Abstract
Objective
Resistance to platinum-based chemotherapy remains a key obstacle in ovarian cancer treatment. This study aims to investigate the role of Uncoordinated 51-like kinase 2 (ULK2) in chemoresistance of ovarian cancer and elucidate its underlying mechanisms using 3D patient-derived organoids.
Methods
Survival analysis was first performed using the Kaplan‒Meier plotter database. Immunohistochemical profiling delineated differential ULK2 expression patterns between chemoresistant and chemosensitive ovarian cancer tissue samples and organoids. ULK2 overexpression was achieved in cisplatin-resistant ovarian cancer organoids via lentiviral vector transduction. Then, we conducted an in-depth examination of the alterations in phosphorylated proteins induced by ULK2 overexpression using phosphoproteomics technology. To investigate the influence of ULK2 on chemosensitivity in ovarian cancer, Cell Counting Kit-8 (CCK-8) and in vivo experiments were conducted. Glycolysis was quantitatively assessed, and the underlying molecular mechanism was systematically investigated.
Results
ULK2 high-expression ovarian cancer exhibited enhanced chemosensitivity and conferred survival advantage. CCK-8 and mouse experiments demonstrated that ULK2 overexpression decreased cisplatin resistance in patient-derived organoids. Gene Ontology (GO) analysis of phosphoproteomics profiling highlighted the predominant role of ULK2 in metabolic processes with experimental validation demonstrating its suppression of glycolysis. Mechanistically, ULK2 attenuated c-Jun expression by phosphorylation of c-Jun at Ser243. Moreover, c-Jun overexpression counteracted the chemosensitivity and glycolytic suppression induced by the ectopic ULK2 expression in ovarian cancer.
Conclusions
ULK2 overcomes cisplatin resistance in ovarian cancer by downregulating glycolysis, a process mediated by phosphorylation-induced c-Jun degradation. These findings emphasized the role of ULK2 as a tumor suppressor, offering novel insights for chemotherapy in ovarian cancer.
Introduction
Ovarian cancer is an aggressive epithelial malignancy and ranks as the most lethal gynecological cancer, evidenced by a 5-year survival rate of merely 49.7%. 1 The generation and progression of ovarian cancer often manifests insidiously, frequently resulting in its detection at advanced stages. 2 Apart from late detection contributes to poor outcomes, the dominant clinical challenge remains platinum resistance, which drives disease progression and recurrence.3,4 However, the mechanisms contributing to chemoresistance in ovarian cancer are intricate and have not yet been entirely elucidated. Therefore, exploring the mechanisms associated with chemoresistance is crucial in ovarian cancer research. This study aimed to fill the existing knowledge gap by examining uncoordinated 51-like kinase 2 (ULK2) through using organoid models.
In recent years, organoid technology has emerged as a promising in vitro model with significant potential in cancer research, particularly in preclinical drug sensitivity testing. 5 By addressing the shortcomings inherent in 2D models, 3D constructs accurately reproduce the morphological and genetic characteristics of the original tumor while preserving the individual heterogeneity specific to the patient compared.6,7 The vast potential of organoids derived from ovarian cancer has been extensively investigated, encompassing both foundational theories and advanced applications. Serving as robust preclinical models, organoids offer critical resources for forecasting patient responses to treatments and assisting in the formulation of individualized therapeutic approaches. 8 We therefore aimed to identify a novel strategy for therapeutic intervention for chemo-resistant ovarian cancer using the patient-derived organoid model.
ULK2 is categorized within the serine/threonine kinase family and plays a crucial role in the regulation of autophagy. 9 An increasing number of evidence has shown that ULK2 expression significantly influences tumor initiation and progression through signaling pathways outside of autophagy. 10 Currently, there have been some studies related to ULK2 in ovarian cancer. For example, ULK2 inhibits the invasion and migration of ovarian cancer via the elevation of IGFBP3. 11 However, systematic research on its function in treatment of ovarian cancer is still lacking. Emerging evidence has identified ULK2 as a molecular regulator of chemoresistance. In non-small cell lung cancer, ULK2 expression can sensitize tumor cells to cisplatin, thereby overcoming drug resistance. 12 Despite the serious threat of chemotherapy resistance to ovarian cancer patient survival, the function of ULK2 in regulating chemosensitivity and the involved signaling pathways remain poorly defined.
Aerobic glycolysis, also known as Warburg effect, serves as a cornerstone of tumor metabolic reprogramming, driving both carcinogenesis and chemoresistance through multifaceted mechanisms.13–15 In ovarian cancer study, it was reported that the TCEB2/HIF1A axis was reported to facilitate the emergence of chemoresistance in ovarian cancer by enhancing glycolytic activity. 16 The findings collectively underscore the therapeutic relevance of dissecting glycolysis in ovarian cancer. However, little information is available regarding the association between ULK2 and glycolysis.
Research indicated that the decreased expression of ULK2 was a hallmark of platinum-resistant ovarian cancer and correlated with poor clinical outcomes. Functional studies demonstrated that the overexpression of ULK2 led to a decrease of chemoresistance in ovarian cancer organoids and mouse xenograft tumor models. Furthermore, ULK2 mediated the suppression of c-Jun expression through phosphorylation-induced its ubiquitin degradation, which in turn resulted in a reduction in glycolytic activity and increased sensitivity to chemotherapy. Collectively, these results emphasized the potential of ULK2 as a therapeutic strategy to inhibit platinum resistance in ovarian cancer.
Methods
Clinical samples and immunohistochemistry (IHC)
Prior to initiating the assay, we first secured ethical approval from Changning Maternity and Infant Health Hospital (Approval No. CNFBLLKT-2023-013; date of approval: April 19, 2023). A total of 80 ovarian cancer samples were obtained from Changning Maternity and Infant Health Hospital (now known as Tongren Hospital) between 2015 and 2020. Eligible patients were aged ≥18 years with a pathological diagnosis of primary epithelial ovarian cancer. Patients were excluded if they had other concurrent malignancies, significant comorbidities, or had received radiotherapy or chemotherapy prior to surgery. Written informed consent was obtained from all participants prior to the collection and use of human samples. After standard deparaffinization and antigen retrieval, tissue sections were exposed to validated primary antibodies, and further incubated with horseradish peroxidase (HRP)-conjugated secondary antibody at ambient room temperature for 30 min. The tissues were then subjected to staining using a DAB horseradish peroxidase color development kit (P0203; Beyotime, Shanghai, China). H-score was calculated using the formula: H-score = ∑(pi × i) = (percentage of weak intensity × 1) + (percentage of moderate intensity × 2) + (percentage of strong intensity × 3). The median H-score was defined as the cutoff for stratifying tumors into ULK2-high and ULK2-low expression groups. IHC of ovarian cancer organoids was performed with the same procedure. ULK2 antibody (PA5-22173) was procured from Thermo Fisher Scientific (Waltham, MA, USA). P-c-Jun (Ser243) antibody was obtained from Absin (abs148022, Shanghai, China), and c-Jun (9165) antibody was sourced from Cell Signaling Technology (Boston, MA, USA). The secondary antibodies used for immunohistochemistry were all HRP-conjugated goat anti-rabbit immunoglobulins, obtained from Abcam (ab6721, Cambridge, United Kingdom).
Immunofluorescence
Samples were fixed with 4% paraformaldehyde (E672002, Sangon Biotech) for 20 min, permeabilized with 0.1% Triton X-100 (20107ES76, Yeasen, Shanghai, China) for 5 min, and blocked with 5% bovine serum albumin (BSA, 36100ES25, Yeasen) for 30 min. The primary antibody was incubated overnight at 4 °C. Following wishes with PBS-T, Alexa Fluor-conjugated secondary antibodies were incubated for one hour at room temperature in the absence of light. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI). Images were acquired using a Nikon fluorescence microscope. P-c-Jun (Ser243) antibody was obtained from Absin (abs148022). c-Jun (9165) antibody were obtained from Cell Signaling Technology.
Organoid generation and culture
Fresh ovarian cancer specimens were dissected to remove necrotic, fibrotic, or clotted regions, followed by three washes with ice-cold Dulbecco's phosphate-buffered saline (DPBS, B640015, Sangon Biotech, Shanghai, China). Tissues were minced into 1–3 mm fragments and transferred to 50 mL conical tubes. After additional DPBS washes, pellets were resuspended in tissue dissociation buffer (D23013-0010, D1Med, Shanghai, China) and incubated at 37 °C for enzymatic digestion. Post-digestion, suspensions were pipetted vigorously to dissociate cells from stromal matrices. Digestion was quenched with basal medium, and suspensions were filtered through 100 μm strainers. Cells were pelleted (200 ×g, 5 min, 4 °C) and resuspended for organoid culture in 24-well plate. Organoids reaching 150 μm in diameter or cultured for 6–7 days were passaged. Chemosensitive organoids were derived from primary ovarian cancer tissues that exhibited sensitivity to cisplatin. Given the substantial heterogeneity of ovarian cancer and the significant biological variation among patient-derived organoids, direct comparisons across different individuals are suboptimal for pinpointing specific molecular mechanisms. To circumvent this limitation, we established chemoresistant organoid by a concentration gradient method in the chemosensitive organoid. A unique advantage of this approach is its provision of a matched, isogenic platform, thereby minimizing confounding effects from genetic heterogeneity.
Cell viability assay
Cell viability was quantified using a cell counting kit-8 (CCK-8, C0037, Beyotime Biotechnology, Shanghai, China). 10 μL of CCK-8 reagent was added to each well of 96-well plates containing 100 μL culture medium, followed by 1 h incubation at 37 °C. Finally, cell viability was assessed at 450 nm using a spectrophotometer.
Quantitative real-time polymerase chain reaction (qRT-PCR) analysis
Total RNA was extracted from ovarian cancer organoids using TRIzol reagent (15596026, Invitrogen, Carlsbad, USA) as per the guidelines provided by the manufacturer. Complementary DNA (cDNA) synthesis was conducted with a reverse transcription kit (Takara) with 1 μg of RNA. The synthesized cDNA was then subjected to qPCR with specific primers for target genes and a PowerUp SYBR Green Master Mix (4367659, ABI, Leuven, Belgium). The qPCR was performed in triplicate using PowerUp SYBR Green Master Mix (Applied Biosystems) on a QuantStudio 5 system with the following cycling parameters: 95 °C for 10 min; 40 cycles of 95 °C for 5 s, 55 °C for 30 s, and 72 °C for 30 s Primer specificity was confirmed by melting curve analysis and amplification efficiency. ULK2 expression was normalized to GAPDH and calculated using the 2ΔΔCt method. Primer sequences: ULK2 5′-TCAAGCATCTTCCAACCTGTTAG-3′; antisense, 5′-TAAACTGTCTGTGCTGCCCTGAT-3′; c-Jun, 5′-AAAGGAAGCTGGAGAGAATCG-3′, antisense, 5′-TGTTTAAGCTGTGCCACCTG-3′; GAPDH (5′-GGAAGCTTGTCATCAATGGAAATC-3′; and antisense, 5′-TGATGACCCTTTTGGCTCCC-3′.
Lentiviral transfection
Human ovarian cancer organoids in the logarithmic growth phase were dissociated into single cells using organoid dissociation reagent (D23031-0050, D1Med) and seeded in 24-well plates at a density of 4 × 104 cells per well. The cultures were transduced with either ULK2-overexpressing lentivirus (Genechem Co., Shanghai, China) or empty vector control virus at a multiplicity of infection (MOI) of 10. To enhance transduction efficiency, HitransG P (Genechem Co.) was added to the system. All lentiviral transduction procedures were meticulously performed according to the manufacturer's protocol. The viral-cell mixture was co-cultured for 6 h in a 37 °C incubator with 5% CO₂, then transferred to pre-cooled PBS and centrifuged at 300 × g for 5 min at 4 °C. The collected ovarian cancer organoids were subsequently embedded for 3D culture. After 7 days, stable clonal populations were selected using 1 µg/mL puromycin treatment for 7 days.
Phosphoproteomics analysis
Phosphorylation mass spectrometry, based on liquid chromatography-tandem mass spectrometry/mass spectrometry (LC-MS/MS), was performed for large-scale qualitative and quantitative analysis of phosphorylation. Samples were lysed using an appropriate volume of SDT [4% (w/v) SDS, 100 mM Tris/HCl, pH = 7.6] for protein extraction. The filter-Aided Proteome Preparation method was employed for trypsin digestion. 17 Mass spectrometry (timsTOF pro2, Bruker, MA, USA) was performed in positive ion mode, with the ion source voltage set to 1.5 kV. Fold Change (FC) > 2.0 and P value <.05 was used to quantify the number of upregulated and downregulated modified peptides.
Analysis of key glycolytic metrics
Glucose uptake and lactate production quantification were performed for measurement of glycolysis. Glucose uptake was measured using a glucose uptake assay kit (ab136955, Abcam). Lactate and ATP production was assessed with specialized kits (S0208S and S0026, Beyotime Biotechnology) according to the manufacturer's instructions. Pyruvic acid level was tested by kt purchased from Sangon Biotech Co. (D799449, Shanghai, China).
Extracellular acidification rate (ECAR) and oxygen consumption rate (OCR)
ECAR represents the rate at which cells release acidic metabolic byproducts. The Seahorse Energy Metabolism Analyzer determines this rate by assessing pH changes resulting from lactate production during glycolysis. On the other hand, OCR signifies the rate of oxygen uptake by cells. The Seahorse Analyzer measures this by evaluating fluctuations in dissolved oxygen levels within the culture medium, which occur due to the activity of oxygen consumption reagents. Following 16-h sensor hydration, cells in XF assay medium underwent sequential treatment with glucose/oligomycin/2-DG for ECAR measurement, and oligomycin/FCCP/rotenone-antimycin A for OCR assessment.
Western blotting (WB)
Western blot analysis was performed on protein extracts from ovarian cancer organoids. Following protein quantification using a BCA assay kit (P0010, Beyotime, Shanghai, China), samples were mixed with loading buffer (P0015, Beyotime), denatured at 100 °C for 10 min, and separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using 10% resolving gels. Electrophoresis was carried out at 60 V until samples entered the resolving gel, then increased to 120 V until completion. Proteins were transferred to polyvinylidene difluoride (PVDF) membrane (88518, Thermo Fisher Scientific) at 300 mA for 60–90 min on ice. After blocking with QuickBlock™ blocking buffer (P0235, Beyotime) for 10 min, membranes were incubated overnight at 4 °C with the following primary antibodies: phospho-c-Jun (Ser243) (abs148022, Absin, Shanghai, China), c-Jun (9165, Cell Signaling Technology), GLUT1 (21829-1-AP, Proteintech, Chicago, IL, USA), HK2 (22029-1-AP, Proteintech), PFKFB3 (13123, Cell signaling technology), LDHA (2012, Cell signaling technology), MCT4 (22787-1-AP, Proteintech), ULK2 (ULK2-201AP, ThermoFisher) or β-Actin (4967, Cell signaling technology). Following three washes with TBS-T, membranes were incubated with the appropriate horseradish peroxidase (HRP)-conjugated secondary antibody (A0208 or A0216, Beyotime) for 2 h. Antibodies were diluted using specialized solutions from Beyotime (P0023A and P0023D). Specifically, the primary and secondary antibodies were diluted to 1:1000 and 1:2000, respectively. Protein bands were visualized using an Enhanced Chemiluminescence (ECL) detection kit (36222ES60, Yeasen).
In vitro kinase assay
Construct for HA-tagged c-Jun was expressed in Escherichia coli and induced with 0.1 mM Isopropyl β-
Co-immunoprecipitation (Co-Ip)
Following three washes with ice-cold PBS, cells were lysed using IP lysis buffer (P0013, Beyotime) maintained on ice for 30 min. The resulting lysates were clarified by centrifugation at 12,000 × g for 15 min at 4 °C. To minimize nonspecific interactions, supernatants were pre-adsorbed with Protein A/G beads for 1 h at 4 °C under constant rotation, followed by a 5-min centrifugation at 12,000 × g and 4 °C. Pre-cleared lysates were subsequently immunoprecipitated with a primary antibody targeting the protein of interest under overnight incubation at 4 °C. The primary antibodies c-Jun (9165, Cell signaling technology) and ULK2 (ULK2-201AP, ThermoFisher) were utilized in the study. Immune complexes were captured by adding Protein A/G beads and then incubated for 4 h at 4 °C. Beads were then pelleted (12,000 × g, 5 min, 4 °C) and subjected to three washes using ice-cold lysis buffer. Proteins were finally eluted from the beads by heating at 95 °C for 5 min in SDS sample buffer (P0015, Beyotime), and the eluates were analyzed by Western blot.
Positron emission tomography/computed tomography (PET/CT)
PET/CT imaging was performed as part of the standard clinical workup. Briefly, after fasting for at least 6 h, patients received an intravenous injection of 18F-Fluorodeoxyglucose (FDG). Before the PET/CT scan, the patients must have a blood glucose level ≤10 mmol/L. Administer 5.55 MBq/kg of 18F-FDG intravenously, and perform scanning from the mid-upper thighs to the cranial vault after 50–60 min of rest. CT settings: 140 kV, 110 mA, 0.5 s/r, 3 mm slice thickness. PET acquisition: 3 min per bed position, with attenuation correction of PET images using CT data. The maximum standardized uptake value (SUVmax) was measured within spherical volumes of interest (VOIs) placed over the primary ovarian tumors, with the highest value used for patients with multiple lesions. Patients were categorized into ULK2-high and ULK2-low cohorts based on the median H-score applied to immunohistochemistry results from their surgical specimens. PET/CT SUVmax was then compared between the two groups.
Construction of the c-Jun Ser243A mutant plasmid
The c-Jun Ser243A mutant plasmid was constructed using a seamless cloning strategy. Briefly, target DNA fragments were amplified with specifically designed primers and subsequently inserted into the enzyme-digested carrier via seamless cloning. The resulting ligation products were then transformed into chemically competent E. coli cells. Single colonies were screened by commercial Sanger sequencing, and clones yielding right sequences were selected as successfully constructed plasmids. Primer sequences used were as follows: 116631FW1-143852: GCCGCGAATTCGAAGTATACCTCGAGGCCACCATGACTGCAAAGATGGAAACGA; 116631RW2-143854: CGGAGCGATCGCAGATCCTTGGATCCTCAAGCGTAATCCGGAACATCGTATGG.
Public database
The association between ULK2 expression and survival in ovarian cancer patients was assessed using the Kaplan‒Meier Plotter database (http://kmplot.com/analysis). URL is available online at http://kmplot.com/analysis. 19
In vivo experiments
Our animal studies adhered strictly to the ARRIVE guidelines 20 and institutional ethical standards for animal welfare, receiving formal approval from the Ethics Committee of Xinhua Hospital (Approval No. XHEC-F-2025-022; date of approval: April 30, 2025). All animal procedures were performed in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals, 8th Edition. 21 The 3R principles (Replacement, Reduction, and Refinement) guided all aspects of our animal experiments. The effect of ULK2 overexpression on platinum chemosensitivity in ovarian cancer was assessed using a subcutaneous xenograft model. 22 Ten female BALB/c nude mice (4 weeks old) with a body weight of 18–20 g were randomly divided into control and ULK2 overexpression groups (n = 5 per group) using a computer-based random number generator and were housed in specific pathogen-free (SPF) conditions with controlled temperature (22 ± 2 °C) in the animal research center of Xinhua hospital. While a formal sample size calculation was not performed, our choice of n = 5 mice per group is consistent with common practice in the field for similar interventions and outcome measures. 23 After 1-week acclimatization, cisplatin-resistant ovarian cancer organoids were harvested and dissociated into single cells. Subsequently, tumor models were established by a subcutaneous injection of cells (6 × 106 in 100 μL PBS) into the right flank of the mice. Tumor formation was monitored three times weekly using digital calipers, with volumes calculated with the formula: V = 1/2 × (length × width2). Once tumors reached 50 mm3, mice were administered weekly intraperitoneal injection of cisplatin (6 mg/kg). Ultimately, after the 4-week study period, all nude mice were euthanized by 100% CO₂ asphyxiation and subsequent cervical dislocation, and a complete necropsy was performed to collect tissues for subsequent analysis. In accordance with animal welfare principles, specific humane endpoints (e.g. tumor ulceration, significant hemorrhage, or a tumor diameter > 2 cm) were established to minimize animal suffering. All experimental procedures, including animal restraint and injection, were performed in a humane manner with focused attention on the alleviation of stress and pain. All tumor measurements and tissue collection were performed by an investigator blinded to the experimental groups. No adverse reactions occurred over the course of the animal study. The animal procedures involved only minimal and transient discomfort and thus anesthesia was not required as per the approved ethical protocol.
Statistical analysis
All statistical analyses were performed using GraphPad Prism 9.0. Data are presented as mean ± SD unless otherwise stated. For comparisons between two groups of normally distributed data, an unpaired, two-tailed Student's t-test was used. In the selection of significantly differentially modified peptides, we applied significance A/B method to assess the statistical significance, as described in Nature Biotechnology. 24 Tumor growth curves were analyzed by two-way ANOVA. Kaplan‒Meier survival analysis with the log-rank test was used to compare outcomes between groups with high versus low ULK2 expression. The staining intensity of ULK2 or c-Jun in immunohistochemistry and immunofluorescence assays was quantified using Image-Pro Plus 6.0 (IPP 6.0) software. Statistical significance was set at p < 0.05.
Results
Low ULK2 expression was linked to chemoresistant ovarian cancer tissues and predicted a poor prognosis for patients
To investigate the correlation between ULK2 and the outcome of ovarian cancer patients, we employed KM online survival analysis tool to examine the association of ULK2 and patient OS, PFS, and PPS. High expression of ULK2 was positively correlated with the OS (HR = 0.57; p = .0034), PFS (HR = 0.47; p = 1.5 × 10−7), and PPS (HR = 0.63; p = 0.047) of ovarian cancer patients. In contrast, low levels of ULK2 were linked to poorer survival outcomes in the patients (Figure 1A–C). Subsequent IHC of ovarian cancer tissue microarrays demonstrated significantly downregulated ULK2 expression in cisplatin-resistant specimens compared to chemosensitive controls (Figure 1D–F). Further survival analysis revealed that ULK2 expression correlated with a trend toward prolonged overall survival in ovarian cancer patients. This was evidenced by hazard ratios (HR) of 0.4778 in the chemosensitive cohort and 0.2549 in the chemoresistant cohort (Figure 1G–H). These findings suggested that ULK2 deficiency may have contributed to the development of chemoresistance in ovarian cancer, ultimately impacting patient survival outcomes.

Prognostic and therapeutic implications of ULK2 in ovarian cancer. (A) Kaplan‒Meier (KM) plotter online network displayed the positive association of ULK2 and the overall survival (OS) in ovarian cancer. HR: Hazard ratio. (B) KM-plotter online network demonstrated a favorable relationship between ULK2 and the progression-free survival (PFS) in ovarian cancer. (C) KM-plotter online network revealed a beneficial correlation of ULK2 and the post-progression survival (PPS) in ovarian cancer. Part labels (A-C) were directly generated by KM-plotter online (http://kmplot.com), and the original data were obtained from GEO database (GSE 1554112, n = 285). (D) Representative immunohistochemical images of ULK2 expression in chemo-sensitive (chemo-S) versus chemo-resistant (chemo-R) ovarian cancer tissues. (E) H-score value of the above immunohistochemistry results. (F) Quantification analysis of ULK2-positive tissues. (G) Prognostic value of ULK2 expression in overall survival of chemosensitive ovarian cancer (n = 56). (H) Association between ULK2 expression and overall survival in chemoresistant ovarian cancer patients (n = 32).
Diminished expression of ULK2 was observed in cisplatin-resistant ovarian cancer organoid models
Organoid models represent an advanced drug screening platform that closely recapitulates human drug responses, and have been widely adopted in cancer drug resistance research. To investigate the role of ULK2 in platinum-based chemotherapy resistance of ovarian cancer, we established paired chemosensitive and chemoresistant ovarian cancer organoids and treated with cisplatin. Within 24 h of culture, distinct irregular spheroids were clearly visible. These structures proliferated and grew in size during culture, developing into larger solid or hollow spherical 3D structures within one week. Despite similar baseline morphology, chemosensitive organoids exhibited pronounced cisplatin-induced apoptosis, resulting in progressive structural disintegration and eventual loss of spherical architecture (Figure 2A). The differential drug responses were validated by CCK-8 assays (Figure 2B–C). Subsequent immunohistochemical analyses revealed that ULK2 was significantly downregulated in chemoresistant ovarian cancer organoids compared to their chemosensitive counterparts (Figure 2D–E). The findings further indicated that ULK2 loss might functionally result in acquired chemoresistance in ovarian cancer.

ULK2 expression level in patient-derived ovarian cancer organoids. (A) Morphological features of chemo-sensitive and -resistant ovarian cancer organoids. (B‒C) Viability assessment by CCK-8 assay confirming differential cisplatin sensitivity in chemo-sensitive (B) and chemo-resistant (C) organoids. Organoids were treated with 8 μg/L cisplatin in different time intervals. (D) ULK2 expression profiles evaluated by immunohistochemical staining in paired chemo-sensitive and -resistant organoid models. E. The immunohistochemical intensity of ULK2 was analyzed using Image-Pro Plus 6.0 (IPP 6.0) software. **p < .01.
ULK2 overexpression restored cisplatin sensitivity in chemo-resistant ovarian cancer organoids, concomitant with extensive remodeling of protein phosphorylation
To delineate the functional role of ULK2 in chemoresistance, we performed lentivirus-mediated ULK2 overexpression in ovarian cancer organoids, with successful transduction confirmed by qRT-PCR (Figure 3A). Functional assessment revealed that ULK2 induction markedly reduced viability in the chemoresistant organoid model (Figure 3B). Considering that ULK2 is a protein kinase that exerts its function by phosphorylating proteins, we conducted phosphoproteomic analysis on ULK2-overexpressing organoid and the control group. A total of 4861 phosphorylated proteins, along with 19016 quantifiable phosphorylated peptide segments were identified. Among the identified 4861 altered proteins, 30 were uniquely present in the ULK2-overexpressing cell line, 28 were exclusively present in the control and the remaining proteins were present in both groups (Figure 3C). Out of the 19016 altered phosphorylated peptide, 345 were specifically found in the ULK2 upregulation cell line, 194 were only detected in the control group, and the rest of the proteins were detected in both groups (Figure 3D). To investigate the distribution of modification sites across various proteins, a comprehensive count of phosphorylation sites present on all identified proteins was conducted. The statistical results show that 71.3% of proteins had two or more modification sites (Figure S1A), and the average distribution of phosphorylation modification sites per 100 amino acids was 49.42 (SFigure S1B). Serine residues represented the predominant phosphorylation modification sites (86.27%), followed by threonine (12.65%) and tyrosine (1.08%) (Figure S1C).
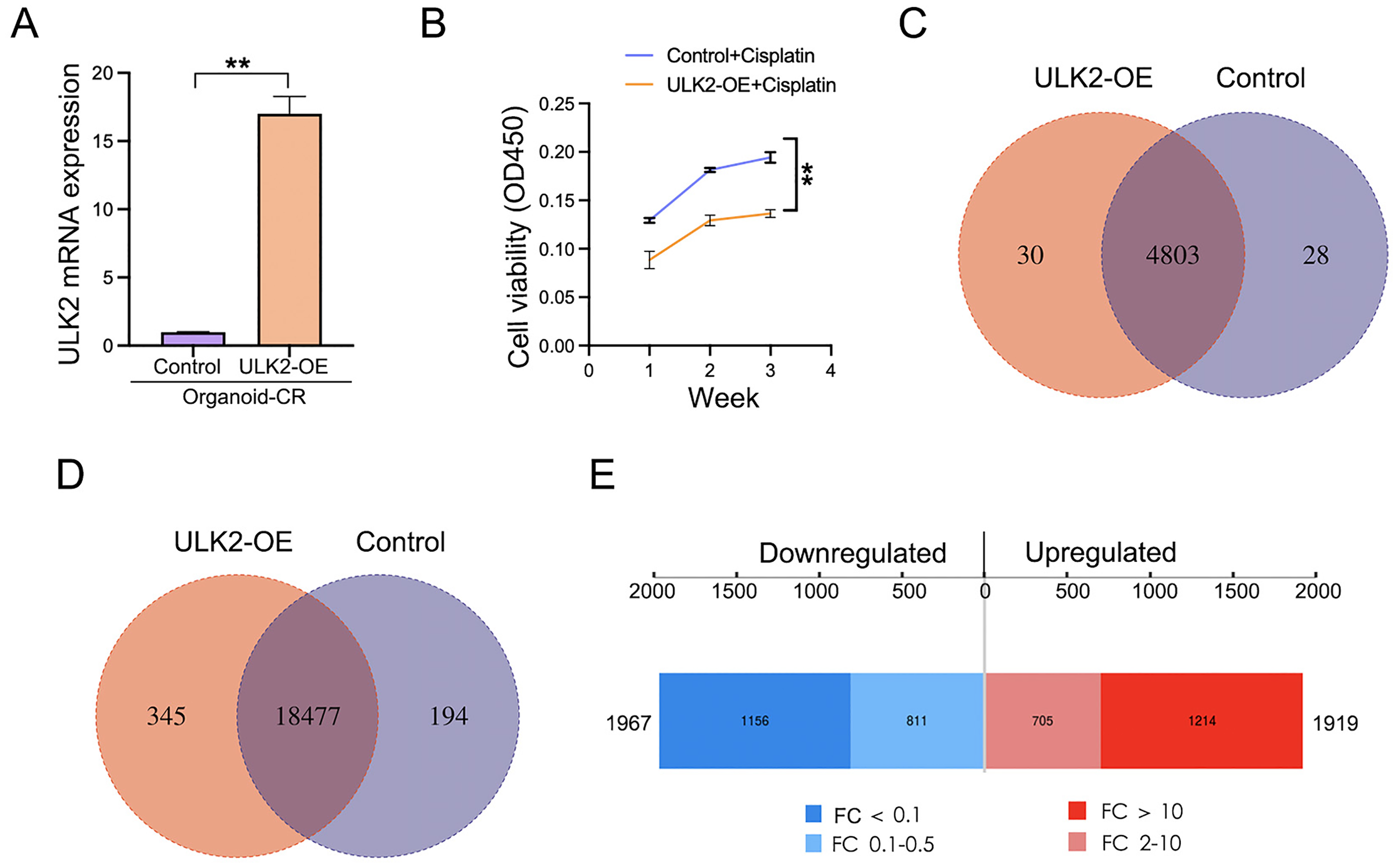
Effects of ULK2 overexpression on chemo-resistant ovarian cancer organoids and the phosphoproteomic alterations. (A) ULK2 overexpression level in chemo-resistant organoids was validated by qRT-PCR. (B) Impact of ULK2 overexpression on chemosensitivity was assessed using CCK-8 assays under cisplatin treatment (8 μg/L). (C) Comparative analysis of phosphoprotein identification between ULK2-overexpressing and control organoids by LC-MS/MS (Venn diagram). (D) Statistical analysis of phosphorylated peptides in ULK2-overexpressing and control organoids (Venn diagram). (E) Upregulated and downregulated peptides induced by ULK2 overexpression (threshold: p < .05, fold change >2).
To identify the differentially expressed phosphorylated peptides (DEPPs) between ULK2 overexpression and the control group, we performed quantitative analysis at the peptide level. The results indicated 3886 significantly DEPPs (Table S1), comprising 1919 upregulated and 1967 downregulated peptides. Out of these, 1214 peptides in the upregulated group exhibited a fold change (FC) exceeding 10, while 1156 peptides in the downregulated group showed a FC below 0.1 (Figure 3E). Additionally, an examination of subcellular localization was carried out on all DEPPs, revealing that 1440 proteins were localized within the nucleus, whereas 290 proteins were found in the cytoplasm (Figure S1D). Structural domain analysis revealed that there were 66 differentially phosphorylated proteins with protein kinase domains (Figure S1E). Based on these findings, we postulated that dysregulated ULK2 expression altered protein phosphorylation, which potentially contributed to chemoresistance in ovarian cancer.
ULK2 exerted inhibitory effects on glycolytic metabolism in ovarian cancer cells
Gene Ontology (GO) enrichment analysis of phosphoproteomic data from ULK2-overexpressing and the control organoids revealed 1149 phosphorylation events associated with metabolic processes, including multiple glycolytic regulators (Figure S2A). These findings suggested ULK2 may have modulated glycolysis. Functional validation demonstrated that ULK2 overexpression significantly reduced glucose uptake, lactate, ATP and pyruvic acid production (Figure 4A–D). Further metabolic flux analysis (ECAR and OCR) furtherly confirmed ULK2-mediated suppression of glycolytic activity (Figure 4E–F). Consistent with its role in attenuating glycolysis, ULK2 upregulation induced the downregulation of key glycolytic effectors, including PFKFB3, LDHA, and MCT4, as demonstrated by Western blot (Figure 4G). Clinically, PET/CT imaging in ovarian cancer patients showed that high ULK2 expression correlated with significantly lower maximum standardized uptake values (SUVmax) of [(18)F]2-deoxy-2-glucose (FDG), supporting its role in glycolytic regulation (Figure 4H–1). Furthermore, within this study, we observed that the application of the glycolysis inhibitor 2-DG to chemoresistant ovarian cancer organoids resulted in a marked reduction in cell viability (Figure S2B). This observation suggested that inhibition of glycolysis had potential for augmenting the sensitivity of ovarian cancer to platinum-based chemotherapy. Therefore, we inferred that ULK2 may have enhanced ovarian cancer chemosensitivity by inhibiting glycolysis. Conversely, patients with low ULK2 expression exhibited high glucose metabolism, leading to ovarian cancer chemoresistance.

The association between ULK2 and glycolysis. (A) Glucose uptake in response to ULK2 overexpression. (B) Lactate production upon ULK2 induction. (C) Evaluation of ATP production subsequent to elevation of ULK2. (D) Assessment of pyruvic acid following ULK2 overexpression. (E‒F) Metabolic profiling of ovarian cancer organoids through extracellular acidification rate (ECAR, E) and oxygen consumption rate (OCR, F) analysis. (G) Glycolytic enzyme expression was assessed by western blot. (H) PET/CT imaging showing representative metabolic activity in ULK2-high (SUVmax 5.6) versus ULK2-low (SUVmax 22.0) expressing tumors. (I) Quantitative analysis demonstrating significantly low SUVmax in ULK2-high group (n = 12) compared to ULK2-low group (n = 13). *p < .05; **p < .01.
ULK2-mediated phosphorylation of c-Jun triggered its ubiquitin-mediated degradation
KEGG pathway analysis of phosphoproteomic data revealed that ULK2 overexpression significantly altered the MAPK signaling pathway, with 38 differentially phosphorylated proteins identified (Figure S2C). The top 10 proteins of the pathway exhibiting the most pronounced increases in phosphorylation levels are listed in Figure S2D. Notably, c-Jun exhibited the most obvious increase in phosphorylation (Ser243). This finding was further validated by Western blotting (Figure 5A) and in vitro kinase assay (Figure 5B). The c-Jun Ser243 phosphorylation has been reported to induce c-Jun degradation in cervical cancer. 25 Given this, we proceeded to examine c-Jun protein expression levels via Western blotting and immunofluorescence. Our results demonstrated that ULK2 overexpression resulted in a decrease in c-Jun levels (Figure 5A, C–E). To delineate the mechanism by which ULK2 regulates c-Jun, we performed sequential molecular and functional investigations. First, co-immunoprecipitation assays verified a direct protein-protein interaction between ULK2 and c-Jun (Figure 5F). We subsequently demonstrated, via cycloheximide (CHX) chase experiments, that ULK2 overexpression substantially accelerated c-Jun degradation (Figure 5G). Crucially, we further established that ULK2 significantly augments c-Jun ubiquitination (Figure 5H), defining a post-translational mechanism for c-Jun destabilization. Functionally, knockdown of c-Jun expression attenuated chemoresistance and suppressed glycolysis in ovarian cancer organoids (Figure 5I–L). Conversely, c-Jun overexpression rescued the suppressed levels of chemoresistance and glycolysis resulting from ULK2 upregulation (Figure 5M–O). Of particular significance, c-Jun Ser243 mutation (c-Jun Ser243A) increased c-Jun protein level and effectively reversed the ULK2-mediated suppression of glucose uptake and cell viability (Figure S2E and Figure 5P–Q). Based on these findings, we conclude that ULK2-mediated phosphorylation of c-Jun at Ser243 promotes its subsequent ubiquitin-dependent degradation. This downregulation of c-Jun impairs glycolytic metabolism and ultimately enhances chemosensitivity in ovarian cancer.

ULK2 modulated c-Jun phosphorylation in ovarian cancer organoids. (A) Immunoblot analysis of phospho-c-Jun (Ser243) and c-Jun expression, with β-actin as loading control. (B) In vitro kinase assay to test the phosphorylation of c-Jun protein by ULK2 protein. (C-D) Quantification of ULK2 and c-Jun immunofluorescence signals was performed using IPP 6.0 software. E. ULK2 and c-Jun were examined by immunofluorescence. (F) Co-IP analysis indicated an interaction between ULK2 and c-Jun in chemoresistant ovarian cancer organoid. (G) Analysis of protein stability by cycloheximide (CHX) chase assay. Western blot of organoid lysates following treatment with CHX (50 μg/mL) for the indicated durations. (H) c-Jun was immunoprecipitated from chemoresistant ovarian cancer organoids and subsequently probed with an antiubiquitin antibody by Western blot. (I) Analysis of c-Jun mRNA expression by qRT-PCR. (J) c-Jun silencing reduced the cell viability of chemo-resistant ovarian cancer organoid. Organoids were treated with 8 μg/L cisplatin in different time intervals. (K) Assessment of glucose uptake following the knockdown of c-Jun. (L) Analysis of lactate production subsequent to c-Jun silencing. (M) Effect of c-Jun overexpression was verified by qRT-PCR. (N) The overexpression of c-Jun counteracted the decrease in cell viability caused by the upregulation of ULK2 in chemo-resistant ovarian cancer organoid. (O) Glucose uptake assay. (P) Effects of ULK2 overexpression and c-Jun S243A mutation on cell viability in chemoresistant ovarian cancer organoids. Organoids were treated with 8 μg/L cisplatin in different time intervals. Q. ULK2 overexpression suppresses glucose uptake, which is rescued by the c-Jun S243A mutation in chemoresistant ovarian cancer organoids. *p < .05; **p < .01.
ULK2 overexpression increased the cisplatin-sensitivity of ovarian cancer in vivo
Drawing from these pivotal in vitro findings, we transitioned to an in vivo experimental approach to comprehensively assess the direct impact of ULK2 overexpression on cisplatin sensitivity in ovarian cancer. The result indicated that ULK2 overexpression significantly improved the therapeutic efficacy of these xenograft models in response to cisplatin treatment, as evidenced by the substantial inhibition of tumor growth and weight (Figure 6A–C). Relative to controls, the experimental group (ULK2 OE) showed a marked reduction in tumor growth: tumor volume was significantly smaller (220.80 ± 56.13, 95% CI: 151.10–290.50 vs. 554.20 ± 186.20, 95% CI: 323.00–785.40; Figure 6B), and final tumor weight was lower (0.25 ± 0.07 g, 95% CI: 0.17–0.34 vs. 0.54 ± 0.13 g, 95% CI: 0.38–0.69; Figure 6C). Furthermore, to elucidate the underlying molecular mechanisms, our subsequent immunohistochemistry and immunofluorescence assay of the xenograft tissues indicated that the elevation of ULK2 resulted in a significant increase in the phosphorylation of c-JUN at Ser243 and a marked decrease in the c-Jun protein level (Figure 6D–H). This molecular evidence strongly corroborated our in vitro findings. Thus, the aggregated data validated the involvement of ULK2 in enhancing cisplatin sensitivity in ovarian cancer by regulation c-Jun. This preclinical evidence establishes a foundation for further investigation of ULK2 as a promising therapeutic candidate.

ULK2 overexpression potentiated cisplatin sensitivity in ovarian cancer in xenograft models. (A) Images of subcutaneous xenograft tumor models established in nude mice. (B) The graph displayed the tumor growth progression observed in the nude mouse subjects. (C) Evaluation of tumor weights was conducted. **p < .01. (D) Immunohistochemistry was performed to assess the association of ULK2, c-Jun and its phosphorylation at Ser243 site. (E) Immunofluorescence analysis was executed to confirm the interaction between ULK2 and p-c-Jun (Ser243) within the subcutaneous xenograft tumors. (F) Quantification of p-c-Jun (Ser243) immunofluorescence signal was performed using IPP 6.0 software. (G) Association between ULK2 and c-Jun was tested by immunofluorescence in the mice models. (H) The immunofluorescence intensity of c-Jun was quantified in ovarian cancer organoids using IPP 6.0 software.
Discussion
Our research demonstrated that ULK2 deficiency was linked to chemoresistant ovarian cancer, and correlated with poor prognosis of the patients. Overexpression of ULK2 led to a reduction in glycolysis and chemoresistance in organoids derived from patients. Furthermore, ULK2 triggers c-Jun degradation by inducing its Ser243 phosphorylation and subsequent ubiquitination. Thus, we inferred that ULK2 might suppresses glycolysis to attenuate cisplatin resistance in ovarian Cancer via c-Jun phosphorylation degradation. These findings emphasized the potential of ULK2 as a therapeutic agent to address platinum resistance in ovarian cancer.
ULK1 and ULK2 are serine/threonine protein kinases, serve as crucial regulators of autophagy initiation. 26 However, emerging studies indicates that ULK1 and ULK2, despite their similarity, can have non-overlapping functions and are differentially regulated in ovarian cancer. For instance, ULK1 is highly expressed in ovarian tumors, and its inhibition has been shown to reduce cell viability in high-grade serous ovarian cancer spheroids. 27 In contrast, ULK2 expression is frequently downregulated in ovarian cancer, 28 suggesting distinct pathogenic roles for these two kinases in the disease. Nevertheless, the functional significance and molecular mechanism of ULK2 in modulating chemosensitivity in ovarian cancer remain unclear.
Given that ULK2 functions as a serine/threonine protein kinase, 29 we hypothesized its involvement in crucial cellular processes via the catalysis of phosphate transfer from ATP to specific substrates. To confirm this, we employed phosphoproteomics to identify alterations in protein phosphorylation profiles and perform associated functional analyses upon ULK2 overexpression. Our findings revealed a significant impact of ULK2 on metabolic processes. As reported by Xi Long et al., 30 metabolic process was significantly enriched in ovarian cancer. Moreover, ULK2 has been shown to be positively associated with IGFBP3, which has been implicated in reducing aerobic glycolysis in ovarian cancer cells by inhibiting the PI3K/AKT pathway. 31 Thus, we suggested that ULK2 may have a potential function in the regulation of glucose metabolism, although its precise functions and regulatory mechanisms remain insufficiently explored. In this study, we confirmed that glycolysis was downregulated following ULK2 overexpression. To mechanistically link ULK2 to glycolytic regulation, we analyzed the protein levels of glycolytic regulators (including GLUT1, HK2, PFKFB3, LDHA and MCT4) following ULK2 overexpression. Notably, ULK2 upregulation led to a marked decrease in the expression of PFKFB3, LDHA and MCT4. This downregulation of critical glycolytic regulators provided a plausible molecular explanation for the observed suppression of glycolysis. The Warburg effect represents a distinctive metabolic reprogramming in cancer cells and is marked by an elevated rate of glycolysis even when oxygen is available. 32 Glycolysis has been associated with tumorigenesis, 33 tumor progression 34 and chemosensitivity.35,36 For example, glycolysis contributes to gastric cancer malignant progression and oxaliplatin resistance. 37 This study revealed that ULK2 attenuated glycolysis, and notably, the glycolysis inhibitor 2-DG subsequently reversed chemoresistance in ovarian cancer organoids. Thus, ULK2 might sensitize ovarian cancer organoids to chemotherapy via inhibition of glycolysis.
Strikingly, our study revealed a marked elevation in the phosphorylation of the key oncogenic protein c-Jun at Ser243 after ULK2 elevation, as identified by phosphorylation mass spectrometry, which was further confirmed by Co-IP and in vitro kinase assay. Building on prior reports that c-Jun phosphorylation at Ser243 promoted its degradation,25,38 we validated our finding by western blotting, which confirmed a reduction in c-Jun protein levels following forced overexpression of ULK2. The c-Jun transcription factor, a central component of the Activator Protein-1 (AP-1) signaling complex, demonstrates oncogenic duality in promoting malignant progression and chemo-therapeutic drug resistance.39–41 For ovarian cancer, it has been established that DGKA confers chemoresistance to platinum in ovarian cancer by stimulating the c-JUN-WEE1 signaling cascade. 42 Our experiments also demonstrated that this reduction in c-Jun significantly diminished the viability of ovarian cancer organoid cells. Collectively, the evidence suggested that ULK2 overexpression may attenuate platinum chemotherapy resistance in ovarian cancer by modulating c-Jun ubiquitin degradation, which was induced by c-Jun phosphorylation at Ser243 site. While an increasing number of studies have highlighted a significant influence of ULK2 on tumor development and apoptosis,43–45 its precise role in ovarian cancer therapy remains uncharacterized. Our finding contributes a new theoretical framework for advancing ovarian cancer therapeutic strategy.
A growing body of preclinical data across tumor types positions c-Jun as a metabolic regulator, though the interplay between c-Jun and tumor glycolysis is both intricate and significant, with c-JUN likely playing varied regulatory functions across different cancer types. For example, c-Jun has been shown to transcriptionally modulate Notch1 in triple-negative breast cancer, 46 which has subsequently been implicated in glycolytic processes in numerous tumor investigations.47,48 Moreover, c-Jun itself can be influenced by pivotal glycolytic enzymes. Notably, PKM2 has been reported to engage with ERK1/2, facilitating its phosphorylation, which subsequently leads to the phosphorylation of the transcription factor c-Jun at ser63 and ser73, thus promoting the metastatic behavior of prostate cancer. 49 Consequently, although several investigations have identified a correlation between c-Jun and glycolysis, a conclusive determination remains elusive. Our results indicated that ULK2 promoted the ubiquitin-mediated degradation of c-Jun via phosphorylation at Ser243; c-Jun led to a marked enhancement in glycolytic activity in the ovarian cancer organoid. Additionally, our study functionally linked glycolysis to cisplatin resistance. Treatment of ovarian cancer organoids with glycolysis inhibitor 2-DG significantly sensitized them to cisplatin-based chemotherapy, establishing a central role for glycolysis in maintaining the chemoresistance. Therefore, our observation implied that ULK2-mediated regulation of c-Jun, along with its subsequent effect on glycolysis, could represent a critical mechanism contributing to chemotherapy resistance in ovarian cancer.
In this study, preliminary phosphoproteomic profiling was performed using only one paired sample set (ULK2 OE vs. control). This approach was adopted to screen candidate differentially phosphorylated proteins for subsequent in-depth molecular investigation, with the primary focus placed on further validation. We acknowledge that the absence of biological and technical replicates represents a methodological limitation. Nevertheless, the key findings were further corroborated through complementary experimental approaches, including Western blotting, co-immunoprecipitation, and in vitro kinase assays.
The in vivo function of ULK2 overexpression was assessed using a subcutaneous xenograft model. While this model has limitations in recapitulating the immune microenvironment, it successfully demonstrated that ULK2 overexpression enhances platinum chemosensitivity in ovarian cancer. This finding was also supported by patient-derived organoid models. Future studies will employ orthotopic transplantation models to build upon these results.
Conclusion
The findings of the present study revealed a favorable correlation between ULK2 expression and ovarian cancer patient survival. Furthermore, we identified that ULK2 downregulated glycolysis process and chemoresistance in ovarian cancer organoid, accompanied by c-Jun degradation. Thus, our study suggested that activating ULK2 may represent a promising therapeutic avenue for overcoming platinum resistance. As we proceed with the discussion, we will delve into the implications of these findings, exploring the potential for ULK2 as a biomarker and its role in guiding treatment strategies for ovarian cancer patients.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261421695 - Supplemental material for ULK2 suppresses glycolysis to attenuate cisplatin resistance in ovarian cancer organoid via c-Jun phosphorylation
Supplemental material, sj-docx-1-sci-10.1177_00368504261421695 for ULK2 suppresses glycolysis to attenuate cisplatin resistance in ovarian cancer organoid via c-Jun phosphorylation by Wei Chen, Xiaoxi Chen, Linlin Wang, Yaoyu Qu, Jin Zhao and Huizhen Sun in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504261421695 - Supplemental material for ULK2 suppresses glycolysis to attenuate cisplatin resistance in ovarian cancer organoid via c-Jun phosphorylation
Supplemental material, sj-docx-2-sci-10.1177_00368504261421695 for ULK2 suppresses glycolysis to attenuate cisplatin resistance in ovarian cancer organoid via c-Jun phosphorylation by Wei Chen, Xiaoxi Chen, Linlin Wang, Yaoyu Qu, Jin Zhao and Huizhen Sun in Science Progress
Footnotes
Acknowledgements
We gratefully thank the company of International Science Editing for their assistance in language improvement.
Ethical considerations and consent to participate
All research involving human participants in this study was conducted in compliance with the Declaration of Helsinki, and the study protocol had obtained prior approval from the Changning Maternity and Infant Health Hospital (now known as Tongren Hospital, Approval No. CNFBLLKT-2023-013) on19 April 2023. Informed consent was secured from all participants.
Animal ethical statement
All animal research conducted adhered to the ARRIVE 2.0 principles 20 and received approval from the Animal Ethics Committee of Xinhua Hospital (Approval No. XHEC-F-2025-022) on 30 April 2025.
Author contributions
Huizhen Sun, Wei Chen and Yaoyu Qu designed the experiments. Jin Zhao, Linlin Wang and Xiaoxi Chen performed the experiments and analyzed the data. Wei Chen, Jin Zhao, Xiaoxi Chen and Huizhen Sun drafted the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Nature Science Foundation of China (82272641 to Huizhen Sun) and Shanghai Municipal Health Commission (202240292 to W.C).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are available in this paper.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
