Abstract
Introduction
Respiratory tract infections are among the most common infectious diseases and can present with symptoms such as fever, cough, expectoration, and dyspnea. 1 These infections not only affect individuals across all age groups but also lead to acute complications. They contribute to significant mortality rates and impose substantial public health and economic burdens. 2 The treatment of respiratory tract infections primarily involves symptomatic management, including rest, antiviral and antibacterial medications, and drugs for cough, phlegm, and fever. Among these, cough and phlegm medications like aminophylline (APL), doxofylline (DXL), ambroxol hydrochloride (AXH), and bromhexine hydrochloride (BXH) (Figure 1) are commonly used to treat acute and chronic respiratory conditions such as acute tracheobronchitis, COVID-19, chronic bronchitis, and chronic obstructive pulmonary disease.3–8

Chemical structure of APL (a), DXL (b), AXH (c), and BXH (d). APL: aminophylline; AXH: ambroxol hydrochloride; BXH: bromhexine hydrochloride; DXL: doxofylline.
APL is used to relieve coughs and asthma symptoms and is frequently used to treat wheezing caused by respiratory tract infections. It also has applications in treating pulmonary hypertension, heart failure, sleep apnea syndrome, fetal distress, and respiratory failure. DXL, a theophylline derivative, has a bronchiectatic effect 10 to 15 times greater than that of AP, although its cardiotonic, anti-inflammatory, and immunomodulatory effects are less pronounced. Additionally, DXL has fewer extrapulmonary adverse reactions compared to APL.4,5 BXH acts as a mucolytic agent, breaking down mucopolysaccharide fibers in sputum to reduce viscosity and facilitate expectoration. 6 AXH, a metabolite of BXH, exhibits a high affinity for lung tissues.7,8
In treating respiratory diseases, combining two or more antitussive and expectorant drugs, either as fixed-dose combinations or separate therapies, can enhance therapeutic efficacy, reduce dosing frequency, and improve patient compliance.9,10 Monitoring the quality and stability of these pharmaceutical products in raw materials, different dosage forms, and finished infusions is necessary to ensure clinical safety. However, the stability data for intravenous infusions of these four antitussive and expectorant drugs are often incomplete or missing in drug package inserts. Although extensive literature exists on the quantitative analysis of the four antitussive and expectorant drugs mentioned,11–22 only a few studies have examined the simultaneous analysis of DXL and AXH or BXH and AXH.11,12 This study aimed to develop a rapid and precise high-performance liquid chromatography (HPLC) method for the simultaneous quantification of APL, DXL, AXH, and BXH. Additionally, the study assessed the stability of these four drugs when mixed with 0.9% sodium chloride injection [0.9% normal saline (NS)] or 5% glucose injection [5% glucose (GS)] over 48 hours using this analytical approach.
Methods
Drugs and reagents
Theophylline, DXL, AXH, and BXH reference standards were purchased from the National Institute for Control of Pharmaceutical and Biological Products (Beijing, China). The purity of Theophylline, DXL, AXH, and BXH were found to be 99.9%, 99.9%, 100.0%, and 99.8%, respectively, according to the manufacturer's certificate. APL injection (2 mL: 0.25 g, lot#2208052, Henan Runhong Pharmaceutical Co., LTD.), DXL injection (20 mL: 0.2 g, lot#2209191, Zhejiang Beisheng Pharmaceutical Co., LTD.), AXH injection (2 mL: 15 mg, lot#22011502, Ruiyang Pharmaceutical Co., LTD.), BXH injection (2 mL: 4 mg, lot#1117220165, Hebei Renhe Yikang Pharmaceutical Co., LTD.), 0.9% Sodium chloride injection (100mL: 0.9 g, lot#22062022, Otsuka Pharmaceutical Co., LTD., China), and 5% glucose injection (100 mL: 5 g, lot# 22012422, Otsuka Pharmaceutical Co., LTD., China) were purchased from Sinopharm Holding Company Limited (Hubei, China). HPLC-grade acetonitrile was purchased from Tianjin Kemiou Chemical Reagent Co., Ltd (Tianjin, China). Potassium dihydrogen phosphate was purchased from the Shanghai Pudong Chemical Reagent Factory (Shanghai, China).
Instruments and chromatographic conditions
The HPLC system (Dionex Ultimate 3000, USA) featured a four-element low-pressure gradient system, column oven, diode array detector, and Chameleon 7 data processing software. Chromatography was performed using an InertSustain C18 column (5 μm, 150 mm × 4.6 mm; Shimadzu Co., LTD, Shanghai, China). The mobile phase consisted of 50 mmol·L−1 potassium dihydrogen phosphate aqueous solution (phase A, pH 4.0) and acetonitrile (phase B), with a flow rate of 0.8 mL·min−1. Table 1 lists the gradient program used for chromatographic separation. The column temperature was maintained at 30 °C, and the injection volume was 20 μL. The maximum absorption wavelengths for APL, DXL, AXH, and BXH were 254, 273, 248, and 245 nm, respectively.
Chromatography buffer gradient.

Preparation of solution
Preparation of standard solutions
The appropriate amounts of APL, DXL, AXH, and BXH were accurately weighed into separate 25 mL bottles. Each was diluted with a mobile phase consisting of phases A and B in a 1 : 4 ratio, and the volume was then adjusted to prepare standard stock solutions of APL, DXL, AXH, and BXH with drug concentrations of 2, 3, 0.9, and 0.8 mg·mL-1, respectively. These solutions were stored in a refrigerator at −20 °C for future use.
Preparation of infusion mixtures
Commercially available ampoules of APL, DXL, AXH, and BXH were diluted with a 0.9% NS or 5% GS infusion to make solutions with final APL concentrations of 1 or 2 mg·mL-1, final DXL concentration of 3 mg·mL-1, final AXH concentrations of 0.15 or 0.3 mg·mL-1, and final BXH concentration of 0.08 mg·mL-1, respectively. They were packaged in 100 or 250 mL polyolefin bags. The final dose and concentration of each drug in the study were chosen according to the drug package inserts, diagnostic and treatment guidelines, and clinical practice guidelines. All infusion mixtures were prepared in triplicate, and each bags were stored for 48 hours at room temperature (25 ± 1 °C) without protection from light.
Preparation specimens
An appropriate volume of the infusion mixtures of the four antitussive and expectorant drugs was transferred to a 10 mL bottle. It was diluted with a mobile phase consisting of phases A and B in a 1 : 4 ratio, and the volume was adjusted as needed. The solution was filtered through a 0.45 μm microporous filter membrane (Jinteng experimental equipment Co., LTD, Tianjing, China), and the resulting filtrate was used as the specimen solution.
Methodology validation
The method was validated for selectivity, linearity, detection limit, quantitation limit, precision, accuracy, stability, and robustness according to the International conference on harmonization of requirements for registration pharmaceuticals for human use (ICH) and the Pharmacopoeia of the People's Republic of China.23,24
Selectivity
The standard solutions of APL, DXL, AXH, and BXH, as well as the specimen solutions and blank control solutions (0.9% NS and 5% GS) were prepared. And 20 μL of each sample were injected according to the chromatographic conditions described above for analysis. The separation between the drugs and their decomposition products was investigated by optimizing the mobile phase composition and column temperature.
Detection limit and quantitation limit
The limits of detection and limits of quantification were determined by diluting the standard solutions of APL, DXL, AXH, and BXH in the blank control solutions to obtain the lowest concentrations that produced signals 3 and 10 times higher than that of the blank sample, respectively.
Linear range
A series of reference solutions containing 0.01 to 0.20 mg·mL−1 APL, 0.003 to 0.06 mg·mL−1 DXL, 0.015 to 0.30 mg·mL−1 AXH, and 0.016 to 0.16 mg·mL−1 BXH were prepared by mixing the standard solutions of the drugs and diluting with mobile phase A and mobile phase B (1 : 4) into different 10 mL bottles using ultrasonic waves. And 20 μL of each sample were injected according to the chromatographic conditions described above. The peak areas of the four antitussive and expectorant drugs were determined, and linear regression was performed with concentration (X) as the horizontal coordinate and peak area (Y) as the vertical coordinate. Linear equations and correlation coefficients (r) were calculated for each drug.
Precision
The standard solutions of the four antitussive and expectorant drugs (APL, DXL, AXH, and BXH at concentrations of 100.0, 30.0, 150.0, and 80.0 μg·mL−1, respectively) were injected six times on the same day. The relative standard deviation (RSD) of the peak areas was calculated to investigate intraday precision. The instrument's day-to-day precision was examined by sampling the same concentration of quality control standard solutions for six consecutive days.
Specimen stability
The stability test for the specimen involved preparing solutions of the four antitussive and expectorant drugs, which were then stored in an automatic injector at room temperature (25 ± 1 °C). The solutions were subsequently injected and analyzed at 0, 1, 2, 4, 6, and 8 hours. The drug content and RSD values were calculated from the peak areas to assess the stability of the specimen solution.
Accuracy
The method accuracy was assessed by determining the recovery of APL, DXL, AXH, and BXH using the standard addition method. Known concentrations of the four reference products were added to the prequantified sample solutions at 3 different concentration levels of 50%, 100%, and 150% of their target concentration, with triplicate determinations. Drug amounts were estimated by measuring the peak areas and fitting these values to the regression equations mentioned earlier. Percentage recoveries were calculated using the following equation:
Robustness
To evaluate the robustness of the developed method, the influence of various factors was investigated. These factors included three chromatographic columns [InertSustain C18 (150 mm × 4.6 mm, 5 μm; Shimadzu Co., LTD, Shanghai, China), Agilent ZORBAX SB-C18 (150 mm × 4.6 mm, 5 μm; Agilent Technology Co. LTD. Beijing, China), and Kromasil C18 (150 mm × 4.6 mm, 5 μm; Akzo Nobel N.V, Amsterdam, the Netherlands)], column temperatures (±1 °C), absorption wavelengths (±2 nm) and flow rates (±0.1 mL·min-1) of the mobile phase. The impact of these variables on the separation and quantification of the four antitussive and expectorant drugs was assessed.
Compatibility stability investigation
The APL, DXL, AXH, and BXH infusion mixtures were exposed to light at room temperature (25 ± 1°C) for 48 hours. Samples were taken at different time points, and changes in color, bubbles, turbidity, and precipitation of the mixed infusion solutions were determined using a clarity detector (YB-2A, Tianjin) with 0.9% NS or 5% GS injection as a blank, according to the standards of the Chinese Pharmacopoeia 2020 edition. The pH value of the infusion mixtures was determined using a pH meter (PHS-3C, Shanghai). An appropriate volume of the compatible liquid was obtained, and the sample was analyzed according to the specimen preparation method and HPLC quantitative analysis method. Each sample was analyzed in triplicate, and the contents of the four antitussive and expectorant drugs at different time points were calculated. The relative percentage content changes of the drugs at different time points were computed, with the content at 0 hours set at 100%. The content results were expressed as mean ± SD, and when the drug content fell below 90%, the drug in the infusion mixtures was considered unstable.
Results
Verification results of analysis method
Under these conditions, the retention times for APL, DXL, AXH, and BXH were 4.2, 6.5, 8.4, and 13.2 min, respectively (Figure 2). The theoretical plate numbers for the chromatographic peaks of each component were higher than 3000, and the degree of separation was >1.5. Table 2 lists the system suitability parameters for APL, DXL, BXH, and AXH using the proposed HPLC method. The results showed that this method has a satisfactory linear range. Precision and accuracy data for both interday and intraday analyses of the four antitussive and expectorant drugs in quality control samples at three levels are shown in Table 3. Both intraday and day-to-day precisions were below 2.1%, indicating that the instrument exhibited good precision. The average recoveries of APL, DXL, AXH, and BXH were 100.4%, 100.3%, 101.2%, and 100.6%, respectively, with RSD values of 1.0%, 0.8%, 0.4%, and 1.9%, indicating an acceptable recovery rate for the method. The RSDS of the peak areas of APL, DXL, BXH, and AXH were 1.4%, 1.0%, 1.6%, and 1.2%, respectively, indicating that the specimen solutions remained stable within 8 hours. The robustness test indicated that small intentional changes in the tested parameters did not affect the stability of the method.

HPLC chromatogram of APL (1), DXL (2), AXH (3), and BXH (4). APL: aminophylline; AXH: ambroxol hydrochloride; BXH: bromhexine hydrochloride; DXL: doxofylline; HPLC: high-performance liquid chromatography.
System suitability parameters for the HPLC-DAD method.
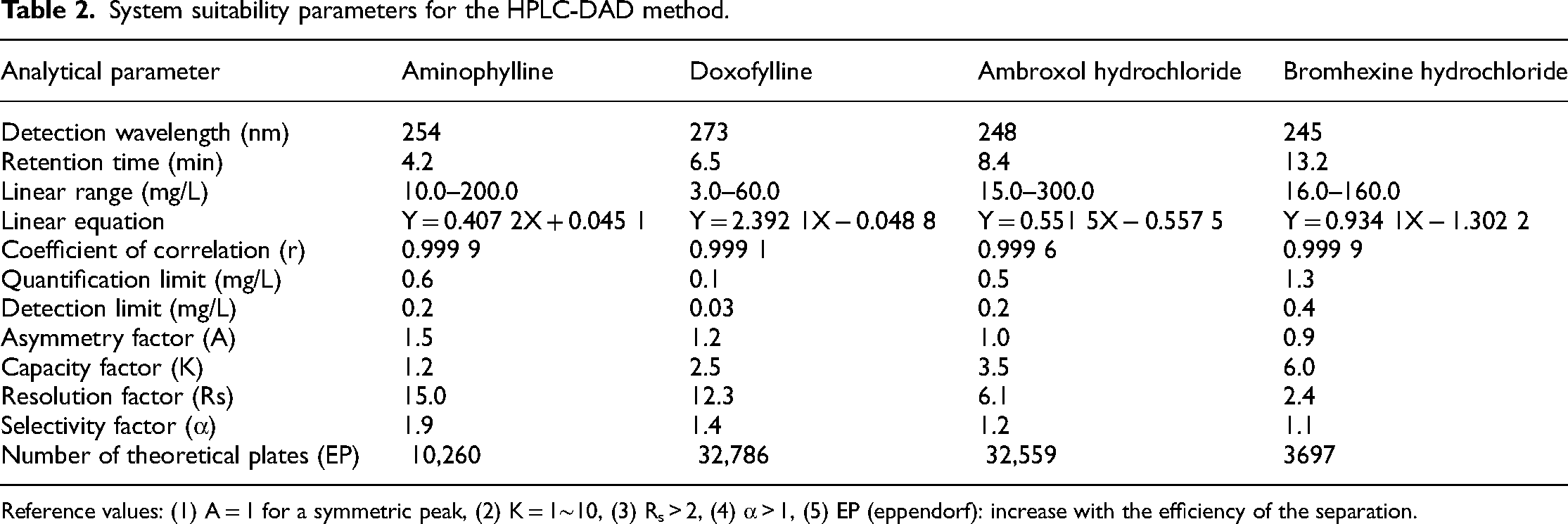
Reference values: (1) A = 1 for a symmetric peak, (2) K = 1∼10, (3) Rs > 2, (4) α > 1, (5) EP (eppendorf): increase with the efficiency of the separation.
Determination results of recovery and precision tests for four the antitussive and expectorant drugs.

RSD: relative standard deviation.
Compatibility stability results
Infusion mixtures of APL, DXL, AXH, and BXH with different prescriptions in 0.9% NS or 5% GS were colorless and clear. No color changes bubbles, turbidity, or precipitation were observed after 48 hours. The average pH values of the APL, DXL, AXH, and BXH mixed solutions in 0.9% NS were 8.9, 6.3, 5.2, and 5.5, respectively. The average pH values of the infusion mixtures in 5% GS were 8.8, 4.8, 5.1, and 4.9, respectively. Under room temperature and light conditions, the pH values of the infusion mixtures fluctuated within ±0.3, indicating that the pH remained relatively stable over 48 h. The contents of the four antitussive and expectorant drugs in the 0.9% NS or 5% GS injections at different time points are shown in Tables 4 and 5, respectively. The tables indicated that the relative percentage of the drugs in the six different prescription-compatible liquids was above 90%, indicating that the infusion mixtures remained stable within 48 hours.
Relative percentage of four antitussive and expectorant drugs in 0.9% NS during 48 hours.

Relative percentage of four antitussive and expectorant drugs in 5% GS during 48 hours.

Discussion
The four antitussive and expectorant drugs—APL, DXL, AXH, and BXH—are widely used in clinical practice, either alone, in fixed-dose combinations, or in combination with intravenous infusion.4–10 In recent years, APL and DXL were used alone or in combination with AXH or BXH for treating respiratory diseases such as chronic obstructive pulmonary disease, chronic bronchitis, and bronchial asthma, yielding good outcomes. Additionally, these four antitussive and expectorant drugs were illegally added as chemical ingredients in traditional Chinese medicine for antitussives and expectorant purposes.12–14 Therefore, establishing an HPLC method for the simultaneous determination of these four drugs held clinical value for evaluating preparation quality, monitoring infusion preparation quality, conducting stability studies, and performing qualitative and quantitative analyses of illegal chemical drugs in traditional Chinese medicine.
According to the literature, numerous studies reported the determination of APL, DXL, AXH, or BXH by HPLC alone, and some studies used HPLC or ultra-violet (UV) methods to determine the content of two components.11–22 Two HPLC methods were developed to determine AXH and BXH simultaneously in pharmaceutical formulations.12,17 In addition, only one method was identified for quantifying DXL and AXH in tablet dosage form. 11 Various ratios, concentrations, and pH values of different mobile phases (methanol-water, acetonitrile-water, acetonitrile-acetic acid aqueous solution, acetonitrile-phosphate buffer, and acetonitrile-potassium dihydrogen phosphate solution) were tested to realize the simultaneous qualitative and quantitative analysis of these four antitussive and expectorant drugs by HPLC. However, the drugs could not be separated and determined simultaneously and quickly with equal elution of the mobile phase. Ultimately, rapid separation of the four drugs was achieved within 20 min using 50 mmol·L−1 potassium dihydrogen phosphate aqueous solution (pH 4.0) and acetonitrile as the mobile phase on an InertSustain C18 column under gradient elution conditions. This method is remarkable for its simple mobile phase, good repeatability, and accurate quantification.
In recent years, more than 2000 central intravenous additive services (CIVAS) of various sizes have been established in domestic medical institutions. The establishment of CIVAS has effectively improved the quality of finished infusions and reduced the incidence of adverse drug events. However, literature on solvent selection, infusion compatibility, and other aspects of widely used injections in clinical practice remains lacking. 25 In clinical practice, USP Chapter 797 classified CIVAS-dispensed infusions as low-risk sterile products, allowing them to be stored at 25 °C for 2 days to ensure microbial safety. 26 A literature search revealed some reports on the stability of APL, DXL, AXH, and BXH in infusions or their compatibility with other drugs. Incompatibility contraindications exist between AXH and BXH and many commonly used injections, such as cephalosporins, penicillins, proton pump inhibitors, traditional Chinese medicine injections, acyclovir, methylprednisolone sodium succinate, and parecoxib sodium27–29 because BXH and AXH are weakly basic and stable under weakly acidic conditions. Hydrochlorates are often used clinically. However, their solubility in water is low. When BXH and AXH were mixed, the pH of the compatible solution changes, drug solubility decreases, and stability is affected, resulting in solution discoloration and precipitation. In addition, most current domestic compatibility stability studies using UV spectrophotometry are inaccurate. The concentration of the infusion solvent or drug is limited, and the stability investigation time is only 6 to 8 hours, which cannot meet real clinical drug delivery needs.
Based on this information, we selected four widely used antitussive and expectorant drugs as research subjects and simulated clinical administration concentrations. We investigated their compatibility and stability over 48 hours when mixed with 0.9% NS and 5% GS. The experimental results showed that the four antitussive and expectorant drugs remained colorless and clear in the infusion mixtures, with the pH value changing by ±0.3 within 48 hours. The relative percentage of drugs ranged from 95.0% to 105.0%. These results indicate that the four antitussive and expectorant drugs are stable at room temperature for 48 h in 0.9% NS or 5% GS.
Conclusion
A simple and sensitive HPLC method was optimized and subsequently used to evaluate the stability of APL, DXL, AXH, and BXH when mixed with 0.9% NS or 5% GS for 48 hours. The developed method was validated to be selective, sensitive, linear, accurate, precise, and suitable for the rapid quantification of the four drugs in infusion samples, bulk drugs, and their dosage forms. The stability study recommends maintaining the stability of the four antitussive and expectorant drugs by diluting them in 0.9% NS or 5% GS injection, preparing them in a licensed CIVAS, and storing them at room temperature for 48 hours.
Footnotes
Authors’ contribution
XM, YF, and BF designed and performed the study in the laboratory and wrote the manuscript. MS and FC analyzed and interpreted the data. MS and BF revised the manuscript and supervised the study. All the authors have read and approved the final manuscript.
Availability of data and materials
Most of the data used to support the findings of this study are included in this article. All other data are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Technology Key Program of Shiyan, China (grant number 21Y75).
