Abstract
Objective
Glioblastoma multiforme (GBM) is the most frequently occurring central nervous system tumor in adults. GBM patients exhibit poor survival outcomes following standard treatment, with drug resistance playing a critical role in disease progression. This study aimed to evaluate the effects of valsartan, a renin−angiotensin system inhibitor, on GBM cell lines with and without temozolomide (TMZ) resistance.
Methods
TMZ-resistant GBM clones were established by repeatedly exposing LN229 and GBM8401 human glioma cells to high doses of TMZ, followed by selective culture of the surviving populations. Tumorigenic potential was assessed using viability, sphere formation, invasion, and apoptosis assays. Gene expression was examined by sequencing and validated by quantitative polymerase chain reaction.
Results
Valsartan reduced the proportion of viable GBM cells regardless of TMZ sensitivity. Flow cytometry and western blotting revealed increased apoptosis in GBM following valsartan treatment. RNA sequencing revealed that valsartan regulates the cell cycle and induces G1-phase arrest, as evidenced by cell cycle flow cytometry. Moreover, APLN, an angiogenesis-related gene, was identified as a potential downstream target whose expression is significantly inhibited by valsartan.
Conclusions
This study revealed that valsartan is a novel strategy for treating gliomas with drug resistance.
Introduction
Glioblastoma multiforme
Glioma is a malignant intracranial tumor that primarily originates from glial cells and is among the most common types of brain tumors. 1 Among primary malignant brain tumors, glioblastoma multiforme (GBM) is the most malignant subtype. It is the most invasive and lethal type of brain tumor and is characterized by a high recurrence rate after treatment and a short survival period. The current standard clinical treatment for GBM includes surgical resection combined with whole-brain radiotherapy and oral chemotherapy with temozolomide (TMZ). 2 However, the survival time for GBM patients remains limited to 12–15 months. 3 Furthermore, at least 50% of patients develop resistance to TMZ following treatment, indicating a need for additional treatment options. 4 The mechanism of TMZ induces DNA mismatches during DNA replication, leading to cancer cell apoptosis.3,5 O 6 -methylguanine-DNA methyltransferase (MGMT) is a DNA repair protein that can repair the mismatches induced by TMZ. The overexpression of MGMT is a key factor contributing to drug resistance in GBM patients.6,7 To date, few treatment strategies that can effectively overcome GBM or its associated drug resistance have been established.
The renin−angiotensin system (RAS) is essential for regulating blood pressure and maintaining ionic balance within the body. 8 Previous studies have shown that hyperactivation of the RAS is associated with oxidative stress, 9 apoptosis, and inflammation. 10 Furthermore, overexpression of RAS components is linked to cancer development and angiogenesis. 11
GBM is a highly vascular tumor characterized by extensive neovascularization. The RAS serves as a key component of the tumor microenvironment and plays a critical role in several cancer hallmarks, including increased angiogenesis, hypoxia, 12 and tumor cell proliferation. 11 Thus, blocking the RAS is a potential antitumor treatment strategy.
Angiotensin II type 1 receptor (AT-1R) blockers (ARBs), such as valsartan, can fully inhibit AT-1R signaling. ARBs exhibit notable anticancer effects both in vitro and in vivo in cancers such as prostate cancer, 13 nasopharyngeal carcinoma, 14 breast cancer, 15 and colorectal cancer. 16 Furthermore, ARB treatment enhances the efficacy of anti-PD-1 therapy in melanoma mouse models. 17 Valsartan stands out for its strong antagonistic action and high binding affinity to AT-1R, suggesting its potential as a therapeutic agent for multiple cancer types. Based on statistical analyses, patients treated with valsartan have a lower incidence of lung cancer. 18 Previous studies have demonstrated that orally administered valsartan can cross the blood−brain barrier, where it affects the brain. 19 These reports inspired us to further investigate the effect of valsartan on brain tumors.
The results of this study demonstrate that valsartan has anti-GBM effects, as evidenced by decreased cell proliferation, induction of cell cycle arrest, induction of apoptosis, and suppression of invasion upon treatment. These findings highlight the potential value of valsartan as a candidate adjuvant in cancer treatment.
Materials and methods
Chemicals
Valsartan (SML0142, Sigma Aldrich, USA) was utilized in this study.
Cell culture
LN229 cells (RRID:CVCL_0393) purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA) and GBM8401 cells (RRID:CVCL_B051) purchased from the Bioresource Collection and Research Center (BCRC, Hsinchu, Taiwan) were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 2% fetal bovine serum (FBS) and 1% penicillin/streptomycin. We generated and sustained LN229 and GBM8401 TMZ-resistant (TMZ-R) clones in our laboratory as previously described by Tsai et al. 20 LN229 and GBM8401 TMZ-R clones were maintained in Roswell Park Memorial Institute (RPMI) 1640 with 10% FBS, 1% penicillin/streptomycin, and 400 μM TMZ for LN229 TMZ-R or 200 μM TMZ for GBM8401 TMZ-R. Normal human astrocytes (NHAs) were maintained in DMEM supplemented with 10% FBS, 1% penicillin/streptomycin, and 1× N-2. All cells were cultured at 37°C in a humidified incubator with 5% CO2. This study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024.
Cell lysis and protein quantification
Cells (5 × 105 cells/well) were seeded in 10 cm dishes overnight and treated with valsartan (0–300 μΜ) for 48 h. RIPA lysis buffer (EMD Millipore Corp, MA, USA) supplemented with protease and phosphatase inhibitors (Sigma Aldrich, USA) was used to lyse the cells. Protein concentration was measured with a DC protein assay kit (Bio-Rad, USA).
Western blotting
Thirty-five micrograms of protein were loaded onto SDS−PAGE gels. Next, samples were transferred to a polyvinylidene difluoride membrane (0.22 μm pore; Millipore, USA). Membranes were blocked with 5% skim milk in TBS-T (Tris-buffered saline with 0.1% Tween 20 buffer) for 1 h. The membranes were incubated with primary antibodies overnight at 4°C, washed with TBS-T, and incubated with secondary antibodies for 45 min at room temperature. Antibodies used in this study are listed in Table 1. Finally, protein bands were detected using Clarity Western ECL Substrate (Bio-Rad, USA) and analyzed with ImageJ software (National Institutes of Health, Bethesda, MD, USA).
The antibodies used in this study.

MGMT: methylguanine-DNA methyltransferase.
Cell viability assay
Cells (3 × 103 cells/well) were seeded in 96-well plates and treated with drugs (0–400 μM TMZ; 0–400 μM valsartan) for 48 h. Cell viability was analyzed using the CellTiter-96 AQ One Solution Cell Proliferation Assay (MTS). After drug treatment, 20 μL of MTS reagent was added to each well, and the samples were incubated for 2 h. Absorbance was measured at 490 nm.
Apoptosis assay
The cells (2 × 105 cells/well) were seeded in 6-well plates overnight and treated with valsartan (0–300 μΜ) for 48 h. After treatment, cells were resuspended in 1× binding buffer (1 × 106 cells/mL) and stained with PE-conjugated Annexin V/7-AAD (#559763, PE-conjugated Annexin V Apoptosis Detection Kit I; BD Biosciences, USA). Cells were analyzed with a FACSCalibur flow cytometer.
Colony formation assay
Cells (2 × 103 cells/well) were seeded in 6-well plates, incubated overnight, and treated with valsartan (0–300 μΜ) for 1 week. After 1 week of incubation, colonies were fixed with methanol for 20 min and stained with 0.005% crystal violet for 1 hour. Photographs were obtained after the samples were washed and air-dried. Colonies larger than 0.1 mm in diameter were counted via ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Cell proliferation assay
Cells (2 × 105 cells/well) were seeded in 6-well plates overnight and treated with valsartan (0–300 μΜ) for 48 h. Cell proliferation was analyzed using BrdU Flow Kits (#559619, BD Biosciences, USA) according to the manufacturer's instructions and analyzed with a FACSCalibur flow cytometer.
Wound healing assay
Cells were seeded in 12-well plates and incubated until they reached at least 90% confluence. A P1000 pipette tip was used to scratch the cells, which were then photographed and treated with valsartan (0–300 μM) for 16 h. After incubation, images were captured under a microscope, and the wound healing rate was calculated using ImageJ software.
Cell cycle analysis
Cells (2 × 105 cells/well) were seeded in 6-well plates overnight and treated with valsartan (0–300 μΜ) for 48 h. Cell cycle was analyzed using propidium iodide (#1932759, Invitrogen) according to the manufacturer's instructions and measured with a FACSCalibur flow cytometer.
Bioinformatics examination
To investigate the expression levels and prognostic value of APLN in glioma, several online bioinformatics platforms were utilized. We used Gene Expression Profiling Interactive Analysis (GEPIA, http://gepia.cancer-pku.cn/) 21 and the Gene Expression Database of Normal and Tumor Tissues 2 (GENT2, http://gent2.appex.kr) 22 to assess gene expression across different stages of gliomas. The association between APLN expression and patient survival was evaluated using GEPIA and the Chinese Glioma Genome Atlas (CGGA, http://www.cgga.org.cn/index.jsp) 23 to generate Kaplan–Meier survival plots. Differential gene expression analysis via GEPIA used a significance cutoff of log₂ fold change > 1 and p < 0.01. Kaplan–Meier survival curves generated by GEPIA were plotted based on the median stratification of APLN expression levels, with statistical significance defined as p < 0.05.
Gene expression profiling and biological pathway analysis
Total RNA extracted from LN229 and LN229 TMZ-R cells was analyzed via next-generation sequencing (Phalanx Biotech Group, Hsinchu, Taiwan). Fold changes were calculated as the ratio of the mean expression values in LN229 TMZ-R cells relative to those in LN229 cells.
RNA isolation
Total RNA was isolated from cells with TRI Reagent® solution and 1-bromo-3-chloropropane (Sigma, USA), followed by reverse transcription into cDNA with the High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific, USA), following the manufacturer's instructions.
Real-time reverse transcription polymerase chain reaction
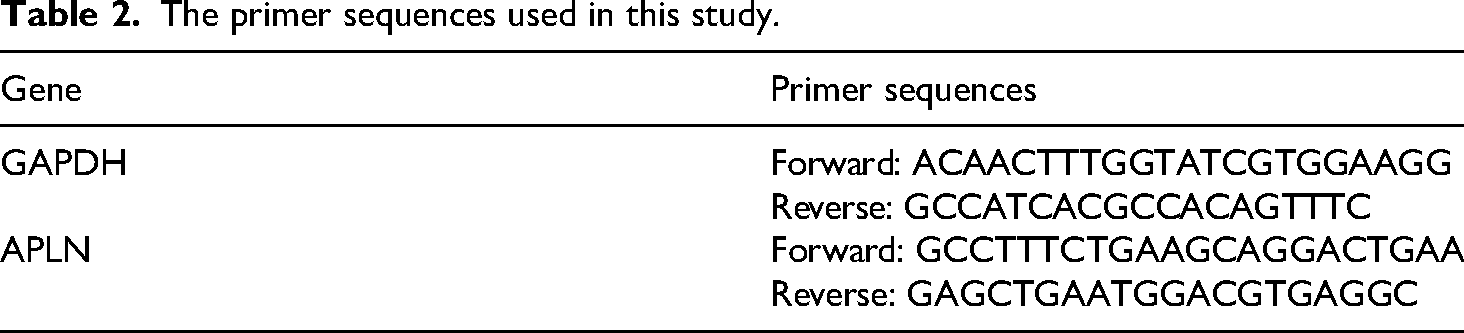
Quantitative real-time PCR was conducted using the IQ2 EVA Green Fast qPCR System Master Mix-HIGH ROX (Biogenesis, Taiwan) on a StepOne™ Real-Time PCR System (Thermo Fisher Scientific, USA). Primer sequences are detailed in Table 2, and GAPDH was used as the internal control for normalization of the results.
The primer sequences used in this study.
Statistical analysis
Statistical analyses were performed using an unpaired two-tailed Student's t test. Specific details regarding the statistical tests employed are provided in the corresponding figure legends. All experiments were performed at least three times unless otherwise specified in the figure legends. Data are presented as the means ± SEMs, with significance levels indicated as ***p < 0.001, **p < 0.01, and *p < 0.05.
Results
A GBM cell line with TMZ resistance was successfully generated
TMZ is an alkylating prodrug that undergoes spontaneous chemical activation in vitro due to its pH-dependent instability. 24 Although originally developed for in vivo use, TMZ does not require enzymatic activation; its conversion into active metabolites occurs efficiently in vitro when exposed to neutral or mildly basic aqueous conditions. 25 To evaluate valsartan as a potential therapeutic agent for treating GBM in both TMZ-sensitive and TMZ-resistant clones, we first developed TMZ-resistant glioma cell lines by repeatedly culturing and selecting surviving LN229 and GBM8401 human glioma cells following exposure to high doses of TMZ. 20 The tolerance of TMZ was measured via MTS assays (Figure 1A). The results showed that the half-maximal inhibitory concentration (IC50) values of TMZ in LN229 and GBM8401 cells were 249.94 and 220.78 μM, respectively. Compared with the parental cells, LN229 TMZ-R and GBM8401 TMZ-R cells had better tolerance to TMZ.

Valsartan decreased the proportion of viable TMZ-S and TMZ-R GBM cells in a dose-dependent manner. (A) TMZ-resistant glioma clones were established through drug exposure. An MTS assay was used to determine the viability of the indicated cells treated with TMZ (0–400 μM for 48 h; n = 3). (B) (Left panel) Effects of treatment with different concentrations of valsartan for 48 h (0–400 µM, n = 3) on the viability of nontumor control cells (NHAs) and TMZ-S and TMZ-R LN229 and GBM8401 clones assessed via MTS assays. Gray represents normal NHAs, red represents LN229 cells, light blue represents LN229 TMZ-R cells, yellow represents GBM8401 cells, light green represents GBM8401 TMZ-R cells, and the black dotted line indicates the IC50. (Right panel) The IC50 value at 48 h was determined from the dose−response curve. (C) Expression of MGMT protein in TMZ-S and TMZ-R glioma cells treated with valsartan at the indicated dose, analyzed by western blotting. Representative images from two independent tests are shown.
Valsartan decreased the proportion of viable GBM cells regardless of TMZ sensitivity
To investigate the effects of valsartan on the viability of GBM cells harboring TMZ-sensitive and TMZ-resistant clones, we treated LN229 and GBM8401 cells with valsartan at concentrations of 0, 100, 200, 250, 300, and 400 μΜ for 48 h and then assessed cell viability via MTS assays (Figure 1B). The results showed that valsartan decreased the proportion of viable cells among both TMZ-sensitive and TMZ-resistant clones. The IC50 values in LN229 and LN229 TMZ-R cells were 229.0 ± 6.7 μΜ and 239.8 ± 4.5 μΜ, respectively; the IC50 values in GBM8401 and GBM8401 TMZ-R cells were 231.6 ± 3.4 μΜ and 290.3 ± 7.2 μΜ, respectively (Figure 1B). Additionally, the IC50 in NHA cells was 359.5 ± 8.2 μΜ (Figure 1B), which was significantly greater than that observed in GBM cells. These findings suggest that valsartan has lower cytotoxicity toward normal control astrocytes than toward GBM cells. Next, we investigated whether valsartan can attenuate drug resistance in GBM cells by determining the expression of MGMT in TMZ-S and TMZ-R LN229 and GBM8401 cells using western blotting. The results indicate that valsartan had no significant effect on MGMT expression (Figure 1C). Taken together, these results suggest that valsartan robustly attenuates drug resistance in TMZ-S and TMZ-R LN229 and GBM8401 cells but not in normal NHA cells.
Valsartan inhibited the proliferation of both TMZ-S and TMZ-R glioma cells
The above results indicate that valsartan effectively decreased viability in TMZ-S and TMZ-R GBM cells. Therefore, we aimed to assess the proliferative capacity of these cells. We treated TMZ-S and TMZ-R GBM cells with valsartan at concentrations of 0, 100, 200, 300, and 400 μΜ for 48 h and stained them with BrdU. The results demonstrated that valsartan inhibited cell proliferation in a dose-dependent manner (Figure 2A–D). We subsequently performed colony formation assays to evaluate the prolonged impact of valsartan. Valsartan dose-dependently inhibited the colony formation of both TMZ-sensitive and TMZ-resistant clones (Figure 3A–D). In summary, our BrdU flow cytometry and colony formation assays indicated that valsartan significantly inhibited the proliferation of GBM cells.

Valsartan dose-dependently inhibited cell proliferation. Cells were stained with BrdU and analyzed by flow cytometry to measure proliferation of LN229 TMZ-S (A), LN229 TMZ-R (B), GBM8401 TMZ-S (C), and GBM8401 TMZ-R (D) clones (0, 100, 200, and 300 μM, respectively, for 48 h; unpaired two-tailed Student's t test; n = 3; *p < 0.05, **p < 0.01, ***p < 0.001).

Valsartan inhibited colony formation in a dose-dependent manner. (A–D) Colony formation of LN229 TMZ-S (A), LN229 TMZ-R (B), GBM8401 TMZ-S (C), and GBM8401 TMZ-R (D) cells treated with 0, 100, 200, or 300 μM valsartan for 14 days (unpaired two-tailed Student's t test; n = 3; **p < 0.01, ***p < 0.001).
Valsartan-induced apoptosis in both TMZ-sensitive and TMZ-resistant glioma cell lines
Since valsartan did not significantly affect MGMT expression (Figure 1C), we investigated whether it induces apoptosis. 26 Both the TMZ-S and TMZ-R GBM cell lines were treated with valsartan (0, 100, 200, or 300 μM) for 48 h, followed by Annexin V and 7-AAD staining. Flow cytometry was used to quantitatively assess the extent of programmed cell death, specifically apoptosis. Compared with the control group, the proportion of apoptotic cells increased in a dose-dependent manner upon valsartan treatment (Figure 4A–D). Additionally, western blot analysis was performed to assess the expression levels of apoptosis-related proteins. The results demonstrated that the expression of the DNA damage marker γ-H2AX increased with increasing valsartan concentration, indicating that increased DNA damage contributes to the induction of apoptosis. Conversely, the expression of the antiapoptotic protein survivin was significantly reduced, further supporting the conclusion that valsartan promotes apoptosis (Figure 4E–4H). Therefore, these findings confirm that valsartan induces apoptosis in both TMZ-S and TMZ-R GBM cells.

Valsartan-induced apoptosis in TMZ-S and TMZ-R glioma cells. (A–D) Apoptotic proportions of LN229 TMZ-S (A), LN229 TMZ-R (B), GBM8401 TMZ-S (C), and GBM8401 TMZ-R (D) clones treated with 0, 100, 200, or 300 μM valsartan, analyzed by flow cytometry based on annexin V and 7-AAD positivity. Bars indicate means ± SDs (unpaired two-tailed Student's t test; n = 3; **p < 0.01, ***p < 0.001). (E–H) Western blots showing γ-H2AX and survivin levels in LN229 TMZ-S (E), LN229 TMZ-R (F), GBM8401 TMZ-S (G), and GBM8401 (H) TMZ-R glioma cells treated with 0, 100, 200, and 300 μM valsartan for 48 h. Bars indicate means ± SDs (unpaired two-tailed Student's t test; n = 3, *p < 0.05, **p < 0.01, ***p < 0.001).
Valsartan inhibited the migration of both TMZ-sensitive and TMZ-resistant glioma cell lines
Enhanced cell migration, a hallmark of aggressive cancers, is closely linked to EMT and reflects the potential for tumorigenesis and metastasis. Therefore, in addition to promoting apoptosis, inhibiting cell migration is also critical for cancer drugs. In this study, we used a wound healing assay to evaluate the cellular migration process. A scratch was made in the monolayer of cells, followed by treatment with different concentrations of valsartan (0, 100, 200, 300 μM) for 16 h. Cells treated with valsartan exhibited slower wound closure, as the healing rate progressively decreased with increasing concentrations of valsartan (Figure 5A–D). Additionally, the expression levels of proteins associated with EMT markers were analyzed by western blotting, and the results revealed that the expression of Snail was inhibited by valsartan (Figure 5E–H). Briefly, these data confirm that valsartan inhibits the migration of TMZ-S and TMZ-R GBM cells by suppressing the expression of Snail.
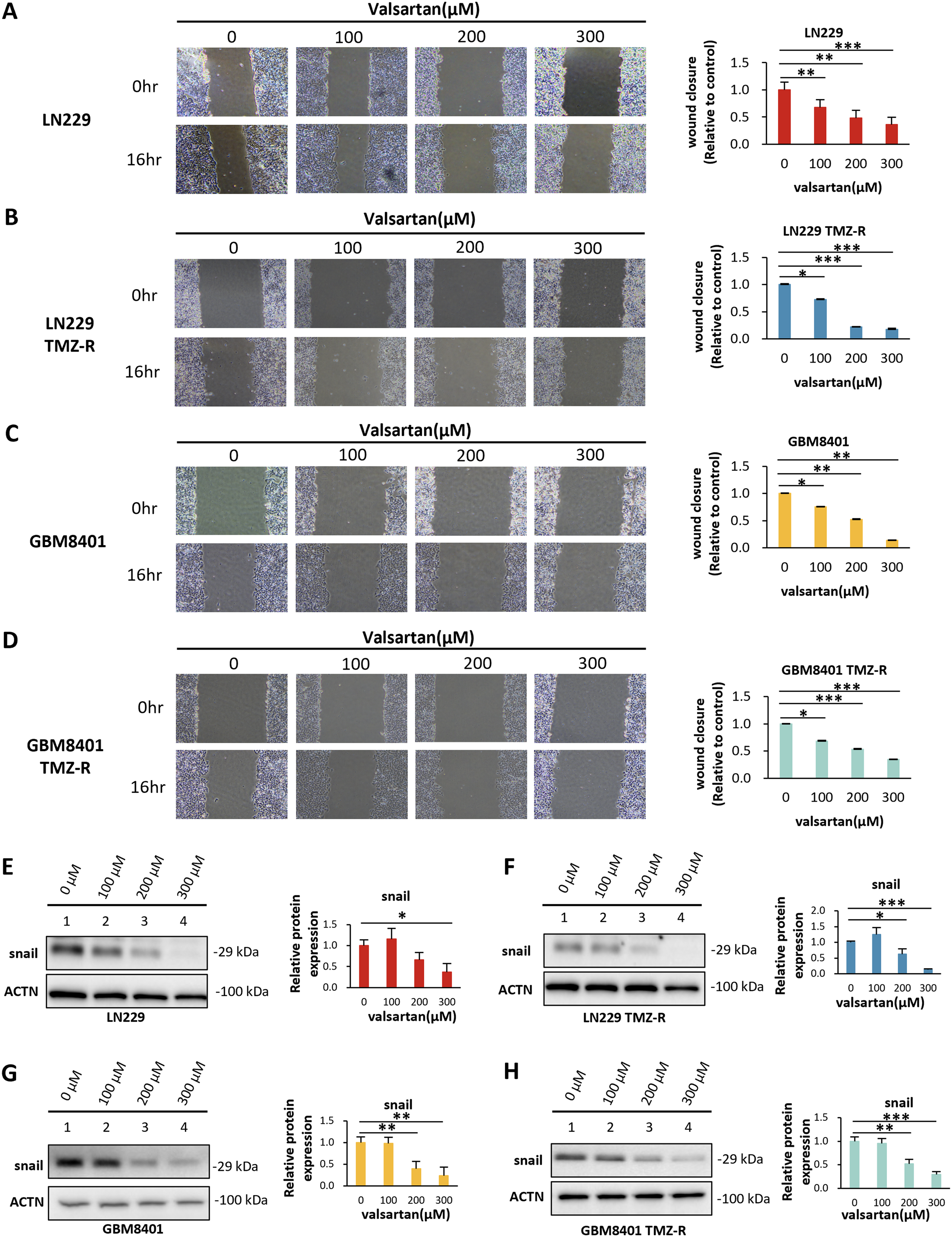
Valsartan inhibited the migration capacity of TMZ-S and TMZ-R glioma cells in a dose-dependent manner. (A–D) The migration capacity of LN229 TMZ-S (A), LN229 TMZ-R (B), GBM8401 TMZ-S (C), and GBM8401 TMZ-R (D) clones was analyzed via a wound healing assay. (E–H) Expression of EMT marker snail in TMZ-S and TMZ-R glioma cells was assessed by western blotting (0–300 μM, for 48 h; unpaired two-tailed Student's t test; n = 3; *p < 0.05, **p < 0.01, ***p < 0.001).
Valsartan-induced G1-phase cell cycle arrest in both TMZ-sensitive and TMZ-resistant GBM clones
The above results indicate that valsartan inhibits tumorigenicity in GBM cells by inducing apoptosis and inhibiting cell proliferation and migration. To determine whether other pathways are affected by valsartan treatment, we used RNA sequencing. The top 10 pathways whose level of enrichment was most affected by valsartan in both TMZ-sensitive and TMZ-resistant clones were identified and compared, revealing a common pathway: the cell cycle (Tables 3 and 4). The effects of valsartan on the cell cycle were subsequently validated. The results demonstrated that the cell cycle was arrested at the G1 phase after valsartan treatment (Figure 6A–D). These findings suggest that valsartan induces G1 phase arrest, preventing cells from progressing to subsequent DNA replication and cell division stages, which likely accounts for the inhibition of cell proliferation and promotion of apoptosis observed in previous experiments.

Valsartan contributed to cell cycle arrest at the G1 phase in TMZ-S and TMZ-R glioma cells. Cell cycle analysis of LN229 TMZ-S cells. (A), LN229 TMZ-R (B), GBM8401 TMZ-S (C), and GBM8401 TMZ-R (D) clones were analyzed by flow cytometry via PI staining (0–300 μM, for 48 h; 6n = 3).
List of the top 10 enriched pathway terms associated with valsartan treatment in LN229 cells.

List of the top 10 enriched pathway terms associated with valsartan treatment in LN229 TMZ-R cells.

TMZ: temozolomide.
Valsartan inhibits the expression of APLN, which is related to angiogenesis
Valsartan functions by blocking the binding of angiotensin to its receptor, thereby inhibiting the RAS. Previous studies have demonstrated a connection between the RAS and vascular function. During our investigation of the genes affected by valsartan, we identified APLN as the most downregulated gene following treatment (Figure 7A). This gene has been shown to be associated with cardiovascular functions and vessel formation. To validate the involvement of the APLN signaling pathway, we analyzed APLN mRNA expression by qPCR. The results revealed that valsartan treatment suppressed APLN expression (Figure 7B–7E). Next, we investigated the relationship between APLN expression and GBM outcomes using publicly available databases. Analysis via GEPIA and GENT2 demonstrated that APLN expression is positively correlated with GBM progression (Figure 7F–7G). Moreover, data from GEPIA and the CGGA revealed a negative correlation between APLN expression and GBM prognosis; that is, high APLN expression was associated with poor outcomes (Figure 7H–7I).

APLN is a potential downstream target of valsartan. (A) RNA sequencing revealed the top 10 upregulated and downregulated genes. (B–E) APLN mRNA expression in LN229 TMZ-S (B), LN229 TMZ-R (C), GBM8401 TMZ-S (D), and GBM8401 TMZ-R (E) clones was measured via RT−PCR after treatment with valsartan (0–300 μM, for 48 h, unpaired two-tailed Student's t test, n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). Expression levels of APLN in different stages of glioma tissue were analyzed via GEPIA (F) and GENT2 (G). Kaplan–Meier survival analysis was performed using data from GEPIA (H) and the Chinese Glioma Genome Atlas (CGGA) (I). T: tumor; N: normal control; LGG: low-grade glioma; HR: hazard ratio; p(HR): pooled hazard ratio; TPM: transcripts per million. TMZ: temozolomide; GEPIA: Gene Expression Profiling Interactive Analysis; GENT2: Gene Expression Database of Normal and Tumor Tissues 2.
Discussion
At present, treatments for GBM are limited. Although bevacizumab, an antiangiogenic agent, can effectively inhibit angiogenesis and reduce tumor size, it does not significantly increase overall survival rates. 27 Moreover, resistance to TMZ decreases treatment efficacy. Therefore, identifying new therapeutic strategies remains a critical and pressing challenge. In this study, we focused on the effect of blocking the RAS via valsartan on GBM clones with and without TMZ resistance.
RAS is expressed in various tissues and affects tumor development by regulating cell proliferation. 28 Previous reports have shown that AT1-R activates multiple pathways, such as the Wnt/β-catenin, MAPK-STAT3 and PI3K-AKT pathways, thereby promoting cancer growth.29,30 RAS inhibitors (RASis) have been shown to suppress tumor growth, metastasis, and cellular proliferation in various cancers. 28 For example, Uemura et al. reported that ARBs inhibit prostate cancer cell proliferation by suppressing MAPK-STAT3 signaling. 31 Reddy et al. reported that RASis reduce K-ras expression, decrease the levels of proliferation markers such as PCNA and PKC-p, and hinder mitosis by modulating gene expression in pancreatic cancer. 32 In this study, we observed that valsartan dose-dependently inhibited the survival, proliferation and colony formation of GBM clones with and without TMZ-R. Moreover, the RNA sequencing data revealed that the MAPK and RAS signaling pathways were significantly altered after valsartan treatment in GBM cells. These data are consistent with previous reports.
TMZ IC₅₀ values in glioma cell lines vary widely, ranging from low to high micromolar ranges. For instance, Yoshino et al. reported TMZ IC50 values for seven malignant glioma cell lines ranging from 22.5 to 441.6 μM. After using a cDNA microarray to explore mechanisms associated with TMZ sensitivity in various glioma cells, they reported that MGMT expression plays a dominant role. 33 Moreover, Perazzoli et al. analyzed TMZ sensitivity in four nervous system cancer cell lines, A172, LN229, SF268, and SK-N-SH. Unlike a previous study, they reported the values of TMZ IC50 positively correlated with the level of MGMT expression. 34 Based on these studies, we evaluated TMZ IC50 and MGMT expression in TMZ-sensitive and -resistant LN229 and GBM8401 cells.
Overcoming TMZ resistance in GBM cells often involves strategies that induce apoptosis. 26 For example, Li et al. reported that pyrvinium pamoate increases the sensitivity of GBM cells to TMZ chemotherapy by downregulating MGMT expression through strong suppression of Wnt signaling and repression of survivin, an antiapoptotic protein. 35 Tsai et al. described SNAP, a nitric oxide donor, as an effective agent for counteracting TMZ resistance in human GBM cells. The mechanism involves suppression of MGMT expression while promoting apoptosis, a process linked to elevated levels of cleaved PARP and cytochrome C, two key apoptosis-related proteins. 20 This study demonstrated that increasing the valsartan dosage triggered apoptotic pathways by reducing survivin expression while promoting the accumulation of γ-H2AX foci, indicating persistent DNA damage.
RNA sequencing revealed that the anti-GBM mechanism of valsartan involves the apelin signaling pathway, as APLN expression was significantly downregulated upon treatment. Previous studies have shown that this pathway plays an essential role in tumor angiogenesis36–38 and that APLN expression is negatively correlated with hepatocellular carcinoma prognosis. 39 Additionally, knockout of APLN in mice has been shown to suppress angiogenesis in the context of malignant melanoma, thereby inhibiting the growth of this vasculature-dependent tumor and improving survival rates in mice. 40 Anne et al. reported that apelin/APLNR signaling is essential for tumor angiogenesis, as evidenced by orthotopic GBM mouse models. Moreover, APLN-knockout mice exhibited significant suppression of vascular-dependent tumor growth. 41 In this study, we confirmed that valsartan suppressed APLN mRNA expression in both parental and TMZ-R GBM cells. Experiments are needed in the future to verify whether apelin signaling is associated with TMZ resistance.
Although the current results demonstrate that valsartan has therapeutic effects on GBM cells, this study has several limitations, including the lack of in vivo validation of these findings. Further validation of apoptosis-related mechanisms through various pathways may also be conducted in the future. While the concentrations of valsartan employed in this study (up to 300 µM) exceed those typically attainable in vivo due to its limited oral bioavailability (∼23%) and pharmacokinetic constraints, 42 they were selected to assess the maximal cytotoxic potential of the compound in GBM cell lines, independent of its established antihypertensive function. To address translational limitations associated with these in vitro concentrations, potential strategies for enhancing central nervous system delivery need to be developed. These include nanoparticle-based formulations and lipid carriers designed to improve blood–brain barrier permeability, 43 as well as localized delivery methods such as convection-enhanced delivery that may achieve higher regional drug concentrations. 44 Furthermore, valsartan may serve as a sensitizing agent in combination with other therapies, such as metformin, a metabolic modulator, 45 and vorinostat, a histone deacetylase inhibitor, 46 both of which have been confirmed to inhibit the growth of GBM cell lines. The dosage of valsartan can potentially be reduced to clinically feasible levels through synergistic interactions with other therapeutic compounds.
Conclusion
In this study, GBM cell lines with and without TMZ resistance were utilized to simulate clinical conditions. The experimental results support the anti-GBM effects of valsartan. However, the mechanism underlying the regulation of TMZ resistance by valsartan requires further investigation.
Footnotes
Acknowledgements
AI-based tools were employed in this study to identify and correct grammatical errors.
Ethics committee
Ethical approval (TSGHIRB No. E202416006) was granted by the Ethics Committee of Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan, on March 12, 2024.
Authors’ contributions
CKT and DYH conceptualized and designed the study. JRH, YK, YLC, CKT, and DYH conducted the research and contributed to the development of the methodology. The initial draft of this manuscript was authored by JRH and CKT. Data interpretation was carried out by JRH, YK, YLC, FCY, CKT, and DYH. All authors have reviewed and approved the final version for submission. The corresponding authors confirm that all listed authors have fulfilled the necessary authorship criteria and that no eligible contributors have been omitted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tri-Service General Hospital, National Science and Technology Council, Ministry of National Defense Medical Affairs Bureau, (grant number TSGH_E_114252, TSGH-C02-112031 and TSGH-C03-113039, NSTC 113-2321-B-016-005 and NSTC 112-2314-B-016−, MND-MAB-D-112075, MND-MAB-D-113058, MND-MAB-D-1130).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding authors, Chia-Kuang Tsai and Dueng-Yuan Hueng, upon reasonable request.
