Abstract
Current guidelines recommend VATS as the first-line treatment for stage II empyema. Patients with concomitant cardiorespiratory failure are considered ineligible for guideline-recommended management. Alternative treatment options are needed. This case report pertains to an obese man in his early 60 s with respiratory failure and decompensated heart failure. He developed an emphyema, which was refractory to the standard antibiotic therapy, thoracocentesis and other chest drainage. As patient was disqualified from surgical treatment, new approach was implemented. Minimally invasive approach of introducing a single-use bronchoscope through an existing chest drain (FOB-VAMT) was performed with support of non-invasive ventilation and analgosedation. Patient's condition improved significantly after the procedure. He remains under regular follow-up at the Pulmonology Outpatient Clinic 2 years after implemented treatment. This case reports that FOB-VAMT may provide an effective therapeutic option for patients disqualified from standard VATS and endotracheal intubation.
Keywords
Introduction
Pleural effusion represents the disturbance between production and resorption of the pleural fluid. Each case requires a wide differential diagnosis including congestive heart failure, cancer, pneumonia, and pulmonary embolism. 1 The amount of pleural infections are rising according to the data gathered from various European and American studies.2,3 Ageing population and chronic comorbidities are believed to be the most important risk factors. However, this rising trend may also be connected to improved access to computed tomography (CT) and bedside ultrasound. Therefore, a proper diagnostic process is essential. 4 Approximately 15% of pleural effusion of bacterial origin can progress to bacterial invasion of the pleural space with a collection of culture positive pus called the empyema5,6 Management of this conditions may pertain empirical antibiotic therapy, thoracosenteses with intrapleural enzyme therapy. Such measures may sometimes prove to be insufficient. It is reported that lung entrapment may occur after 10–14 days of ongoing pleural infection. 7 Surgical intervention of draining, debridement and decortications by means of video-assisted minimal access thoracis surgery (VATS) is well established treatment option with outcomes superior to chest tube in the case of organized empyema.8,9 Optimal timing for the surgery is also crucial, as better 90-day post-operative survival among patients with stage 3 emphysema is observed when the procedure is performed within approximately 7 days since admission 10 VATS can be performed with triportal or uniportal approach. Uniportal VATS may be less painful procedure while being similarly safe and feasible as its triportal counterpart.11,12 Evolution of the VATS technique and thoracic anesthesia were paralelled. Awake and monitored anestesia care led the way for the nonintubated uniportal VATS, which has the potential to prevent complications related to invasive ventilation as well as general anesthesia.7,13 Nevertheless, the morbidity and mortality after VATS are still significant. Therefore not all patients with empyema are surgical candidates. Other ways to access intrapleural space in less invasive way were researched. In 1988, Japanese study assessing 20 patients proved that a flexible bronchoscope can be inserted through the chest drain for diagnostic purposes. 14
To our knowledge, therapeutic use of this method has not been broadly researched.
Case presentation
The reporting of this case report conforms to CARE guidelines 15
Patient information
In December 2022, an obese man in his 60 s with a history of chronic respiratory failure (CRF), persistent atrial fibrillation, arterial hypertension, and chronic kidney disease was admitted to the Emergency Department (ED) of Upper Silesian Medical Centre in Katowice due to progressive dyspnea. The patient was subsequently transferred to the Intensive Cardiac Care Unit (ICCU) for management of newly diagnosed decompensated heart failure with reduced left ventricular ejection fraction (LVEF), corresponding to NYHA class IV.
On admission clinical presentation included severe resting dyspnea (mMRC grade IV), tachypnea (24 breaths/min), SpO₂ of 88% on 2 L/min nasal oxygen, and blood pressure of 140/100 mm Hg. He was in atrial fibrillation/flutter with a rapid ventricular response (130–140 bpm) and was in exacerbated respiratory distress.
Clinical findings
Transthoracic echocardiography revealed severe left ventricular systolic dysfunction with a markedly reduced LVEF of 15%, an enlarged left ventricular end-diastolic diameter (LVEDD) measuring 60 mm, biventricular and right atrial enlargement, moderate tricuspid regurgitation and trace pericardial effusion. The findings were consistent with low cardiac output syndrome and right heart failure with ascites. Lung ultrasound (LUS) showed 8 cm of pleural fluid in the left pleural cavity with secondary atelectasis.
Timeline
The exact timeline is presented in Figure 1.

Timeline of the clinical course.
Diagnostic assessment
On admission, the patient presented with elevated inflammatory markers and cholestatic enzymes. Arterial blood gas analyses revealed features of chronic respiratory failure with metabolically compensated hypercapnia. Hypoxemia and compensated respiratory acidosis were also observed during patient's stay at the Intensive Cardiac Care Unit and on admission to the Department of Lung Diseases.
During hospitalization, infections with Clostridium difficile and Enterococcus faecalis were identified. In a subsequent culture, Escherichia coli was isolated from a lower respiratory tract specimen. In the Emergency Department in the bedside chest X-ray the image indicated a significant accumulation of fluid in the left pleural cavity and atelectatic changes. Thoracocentesis was performed and the obtained pleural fluid was evaluated as exudate according to Light's criteria.
Chest CT performed after initial thoracocentesis revealed a massive accumulation of fluid in the left pleural cavity, compressing the left lung parenchyma. Additionally, two emphysematous bullae were identified in the middle zone of the left lung. A small amount of fluid was also noted in the right pleural cavity. The mediastinum was shifted towards the right side. Radiological course presented by subsequent CHEST CT is demonstrated in Figure 2. Second thoracocentesis was performed. It was decided to make cardiothoracic consultation. Chest drainage was inserted under the guide of ultrasound as its result.
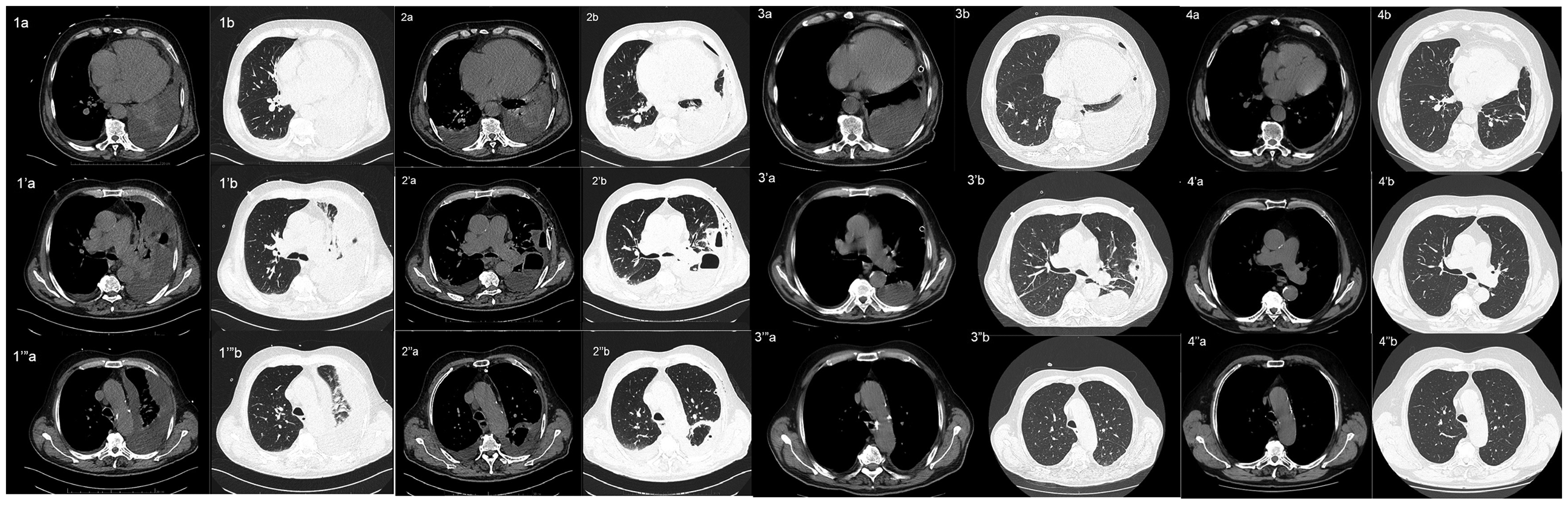
Radiological course presented by chest computed tomography images of the thorax, presented in mediastinal (a, a’, a’’) and lung (b, b’, b’’) windows, at sequential anatomical levels.a, b. At the level of the lung bases. ‘a, ‘b. At the level of the tracheal bifurcation. “a, “b. At the level of the aortic arch.
The pleural fluid obtained during the second thoracocentesis was found to be purulent, and stage II/III pleural empyema was diagnosed.
Follow-up chest CT performed after three days of drainage revealed a significant reduction in left pleural fluid volume, but demonstrated a newly visualized encapsulated collection in the upper left oblique fissure and a thick-walled, fluid-containing emphysematous bulla in the posterior left lung, suggestive of a partially evacuated abscess. Extensive atelectatic-inflammatory changes were noted in the left lung, with associated bronchial narrowing and truncation.
Improvements in the pulmonary parenchyma on the left side, as observed in follow up chest X-rays and CT scans, did not correlate with the patient's clinical condition or blood gas parameters. Subsequent chest CT revealed an empyema in the left pleural cavity.
Therapeutic intervention
Empirical antibiotic therapy was initiated with levofloxacin and cefuroxime. Based on subsequent microbiological results (Clostridium difficile and Enterococcus faecalis), targeted antibiotic therapy with vancomycin, metronidazole, and ampicillin was administered, resulting in a clinical improvement. Pharmacotherapy also consisting of diuretics (furosemide, spironolactone) and nitroglycerin was administered. Thoracocentesis was performed twice and total amount of 4.5 L of blood-stained fluid was obtained. The second thoracocentesis was complicated by an iatrogenic pneumothorax, requiring chest drainage. The patient was evaluated by thoracic surgeons and anesthesiologists, with the volume of pleural drainage over the past 12 days assessed at approximately 300 mL of transudative fluid per day, increasing to 550 mL/day on the day prior to consultation. Thoracic surgery consultation disqualified the patient from any major surgical intervention beyond drainage. Conservative management was recommended, with a suggestion to insert a Heimlich valve if pleural drainage continued to exceed 300–400 mL/day.
Anesthesiology consultation concluded that intubation and mechanical ventilation were not appropriate due to the patient's comorbidities and established respiratory failure. Non-invasive ventilation (NIV) and the use of a Heimlich valve were recommended as only feasible management strategies. Due to type two respiratory failure NIV was instituted as adjunctive supportive therapy and was well tolerated.
Pleural drainage with a Heimlich valve was reinserted and maintained. However, due to lack of evidence supporting its effectiveness and radiological confirmation of drain displacement, the decision was made to remove the drain couple of days after insertion.
The patient was subsequently transferred to the Department of Lung Diseases. Fiberoptic bronchoscopy (FOB) was performed. Subsequently, targeted antibiotic therapy with meropenem and amikacin, guided by bronchoalveolar lavage (BAL) culture results, was initiated, resulting in a decrease in inflammatory markers. Alternative management of pleural empyema was implemented by performing medical thoracoscopy utilizing a single-use flexible bronchoscope (FOB-VAMT) through the introduction of aforementioned bronchoscope via the pleural drain. Signed consent to treatment from the patient was obtained beforehand. During the procedure the patient was under respiratory support of NIV due to his disqualification from intubation. Figure 3 presents the procedure itself.
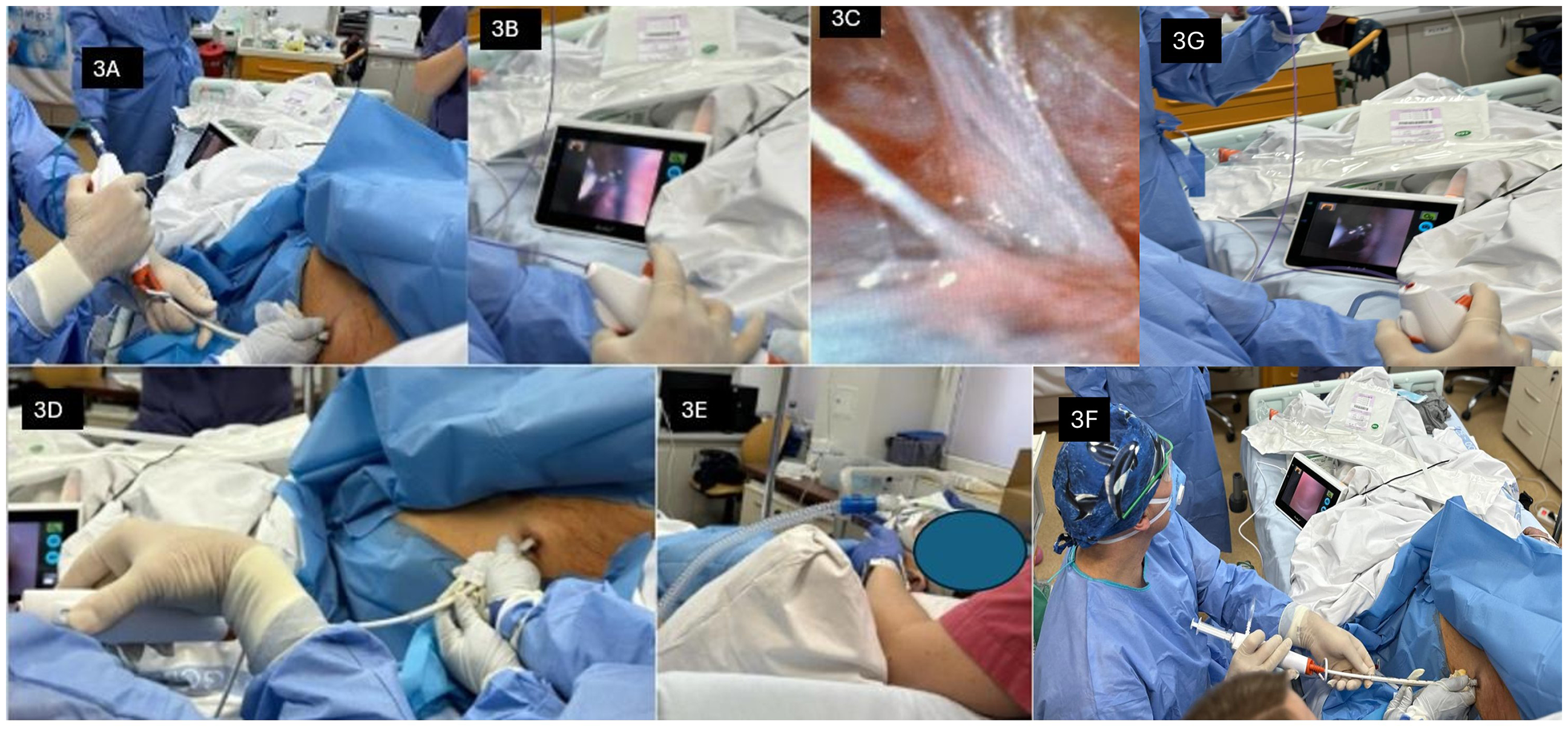
Course of the FOB-VATS procedure. 3A. Fiberoptic bronchoscope inserted through drainage tube. 3B. Bronchoscopic scissors used to cut pleural fibrin adhesions. 3C. Pleural fibrin adhesions. 3D. Aseptic environment was ensured during the procedure. 3E. NIV usage to prevent respiratory failure. 3F Overall view of the procedure and equipment. 3G Bronchoscopic forceps working within the pleural cavity.
Fiberoptic bronchoscope (Ambu® aScope™ 4 Broncho, Ambu® aView™; width 241 mm, height 175 mm, thickness of 33.5 mm) was used to perform FOB-VAMT. The procedure was performed twice over four days. The first procedure was performed with non-invasive ventilation (NIV) under shallow analgosedation with fentanyl and midazolam administered in fractionated doses. Respiratory support was provided with NIV in Spontaneous/Timed (ST) mode using the ResMed Astral 150 (ResMed, USA), with pressure support of 18–24 cmH₂O, inspiratory time of 0.9–1.1 s, and a backup rate of 18 breaths/min. Oxygen delivery was titrated according to clinical status. Bronchoscopic forceps were used to dissect pleuro-pulmonary adhesions while maintaining sterility.
The 30-minute procedure achieved substantial, although incomplete, removal of pleural adhesions.
During the second procedure, pleural expansion was achieved by intrapleural oxygen insufflation via the FOB suction channel using a 20 mL syringe and an oxygen line. Adhesions were removed, and bacteriological cultures were obtained. T-PA and DNase were carried out in accordance with the summary of product characteristics and the European Respiratory Society (ERS) guidelines, with the aim of removing residual pleural adhesions.
Excessive oxygen administration intermittently induced transient episodes of hypotension and oxygen desaturation, effectively managed through pleural air evacuation, adjustment and optimization of NIV settings, and judicious fluid resuscitation.
A 28 Fr pleural drain was placed via the posterior port under thoracoscopic guidance, followed by lung re-expansion. The drain was removed upon complete re-expansion, absence of air leak, and fluid drainage below 200 mL/day.
Post-procedural CT confirmed significant left lung expansion, and TTE showed an increase in LVEF to 31%, attributed to reduced intrathoracic pressure. The patient was transferred in stable condition to the Department of Thoracic Surgery, where ultrasound-guided pleural puncture yielded 200 mL of fluid. No further accumulation was observed in subsequent days.
Follow up and outcomes
Patient has spent 13 days in the cardiac ICU, followed by 23 days in the Pulmonology ward. On 35th day of his overall hospital stay he was transferred to the Thoracic Surgery Department. He was discharged home after overall hospital stay of 40 days.
Following discharge, the patient underwent three hospitalizations in the Pulmonology Department; first for atrial fibrillation with suspected circulatory decompensation and ESBL-positive E. coli infection, and later for scheduled follow-up. In both cases patient was discharged in stable condition.
The patient remains under regular follow-up at the Pulmonology Outpatient Clinic. Most recent CT performed 2 years after FOB-VAMT showed small calcifications in the lower uvula, thickening of the left oblique fissure, scattered sub-4 mm blurred nodules, and periseptal emphysema in the upper lobes. The bronchial tree and trachea remained patent, with no pleural or pericardial effusion.
The patient is currently treated for COPD with ICS/LABA/LAMA via metered-dose inhaler. Recent spirometry showed moderate obstruction with preserved patency and reduced vital capacity (FEV₁/FVC 0.58; FEV₁ 1.73 L; FVC 2.99 L; Global Lung Initiative reference). During the six-minute walk test, the patient covered a distance of 550 m, without clinically significant desaturation. His arterial blood gases are within normal limits.
Discussion
To the best of our knowledge, no previous reports have described medical thoracoscopy utilizing a single-use flexible bronchoscope (FOB-VAMT) in a patient with stage II empyema and concomitant cardiorespiratory failure deemed ineligible for guideline-recommended management. However, intrapleural examinations by the flexible bronchoscope were performed before, as it was reported by Tonotsuka et al. 14 This paper pertained 20 patients, who underwent diagnostic process due to pleural effusion. In our case report transient episodes of oxygen desaturation was noted. Japanese publication also reported decrease of blood oxygenation, as the anatomical shunt rate and alveolar hypoventilation may occur during the procedure.
The choice of a disposable bronchoscope was justified by many reasons. Due to the numerous intrapleural manipulations regarding the adhesions, the bronchoscope could be damaged beyond repair. A loss of reusable equipment is a greater loss economically wise. Furthermore, it was more comfortable for the operator to use a detachable screen that could be positioned according to the patient's position.
Choice of this method for our patient required critical comparison of available minimally invasive options. Percutaneous catheter drainage was advised as a conservative treatment. Despite numerous days, it did not provide the resolution of aforementioned changes mostly because of pleural adhesions. Removing them was most crucial part of treatment, but it required more invasive, intrapleural intervention. In Poland, VATS is the procedure performed under general anesthesia, which was not permissible by consulting anesthesiologist. Other minimally invasive option would be medical thoracoscopy. However, such method would require additional entry to the pleural cavity. Patient already has working chest tube with wide lumen, which could serve as a uniportal access without causing another wound. Medical thoracoscope is equiped with the camera to provide intrapleural vision. Using flexible bronchoscope, operator could not only see what is inside, but also remove adhesions by forceps placed through the working channel of the bronchoscope. Additional advantage was the ability to insufflate and desufflate according to clinical status of the patient.
According to AATS 2017 Guidelines, VATS is recommended as the first-line treatment for all patients with stage II acute empyema. The procedure is intended to achieve complete removal of potentially infected fluid and/or material, as well as full re-expansion of the lung. Key technical considerations for both approaches include ensuring safe access to the thoracic cavity, effective pleural space drainage, and performing maneuvers that allow the lung to fully expand. Avoidance of rib distraction is more important than the number or size of port incisions. 16
The initial antibiotic regimen for pleural infections is almost always empirical due to the need for prompt initiation of antimicrobial therapy. However, in practice, a complete microbiological workup, including culture and antibiogram, is typically performed as quickly as possible. It is recommended to continue anaerobic coverage since these organisms are difficult to culture, yet frequently cause infections in the pleural space. 8 Treatment strategies are generally derived from lung abscess management, with a widely accepted recommendation of a minimum duration of 3 weeks, guided by clinical, biochemical, and radiological responses.8,17 In practice, prolonged antibiotic courses are used in cases of hospital-acquired infections or infections following surgical procedures18,19
Patients with pleural infections often present with a significant burden of comorbidities, which contributes to prolonged hospitalizations. In this group the prevalence of pre-existing comorbidities is notably high, reaching up to 72% with chronic respiratory and cardiovascular diseases being the most prevalent contributors along with notable rates of malnutrition and alcohol dependence observed in this population, highlighting the importance of dietary supplementation during treatment.5,20 Additionally, the prevalence of diabetes in this patient group is five times higher than in the general population.5,8,21 Concurrent malignancy rates in patients have been reported as high as 30% with an average prevalence of 12–13%, underscoring the need to avoid diagnostic anchoring, especially during prolonged clinical courses2,5,22
Non-general anesthesia surgical options include rib resection, washout, and large-bore tube placement, as well as open window thoracostomy with or without vacuum-assisted closure. These procedures involve intrapleural saline irrigation, medical thoracoscopy, and the implementation of an indwelling pleural catheter along with prolonged antibiotic therapy. 16 23–25 Medical thoracoscopy remains infrequently employed due to its comparatively lower efficacy and the potential for exacerbation of respiratory insufficiency.
A growing body of evidence indicates that delays in pleural infections significantly worsen outcomes, emphasizing the need for early initiation of intrapleural enzyme therapy (IET) alongside prompt surgical consultation. 26 Early imaging should be incorporated into the management plan to guide further treatment, and avoiding surgery should not be the primary goal.
The landmark MIST-2 RCT established that the combination of tPA and DNase resulted in enhanced radiographic clearance (primary outcome) and significant reductions in the rates of surgical referral (77%) and length of stay (6.7 days) when compared to placebo (secondary outcome)27,28
Given that treatment delays are associated with worse outcomes, initiating IET within 48 h of standard care (chest tube drainage and antibiotics) is considered a potentially surgery-sparing approach when there is ongoing evidence of treatment failure. In the absence of direct superiority data, surgical referral is typically considered concurrently with the commencement of IET.8,29
Better 90-day post-operative survival among patients with stage III emphyema is observed when the procedure is performed within approximately 7 days since admission. Study published by Lin et al. also states that mortality increases threefold thereafter (14.2% vs 44.6%, p < 0.001). 10 Our patient was diagnosed with stage II emphyema. His general condition and comorbidities prohibited prompt surgical interventions.
The advantages of performing VATS include significantly reduced postoperative pain. In Nomori's study, it was demonstrated that the pain level after video-assisted thoracoscopic surgery was twice as low (VAS 2–4) compared to Dabir's study (classical thoracotomy, VAS 6–8). 30
Surgical intervention should be considered if, despite the use of drainage and antibiotic therapy, signs of infection persist for more than 7 days and pleural fluid remains present. In such cases, the possible surgical treatment options include VATS, open pleural cavity drainage, thoracotomy, and decortication. There are potentially life-threatening procedure associated risks that the team performing the VATS procedure should be prepared for. Possible complications following VATS include persistent air leaks and the formation of subcutaneous emphysema, incomplete lung re-expansion after the procedure, bleeding, and postoperative pain.
The patient was disqualified from surgical management following anesthesiological evaluation, which revealed decompensated chronic respiratory failure and contraindications to one-lung ventilation (OLV). Thoracic surgery consultation further identified severe biventricular dysfunction (LVEF 15%) and an inability to tolerate double-lumen endotracheal intubation. Given the necessity of OLV for thoracoscopic access-entailing intentional lung collapse and associated right-to-left intrapulmonary shunting-initiation of VATS in this context posed an unacceptably high risk of worsening hypoxemia and circulatory compromise. While pleuro-pulmonary adhesions were present, their morphology was not deemed prohibitive for VATS, as they were limited to thin, non-fibrous adhesions amenable to blunt dissection. Nonetheless, the anticipated hemodynamic impact of intraoperative pneumothorax in a patient with severely impaired cardiac reserve precluded safe surgical intervention. FOB-VAMT has potential to be less straining for the circulatory system. Moreover, midazolam and fentanyl constitutes a satisfactory analgosedation while NIV is clinically appropriate approach during FOB-VAMT procedure, which does not involve thoracic expansion. On the other hand, limitations such as narrow working channel, reduced visibility and restrictions of the bronchoscope itself must be acknowledged. More prospective studies pertaining FOB-VAMT are necessary.
Conclusions
Minimally invasive approach of FOB-VAMT may provide an effective therapeutic option for patients with certain intrapleural illnesses, who are disqualified from standard VATS and endotracheal intubation. Case report of our patient suggests that this method may be utilized safely in patient groups with some contraindications. However, randomized control trials are needed to develop dedicated guidelines.
Patient's perspective
The patient reports the feeling of unease and hopelessness through the whole diagnostic process. He reports that after being hospitalized for almost half a year he only felt senselessness and he stopped believing that he could be really treated as he was disqualified from procedures. Later on when the decision was made to perform the FOB-VAMT procedure, he felt for the first time that someone really wanted to take care of him.As he reports after the procedure he felt relief and a significant improvement in his well-being, as the dyspnea rapidly vanished. Thanks to the procedure his state improved.
Signed consent to publish this case was obtained.
Footnotes
Acknowledgements
Authors would like to acknowledge the medical staff of all aforementioned Department, who took part of this particular patient's care.
Ethics approval and consent to participate
Ethics committee of Medical University of Silesia stated that case reports do not need to be approved by a review board.
Consent for publication
Patient's consent was obtained and downloaded as a supplementary file
Author contributions
![]() . Also serving as a corresponding author, submitted previous versions and corrections.
. Also serving as a corresponding author, submitted previous versions and corrections.
![]() as well as preparing the radiological descriptions.
as well as preparing the radiological descriptions.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical University of Silesia in Katowice, (grant number BNW-1-121/N/5/K).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data and material could be made available only in accordance to the European General Data Protection Regulation.
