Abstract
Objective:
This study investigated the fungal contamination profile of cocoa beans from cocoa-growing regions in Ghana, with particular emphasis on the potential impact of ochratoxigenic Aspergillus species.
Methods:
A total of 104 fermented and dried cocoa beans were randomly collected from farmers for analysis. Fungal isolation was conducted using potato dextrose agar and malt extract agar media. Species-level identification was performed through macroscopic and microscopic techniques. Ochratoxin A (OTA) quantification and confirmation were carried out using high-performance liquid chromatography with fluorescence detection and liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Results:
The investigation revealed 163 fungal isolates representing 14 distinct species across 10 genera. Aspergillus species, including Aspergillus flavus, Aspergillus niger, Aspergillus parasiticus, and Aspergillus fumigatus, were predominant. No statistically significant differences in fungal species occurrence were observed among the different cocoa-growing regions. However, certain species exhibited preferential growth on specific media, emphasizing the importance of medium selection in mycological studies. Of the 14 fungal species tested, only Aspergillus niger demonstrated the ability to produce OTA in vitro, as confirmed by LC-MS/MS analysis.
Conclusion:
This discovery holds substantial implications for both the cocoa industry and public health, considering that OTA is a potent mycotoxin subject to strict regulatory limits in food commodities. It is crucial to implement enhanced monitoring and control measures to mitigate fungal contamination in cocoa beans, particularly targeting OTA-producing species. The study also highlights the need for improved post-harvest handling practices, optimized storage conditions, and potential biocontrol approaches.
Introduction
Cocoa (Theobroma cacao L.), historically indigenous to the Amazon basin and tropical regions of Central and South America, has emerged as a vital cash crop in Ghana. 1 Its cultivation and processing significantly contribute to the nation's economy and the well-being of cocoa-producing farmers and local communities. Cocoa serves as a raw material for diverse products, including confectionery, cocoa beverages, and chocolates. 2 The cocoa value chain involves several stages, from cultivation and fermentation to drying, roasting, and manufacturing.
Historically, Ghana played a pivotal role in global cocoa production, contributing 30–40% of the world's output between 1911 and 1976. 3 However, a decline in production during the 1970s nearly led to the collapse of the cocoa sector in the early 1980s.4,5 Subsequent reforms facilitated recovery, and by the 1990s, production had nearly doubled. Currently, Ghana ranks as the second-largest cocoa producer globally, contributing approximately 25% of the total cocoa output. 6
Despite its economic importance, cocoa cultivation faces multifaceted challenges. These challenges include pests, diseases associated with fungal infections, and the impact of climate change. Among the fungal pathogens affecting cocoa, filamentous fungi that target cocoa beans and pods are of particular concern. Some of these fungi produce mycotoxins, with ochratoxin A (OTA) being especially noteworthy due to its widespread occurrence and acute toxicity. 7
Conventionally, fungi identification relies on colony morphology, encompassing characteristics such as size, shape, color, and sporulation patterns. 8 Additionally, microscopic examination of fungal hyphae and spores is a common method for species identification. These morphological techniques have been extensively employed in several studies.9–12
OTA is mainly synthesized by Aspergillus and Penicillium species13–15 and is found in various foods like cocoa beans, coffee, cereals, dried fruits, and spices.15–19 Its production varies with climate: Aspergillus species thrive in warm and temperate regions, 20 while Penicillium species are more common in cold areas. 21 Water activity (aw) also affects OTA production, with higher levels at 0.98aw. 22 Optimal OTA synthesis occurs between 25 and 30 °C for certain Aspergillus species. Aspergillus ochraceus is a key OTA producer in warmer regions, whereas Penicillium verrucosum is prevalent in colder climates. 23 Various Aspergillus species, including Aspergillus alliaceus, Aspergillus sclerotiorum, Aspergillus sulphureus, Aspergillus albertensis, Aspergillus auricomus, and Aspergillus wentii, produce OTA. 24 Notably, Aspergillus alliaceus contaminates figs in California. 25 Other OTA-producing species include Aspergillus niger, Aspergillus lacticoffeatus, Aspergillus sclerotioniger, Aspergillus steynii (found in coffee), and Aspergillus welwitschiae.26–28 Recently, a novel OTA-producing fungus, Aspergillus affinis, was identified in decomposing leaves. 29
Penicillium verrucosum, Penicillium nordicum, and Penicillium expansum are known OTA producers. 30 Penicillium nordicum typically contaminates sodium chloride and protein-rich foods like cheese and dry-cured meat, while Penicillium verrucosum is found in cereals. 31 Other species, such as Penicillium chrysogenum, Penicillium glycyrrhizacola, and Penicillium polonicum, produce OTA on fresh or dried liquorice, with Penicillium chrysogenum being a significant producer. 32 Conversely, Penicillium brevicompactum, Penicillium crustosum, Penicillium olsonii, and Penicillium oxalicum produce OTA at much lower levels. 33
OTA was initially isolated as a secondary metabolite from Aspergillus ochraceus in 1965. 34 OTA-producing fungi contaminate a diverse range of food commodities, impacting both human health and economic stability. OTA is a polyketide-derived secondary metabolite with a unique structure. It consists of a dihydro-isocoumarin moiety linked to an L-β-phenylalanine via an amide bond. The complete chemical name is L-phenylalanine-N-[(5-chloro-3,4-dihydroxy-8-hydroxy-3-methyl-1-oxo-1H-2-benzopyrene-7-yl) carbonyl]-R-isocoumarin. 35 OTA exhibits potent nephrotoxicity and other adverse effects, including hepatotoxicity, teratogenicity, and immunosuppression. Carcinogenic studies have linked OTA to kidney and liver cancers. 36 The International Agency for Research on Cancer (IARC) and the World Health Organization (WHO) classify OTA as a group 2B human carcinogen. 37 Additionally, OTA has been tentatively associated with Balkan endemic nephropathy (BEN) and urinary tract tumors. 38 In response to the recognized risks associated with OTA, several regulatory entities, including the European Union, the Brazilian Health Agency (ANVISA), and the Codex Alimentarius Commission, have instituted permissible limits for OTA in food commodities. These limits apply specifically to products derived from cocoa, such as cocoa beans and cocoa powder. Additionally, a tolerable daily intake (TDI) guideline has been established, recommending no more than 5 ng of OTA per kilogram of body weight per day. 18 These regulatory measures serve to protect public health by mitigating exposure to this mycotoxin.
Recent evidence shows increasing OTA contamination in various food commodities and products, including cocoa beans, coffee, spices, and beer. 39 OTA in cocoa-derived products is due to molds in cocoa beans. Banahene et al. 40 reported OTA in Ghanaian cocoa beans, emphasizing the need for further investigations. Environmental factors, such as climate and poor agricultural practices (e.g. inadequate harvesting, drying, and storage), contribute to OTA presence. 41
The problem is that, while previous mycological studies have shed light on the fungal populations associated with Ghanaian cocoa, there remains a significant gap in the comprehensive understanding of ochratoxigenic fungi across the cocoa-growing regions of Ghana. This lack of knowledge poses a challenge to sustainable cocoa production, as the impact of these fungi on cocoa safety and quality is not fully understood. Identifying ochratoxigenic fungal contaminants and developing effective interventions are crucial for ensuring public health safety and maintaining the quality of cocoa. Therefore, this study aims to characterize the fungal profile in cocoa beans from various cocoa-growing regions of Ghana, with a particular focus on assessing the OTA-production capacity of the fungal isolates in vitro.
Materials and methods
Materials
Sample collection
From October 2020 to September 2021, fermented and dried cocoa beans (n = 104) were collected directly from farm gates of individual cocoa farmers across the cocoa-growing regions of Ghana. The sampled regions included Ashanti, Brong Ahafo, Central, Eastern, Western North, Western South, and Volta (Figure 1), collectively accounting for 100% of cocoa production in Ghana. The regions under study exhibit peak rainfall periods in May/June and October, with mean annual precipitation ranging from 1100–1850 mm. Furthermore, Ashanti, Brong Ahafo, Central, Eastern, Western North, Western South, and Volta regions demonstrate the following mean annual temperature and relative humidity values: 24–30 °C and 85%; 23–34 °C and 85%; 25–32 °C and 81%; 24–29 °C and 84%; 24–34 °C and 87%; 24–34 °C and 87%; and 25–33 °C and 80%, respectively. Except for the Volta region, which comprises only two cocoa-growing districts, four districts were selected from each of the remaining regions, representing the primary cocoa-growing areas. Four samples were randomly collected from different farmers within each of the 26 district locations, resulting in a total of 104 samples, each weighing approximately 200 g. These samples were promptly placed in sterile paper bags and stored at 4 °C before being transferred to the laboratory for further analysis.

Map of Ghana showing the cocoa-growing regions where the cocoa beans were sampled.
Reagents
Solvents and chemicals were sourced as ACS grade or equivalent (Carlo Erba, Val-de-Reuil, France). For HPLC or LCMS analysis, LCMS-grade methanol, acetonitrile, and acetic acid were obtained from Chem-Lab Analytical (Zedelgem, Belgium). Ultra-pure water with 18.2 MΩ/cm resistivity and 0.1 µm sterile filtration was supplied by Evoqua Water Technologies (Barsbuttel, Germany). Ammonium formate (NH4F) from Park Scientific Limited (Northampton, UK) served as the mobile phase additive. The calibration standard for OTA was prepared by diluting a 10 µg/mL stock solution obtained from Biopure (Tulin, Austria).
Methods
Isolation of fungal mycoflora
A ten-gram (10 g) sample of cocoa beans was carefully weighed and immersed in a sodium hypochlorite solution (0.6%). The mixture was agitated for 2 minutes using an orbital shaker. Subsequently, the sample underwent thorough rinsing with sterile distilled water and was homogenized in a laboratory blender. An aliquot of 1 g of the homogenized sample was transferred into a McCartney tube containing 9 mL of sterile 0.1% peptone water. After allowing the mixture to stand for 30 min, it was gently shaken to obtain a uniform homogenate. Serial dilution was carried out to achieve a concentration of 0.01 g/mL (10−2) using sterile 0.1% peptone water. Each uniformly mixed dilution (1 mL of aliquot) was then transferred onto Petri plates (with three replicates for each medium). To each plate, 20 mL of liquefied medium, specifically Potato Dextrose Agar (PDA) and Malt Extract Agar (MEA) were added. The mixture was gently swirled and allowed to solidify, and then incubated aseptically at 25 °C for 7 days. Fungal colonies that developed were observed, and selected colonies were sub-cultured. Briefly, the sample from the fungal source was collected using a sterile loop and suspended in a sterile water solution to create a spore suspension. Serial dilution of the spore suspension was performed to reduce the concentration, making it easier to isolate single spores. Aliquot of 1 mL of the diluted spore suspension was spread onto a malt extract agar plate using a sterile spreader and further incubated at 25 °C for 7 days. The resulting pure cultures were stored at 4 °C for subsequent identification and analysis. The frequency and domination of fungi species was calculated as:
Identification of fungal isolates
The purified fungal colonies were meticulously examined to the species level using both macroscopic and microscopic techniques. These procedures followed established protocols outlined in previous studies.42–45 Specifically, the Leica DM750 high-definition digital microscope, in conjunction with the Leica Application Suite (LAS) version 4.12.0, facilitated the observation of morphological characteristics within the samples. Photographic documentation was also captured during this process.
Ochratoxin a production and extraction from culture media
OTA production was investigated in the identified isolates, following established protocols with slight modifications.46,47 Briefly, three agar plugs (Ø = 6 mm) were excised from the inner, middle, and outer areas of the media of each colony of identified species. These agar plugs contained fungal mycelium and metabolites. Next, OTA was extracted from the agar plugs using a 2 mL mixture of 98% methanol and acetic acid. The extraction was performed in darkness using an ultrasonic bath for 1 hour. The resulting extracts were vortexed and filtered through a 0.22 µm syringe filter and stored at −18 °C until further analysis.
Determination of ochratoxin a using high-performance liquid chromatography
A Shimadzu Prominence Series HPLC system equipped with a fluorescence detector (FLD) (Shimadzu Corporation, Kyoto, Japan) was utilized. OTA detection occurred at excitation and emission wavelengths of 330 nm and 460 nm, respectively. A reverse-phase (RP) analytical column (Mightysil RP-18 GP, 5 µm, 4.6 × 150 mm, Kanto Chemical Company, Tokyo, Japan) was employed. The mobile phase consisted of acetonitrile, water, and acetic acid (55:43:2, v/v/v) at a flow rate of 0.8 mL/min. The injection volume was 40 µL and the column oven temperature was 40 °C. The limit of detection for OTA was 1 ng/mL, based on a signal-to-noise ratio of 3:1. A standard calibration curve was constructed using pure OTA standards (ranging from 1 to 20 ng/mL) with a correlation coefficient (R2) value of 0.998. The calibration was used for OTA quantification in the sample extracts.
Ochratoxin a confirmation
Sample extracts containing OTA, detected using HPLC-FLD, were subjected to confirmation analysis by liquid chromatography-tandem mass spectrometry (LC-MS/MS) in the multiple reaction monitoring mode. The methodology followed the procedures outlined by Banahene et al. 40 Specifically, a Shimadzu Nexera Series separation module coupled with an 8060NX triple quadrupole mass spectrometer (Shimadzu Corporation, Kyoto, Japan) was employed. The separation of OTA was achieved using a Mastro2 PFP column (150 × 2.0 mm, 3 µm) from Shimadzu Corporation (Kyoto, Japan). The mobile phase consisted of a 0.15 mM NH4F aqueous solution (solvent A, approximately pH 6) and a 0.15 mM NH4F methanol solution (solvent B). OTA elution occurred at a flow rate of 0.4 mL per minute in a gradient mode: solvent B was initially held at 15% for 1 min, increased to 100% over 2 min, decreased back to 15% over 4 min, and finally stopped at 12 min. The injection volume was 2 µL, and the column oven temperature was maintained at 40 °C. For electrospray ionization (ESI), positive ion mode conditions were as follows: source temperatures (interface, heat block, and desolvation line) at 400 °C, 400 °C, and 250 °C, respectively; gas flow was (nebulizer, heating, and drying) set to 2, 15, and 3 L/min, respectively; capillary voltage at 4.0 kV; and collision gas (argon) pressure at 270 kPa. The LC-MS/MS analysis of OTA utilized a precursor ion with m/z 404.30, and three specific transitions were monitored: m/z 239.00, 358.15, and 221.10. Data generated from the OTA analysis were acquired and processed using LabSolutions software (version 5.99) and LabSolutions Insight (version 3.7) from Shimadzu Corporation (Kyoto, Japan).
Statistical analysis
The data were statistically analyzed using IBM SPSS software (version 27, SPSS Inc., Chicago, USA) to assess the effect of sample origin and the cultivation media by analysis of variance, followed by Tukey's Honestly Significant Difference (HSD) post hoc test to evaluate differences among means. Statistical significance was considered when p-values were < 0.05.
Results and discussion
Fungal isolates: occurrence and diversity
In this study, we investigated the fungal mycoflora associated with cocoa beans obtained from seven cocoa-growing regions of Ghana. The identified fungal species included Aspergillus (Aspergillus flavus, Aspergillus niger, Aspergillus parasiticus, and Aspergillus fumigatus), Cladosporium (Cladosporium sphaerospermum and Cladosporium macrocarpum), Absidia corymbifera, Alternaria alternata, Byssochlamys nivea, Eurotium herbariorum, Mucor racemosus, Penicillium brasilianum, Fusarium species, and Rhizopus stolonifer (Table 1). Our investigation yielded 163 fungal isolates, representing 14 distinct species (Figure 2) across 10 genera. In a comprehensive examination of the identified fungal isolates, the macroscopic and microscopic morphological characteristics of each species were observed. Aspergillus flavus was noted for its dense felt of yellow-green conidiophores, each bearing phialides directly on a globose to sub-globose vesicle. Rhizopus stolonifer presented with whitish mycelium, brownish sporangiophores, and brown-black sporangia, all accompanied by columella. Mucor racemosus initially appeared white but transitioned to a brownish-gray hue after one week, with sporangiophores associated with columella and hyphae. Aspergillus niger displayed a compact white basal felt, overlaid with a dense layer of black conidiophores and a globose conidial head arranged in a rosette pattern. Alternaria alternata exhibited a black to olivaceous-black appearance with a suede-like to floccose texture, forming branched acropetal chains of multicellular beaked conidia. Absidia corymbifera was characterized by rapid growth, presenting a floccose, light grayish appearance with terminal sporangiospores. Aspergillus parasiticus featured a dense felt of green conidiophores, with phialides borne directly on a sub-globose vesicle. Byssochlamys nivea was distinguished by white ascomata in the basal felt, obscured by floccose to funiculus overgrowth, and included conidial structures and ascospores. Cladosporium macrocarpum had a velvety texture covered with grayish aerial mycelium, while Cladosporium sphaerospermum exhibited a velvety olive-green to olivaceous-brown appearance with acropetal conidia and conidiophores. Eurotium herbariorum displayed broad zones of dull green to gray-green, spreading broadly and irregularly, with conidial structures including conidiophores and conidia. Aspergillus fumigatus featured a dense felt of dark green conidiophores intermixed with aerial hyphae bearing conidiophores, with conidia borne in chains directly on the vesicle. Penicillium brasilianum exhibited a velvety colony texture, grayish-green with whitish margins and hyaline mycelium, with long, biverticillate conidiophores and finely rough-walled to rarely echinulate conidia. Finally, Fusarium species was characterized by brownish-white cottony mycelium growth with smooth margins, with non-chain, mostly non-septate microconidia ranging from ellipsoidal to cylindrical.

Macroscopic and microscopic (magnification: ×100; scale bar: 100 µm) plates of the isolated fungi on malt extract agar (MEA) : (a) Aspergillus flavus, (b) Rhizopus stolonifer, (c) Mucor racemosus, (d) Aspergillus niger, (e) Alternaria alternata, (f) Absidia corymbifera, (g) Aspergillus parasiticus, (h) Byssochlamys nivea, (i) Cladosporium macrocarpum, (j) Cladosporium sphaerospermum, (k) Eurotium herbariorum, (l) Penicillium brasilianum, (m) Fusarium spp., and (n) Aspergillus fumigatus.
Fungal species from cocoa beans obtained from the various cocoa-growing regions on PDA and or MEA media.

Note: PDA: potato dextrose agar; MDA: malt extract agar.
As summarized in Tables 2 and 3, Aspergillus niger, Aspergillus fumigatus, and Aspergillus flavus emerged as the predominant species. Fungal species occurrence variations in the regions were observed, with Aspergillus niger being highly prevalent in the Western South and Central regions, while Aspergillus fumigatus dominated in Brong Ahafo, Ashanti, and Eastern regions (Figure 3). However, it is noteworthy that no statistically significant differences exist in fungal species occurrence among the different regions as indicated by the p-value (0.4482 > .05). Furthermore, Tukey's honestly significant difference post hoc test was performed to compare the mean occurrence between pairs of regions. The results showed that no statistically significance differences were found between any pair of regions (all p-adjusted values >0.05). The largest mean difference was observed between Brong Ahafo and Central regions (mean difference = −6.2279), but it was not statistically significant (p-adjusted = 0.5348). Additionally, the smallest mean difference was between the Central and Western North regions (mean difference = −0.022; p-adjusted = 1.0). Conversely, the extremely low p-value (5.2e-08 < 0.05) indicates that there are highly statistically significant differences in occurrence among different fungal species. These findings imply that different fungal species have varying levels of prevalence. Furthermore, the Eastern region exhibited the highest fungal diversity, while the Volta region had the lowest diversity. This diversity pattern may be linked to climate, mycoflora, and post-harvest practices. Furthermore, the diversity in fungal species occurrence among cocoa beans from different locations within the regions can be attributed to several other factors. For instance, farmers’ storage practices at various collection points significantly impact fungal colonization. Previous studies have underscored the impact of storage conditions on the occurrence of fungal species in cocoa beans.41,48,49 Furthermore, variations in the origins of cocoa beans, whether from distinct farms or different regions, may contribute to the observed differences.
Mean occurrence of fungi in the cocoa beans obtained from the various cocoa-growing regions on potato dextrose agar media.

Note: Data are presented as mean ± standard error (mean ± SE); Afv: Aspergillus flavus; Rsn: Rhizopus stolonifer; Mrm: Mucor racemosus; Ang: Aspergillus niger; Aan: Alternaria alternata; Acb: Absidia corymbifera; Aps: Aspergillus parasiticus; Bnv: Byssochlamys nivea; Cmc: Clasdoporium macrocarpum; Css: Clasdoporium sphaerospermum; Ehb: Eurotium herbariorum; Pbl: Penicillium brasilianum; Fsp: Fusarium spp.; Afm: Aspergillus fumigatus.
Mean occurrence of fungi in the cocoa beans obtained from the various cocoa-growing regions on malt extract agar media.

Note: Data are presented as mean ± standard error (mean±SE); Afv: Aspergillus flavus; Rsn: Rhizopus stolonifer; Mrm: Mucor racemosus; Ang: Aspergillus niger; Aan: Alternaria alternata; Acb: Absidia corymbifera; Aps: Aspergillus parasiticus; Bnv: Byssochlamys nivea; Cmc: Clasdoporium macrocarpum; Css: Clasdoporium sphaerospermum; Ehb: Eurotium herbariorum; Pbl: Penicillium brasilianum; Fsp: Fusarium spp.; Afm: Aspergillus fumigatus.

Diversity in the isolated fungal species across the cocoa-growing regions of Ghana.
Consistent findings across multiple studies have highlighted the contamination of cocoa beans by various fungi species.50–55 Stephanie et al. 56 reported the occurrence of Aspergillus species alongside other fungi in dry fermented cocoa beans from three main cocoa-producing regions in southeastern Cote d’Ivoire. Similarly, Akinfala et al. 57 documented diverse fungal isolates in two cocoa bean hybrids in Nigeria, emphasizing the dominance of Aspergilli.
The occurrence of the various fungal species on cocoa beans obtained from different cocoa-growing regions in Ghana is not unexpected. Throughout the cocoa bean handling process, from harvest to storage, multiple stages provide opportunities for mold infestation. While diseased and rotten pods are known to harbor fungal spores and mycelia, even dry cocoa beans obtained from healthy pods can carry these contaminants. 58 Under favorable conditions, these spores and mycelia germinate, posing quality and safety risks. Processes involved in cocoa bean production, including pod breaking, fermentation, and drying, introduce fungal spores into the final dried cocoa beans. 59 In the studied cocoa-growing regions of Ghana, cocoa farmers follow a series of post-harvest processing practices to ensure the quality of their beans. Harvesting occurs every three to four weeks during the main crop season (October–April) and light crop season (May–August), using machete or cocoa hook to cut ripe pods. The pods are then collected and manually broken to extract the beans. Fermentation follows, lasting about six days, during which the beans are turned every two days. After fermentation, the beans are sun-dried on raised beds for about eight days, ensuring they reach the optimal moisture content of approximately 7%. After drying, the beans are bagged and stored prior to purchase by Licensed Buying Companies (LBCs). Proper adherence to Ghana Cocoa Board and Codex Alimentarius Commission recommendations during these steps minimizes fungal loads to acceptable levels. However, if drying and storage are inadequate, cocoa beans obtained directly from farm gates may be contaminated with fungal spores or mycelia. Moisture content, temperature, humidity, and other environmental factors significantly influence fungal germination. 41 The presence of fungi in cocoa beans adversely affects quality, diminishing their aesthetic value and posing safety concerns. For instance, dry fermented cocoa beans with over 3% internal fungal contamination are considered unsafe for consumption and are therefore rejected based on marketable quality (cut test) standards. 60 Dano et al. 59 observed that the cocoa pulp within undamaged cocoa pods remains microbiologically sterile. However, the hygiene practices during cocoa bean handling, from pod breaking to seed extraction, fermentation, and drying, vary significantly among individual cocoa farmers. Consequently, contamination by different fungi during post-harvest processes is likely. In their study, Dewayani et al. 61 underscored the role of inadequate handling practices in contributing to mold contamination of cocoa beans. These practices include contamination from pod surfaces, worker hands, machetes, insects, and work environments. Furthermore, the authors emphasized that physical damage to cocoa beans and poor plant health can also increase their susceptibility to fungal contamination. Ochratoxigenic fungi, particularly Aspergillus species are prevalent fungus in cocoa beans, influenced by both climatic and environmental conditions, as well as post-harvest processing practices in cocoa-growing regions of Ghana. 60 The warm and humid climate of the studied regions provides an ideal environment for several fungi. The tropical conditions, characterized by high temperatures and substantial rainfall, facilitate the growth of these fungi. Variations in climate, such as increased temperatures and changes in rainfall patterns, can further enhance conditions favorable to Aspergillus species, potentially leading to higher contamination rates in cocoa beans. Additionally, certain Aspergillus strains are commonly found in tropical soils and vegetation. 62 The natural environment in these regions serves as a reservoir for the fungus, which can easily transfer to cocoa pods and beans. Effective fermentation is crucial in mitigating fungal contamination. However, inadequate management of the fermentation process can create conditions conducive to their proliferation. Poor aeration and high moisture levels during fermentation can promote fungal growth. Proper drying of cocoa beans is essential to reduce moisture content to around 7%, preventing fungal growth. 63 Delays or improper drying can leave residual moisture, supporting the growth of the fungi. Post-harvest storage conditions significantly influence fungal contamination. Cocoa beans stored in humid or poorly ventilated environments are more susceptible to these organisms. Mitigating both environmental factors and post-harvest practices is crucial in minimizing the risk of Aspergillus contamination in cocoa beans, thereby ensuring the quality and safety of the final product.
Recommendations include excluding damaged and diseased pods during fermentation. Additionally, sucrose and glucose promote fungal proliferation, while lactic acid and acetic acid produced by bacteria inhibit fungi growth.55,64,65 Proper fermentation, involving the breakdown of sugars, helps mitigate fungal proliferation. 66 Understanding the fungal occurrence, diversity, and ecological factors affecting cocoa beans is crucial for quality control and food safety. Future research should explore safe and natural alternatives to synthetic fungicides, emphasizing sustainable practices that safeguard both consumer health and the cocoa industry.
Fungal species occurrence on potato dextrose agar and malt extract agar media
The present study also highlighted the variations in the prevalence of fungal species in two different cultivation media: Potato Dextrose Agar (PDA) and Malt Extract Agar (MEA). A distinct pattern of fungal distribution between these two media was revealed. Certain species exhibited higher prevalence in PDA, while others were more abundant in MEA (Figure 4). Among the species identified, Aspergillus fumigatus exhibited the highest mean occurrence, with 22.67% on PDA and 25.26% on MEA. In contrast, Cladosporium shpaerospermum had the lowest mean occurrence on PDA (0.16%), while Mucor racemosum showed no occurrence on MEA (0.0%). Particularly, some fungi displayed differences in occurrence between PDA and MEA media. For instance, Eurotium herbariorum had a minimal occurrence of 0.77% on PDA but a much higher occurrence of 22.19% on MEA.

Occurrence of the identified fungal species in potato dextrose agar (PDA) and malt extract agar (MEA) media. Data points represent the mean occurrence of the identified fungal species (n =3) ± standard error.
Although statistical analysis did not detect a significant difference in the overall occurrence, variations observed suggest that nutritional composition plays a crucial role. Among the fungal species analyzed, Alternaria alternata, Absidia corymbifera, Aspergillus flavus, Aspergillus niger, Aspergillus parasiticus, and Byssochlamys nivea, were predominantly distributed in PDA. In contrast, Aspergillus fumigatus, Cladosporium macrocarpum, Cladosporium sphaerospermum, Eurotium herbariorum, Fusarium species, Penicillium brasilianum, and Rhizopus stolonifer exhibited higher occurrence in MEA. The lack of statistical significance (p = 0.1619, p > 0.05) suggests that medium type alone does not account for these differences. The observed variation in fungal species occurrence likely stems from the distinct nutritional profiles of PDA and MEA. 67 PDA, commonly used for the cultivation of luxuriant fungal growth, contains dextrose and dehydrated potato infusion. Its acidic environment, achieved through acid or antibiotic additives, inhibits bacterial growth. In contrast, MEA contains a high concentration of maltose, which makes it suitable for the growth of fungi. Generally, MEA is used as a growth medium to isolate and cultivate fungi from a wide range of sources. It contains carbon, protein, and nutrient sources (dextrin, glycerol, and gelatine) essential for fungal growth. Additionally, MEA contains a digest of animal tissues (peptone) which provides significant quantities of amino acids and nitrogenous compounds for the growth of fungi. This rich source of nutrient milieu favors fungal growth and metabolism. Consequently, the nutritional requirements of specific fungal species influence their distribution across these media. 68
OTA production and confirmation
In this study, we investigated the OTA production capabilities of the identified fungal species. Chromatograms from OTA-positive and OTA-negative species, along with an OTA standard, were analyzed (Figure 5). Among the 14 distinct fungal species tested, only Aspergillus niger demonstrated OTA production (Table 4). Our findings corroborate the work of Rahayu et al., 66 who also identified Aspergillus niger as an OTA producer. Similarly, Nugroho et al. 69 reported OTA production associated with Aspergillus niger.

HPLC (high-performance liquid chromatography) chromatogram of Aspergillus niger (ochratoxin A (OTA)-positive), the OTA-negative isolate, and the OTA standard (10 ppb).
OTA production by Aspergillus niger grown on malt extract agar medium after 7, 14, and 21 days of incubation at 25°C.

Note: OTA: ochratoxin A; MEA: malt extract agar.
To confirm OTA production by Aspergillus niger, we employed liquid chromatography-tandem mass spectrometry (LC-MS/MS). By comparing the mass spectra of existing literature40,70 and an authentic OTA standard, we identified an [M+H]+ OTA precursor ion at m/z 404.30, along with product ions at m/z 239.00, 358.15, and 221.10 (Figure 6). The retention time and mass spectra of OTA from the culture media extract were identical to the authentic standard, providing unambiguous confirmation of OTA presence in the Aspergillus niger culture filtrate.
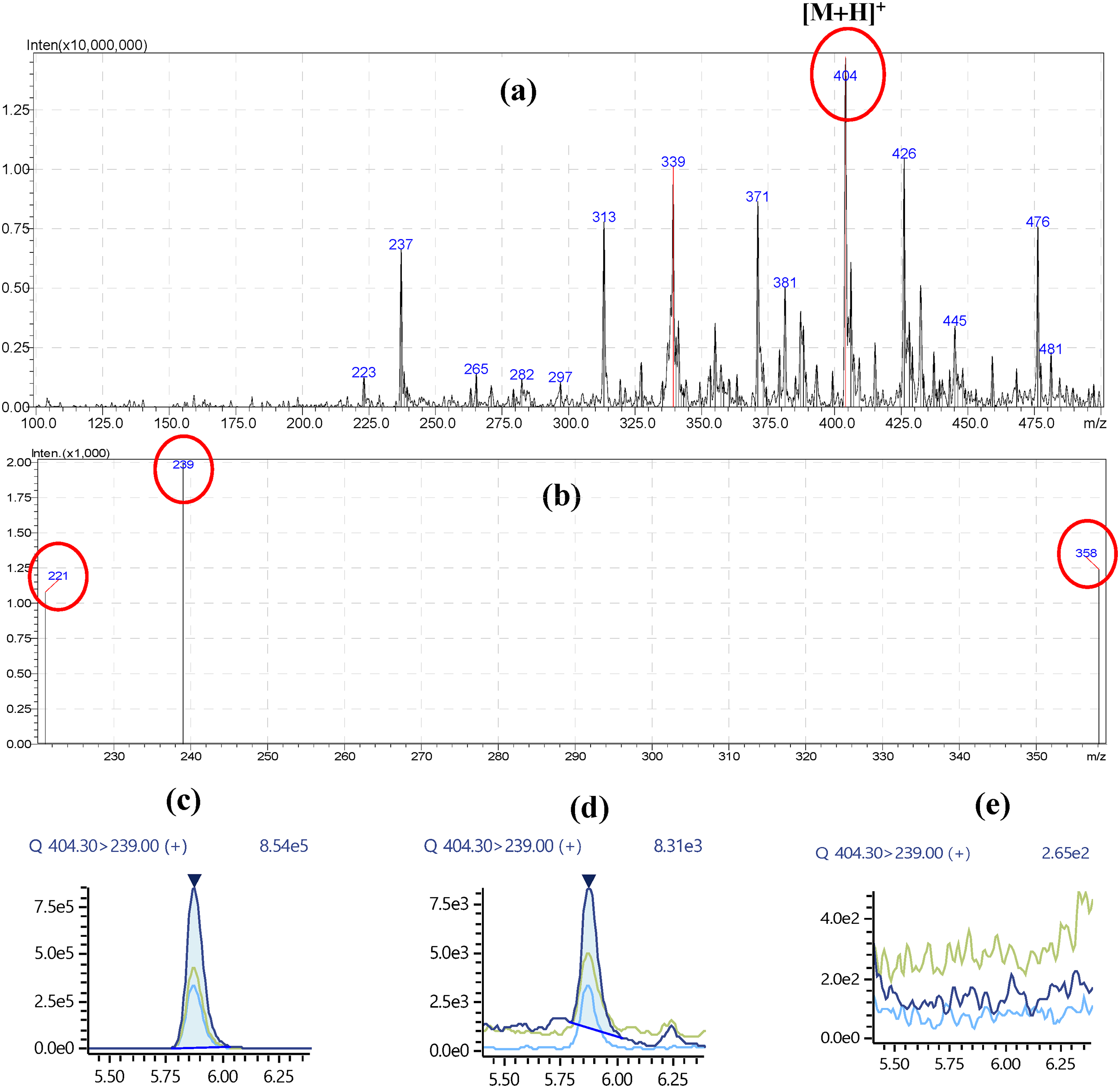
Liquid chromatography-tandem mass spectrometry (LC-MS/MS). (a) Mass spectra of the ochratoxin A (OTA) standard showing the precursor ion (m/z 404.30). (b) Mass spectra of the OTA standard showing the product ions (m/z 239.00, 358.15, and 221.10). (c) OTA standard (10 ppb) peak at a retention time of 5.855 min. (d) Aspergillus niger (OTA-positive) culture filtrate peak at a retention time of 5.855 min. (e) OTA-negative isolate culture filtrate with no peak.
Previous studies51–53,55 reported the occurrence of several fungal species, including Aspergillus niger, Aspergillus flavus, Aspergillus fumigatus, Penicillium verrucosum, and Aspergillus ochraceus, in cocoa beans from various regions. While Aspergillus niger is recognized as an ochratoxigenic species,66,69 other fungi isolated from cocoa beans have not been directly linked to OTA production. Instead, some of these fungi are known to produce different mycotoxins. For example, Aspergillus flavus and Aspergillus parasiticus are responsible for aflatoxin production, while Fusarium species produce zearalenone, deoxynivalenol, and fumonisins.50,51,53,71
Figure 7 illustrates the mapping between the studied regions and each fungal isolate, particularly Aspergillus niger. This finding highlights the extensive incidence of Aspergillus niger across multiple regions, demonstrating its adaptability to diverse geographical areas within the studied regions.

Mapping of the isolated fungi distribution across the studied regions.
The presence of OTA-producing Aspergillus niger on cocoa beans raises concern for cocoa producers, consumers, and the trade industry. Aspergillus niger presence was detected at all processing stages, with varying isolation frequencies: 3.92% during fermentation, 14.8% at the drying stage, and 26.15% during storage. 72 However, it is essential to note that the mere presence of ochratoxigenic fungi in food commodities does not guarantee OTA synthesis. Environmental factors, such as temperature, water activity, pH, oxygen availability, and ion presence, play a pivotal role in OTA production across ochratoxigenic fungal species.73,74
In this study, the Western South region exhibited the highest mean occurrence of the ochratoxigenic strain Aspergillus niger in the analyzed cocoa bean samples. This result aligns with the findings of Banahene et al., 40 who also reported the highest occurrence of OTA contamination in cocoa bean samples from the same region.
The presence of non-mycotoxigenic (OTA) fungal isolates on cocoa beans can significantly impact their quality in both beneficial and detrimental ways. Certain non-mycotoxigenic fungi serve as biocontrol agents, inhibiting the growth of pathogenic fungi, which can reduce the incidence of mycotoxins in cocoa beans and thereby enhance overall bean quality. 75 Conversely, despite not producing mycotoxins, some non-mycotoxigenic fungi can still cause spoilage, leading to off-flavors, discoloration, and other quality defects in the beans. 76 The influence of non-mycotoxigenic fungal isolates on cocoa beans is multifaceted; while they can enhance fermentation and suppress the formation of mycotoxins and diseases, they can also cause spoilage and quality deterioration under favorable conditions. Therefore, effective management of these fungi through proper post-harvest practices is essential to maintain the high quality of cocoa beans.
It is crucial to acknowledge that in this study, both macroscopic and microscopic morphological fungal identification techniques lack the specificity required for distinguishing closely related species, thereby complicating accurate species-level identification. These methods also depend heavily on the mycologist's expertise and experience, potentially leading to subjective and inconsistent results. However, incorporating molecular identification techniques would enhance the accuracy of identifying fungal isolates. Molecular identification of fungal isolates involves the extraction of fungal deoxyribonucleic acid (DNA), followed by amplification of specific genetic markers using polymerase chain reaction (PCR). Commonly targeted regions include the internal transcribed space regions, which are highly variable among different fungal species. Sequencing the amplified DNA allows for comparison with reference databases, enabling accurate species-level identification. This approach provides greater specificity and reliability compared to traditional morphological methods, facilitating the accurate classification of fungal isolates. Furthermore, an inadequate sample size from the studied regions may limit the generalizability of the findings, as a small or non-representative sample may not reflect the full diversity of fungal species in the environment. Future studies should incorporate molecular identification techniques and larger sample sizes to validate the study's conclusions.
Conclusion
The study examined the fungal mycoflora of cocoa beans from cocoa-growing regions of Ghana, analyzing 163 fungal isolates across 14 species and 10 genera. Dominant species included Aspergillus niger, Aspergillus fumigatus, and Aspergillus flavus. Despite regional diversity, no significant differences in species occurrence were found, indicating a consistent fungal profile nationwide. Notably, Aspergillus niger was the only species producing ochratoxin A (OTA) in vitro, highlighting the need for stringent monitoring due to OTA's health risks. The findings emphasize a multifaceted approach to cocoa bean safety, suggesting future research on mitigating fungal contamination, particularly OTA producers. Investigations into post-harvest practices, storage conditions, and safe, natural and sustainable biocontrol methods are recommended. The study underscores the importance of understanding environmental, agricultural, and genetic factors influencing fungal colonization, aiming to develop resilient cocoa varieties and targeted antifungal strategies. Continuous monitoring and evidence-based practices are crucial for maintaining cocoa quality and safety amid growing global demand.
Footnotes
Acknowledgments
The authors express their gratitude to Mr George Ashong Akwetey from the Department of Plant and Environmental Biology, University of Ghana, for his valuable assistance with the microbiology analysis aspect of this study. Additionally, the first author acknowledges the Ghana Cocoa Board for sponsoring his doctoral studies.
Authors’ contribution
Joel Cox Menka Banahene contributed to writing‒original draft, resources, project administration, methodology, formal analysis, data curation, and conceptualization. Isaac Williams Ofosu (main supervisor) contributed to conceptualization, writing‒review and editing, and supervision. Herman Erick Lutterodt contributed to review and editing and supervision. William Otoo Ellis contributed to review and editing and supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study did not involve human participants, animal subjects, or any form of personal data collection. Therefore, ethical approval and informed consent were not required.
Data availability
The data utilized to support this study's findings have been provided in the manuscript.

