Abstract
Objective
To investigate the physical and chemical stability of cefminox sodium, cefuroxime sodium, cefoxitin sodium, ceftizoxime sodium, cefmetazole sodium, ceftazidime and cefamandole nafate when continuously infused with 0.9% sodium chloride (NS) and 5% glucose (GS).
Methods
Seven cephalosporin antibiotics were reconstituted in 50 and 100 mL of NS and GS, and stored at room temperature. Samples were collected at 0, 2, 4, 8, and 24 hours to evaluate changes in appearance, pH, insoluble particle count, and drug content.
Results
The admixture solutions of cefuroxime with both solvents turned yellow at 8 hours. The relative percentage content of the low-concentration group in NS dropped to 82.97% at 24 hours. When cefamandole nafate was admixed with NS, contents of both high and low concentrations dropped below 90% at 8 hours and below 60% at 24 hours. The pH change of the high-concentration group exceeded 1.0 pH unit at 24 hours. For cefoxitin sodium, the pH change of all concentration groups admixed with both solvents exceeded 1.0 pH unit at 24 hours. The high-concentration group in NS declined to 87.72%. The admixture solutions of cefminox with both solvents showed slight discoloration at 24 hours. When cefminox was admixed with GS, the contents of both concentration groups dropped below 90% at 2 hours. All other drug–solvent–concentration combinations had all indicators within the specified limits.
Conclusion
When admixed with NS, cefmetazole, ceftazidime, and ceftizoxime are stable for 24 hours continuous infusion; cefoxitin and cefminox for up to 8 hours; and cefuroxime and cefamandole nafate for 4 hours. When admixed with GS, cefmetazole, ceftazidime, and ceftizoxime are stable for 24 hours; cefoxitin for up to 8 hours; cefuroxime for 4 hours; and cefminox has a stability period of less than 2 hours, so GS is not recommended as it is continuous infusion solvent.
Introduction
Antimicrobial resistance (AMR) has emerged as a significant global health threat, as infections caused by multidrug-resistant pathogens markedly increase morbidity and mortality.1,2 Among the key therapeutic agents against bacterial infections, cephalosporins are commonly used β-lactam antibiotics, which rely on the duration of free drug concentrations above the minimum inhibitory concentration (T > MIC) for optimal efficacy. 3 However, the rising prevalence of AMR and varying pharmacokinetics among patients have led to increased MICs, necessitating the optimization of pharmacokinetic and pharmacodynamic parameters to improve therapeutic outcomes.4,5 Furthermore, the risk of incompatibility between cephalosporins and co-administered drugs or solvents poses additional safety concerns. For instance, ceftriaxone can cause severe adverse reactions when admixed with certain agents, leading to potentially fatal outcomes. 6
Traditionally, cephalosporins are administered as short-term intravenous infusions. While some cephalosporins exhibit acceptable stability, they are generally considered prone to degradation and thus need to be prepared and used immediately, resulting in drug wastage and an increased risk of microbial contamination during preparation in hospital settings. To enhance bactericidal efficacy and achieve prolonged duration of T > MIC, extended infusion strategies (prolonged or continuous infusions) have been proposed. These methods extend drug exposure and help maintain steady-state serum concentrations, thereby optimizing therapeutic efficacy.7–10 Current international guidelines recommend the use of extended β-lactam infusions for patients with septic shock or hospital-acquired infections.11,12 Although meta-analyses have demonstrated the benefits of extended infusions in both critically and noncritically ill patients, their practical application remains limited due to insufficient stability data and the absence of validated maximum infusion durations, which further increases the risk of medication use.13,14
Consequently, rigorous compatibility and stability assessments are essential. These studies can identify potential incompatibility, prevent patient harm, and provide guarantees for safe medication use. 15 While previous research has evaluated the stability of specific cephalosporins, such as cefamandole nafate, cefoxitin, ceftazidime, and cefuroxime, these studies often lacked comprehensive analysis across different solvents and concentrations.16–19 Therefore, this study systematically investigated the physical and chemical stability of seven commonly used cephalosporins when reconstituted in 0.9% sodium chloride and 5% glucose injections under simulated clinical conditions over a 24-hour period. We aimed to establish evidence-based maximum safe infusion durations for each cephalosporin to mitigate the risks of efficacy loss or toxicity due to unnecessarily prolonged administration. Additionally, this study seeks to optimize resource utilization in clinical practice, providing a scientific foundation for the effective implementation of extended infusion strategies.
Instruments and reagents
Instruments
The samples were analyzed using an Acquity UPLC H-Class system (Waters Corporation, USA) combined with an FE20 laboratory pH meter (Mettler-Toledo, Switzerland), a GWF-8JD particle analyzer (Tianjin Tianhe Analytical Instruments Co., Ltd, China), and an AB204-S standard electronic analytical balance (Mettler-Toledo, Switzerland).
Drugs and reagents
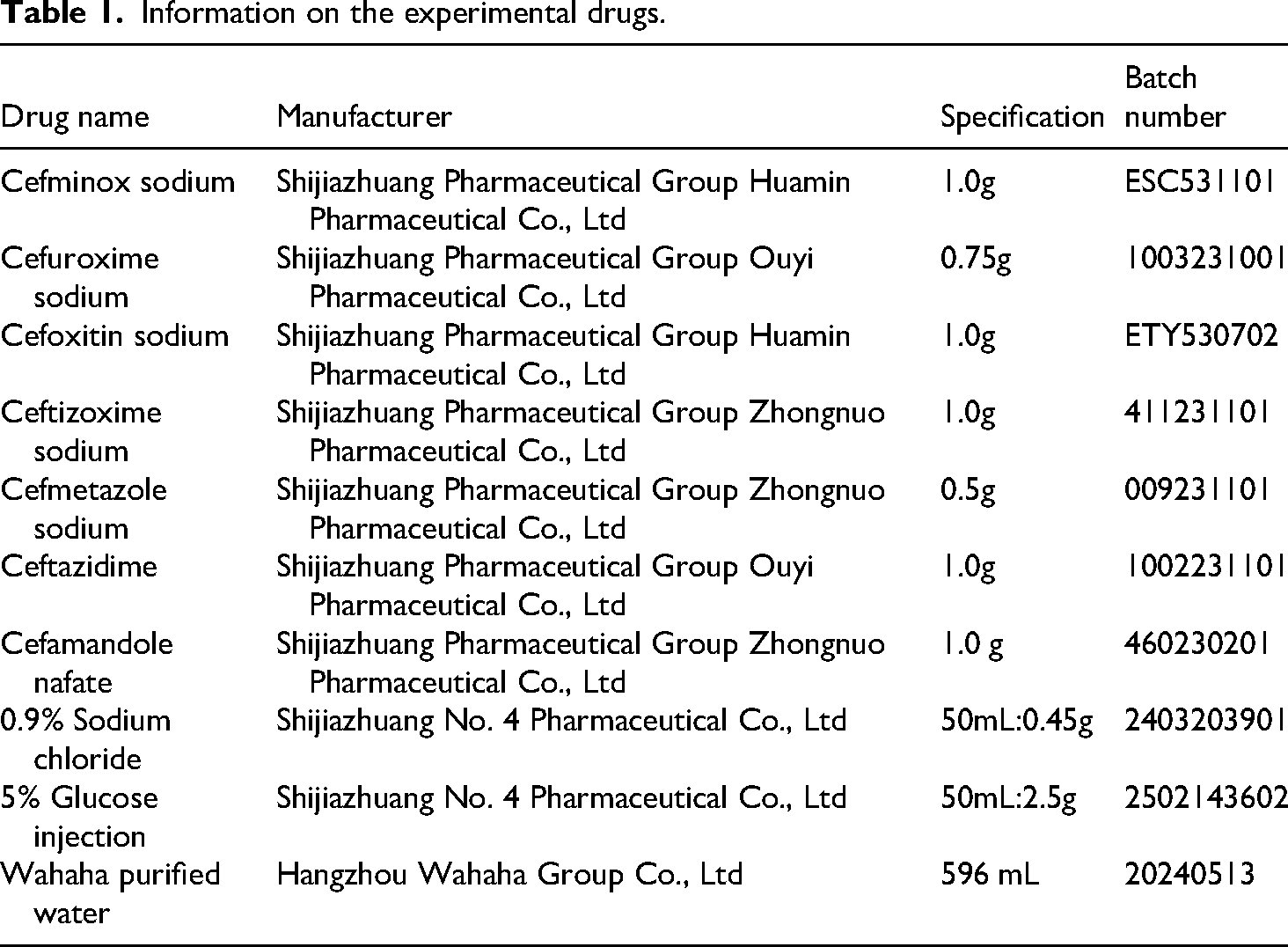
The information of the drugs used in the experiment is shown in Table 1. The seven cephalosporin reference standards were purchased from the National Institutes for Food and Drug Control, China, and their batch numbers and contents are listed in Table 2. The water used in the experiments was Wahaha purified water. The chromatographic solvents, methanol and acetonitrile, were purchased from Thermo Fisher Scientific (China) Co., Ltd.
Information on the experimental drugs.
Information on the seven cephalosporin reference standards.

The package insert states that cefminox sodium should be reconstituted immediately before use and the reconstituted solution should be used as soon as possible. For ceftazidime, intravenous injections diluted with normal saline, 5% glucose injection, or sodium lactate should not be stored at room temperature for more than 24 hours. After reconstitution, ceftizoxime sodium solution should not be left at room temperature for more than 7 hours, nor stored in a refrigerator for more than 48 hours. No information regarding the in-use period of the reconstituted drug is provided in the package inserts of cefmetazole sodium, cefuroxime sodium, cefamandole nafate, and cefoxitin sodium.
Methods
Chromatographic conditions
The chromatographic column Titank C18 (150mm × 2.1 mm; 3μm) was provided by Phenomenex (Guangzhou Philomon Scientific Instrument Co., Ltd). The flow rate was set at 0.5 mL/min. The analytes were detected photometrically at 254 nm. The column temperature was maintained at 35°C. The injection volume was 10 μL. Isocratic elusion was maintained with mobile phase compositions adjusted as follows: 76% water–24% acetonitrile for cefamandole nafate and 87% water–13% acetonitrile for the remaining cephalosporins. The study was conducted inaccordance with the Declaration of Helsinki (1975), as revised in 2024.
Solution preparation
Preparation of reference standard solutions
The reference substances of cefminox sodium, cefuroxime sodium, cefoxitin sodium, ceftizoxime sodium, cefmetazole sodium, ceftazidime, and cefamandole sodium were accurately weighed. Each weighed substance was transferred into separate 100 mL volumetric flasks, dissolved in methanol (except cefotetan sodium which was dissolved in phosphate buffer). The solutions were diluted to the mark with water, admixed thoroughly, and the reference stock solutions were obtained with the following concentrations: cefminox sodium 1.035 g/mL, cefuroxime sodium 1.117 mg/mL, cefotetan sodium 0.495 mg/mL, ceftizoxime sodium 1.050 g/mL, cefmetazole sodium 1.0175 mg/mL, ceftazidime 1.0125 mg/mL, and cefamandole nafate 1.090 g/mL.
Preparation of test solutions
This study selected two different concentrations of seven drugs in two solvents to prepare test solutions, referencing common clinical usage dosages and the practical application of multichannel micropumps in clinical settings. Groups A and C were prepared as high-concentration solutions mimicking intensive care unit (ICU) environments, while groups B and D investigated the stability of conventional clinical treatment concentrations. For example, two vials of cefminox sodium for injection were respectively added to 50 and 100 mL of 0.9% sodium chloride injection (non-polyvinyl chloride [PVC] infusion bags), and admixed uniformly to obtain cefminox sodium test solutions A and B at concentrations of 20 and 10 mg/mL. Using the same method, test solutions C and D of cefminox sodium compatible with 5% glucose injection were prepared. Test solutions of the remaining six drugs were configured using the same procedure. Details on drug specifications, solvent volumes, and test solution concentrations are provided in Table 3.
Preparation of test solutions.

GS: 5% glucose injection; NS: 0.9% sodium chloride injection.
Preparation of negative reference solution
The 0.9% sodium chloride injection and 5% glucose injection from the same batch was used as the negative reference solution.
Methodological validation
Since the high performance liquid chromatography (HPLC) detection method used in this study was newly established by our team. The methodology validation was conducted in accordance with the Guiding Principles for Validation of Analytical Methods (General Rule 9101) in the 2020 edition of the Chinese Pharmacopoeia. 20
System suitability test
The reference, test, and negative control solutions prepared as described in section “Solution preparation” were injected under the chromatographic conditions detailed in section “Chromatographic conditions.”
Linearity range and sensitivity
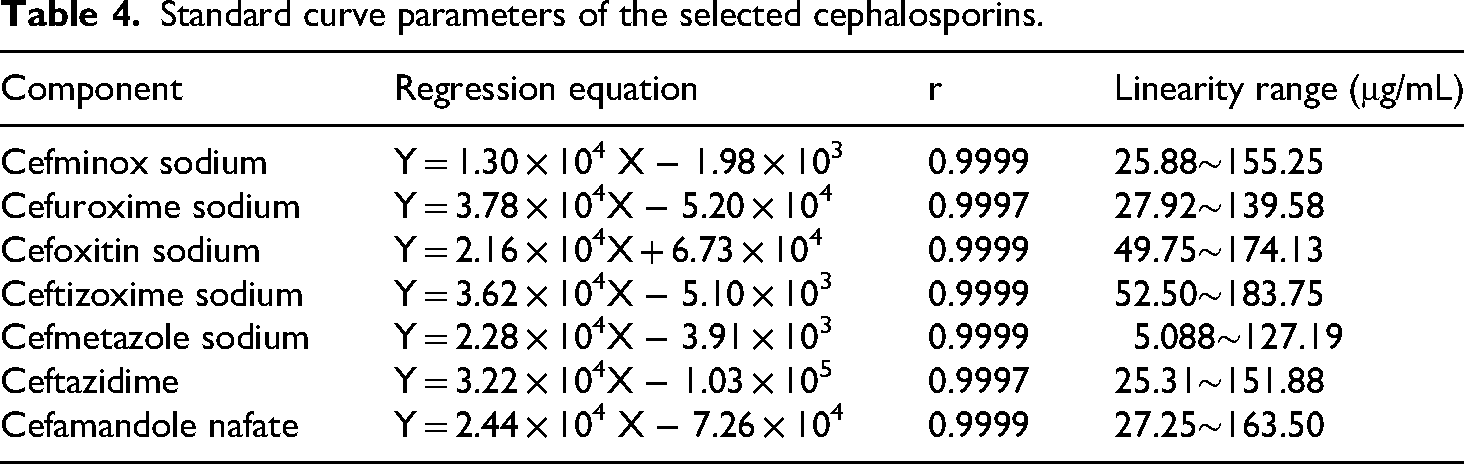
Appropriate amounts of each reference solution were accurately transferred and diluted with purified water to prepare a series of linear working solutions with different concentration gradients. The concentrations of the reference solutions for each antibiotic were as follows: cefminox sodium (25.88, 51.76, 77.64, 129.40, 155.25 μg/mL), cefuroxime sodium (27.92, 55.84, 83.76, 111.68, 139.58 μg/mL), cefoxitin sodium (49.75, 74.625, 99.50, 124.375, 174.13 μg/mL), ceftizoxime sodium (52.50, 78.75, 105.00, 131.25, 183.75 μg/mL), cefmetazole sodium (27.92, 55.84, 83.76, 111.68, 139.58 μg/mL), ceftazidime (25.31, 50.62, 75.93, 126.55, 151.88 μg/mL), and cefamandole nafate (27.25, 54.5, 81.75, 136.25, 163.50 μg/mL). The solutions were injected in order of increasing concentration under the chromatographic conditions described in section “Chromatographic conditions.” The chromatographic peak areas (Y-axis) and concentrations (X-axis) were recorded, and linear regression analysis was performed to establish the standard curve equations for each component.
Standard curve parameters of the selected cephalosporins.
Instrument precision test
The reference solutions and admixed reference solutions were accurately injected six times consecutively, and the relative standard deviation (RSD) values of the peak areas for the seven components were calculated.
Reproducibility test
For each drug from the same batch number, seven test solutions were prepared according to the method described in section “Preparation of test solutions.” The samples were injected and analyzed under the chromatographic conditions specified in section “Chromatographic conditions,” and the RSD values of the peak areas for each component were calculated.
Recovery test
Six aliquots of the test solutions were accurately measured, diluted, and spiked with the reference solution to prepare spiked recovery samples. The samples were analyzed under the chromatographic conditions described in section “Chromatographic conditions.” Peak areas were recorded, and recovery rates were calculated.
Stability test
Seven test solutions at two concentrations in two solvents were prepared in triplicate according to the method described in section “Preparation of test solutions.” After preparation, the solutions were stored at room temperature, and their appearance, pH, insoluble particle count and relative percentage content were evaluated at 0, 2, 4, 8, and 24 hours. Before drug preparation, each drug was stored in accordance with the storage conditions specified in its package insert.
Criteria for determining drug compatibility (Figure 1): If the results of physical and chemical stability are different, they shall be reported separately.20–24
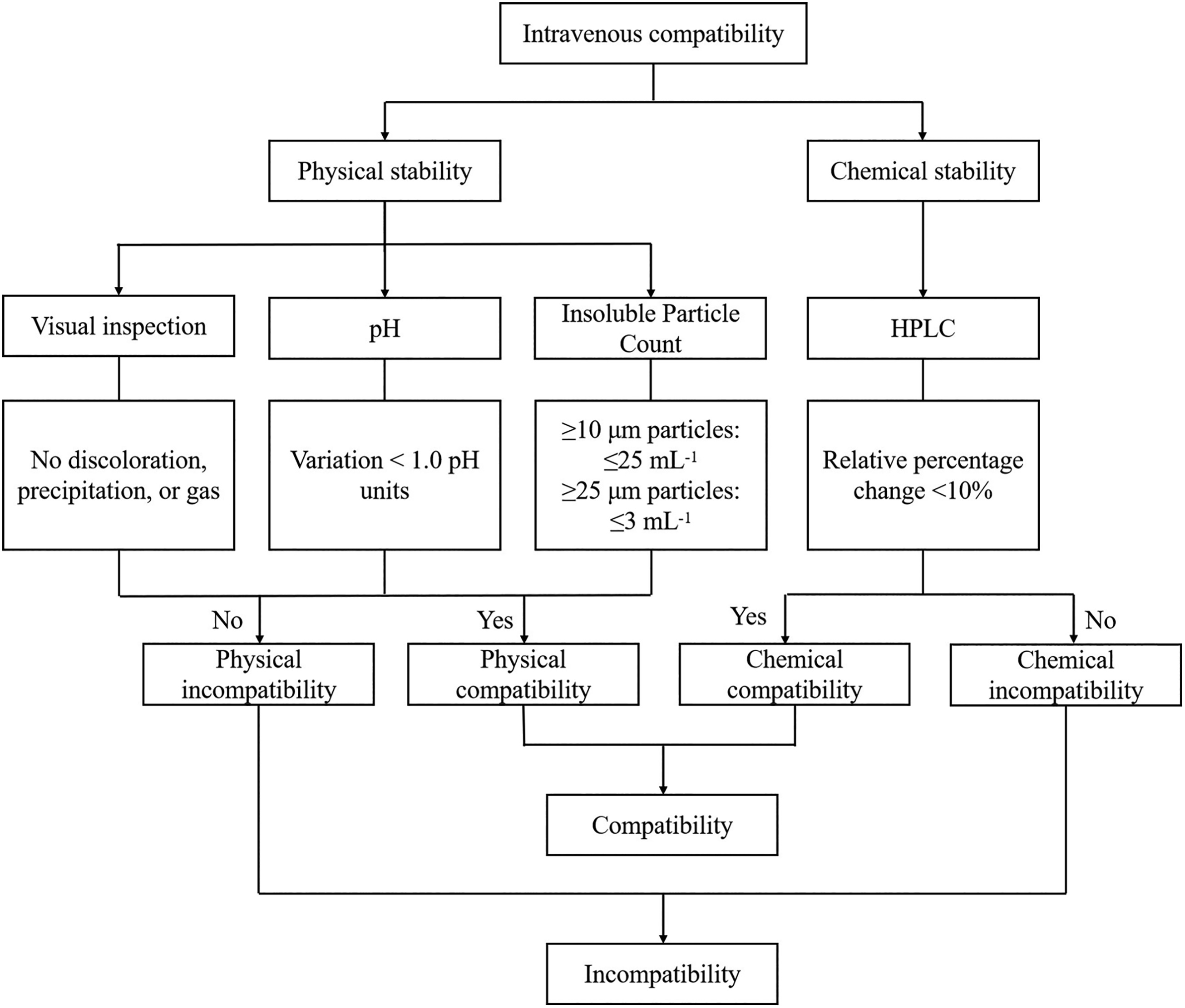
Criteria for determining intravenous drug compatibility.
Appearance and pH changes
The visual inspection method was conducted in accordance with the General Requirements of the Fourth Part of the Chinese Pharmacopoeia (2020 edition): clarity of solution test and visible foreign matter tests under the characterization methods. 20 The appearance of the test solutions was visually assessed by unaided eye visual observation at 0, 2, 4, 8, and 24 hours at room temperature, and the pH of each solution was measured. The acceptable range for pH was set as a change of less than 1 pH unit compared with the measured value at 0 hours. The method for pH determination was as follows: at each time point, 3 mL of the test solution was taken from the infusion bag and placed in a PVC tube, followed by pH measurement. The pH value can serve as an auxiliary reference index for stability evaluation. If the pH change exceeds this range, it may indicate drug degradation in the solution, changes in the properties of excipients, or potential reactions, requiring further investigation of other key indicators such as solution clarity and drug content.
Insoluble particle count
The change in particle count can reflect the stability status of the drug. Increased particles often indicate decreased drug stability, which may be accompanied by the formation of degradation products or changes in system compatibility. Meanwhile, particles may induce adverse reactions such as vascular embolism and inflammatory reactions. The insoluble particle values of the seven cephalosporin drugs were measured using an insoluble particle analyzer based on the light obscuration method. Glass beakers were rinsed with pure water immediately before use. The test solution was slowly added to the beaker and allowed to stand until the solution was stable and free of bubbles, after which detection was performed. The insoluble particle inspection method followed the 2020 edition of the Chinese Pharmacopoeia for the inspection of insoluble particles in drugs, the number of insoluble particles with a diameter of 10 μm or larger in intravenous injections with a labeled volume of 100 mL or more should not exceed 25, and the number of particles with a diameter of 25 μm or larger should not exceed 3. 20
Relative percentage content changes
At the specified time points, after the test solutions were withdrawn, they were first diluted with water. The low-concentration group was diluted 100-fold, and the high-concentration group was diluted 200-fold. The test solutions were withdrawn and injected under the chromatographic conditions described in section “Chromatographic conditions.” The chromatographic peak areas at 0 hours were set as 100% reference were recorded, and the relative percentage content at each subsequent time point was calculated.
Results
Methodological validation
System suitability test
The resulting chromatograms are presented in Figure 2. The negative control solution exhibited no interference with the sample signals. The method demonstrated satisfactory system suitability.
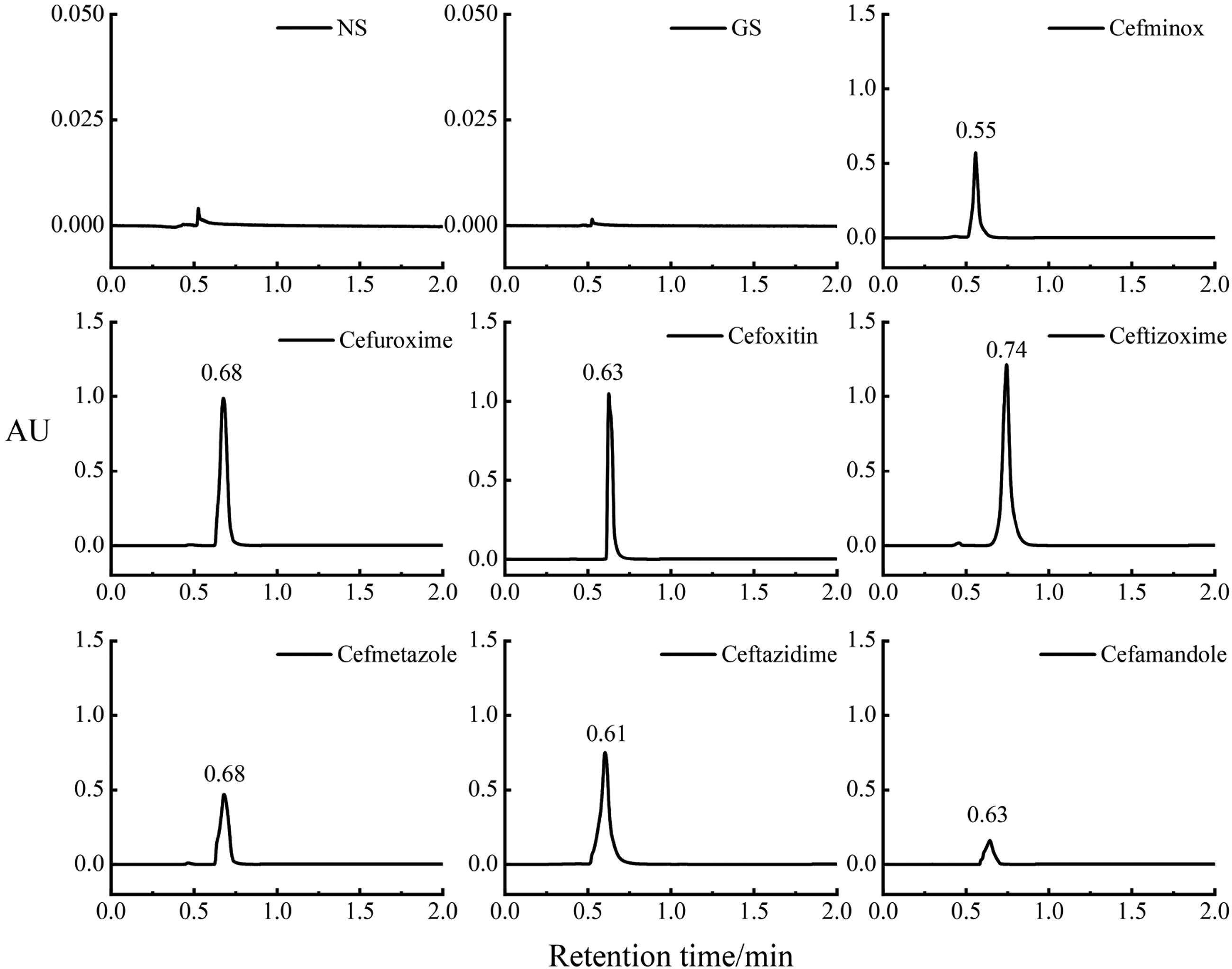
Chromatograms of the seven cephalosporin antibiotic reference standards and the negative control solution. GS: 5% glucose injection; NS: 0.9% sodium chloride injection.
Linearity range and sensitivity
The regression equations for the seven cephalosporin antibiotics were listed in Table 4, with good linearity demonstrated within their respective concentration ranges.
Instrument precision test
The RSD values for the peak areas of seven cephalosporin antibiotics were determined to be within 0.09% to 0.37%, demonstrating satisfactory instrument precision, with detailed data provided in Table 5.
Results of precision, repeatability, and recovery (n = 6).

RSD: relative standard deviation.
Reproducibility test
The RSD values for the peak areas of the seven test solutions ranged from 0.42% to 2.43%, indicating good method repeatability. The detailed data were provided in Table 5.
Recovery test
Recoveries of the seven drugs were achieved within 98.64% to 101.25%, demonstrating satisfactory method accuracy, with detailed results systematically summarized in Table 5.
Stability test
Two concentrations of seven test solutions were prepared in triplicate according to the method described in section “Preparation of test solutions.” The solutions were stored at room temperature, and their appearance, pH, insoluble particle count, and relative percentage content were evaluated at 0, 2, 4, 8, and 24 hours.
Appearance and pH changes
All seven test solutions remained clear and transparent without visible precipitation for 24 hours at room temperature. When admixed with 0.9% sodium chloride and 5% glucose, the low-concentration solution of cefuroxime sodium showed slight yellowing after 24 hours, while the high-concentration solution exhibited obvious yellowing after 8 hours. For cefminox sodium, the low-concentration solution showed no obvious color change, whereas the high-concentration solution developed slight yellowing after 24 hours. When admixed with 5% glucose injection, the color changes of the two drugs over 24 hours are shown in Figure 3. The pH changes of the high-concentration group of cefamandole nafate in 0.9% sodium chloride, and cefoxitin sodium in both 0.9% sodium chloride and 5% glucose at 24 hours exceeded 1.0 pH unit which is beyond the specified limit. In contrast, the pH values of cefminox sodium, cefuroxime sodium, ceftizoxime sodium, cefmetazole sodium, and ceftazidime sodium remained within the specified limits. The results are shown in Figure 4.

The changes in appearance of two cephalosporin antibiotics in 5% glucose injection over 24 hours (a. cefuroxime group C, b. cefuroxime group D, c. cefminox group C, d. cefminox group D).
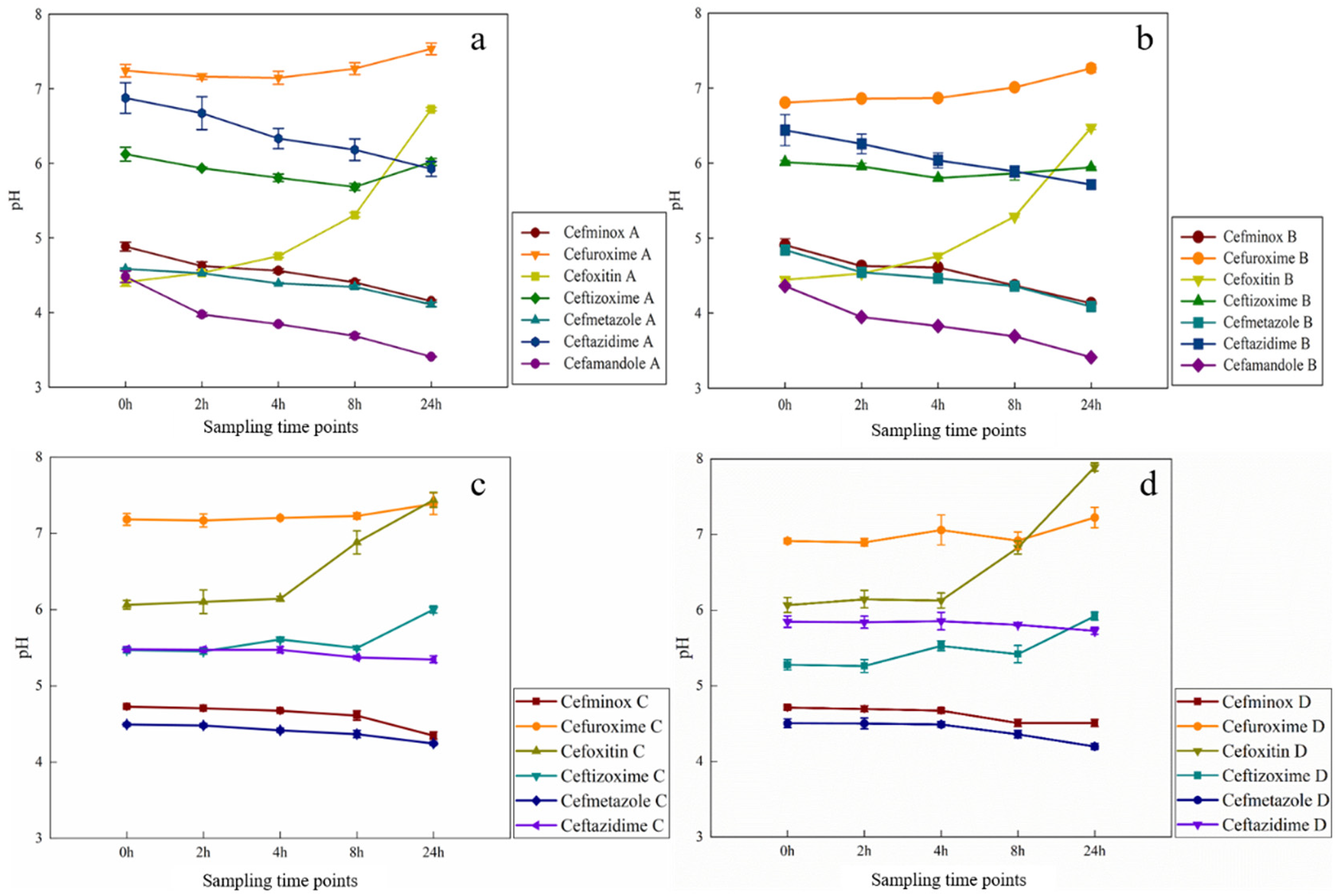
The changes in pH values of seven cephalosporin antibiotics over 24 hours (a. 50 mL NS, b. 100 mL NS, c. 50 mL GS, d. 100 mL GS). GS: 5% glucose injection; NS: 0.9% sodium chloride injection.
Insoluble particle count changes
The results demonstrated that the number of insoluble particles of all test solutions remained within the specified limits for 24 hours, as illustrated in Figure 5.

The changes in insoluble particles of seven cephalosporin antibiotic over 24 hours (a. ≤ 25µm, b. ≤ 10µm).
Relative percentage content changes
The results demonstrated that the content of cefamandole nafate in groups A and B decreased by more than 10% after 8 hours and dropped to below 60% at 24 hours. The relative percentage content of cefuroxime sodium in group B dropped to 82.97% at 24 hours. The relative percentage content changes of cefoxitin in group A dropped to 87.72% at 24 hours. For cefminox sodium, the content in group C dropped to 60.67% and in group D to 45.20% at 2 hours, the content in group C dropped to 41.56% and in group D to 24.51% at 24 hours. In contrast, the changes for the other three substances remained within 10%, thereby meeting the specified criteria. The results are illustrated in Figure 6.
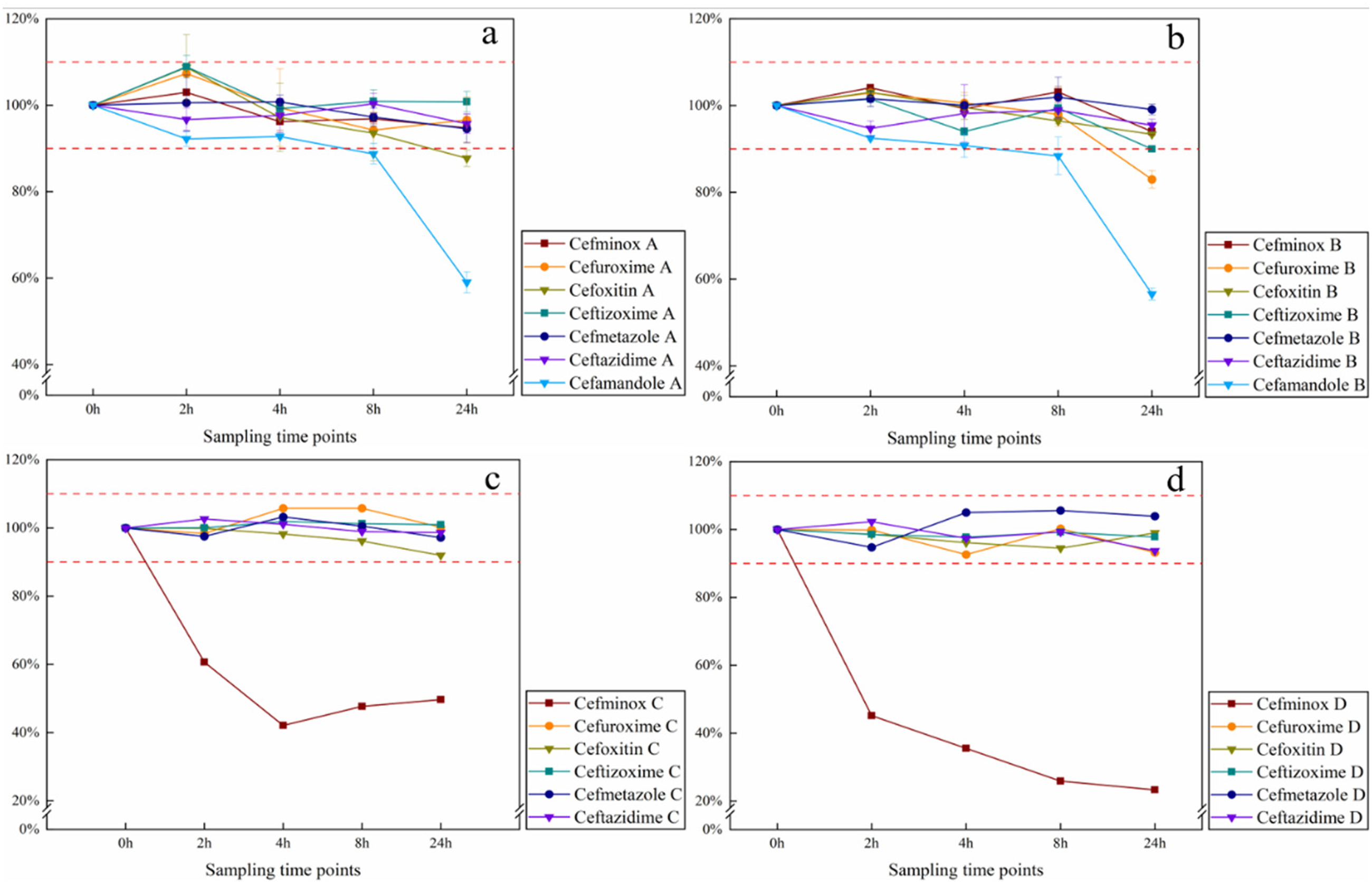
The changes in the content of seven cephalosporin antibiotics over 24 hours (a. 50 mL NS, b. 100 mL NS, c. 50 mL GS, d. 100 mL GS). GS: 5% glucose injection; NS: 0.9% sodium chloride injection.
Discussion
The selection of test concentration and diluent
This study systematically investigated the stability of seven antimicrobial agents in 0.9% sodium chloride injection and 5% glucose injection, with a particular focus on their performance at both standard and elevated concentrations. The selected drug concentrations were clinically relevant, reflecting typical usage in routine treatment as well as in ICU settings, where infusion pumps often necessitate higher drug concentrations. Drugs were prepared by dissolving a full daily dose in 50 mL solution for continuous infusion, which may accelerate drug degradation. These elevated concentrations may significantly influence the physicochemical stability of the agents, potentially leading to increased degradation rates and reduced stability. 25 Such effects are likely attributable to the heightened propensity for aggregation or chemical reactions at higher concentrations, particularly during prolonged infusion periods. By assessing stability across a range of concentrations, this study provides robust and comprehensive data to support extended infusion protocols, thereby ensuring the safety and efficacy of these agents in diverse clinical applications.
Pharmaceutical diluents commonly employed in clinical practice include 0.9% sodium chloride injection, 5% dextrose injection, and dextrose sodium chloride injection. Comparative studies have established that dextrose, as a chemically more complex compound than sodium chloride, exhibits greater propensity to induce drug degradation through chemical interactions. In an experimental investigation of the effects of three diluents on ceftazidime stability under different storage conditions, 0.9% sodium chloride solution exhibited the highest stability under refrigerated or frozen storage. 26 Additionally, glucose solutions were found to promote ceftaroline degradation more significantly than sodium chloride solutions at elevated temperatures.27,28 Although drug monographs (e.g. cefminox sodium) specify that cephalosporins may be reconstituted with 5% to 10% glucose injection or 0.9% sodium chloride injection, sodium chloride solution is more commonly used in clinical practice. Therefore, this study exclusively evaluated the stability of seven cephalosporins when reconstituted with 0.9% sodium chloride injection and 5% glucose injection.
Hydrolysis characteristics and stability
All test solutions remained clear and transparent without visible precipitation at room temperature for 24 hours, consistent with the findings of Jenkins and Das Gupta.16,29 These studies confirmed that most cephalosporin antibiotics exhibit good physical stability under room temperature.
Cephalosporin antibiotics contain a β-lactam ring structure that is prone to hydrolytic ring-opening, with their degradation rate being significantly influenced by pH, temperature, and light exposure. The hydrolytic cleavage may generate inactive degradation products, whose reduced antimicrobial activity combined with potential toxicity increase may compromise clinical efficacy. 30 The structural formulas of the seven cephalosporin antibiotic are shown in Figure 7. HPLC is widely employed for quantifying cephalosporins and monitoring their degradation products. For instance, ceftriaxone sodium injection may undergo (Z/E) isomerization during storage, yielding the toxic E-isomer as a degradation product, necessitating chromatographic monitoring of the toxic isomer content.31,32

Chemical structures of seven cephalosporin antibiotic.
Cefamandole nafate
Significant pH changes beyond the specified limits were observed for cefamandole nafate when reconstituted with 0.9% sodium chloride at room temperature. The pH of cefamandole nafate at 0 hours was 4.48 ± 0.08, and it decreased to 3.41 ± 0.01 at 24 hours, with a total change of 1.07 units. The three parallel samples showed a highly consistent trend in pH change (RSD < 2%). Das Gupta reported that during storage at room temperature, the pH of cefamandole nafate sodium decreased from 7.0 to 3.8, aligning with the results of this study. 16 This may be related to the chemical structure and hydrolysis characteristics of cefamandole sodium. Specifically, the ester group of cefamandole nafate sodium is prone to hydrolysis in aqueous solution, resulting in the formation of cefamandole and formate ions, which contribute to the observed pH changes. Such pH variations may compromise the stability and biological activity of these drugs.
Furthermore, the content of cefamandole nafate decreased to approximately 60% within 24 hours at both concentrations. As shown in Figure 8, the chromatographic peak response at 0.70 minutes gradually increased over time, indicating that this peak corresponds to the hydrolysis product of cefamandole nafate. Different degradation products may have different chemical properties, potentially appearing as separate peaks in chromatographic analysis. Faouzi investigated the stability of cefamandole nafate in PVC infusion bags and reported that the content of cefamandole nafate decreased to approximately 41.5% in 0.9% sodium chloride, while the total concentration of cefamandole nafate and its hydrolysis product, cefamandole, remained almost unchanged. 33 These findings further corroborate the stability results of cefamandole nafate observed in this study. However, studies suggest that this degradation is not clinically significant, as cefamandole and cefamandole nafate have equivalent antimicrobial activity, and no toxicological differences have been observed between them. 33 Nonetheless, special attention is required during the preparation and storage of cefamandole nafate injection solutions to avoid potential clinical risks associated with degradation products.

Changes in the content of cefamandole esters (1. unknown product; 2. cefamandole).
Cefoxitin sodium
Cefoxitin sodium exhibited pH changes exceeding the specified limits in both 0.9% sodium chloride and 5% glucose at room temperature. Consistent with our findings, Das Gupta reported a pH increase from 4.7 to 7.2 in cefoxitin sodium solutions. 29 As a semisynthetic cephamycin antibiotic, cefoxitin sodium contains the critical 7α-methoxy group (-OCH3), which confers enhanced stability against β-lactamases in Gram-negative bacteria. 34 This structural moiety may participate in oxidative/hydrolytic reactions, generating small acidic molecules (e.g. formic acid). Moreover, the thioether group can be oxidized to sulfonic acid derivatives under oxidative conditions, thereby increasing solution acidity and contributing to pH reduction. The molecular structure is illustrated in Figure 7. This pH alteration may compromise pharmaceutical stability and bioactivity, necessitating stringent storage condition controls for cefoxitin sodium in clinical applications.
Cefuroxime sodium
When reconstituted with 0.9% sodium chloride in the experiment, the low-concentration group of cefuroxime sodium showed a relative percentage content decrease exceeding 10% within 24 hours, with pH values ranging from 6.81 to 7.26 during this period. In contrast, the high-concentration group exhibited less than 10% variation in relative content, accompanied by pH values between 7.15 and 7.54 over 24 hours. The hydrolytic degradation of cefuroxime sodium's β-lactam ring demonstrates pH-, temperature-, and diluent-dependent stability characteristics. Previous studies have documented accelerated degradation rates in 0.9% sodium chloride solution under elevated temperatures, while refrigeration effectively prolongs drug activity retention. Gupta VD's experiments demonstrated optimal chemical stability of cefuroxime in reconstituted solutions at near-neutral pH (6.0–7.5), beyond which hydrolytic reactions were significantly accelerated. 35 These findings suggest concentration-dependent stability behavior in cefuroxime sodium solutions.
Cefminox sodium
When cefminox sodium was admixed with 5% glucose injection, the relative percentage content in the high-concentration group dropped to 41.56%, and that in the low-concentration group dropped to 24.51%. The 5% glucose injection is typically a weakly acidic environment, in which the β-lactam ring of cefminox sodium is prone to hydrolytic ring-opening reactions, leading to drug structure destruction. Xu successfully isolated and identified 13 impurities and degradation products of cefminox sodium. 30 After cefminox sodium loses the 5-mercapto-1-methyltetrazole group, its β-lactam ring further undergoes hydrolysis to form a degradation product with a five-membered lactone ring. This is consistent with the observation in this study that the weakly acidic 5% glucose injection promotes the ring-opening of the β-lactam ring. The methoxy group (-OCH3) at the C-7 position increases the steric hindrance of the β-lactam ring, but it also makes the ring more susceptible to intramolecular nucleophilic attack in an acidic environment, resulting in ring rupture. The D-cysteine at the end of the side chain contains free amino and carboxyl groups, which can form zwitterions in an acidic environment, promoting intramolecular cyclization or polymerization reactions to generate inactive polymers. Although Xu did not directly observe the cyclization products mediated by D-cysteine, they obtained relevant results through UV data and other methods. 30 They found that changes in the side chain structure of cefminox sodium lead to significant alterations in the UV absorption characteristics of impurities. This suggests that reactions of the side chain groups directly affect the conjugated system and activity of the drug molecule. Additionally, the dimeric impurities identified in their study also indirectly indicate that cefminox sodium has a tendency to polymerize. This finding further supporting the speculation in this study that side chain-mediated polymerization reactions occur. It is speculated that the combination of the 7α-methoxy group and the D-cysteine side chain in cefminox makes it face dual degradation pathways of β-lactam ring hydrolysis and side chain cyclization in an acidic environment, leading to significant degradation within 24 hours when compatible with 5% glucose injection.
Study limitations
This study was an in vitro compatibility and stability study that only simulated the physicochemical changes between antimicrobial agents and solvents. It did not include the effects of in vivo physiological environments (e.g. blood pH, enzyme systems, protein binding rate) on drug stability and efficacy. This creates limitations when extrapolating conclusions to clinical use. Subsequent in vivo experiments are needed for verification. Study did not conduct control experiments on stability under 2°C to 8°C refrigeration. Thus, it cannot clarify how refrigeration affects strict storage conditions. Another limitation is that formal stability-indicating studies, such as forced degradation under stress conditions (e.g. acid, alkali, and oxidation), were not conducted. Consequently, the method's ability to separate the parent drug from all potential degradation products was not fully characterized. Additionally, experimental constraints prevented it from investigating the potential impact of microbial contamination on drug stability. Microbial growth can lead to drug degradation and more particles. This, in turn, impacts the accuracy of results and the assessment of medication safety.
Conclusion
This study systematically evaluated the stability of seven β-lactam antibiotics over 24 hours under conditions simulating common drug concentrations in ICU. The results showed that at room temperature, cefmetazole sodium, ceftazidime, and ceftizoxime sodium for injection remained stable within 24 hours in 0.9% sodium chloride; cefminox sodium and cefoxitin sodium for injection were stable within 8 hours after admixing; cefuroxime sodium and cefamandole nafate for injection were stable within 4 hours after admixing. In 5% glucose, cefmetazole sodium, ceftazidime, and ceftizoxime sodium for injection remained stable within 24 hours; cefoxitin sodium for injection remained stable within 8 hours; cefuroxime sodium for injection remained stable within 4 hours; cefminox sodium was stable for less than 2 hours, so 5% glucose was not recommended as the solvent for its continuous infusion. These findings provide a scientific basis for optimizing the infusion duration of β-lactam antibiotics in clinical settings.
Footnotes
Ethical approval
This work did not require ethical approval from a human subject or animal welfare committee.
Authors’ contribution
ZD is responsible for research design and guidance; LZ, LF, and JA for research design and paper writing; and YW and XG for conducting experiments and organizing data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical Science Research Project of Hebei (grant number 20250307).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data utilized in this study are available upon contact with the corresponding author.
