Abstract
Objective
Keratinocyte cancers (KCs) account for significant healthcare burden in regional Australia. Effective surgical management relies on accurate pre-operative biopsy to inform risk status and guide clinical excision margins. Through retrospective analysis, we aim to describe correlation rates of pre-operative biopsy and final pathology in head and neck KC excisions.
Methods
From July 2023 to June 2024, 137 KC excisions performed in a regional Australian Otolaryngology Head and Neck surgery outpatient skin unit were analysed. Patient demographics, lesion characteristics, preoperative biopsy, and final histopathology were examined.
Results
Excisions were undertaken for 101 basal cell carcinomas (BCCs), 30 squamous cell carcinomas (SCCs), and six intra-epidermal carcinomas. Nineteen excisions were performed without pre-operative biopsy and 15 further excisions were re-excisions for close or involved margins and were not included in correlation analysis. Of the remaining 97 KC, there was concordance between biopsy and formal pathology risk status in 47.4%. Upgraded (higher) risk formal pathology was reported in 19.6% while downgraded (lower) risk formal pathology was reported in 8.2%. Within the higher risk group, three BCC excisions reported close margins when their lower risk subtype (if confirmed) would have been considered clear. There was no residual malignancy in 24.7% of KC excisions. Pathologic concordance rates after subgroup analysis were 58.7% for BCCs and 9.1% for SCCs.
Conclusion
Accurate pre-operative biopsy to guide treatment options and inform clinical margins is especially important for head and neck KCs, where functional and cosmetic constraints are greatest. The correlation rate for surgically excised head and neck KCs has not been previously investigated in the Australian regional setting. This work highlights the importance of counselling patients on the potential for higher risk final pathology when discussing the risk of requiring further treatment.
Introduction
Australia faces the highest incidence of keratinocyte cancers (KCs) globally.1,2 The management of KCs accounts for an estimated one million episodes of care per year nationally 3 and this burden is greatest in Queensland, recording 37,000 hospital-based episodes of care in 2023–2024. 4 KCs equalled breast cancer as the largest oncologic economic burden in 2015–2016, totalling $1 billion or 10% of the national cancer-related health budget. 5 Previously termed non-melanoma skin cancers, KCs comprise basal cell carcinomas (BCCs) and squamous cell carcinomas (SCCs). 3 A non-invasive pathology, intra-epidermal carcinoma (IEC) or SCC in situ, is considered a precursor to invasive SCC. 3
Skin biopsy is an essential tool to confirm lesion type and subtype and identifying any high-risk features, therefore guiding further management. Punch, shave, incisional, and excisional biopsies may be utilised in sampling non-pigmented lesions depending on the involved site.6,7 Biopsy confirmation of KC type and subtype prior to definitive surgical excision is highly preferable for the head and neck, where anatomical and functional constraints demand judicious clinical margin measurement. This must be balanced with the goal of complete lesion excision.
There is a paucity of Australian literature examining the degree of correlation between pre-operative KC biopsy and formal pathology from traditional surgical excisions. It is hypothesised that a substantial portion of excised KCs demonstrate unexpected pathologic subtype and risk profile, which may have otherwise altered clinical margins. The importance of accurate pre-operative biopsy in guiding surgical excision is heightened in the regional Australian setting, where patient travel, lesion multiplicity, and barriers to ongoing follow up pose challenges for lesion re-excisions and surveillance.8,9
Methods
We conducted a retrospective analysis of 137 skin lesions excised under local anaesthetic in the outpatient day surgical setting in a regional Queensland specialist service from July 2023 to June 2024. Patients were referred to the Toowoomba Hospital department of Otolaryngology Head and Neck Surgery, largely by primary care providers, and triaged onto dedicated skin excision lists following clinical review. Patients were included when excision pathology confirmed KC, or where pre-operative biopsy indicated KC even where no residual malignancy was then detected. Melanomas and known benign skin and mucosal pathologies were excluded from this study.
Patient demographics, lesion characteristics, and operative techniques were recorded. Pathologic assessment of excision specimens was performed by Pathology Queensland, and these reports were analysed. Formal pathology was then compared to pre-operative biopsy reports, excluding re-excisions specimens for correlative analysis. Patient data were de-identified. Lesions excised without pre-operative biopsy were recorded but were unable to be analysed for pathological correlation.
The risk status of KC subtypes was defined with guidance from the Australian Cancer Council Clinical Practice Guidelines. 3 Lower risk subtypes of BCC include nodular (sometimes referred to as solid), nodulocystic, and superficial (multifocal). Higher risk subtypes include infiltrative, micronodular, sclerosing (fibrosing) or morpheaform (morpheic), and basosquamous. Well-differentiated (WD), moderately differentiated (MD), and poorly differentiated (PD) SCCs have been considered low, intermediate, and high-risk subtypes, respectively, for this analysis. Agreement between biopsy and final pathology was defined as concordance of clinical risk status. Perineural invasion (PNI) was also recorded.
Study data was collected with approval, including waiver of written consent, from the Darling Downs Hospital and Health Service (DDHHS) Human Research Ethics Committee (LNR/2023/QTDD/97361). Our study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024. 10 This work has been reported with guidance from the TRIPOD statement under the Equator Network Guidelines. 11
Results
Patient demographics
In the 12-month study period, 119 patients underwent excision of 137 skin lesions. Eighty-one patients (68.1%) were male, while 38 (31.9%) were female. The mean age was 70.7 years (range 39–93 years). There were 36 patients (30.3%) who lived more than 100 km from the Toowoomba Base Hospital.
Lesion characteristics
Lesions within the high-risk anatomical H-zone of the face comprised 73.7% of all excisions with the most common site being the nose, accounting for 14.0%. The most common site outside of the H-zone was the cheek, representing 4.4%. Lesion defects were closed primarily for 84 cases (61.3%). Full-thickness skin grafts and local flaps were employed for 30 (21.9%) and 23 (16.8%) cases, respectively.
BCCs were recorded as the indication for lesion excision in 101 cases (73.7%). SCCs and IECs represented the remaining 30 (21.9%) and six (4.4%) excisions, respectively. These data represent the pathologic indication for excision and therefore do not account for negative residual rates, analysed below. Three lesions (all BCCs) demonstrated PNI of nerves 0.1 mm diameter or greater on final pathology (conferring T3 disease per American Joint Committee on Cancer Staging Manual, 8th edition). 12
Pooled KC correlation
Of the 131 KC excision specimens, there was no preoperative biopsy for 19 lesions. Fifteen excisions represented re-excisions due to involved or near margins on previous attempt and were excluded from biopsy correlation analysis. The remaining 97 KC excisions underwent correlative analysis as shown in Figure 1.

Breakdown of KC excisions included in correlative analysis and rates of concordance when compared to pre-operative biopsy.
No residual malignancy was demonstrated in 24.7% of excisions (24/97) despite confirmatory pre-operative biopsy. Formal histopathology reported higher risk KC subtypes than suggested by biopsy in 19.6% of cases (19/97). Lower risk KC subtypes were confirmed in 8.2% (8/97). Agreement between KC subtype risk profile on final pathology and pre-operative biopsy was reported in 47.4% (46/97).
BCC correlation sub-analysis
Formal pathologic subtypes of the 101 BCC excisions are shown in Table 1. Nodular BCC was the most common low risk subtype, reported in 36.6% (37/101) of specimens, while infiltrative was the most frequent high-risk subtype, reported in 34.7% (35/101). Where high and low risk BCC subtypes were co-existent, the higher risk pathology took precedence for correlation analysis. The cumulative total for BCC subtypes is greater than 100% given co-existent subtypes in some specimens.
Type and subtype results for 131 pathologically confirmed keratinocyte cancer excisions.
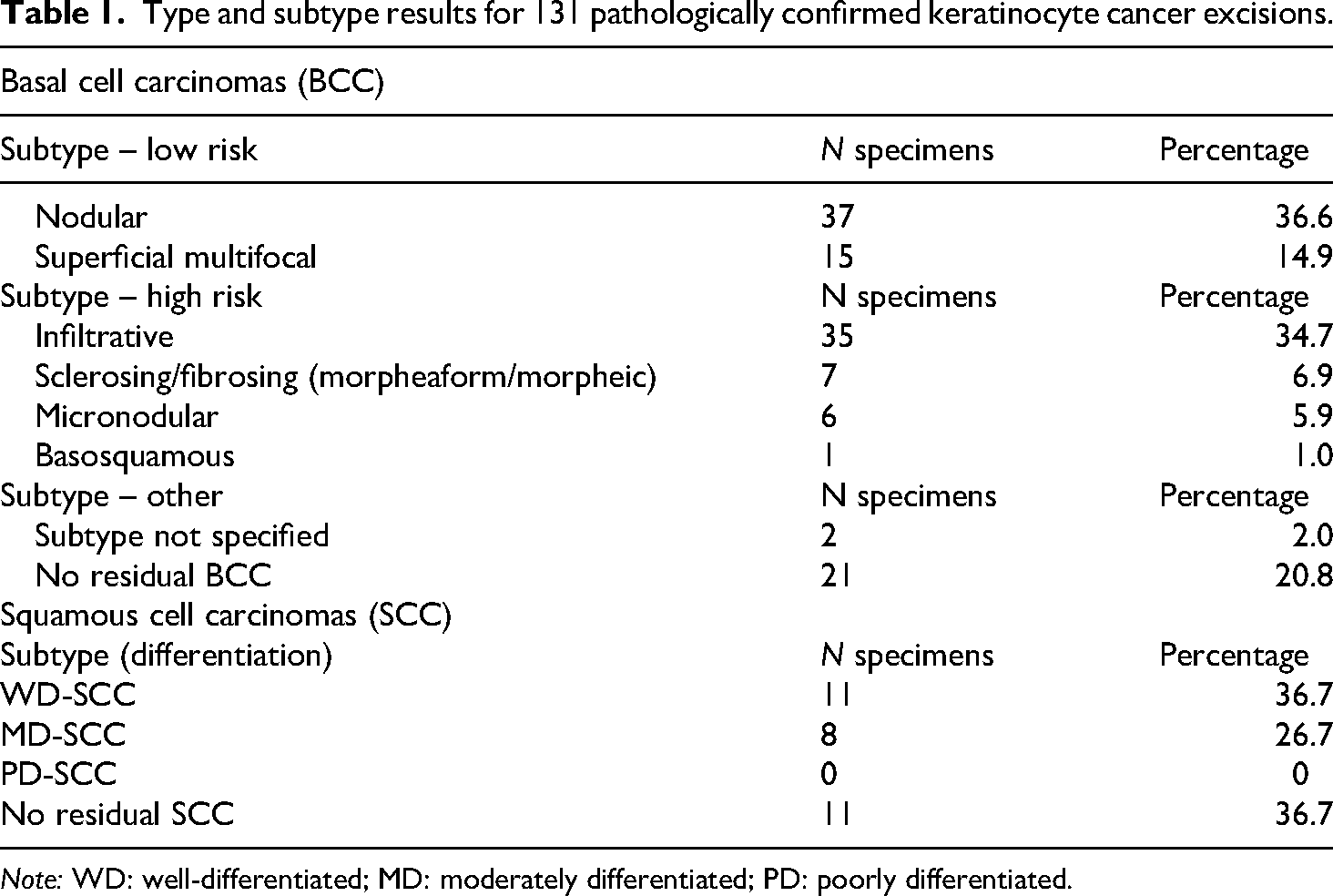
Note: WD: well-differentiated; MD: moderately differentiated; PD: poorly differentiated.
There was no pre-operative biopsy for 14 BCC excision specimens (13.9%), and these were excluded from correlation analysis. Twelve excisions were re-excisions of previous close or involved margins and were excluded from biopsy correlation analysis. The remaining 75 BCCs were analysed. There was subtype risk concordance in 58.7% (44/75) of BCC excisions. A lower risk subtype than expected was demonstrated on formal pathology in 5.3% (4/75). There were an additional 20.0% (15/75) of BCC excisions that demonstrated no residual malignancy. Higher risk pathology than expected (i.e. when pre-operative biopsy confirmed a low-risk subtype) was demonstrated in 16.0% (12/75) of excision specimens. Three of these excisions (25%) were then considered to have close margins, where they would have otherwise been considered clear excisions of the lower risk BCC subtype.
SCC correlation sub-analysis
Formal pathologic subtypes (grading) for 30 excised SCCs are also outlined in Table 1. No SCCs in this series demonstrated PNI of nerves in the dermis or nerves 0.1 mm in calibre or larger.
There was no pre-operative biopsy for five SCC excisions (16.7%). Three specimens represented re-excision for previous close or involved margin and were also excluded. The remaining 22 SCC excision specimens were included in analysis. There was agreement on degree of differentiation in two excisions (9.1%). Poorer than expected subtype (differentiation) on formal pathology was confirmed in seven specimens (31.8%) while four excisions (18.2%) confirmed lower risk differentiation than biopsy suggested. The remaining nine lesions (40.9%) demonstrated no residual malignancy, with or without residual IEC.
Re-excisions
Of the re-excisions for close or involved margins on previous attempt, no residual malignancy was demonstrated was demonstrated in 40% (6/15). This includes a negative residual rate of 41.7% (5/12) for BCCs and 33.3% (1/3) for SCCs. When re-excisions are included in the total excision data set, the pooled negative residual rate increases slightly to 26.8%.
Discussion
To the authors’ knowledge, analysis of head and neck KCs from traditional surgical excisions and their pre-operative biopsy correlates in the Australian setting has not been previously published. Our cohort includes a majority male patients (68.1%), consistent with the elevated risk of KCs in men.3,13 BCCs were approximately three times as common as SCCs in our population, which is similar to the expected ratio for head and neck KCs. 3
Our study demonstrates that pre-operative biopsy correctly identified KC subtype risk status in 47.4% of cases. This rate was much higher for BCCs (58.7%) than SCCs (9.1%), although the latter was limited by low case numbers. For biopsy proven KC, our data suggest a 24.7% chance of finding no residual malignancy in the formal excision specimen, even after excluding re-excision specimens performed for close or involved margins. Whilst these negative residuals should not be considered ‘pathologic discordance’ when comparing to pre-operative biopsy, they were included in analysis to reflect a realistic cohort of patients undergoing formal KC surgical excision.
Of note, biopsy underestimated KC risk status in nearly a fifth (19.6%) of cases. Of the 12 high risk BCCs specimens that were biopsied to be low risk subtypes, three (25%) resulted in narrow excision margins (defined here <1 mm). Had the low-risk biopsy subtype been confirmed (nodular BCC), these excisions would have otherwise been considered clear. All three patients have elected to undergo close clinical surveillance, declining re-excision.
A recent article examined a similar research question in the context of Mohs micrographic surgery. Stewart et al. analysed the biopsy discrepancy rate to formal KC excisions performed by a single Mohs micrographic surgeon in Sydney. 14 This study reported an overall KC type and subtype concordance of 58.0%, similar to our cohort here. 14 Another study, also examining Mohs micrographic surgery, found 50.5% of lesions changed aggressiveness with 33.0% becoming higher risk lesions than initially biopsied. 15 This study excluded Mohs excisions that reported clear margins on first stage, potentially accounting for higher proportion of excisions with upgraded risk status when compared to our cohort. 15
Detecting high risk features pre-operatively and maintaining a low rate of involved or close margins can reduce repeated procedures and specialist follow-up appointments. This would minimise the burden of KCs on both the individual patient and broader health system. Cancer Council Australia supports pre-treatment biopsy of suspected KCs to confirm diagnosis, inform prognosis, and maximise cure rate with reduced treatment-related morbidity. 3
Pre-operative biopsy is invaluable in defining lesion risk and informing surgical planning, especially when marking clinical excision margins. The Cancer Council Australia guidelines suggest clinical margins of 2–3 mm for low-risk BCCs and greater than 5 mm for high-risk BCCs.3,16 This extends to 4 mm for low-risk SCCs and 6 mm for high-risk SCCs.3,16 However, international guidelines vary widely and there is no universally accepted cut-off. 17 Pragmatically, margin determination represents a balance between disrupting functional and cosmetic subunits, long-term complications (e.g. ectropion, nasal obstruction), and tolerance for closer pathologic margins. Lesion type, subtype, and any additional high-risk features (e.g. PNI) can inform this balancing act in conjunction with shared decision making.
Pathologic classification of BCCs is complex, with up to 66 subtypes reported in the literature. 18 One study from Turkey in 2014 demonstrates largely similar distribution of histopathologic BCC subtypes from the head and neck, with the nodular subtype predominating. 19 However, this study did not report any infiltrative BCC subtypes, likely reflecting geographic differences in dermatopathology reporting.18,19 Another correlative analysis study reports a kappa statistic of 0.301 for BCC subtypes amongst six dermatopathologists, which increased to 0.699 with the dichotomy of low- and high-risk BCCs. 18 McKenzie et al. (2016) have endorsed simplified pathological reporting, stating ‘low-risk’ or ‘high-risk’ BCC with subtype pattern qualifier thereafter to increase clinical utility, aid prognostication, and improve inter-rater agreement. 20 This nomenclature is also used in the Cancer Council Australia Clinical Practice Guidelines for KC. 3 For this reason, if risk status remained the same from biopsy to formal excision pathology, even if specific subtype additions or omissions were noted, this was defined as concordance in our study.
The grading of cutaneous SCC is similarly nuanced. Poorly differentiated SCC is a well-recognised high-risk pathology,3,21 conferring a five-fold increase in metastasis and disease-specific mortality in a recent meta-analysis. 22 While MD-SCC is sometimes grouped with WD-SCC as low-risk grading, 3 other authors consider MD-SCC as intermediate risk given its poorer prognostic features when compared to WD-SCC.21,23,24 As a result, this study considered a WD-SCC on biopsy then confirmed to be MD-SCC on excision to be pathologically discordant.
Clinically, head and neck KCs are considered higher risk than other body sites, especially in anatomical cleavage lines (H-zone) of the face.3,25 While this study focusses on pathobiology risk status, the reality of clinical KC is that of a risk continuum, considering every aspect of the patient's presentation. 26
This study has several limitations. Analysis was undertaken in a single regional Australian specialist surgical centre with some of the highest global KC rates, representing an inherently high-risk population. In addition, our study analysed traditional surgical excisions under local anaesthetic only, as our centre does not perform Mohs micrographic surgery, though previous work on this topic is noted.14,15 These two limitations may reduce generalisability of our findings. On subgroup analysis, the small case number of SCCs limits our confidence in the identified biopsy correlation trends specific to SCC. Finally, it is sometimes difficult to judge the intent of pre-operative tissue sampling and whether a narrow margin excision represents biopsy or failed primary surgical excision. Every attempt was made to analyse macroscopic lesion descriptions, biopsy method, and referral letters to ascertain this intent and exclude KC excisions that are indeed re-excisions.
Conclusion
Our work outlines the previously undefined correlation for head and neck KCs surgically excised in a regional Australian centre when compared to pre-operative biopsy. Whilst our data suggest biopsy may occasionally overestimate pathological risk status, of greater clinical importance is the potential to underestimate KC subtype risk and alter the interpretation of margin tolerance. Further research examining a greater number of SCC excisions would help to clarify the low correlation rate demonstrated here. Additional work analysing consistency of measured clinical margins to biopsy risk status and KC clearance rates would also be informative. The limitations of pre-operative biopsy must be acknowledged by the treating surgeon and should be considered in shared decision making and consent processes.
Footnotes
Acknowledgements
The authors would like to thank Toowoomba Hospital ENT outpatient staff for their support and commitment to the FLAME clinic.
Author contributions
Timothy Sapsford: concept and design, collection and assembly of data, data analysis and interpretation, manuscript writing, and final approval of manuscript; Zachary Wilson: collection and assembly of data, data analysis and interpretation, manuscript writing, and final approval of manuscript; Daniel Anderson: concept and design, manuscript writing, and final approval of manuscript.
Data availability
Data utilised in this study are available with contact to the corresponding author.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics
Data was collected with approval from the DDHHS Human Research Ethics Committee (LNR/2023/QTDD/97361). This approval includes the waiver of written consent.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
