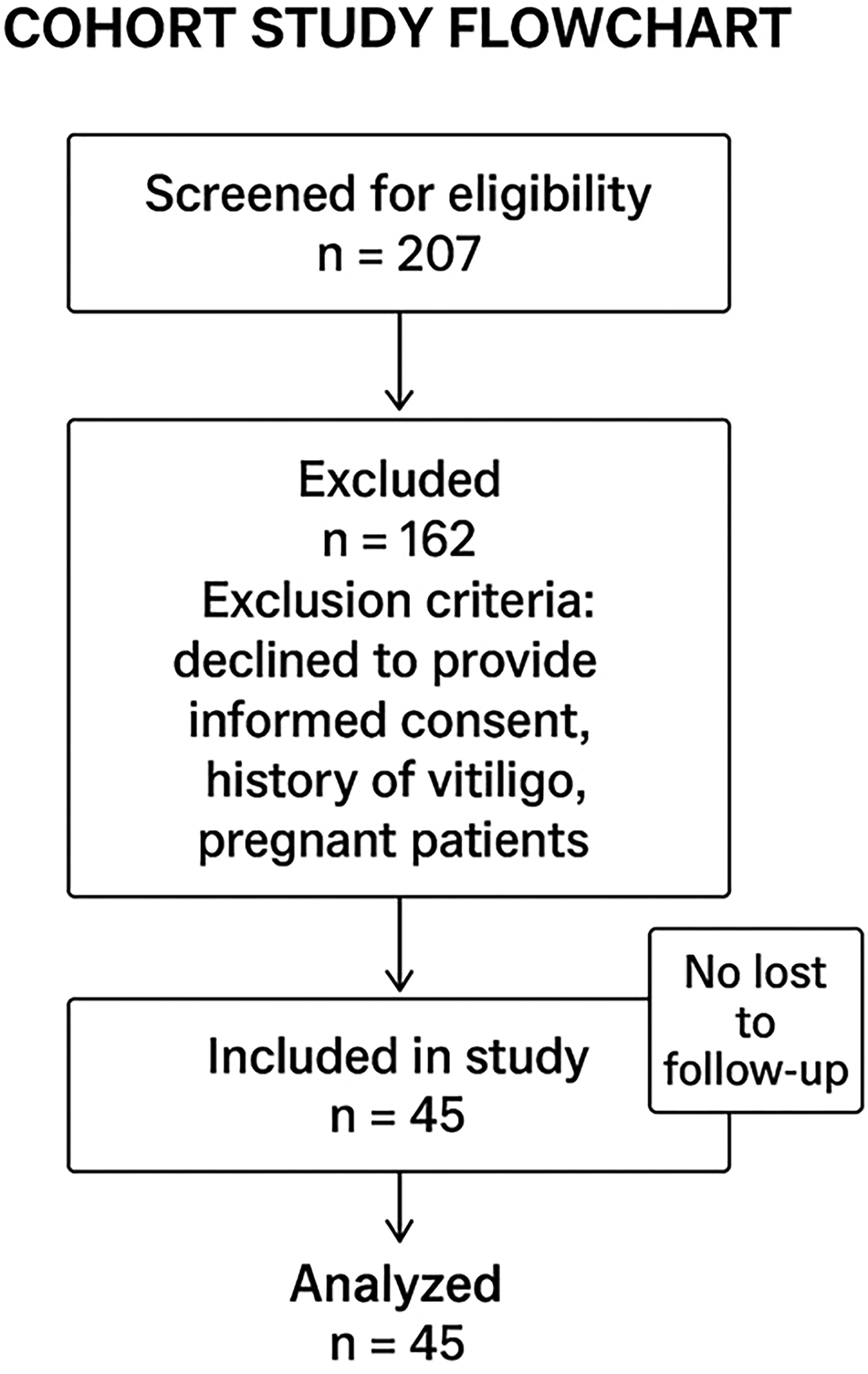
Flowchart.
Regarding the timeline of these reactions and recovery rates, a comprehensive review of 87 studies examining the side effects of extra-articular corticosteroid injections indicated that skin-related adverse effects typically appear within 2 to 4 months after the injection, although delayed reactions up to 10 months have also been documented. Soft-tissue atrophy and hypopigmentation generally resolve spontaneously within 9 to 12 months after injection, although in some cases, these changes may persist permanently. Notably, this data is derived from a relatively limited number of case reports.3,6–9 Currently, there is a lack of robust, prospective studies that provide accurate information on the timing of reactions, recovery rates, and duration of recovery following such adverse effects.
Research objective
Given the challenges in accurately quantifying skin changes following corticosteroid injections, we aimed to determine the incidence, timeline, and recovery rates of these skin changes. Additionally, we evaluated the influence of variables such as sex, age, and skin shade in patients receiving injections into the dorsal wrist and elbow regions.
Materials and methods
This prospective cohort study was approved by the Emek Helsinki Committee (IRB EMC-0163-21, Afula, 20.3.22) and was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024. The reporting of this study conforms to STROBE guidelines. 10 All patients attending our outpatient hand clinic who received extra-articular corticosteroid injections into the dorsal wrist or elbow region between March 2022 and March 2024 were invited to participate. We received signed consent from all patients in our study. All patient details were de-identified.
The inclusion criteria were all patients receiving corticosteroid injections into the dorsal wrist or elbow region. Specific indications for dorsal wrist and elbow injections that were included in our study were De Quervain's tenosynovitis, CMC1 osteoarthritis, and lateral epicondylitis. A total of 43 patients were enrolled in the study. Two patients received bilateral injections and were considered as two separate cases, yielding a total of 45 injections. The exclusion criteria included individuals who declined to provide informed consent, those with a history of vitiligo, and pregnant patients.
Demographic and clinical data, including age, sex, diagnosis, injection date and site, and skin shade, were collected for all consenting participants. Individuals were categorized into three groups based on the skin shade: light, tan, and brown (Table 1).
Baseline table.

Skin phenotype classification
Skin phenotype was initially categorized by the treating hand surgeon at the time of injection into three groups: light, tan, and brown. Classification was based on visual assessment. During follow-up, the primary investigator reassessed skin phenotype as part of ongoing evaluation. There were no discrepancies between the initial classification by the hand surgeon and the follow-up assessments by the primary investigator.
Although a validated scale such as the Fitzpatrick phototype system was not formally applied, the study categories correspond approximately to the following Fitzpatrick types:
11
Light: Types I and II Tan: Types III and IV Brown: Types V and VI
This mapping provides a standardized reference point to improve interpretability and generalizability. Nonetheless, the use of visually assigned, nonvalidated categories may introduce subjectivity and is acknowledged as a study limitation.
Injection technique and product details
All injections were performed by a fellowship-trained hand surgeon using 1 mL of betamethasone dipropionate/sodium phosphate (Diprospan, 5 mg + 2 mg/mL), a mixed soluble/particulate corticosteroid preparation. No diluent or anesthetic was added to the injection.
A 25-gauge needle was used for all procedures. The injection approach varied by condition:
For carpometacrpal joint (CMCJ1) injections, the needle was inserted perpendicularly to the skin surface, targeting the intra-articular space. For De Quervain's tenosynovitis, injections were delivered at an oblique angle into the first dorsal extensor compartment. For lateral epicondylitis, the approach was perpendicular to the skin, with the steroid deposited approximately 2 mm superficial to the lateral epicondyle, overlying the origin of the common extensor tendon.
Ultrasound guidance was not used. Prior to injection, the skin was disinfected with 70% isopropyl alcohol. To reduce the risk of intradermal tracking, the area was allowed to dry fully before injection. After the injection, the site was gently massaged for a few seconds, and a standard adhesive bandage was applied. No additional compression, dressings, or activity restrictions were routinely advised.
Patients were followed up monthly via telephone for 1 year. Patients who exhibited a reaction to the injection were invited for in-person clinical assessment, including photographic documentation. Hypopigmentation was defined as a visible lightening of the skin compared to surrounding areas, confirmed under standardized lighting conditions. Subcutaneous fat atrophy was defined as a visible depression or thinning of the skin surface, confirmed by palpation.
These patients were subsequently monitored monthly for up to 18 months after reaction or until they fully recovered.
Outcome assessment
All assessments were conducted by the primary investigator, a senior orthopedic resident (postgraduate year 5) with clinical experience in musculoskeletal evaluation and procedural techniques. The assessor was not blinded to the injection site during the evaluation.
Interrater agreement
Interrater reliability was not assessed, as all evaluations were performed by a single assessor.
Photography protocol
Photographs were taken using an Android smartphone without digital zoom, from a consistent distance of approximately 25–30 cm. Natural lighting was used for all images, and no flash or additional lighting equipment was applied. Background and camera settings were not standardized or calibrated.
Follow-up pathway and detection bias mitigation
All patients were followed monthly via telephone by the primary investigator throughout the study period. In addition, patients were provided with the investigator's direct phone number and instructed to text immediately if they noticed any skin changes or adverse effects at the injection site.
If a patient did not initially answer a scheduled call, daily follow-up attempts were made via both phone calls and text messages until contact was re-established. All patients responded within a few days each month, and no participants were lost to follow-up.
Triggers for in-person review included any patient-reported change in skin coloration or visible skin depression around the injection. Patients who reported such symptoms were scheduled for an in-person assessment, which occurred within 1 month of the initial report. The date of onset was documented based on when the patient first noticed the skin change. In all cases, the skin reaction was first confirmed by photographs sent via text message, followed by an in-clinic evaluation.
Following a confirmed positive reaction, patients continued to receive monthly follow-up calls until full resolution of symptoms or up to a maximum of 18 months postreaction. Recovery was defined as the patient reporting that the skin appearance had returned to normal, supported by a confirming photograph. Patients who met the criteria for full recovery were not reassessed in clinic.
This protocol combined active surveillance—with investigator-initiated monthly follow-up—with patient-initiated reporting, designed to capture adverse events in a timely and systematic manner. However, the reliance on telephone-based screening may have limited the detection of subtle or asymptomatic findings that could be more readily identified during physical examination. This potential for detection bias is acknowledged as a study limitation.
Statistical analysis
R software (version 4.1.3; R Foundation for Statistical Computing, Vienna, Austria) was used for all statistical analyses. Continuous variables were summarized as means and standard deviations, whereas categorical variables were reported as frequencies and percentages. Fisher's exact test was used to compare the incidence rates of hypopigmentation and subcutaneous fat atrophy because of the presence of low expected cell counts, which rendered the chi-square test unsuitable. Pairwise comparisons between positive reactions and the different skin shade categories were adjusted using Bonferroni correction. The two-tailed unpaired Student's t-test was used for continuous variables, and the nonparametric Mann–Whitney U test was applied when the assumption of normality was not met. Unadjusted odds ratios (ORs) and 95% confidence intervals (CIs) were calculated as measures of association between hypopigmentation and fat atrophy, with continuity correction applied (Haldane–Anscombe correction for zero cells). Covariates included a priori in multivariable models were age, sex, skin shade, and injection site. Quasi-complete separation in male participants led to unstable estimates for sex, which was noted and reported as such. Multiplicity of comparisons for skin shade was addressed using the Benjamini–Hochberg false-discovery rate method, and analyses were labeled exploratory when appropriate. Multivariable logistic regression was performed to estimate adjusted ORs for the association between positive hypopigmentation reactions and age, gender, skin shade, and injection site. Kaplan–Meier survival curves were generated to compare the time to onset/recovery between wrist and elbow injections, and statistical significance was assessed using the log-rank test. A P-value <0.05 was considered statistically significant.
Results
A total of 43 patients were enrolled in the study. Two patients received bilateral injections and were considered as two separate cases, yielding a total of 45 injections. Of these, 33 (73.3%) cases involved the dorsal wrist region, whereas 12 (26.7%) cases involved the elbow region. The cohort consisted of 33 (73.3%) women and 12 (26.7%) men, with a mean age of 50.0 years (range: 18–80 years). Regarding skin shade distribution, 19 patients had light skin (42.2%), 15 had tan skin (33.3%), and 11 had brown skin (24.5%).
Hypopigmentation
Hypopigmentation was observed in 17 of 45 cases (37.8%). In the unadjusted analysis, among injections at each site, hypopigmentation occurred in 16 of 33 dorsal wrist injections (48.5%) and in 1 of 12 elbow injections (8.3%), respectively. This difference was statistically significant (P = 0.017), with an unadjusted OR of 9.90 (95% CI: 1.14–93.8). After adjustment for potential confounders, the association was attenuated and no longer statistically significant (adjusted OR = 3.53, 95% CI: 0.34–83.18). The loss of statistical significance likely reflects limited precision due to the small sample size (Table 2).
Outcomes by site and sex: hypopigmentation and fat atrophy.

OR: odds ratio; CI: confidence interval.
All 17 cases of hypopigmentation occurred in women, with no cases observed among men (unadjusted P = 0.013). The unadjusted OR was 0.037 (95% CI: 0.002–0.689), indicating substantially lower odds of hypopigmentation among men compared with women. When sex was entered into the multivariable logistic regression model, the effect remained near zero (adjusted OR ≈ 0), as no male participants developed hypopigmentation. The wide CI reflects the zero cell and small sample size, which also contributed to the loss of statistical significance in the adjusted model.
In the multivariable logistic regression analysis, increasing age was independently associated with a reduced likelihood of developing hypopigmentation (OR = 0.95, 95% CI: 0.89–0.99). No significant associations were observed for skin shade or injection site. The model also suggested markedly lower odds of hypopigmentation among male participants (OR ≈ 0); however, this estimate was unstable and likely reflects quasi-complete separation due to the limited number of positive cases in males.
No statistically significant association was found between hypopigmentation and age or skin shade (P = 0.100 and P = 1.000, respectively). When included in the multivariable model, these variables remained nonsignificant. To account for multiple comparisons of the shade variable, P-values were adjusted using the Benjamini–Hochberg false-discovery rate method. No statistically significant differences were observed between skin shade groups, and all adjusted P-values remained nonsignificant. Therefore, these analyses should be considered exploratory.
Subcutaneous fat atrophy
Subcutaneous fat atrophy was noted in 3 of the 45 (6.7%) cases. Two of these occurred in patients who also developed hypopigmentation, whereas one occurred independently. All three cases were associated with wrist injections, and none of the cases were associated with elbow injections (P = 0.550). All the affected individuals were women (P = 0.550). No statistically significant association was observed between subcutaneous fat atrophy and age or skin shade (P = 0.100 and P = 1.000, respectively).
Timing of reactions and recovery
The mean time to onset of hypopigmentation and subcutaneous fat atrophy after injection was 5.94 weeks (SD = 1.73) and 5.33 weeks (SD = 1.89), respectively. All reactions developed between 4 and 8 weeks after injection, except for one case of hypopigmentation that appeared at 10 weeks.
All 17 hypopigmentation cases resolved completely, with a mean recovery time of 7.14 months (range: 2–18 months). Among the three cases of subcutaneous fat atrophy, only one was resolved after 14 months, whereas two remained unresolved after 18 months of follow-up. Given the small number of fat atrophy events, differences in recovery rates between hypopigmentation and fat atrophy are presented descriptively and should be interpreted with caution. Kaplan–Meier curves (Figure 2) illustrate the time to onset and recovery of hypopigmentation and indicate the number of patients at risk and censoring points over time. No significant associations were found between the recovery rate and age or skin shade (Figure 3).
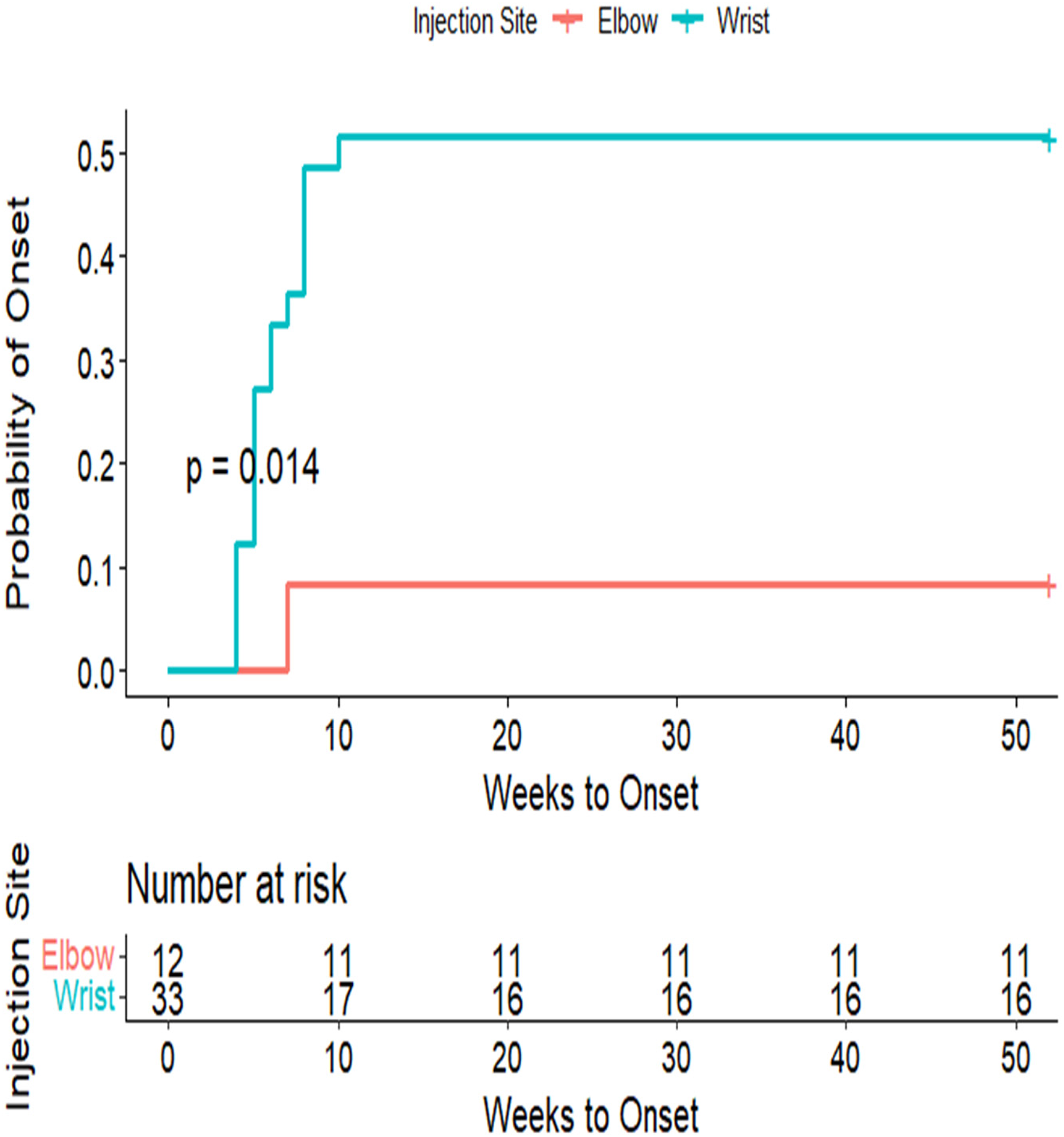
Kaplan–Meier curves: (a) time to onset and (b) time to recovery. Hypopigmentation: time to onset: Kaplan–Meier analysis suggested earlier onset of hypopigmentation following wrist region injections compared with elbow region injections (log-rank P = 0.014). Approximately 50% of patients who received wrist region injections developed hypopigmentation within the first 10 weeks (median = 10 weeks; 95% CI: 7–NA). Only one event occurred at the elbow site, preventing estimation of a median onset time. Given the small number of events at the elbow site, results are presented descriptively, and formal comparisons should be interpreted with caution. Censoring rules and numbers at risk are indicated below each plot. Hypopigmentation: time to recovery: among patients with wrist region injections, the median time to recovery was approximately 14 months (95% CI: 9–NA), whereas the median time to recovery at the elbow site could not be estimated because fewer than half of patients experienced the event (1 of 12). The survival curve for the elbow site shows a gradual decline with wide confidence limits, reflecting limited events and notable censoring, and numbers at risk and censoring points are presented below the curves for transparency. CI: confidence interval.

(a) Skin hypopigmentation of the dorsal wrist. (b) Skin hypopigmentation and subcutaneous fat atrophy.
Discussion
The pathophysiology underlying corticosteroid-induced skin changes is not fully understood; however, histopathological analyses of the affected tissues provide some insight. Subcutaneous tissue biopsies of specimens from sites of steroid injection have demonstrated a reduction in both the number and size of adipocytes, possibly owing to the presence of “lipophage-like” macrophages.6,12 Additionally, punch biopsies of hypopigmented skin have revealed a decrease in melanin-containing cells, 13 and studies have shown impaired melanocyte function at the cellular level. 14
Current knowledge regarding the treatment of steroid-induced cutaneous atrophy with saline injections is primarily based on case reports and small case series, rather than large-scale clinical studies or randomized controlled trials. The most cited evidence consists of case series demonstrating successful reversal of atrophy with serial intralesional saline injections, with complete resolution observed in a handful of patients over several weeks.15,16
Our study showed a higher incidence of hypopigmentation following corticosteroid injections into the dorsal wrist region than into the elbow region, consistent with the limited prospective literature. In unadjusted analyses, this difference was statistically significant; however, after adjustment for potential confounders, the association was attenuated and no longer statistically significant, likely reflecting limited precision due to the small sample size. To our knowledge, this is the first prospective study directly comparing the incidence of hypopigmentation between these two anatomical regions, providing valuable descriptive data while highlighting the need for cautious interpretation of effect sizes.
The predominance of cutaneous reactions at the wrist in this study suggests that local anatomical factors play a significant role in susceptibility. The dorsal wrist has relatively thin subcutaneous tissue, particularly compared to the lateral elbow, potentially increasing the risk of steroid diffusion into the dermis and subsequent atrophy or pigmentary change. In contrast, the elbow region may offer more soft-tissue buffering, which could reduce exposure of the skin to corticosteroid effects. The use of a particulate corticosteroid formulation (betamethasone dipropionate) may also contribute via a depot effect, leading to prolonged local exposure.
The observation that all positive skin reactions occurred in female patients, with none reported in male patients, is noteworthy. In unadjusted analyses, this sex-based disparity was statistically significant; however, after adjustment for potential confounders, the association was attenuated and no longer statistically significant, likely due to the small sample size and the zero events among males. This effect instability suggests caution in interpreting the sex difference. Further investigations are warranted to explore potential biological or procedural contributors to these differences.
In contrast to the high incidence of hypopigmentation, the rate of subcutaneous fat atrophy in our cohort was relatively low. Importantly, many prior studies have not clearly distinguished between hypopigmentation and soft-tissue atrophy, often reporting them collectively as a single complication. Our findings highlight the importance of separating these two adverse effects as they differ in frequency, recovery profile, and likely underlying mechanisms.
Regarding the relationship between skin reactions and skin shade, conventional wisdom, largely based on case reports, suggests an increased risk in individuals with darker skin.17,18 However, we did not find a statistically significant association between skin shade and the incidence of either hypopigmentation or subcutaneous atrophy. We propose that this commonly held belief may be because of the greater visual contrast of hypopigmentation against darker skin rather than a true increase in incidence.
In a prospective study on De Quervain's tenosynovitis by Anderson et al., 31% of patients developed skin changes, ranging from mild lightening to frank atrophy. 4 Notably, hypopigmentation and subcutaneous atrophy were collectively labeled as “atrophy,” defined broadly as skin discoloration, dimpling, or loss of subcutaneous tissue. However, the specific timing of onset, recovery rates, and stratification by sex or skin shade were not reported. Similarly, Shin et al. reported skin complication rates of up to 78.9% 3 months after injection, again combining hypopigmentation and atrophy into a single category without further differentiation or demographic analysis. 5
Prospective data on hypopigmentation following elbow injections are limited. In a study by Tonks et al., only one (4.2%) case of hypopigmentation and atrophy was reported among 24 patients treated for lateral epicondylitis. 19 Similarly, Bisset et al. reported hypopigmentation and atrophy rates of 3% and 1.5%, respectively, in a cohort of 65 patients. 20 However, none of these studies provided information on the onset timing, recovery rates, or associations with sex or skin shade.
Limitations and practical implications
This study has several limitations. First, the sample size was relatively small, particularly for subgroup analyses of subcutaneous fat atrophy, which may limit the generalizability of the findings. Second, although patients were followed monthly, reliance on telephone-based follow-up for initial screening of skin changes may have introduced observer bias or delayed the recognition of subtle reactions. Third, outcomes were assessed by a single, nonblinded assessor, and the photography protocol (lighting, background, and device) was not standardized. These factors may have introduced misclassification or detection bias, likely biasing results toward underdetection of subtle changes. Fourth, all injections were performed by a single fellowship-trained hand surgeon at a single center, which may reduce external validity. Outcomes may differ when performed by injectors with varying levels of experience or in different clinical settings. Fifth, all injections used a single corticosteroid formulation—betamethasone dipropionate/sodium phosphate (Diprospan). Other corticosteroid compounds, particularly those with different solubility or particle characteristics, may have a different risk profile. The study did not include comparative data on alternative formulations. Sixth, although patients were categorized into broad skin shade groups (light, tan, brown), more precise dermatological skin typing—such as using the Fitzpatrick phototype scale—may have provided a more nuanced understanding of the relationship between skin phenotype and cutaneous complications. Finally, the study focused on specific anatomical regions (dorsal wrist and lateral elbow), and the findings may not be generalized to other injection sites with different tissue characteristics or cosmetic significance.
Despite these limitations, the study offers several practical insights for clinicians. Site selection remains an important consideration, particularly in regions with thin subcutaneous tissue or limited soft-tissue padding, where the risk of steroid-induced skin changes may be higher. Patient counseling should include discussion of the potential for skin discoloration, atrophy, or contour deformities, particularly in individuals with darker skin phenotypes or in cosmetically sensitive areas. To minimize risk, injection techniques should include measures such as ensuring proper depth (e.g. avoiding intradermal injection), selecting appropriate needle size, allowing antiseptic to dry before injection, and avoiding excessive postinjection pressure. While this study did not use image-guided techniques, the use of ultrasound guidance in certain anatomic areas may help improve precision and reduce complications. Finally, consideration of alternative corticosteroid formulations—particularly nonparticulate agents—may be warranted in high-risk scenarios, although further comparative studies are needed to guide formulation-specific risk stratification.
Conclusions
This prospective cohort study provides new insights into the incidence, timing, and recovery of corticosteroid-induced skin complications following extra-articular injections into the wrist and elbow regions. After adjustment for potential confounders, hypopigmentation was attenuated but remained more commonly observed following wrist injections and in women, likely reflecting limited precision related to the small sample size. Subcutaneous fat atrophy remained rare. No statistically significant associations were observed between adverse skin reactions and age or skin shade in either unadjusted or adjusted models, challenging the assumption that individuals with darker skin tones are inherently at greater risk than those with lighter tones.
This study is the first to provide data defining a structured timeline for the onset and resolution of corticosteroid-induced skin changes. Nearly all reactions occurred between 4 and 8 weeks after injection, with a single case emerging at 10 weeks. In this cohort, hypopigmentation resolved fully in all cases, with recovery times ranging from 2 to 18 months. These findings offer clinicians a clear framework to inform patients about the expected timing of skin changes and the likelihood of recovery, helping set realistic expectations. Patients should be counseled that while most cases resolve completely, skin changes may be cosmetically distressing during recovery, particularly in visible or cosmetically sensitive areas.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251409838 - Supplemental material for Cutaneous complications following extra-articular corticosteroid injections: A prospective cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504251409838 for Cutaneous complications following extra-articular corticosteroid injections: A prospective cohort study by Aryeh Weinstein, Konstantyn Drizlikh, Nimrod Rozen, David Rothem and Guy Rubin in Science Progress
Footnotes
Ethics approval and consent to participate
The study and all experimental protocols were approved by the Emek ethical committee (IRB EMC-0163-21, Afula, 20.3.22). All methods were carried out in accordance with relevant guidelines and regulations.
Consent for publication
Written informed consent was obtained from the patients.
Author contributions
AW: conception and design, obtaining data, revising the article, and final approval.
KD: analysis and interpretation of data, revising the article, and final approval.
NR: obtaining data, revising the article, and final approval.
DR: obtaining data, revising the article, and final approval.
GR: conception and design, obtaining data, revising the article, and final approval.
All authors read and approved of the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
De-identified data and analysis code are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
